CleanCap Co-Transcriptional Capping: Revolutionizing mRNA Synthesis for Therapeutics and Vaccines
This article provides a comprehensive overview of CleanCap co-transcriptional capping technology, a transformative approach for mRNA synthesis.
CleanCap Co-Transcriptional Capping: Revolutionizing mRNA Synthesis for Therapeutics and Vaccines
Abstract
This article provides a comprehensive overview of CleanCap co-transcriptional capping technology, a transformative approach for mRNA synthesis. Tailored for researchers, scientists, and drug development professionals, it covers the foundational science behind the 5' cap's role in mRNA stability and translation. It details practical methodologies, optimization strategies to maximize yield and cost-efficiency, and presents comparative data and case studies validating its superior performance over legacy capping methods. The content synthesizes the latest advancements to serve as a essential guide for implementing this efficient technology in therapeutic and vaccine development pipelines.
The mRNA 5' Cap: Understanding the Critical Role of Cap Structures in Therapeutic Efficacy
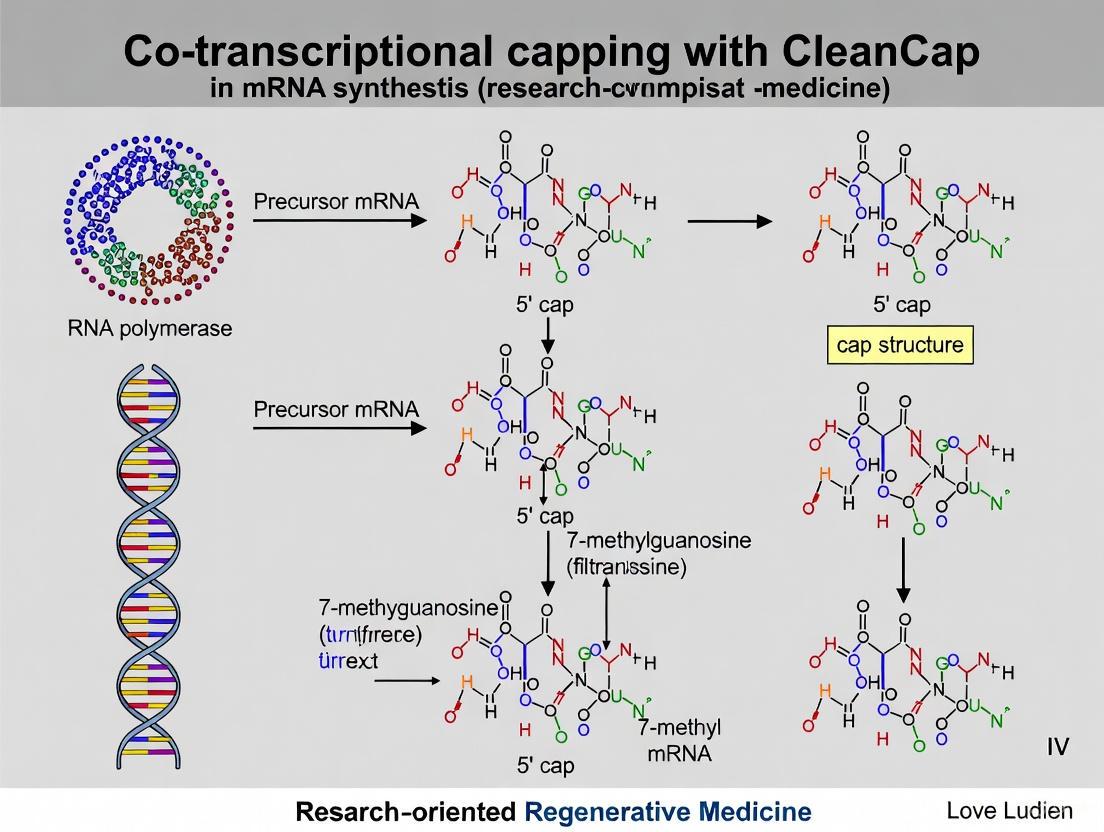
The 5' cap is an evolutionarily conserved, modified nucleotide structure found at the 5' end of eukaryotic messenger RNA (mRNA). This critical modification consists of a 7-methylguanylate (m7G) connected to the initial nucleotide of the RNA via a unique 5' to 5' triphosphate linkage (m7GpppN) [1] [2]. Beyond this basic "cap-0" structure, further modifications frequently occur: methylation of the 2'-O position of the first transcribed nucleotide ribose yields a "cap-1" structure (m7GpppNm), while methylation of the second nucleotide creates "cap-2" [1] [3]. For synthetic mRNA, achieving the cap-1 structure is particularly crucial as it is a key identifier for the innate immune system, helping to distinguish self from non-self RNA [3] [2].
This Application Note examines the biological functions of the 5' cap, emphasizing its indispensable role in regulating mRNA stability, translation efficiency, and immunogenicity. With a specific focus on co-transcriptional capping with CleanCap technology, we provide detailed protocols and data to guide researchers in optimizing mRNA synthesis for therapeutic and vaccine development.
Biological Functions of the 5' Cap
The 5' cap is not a single entity but a complex structure whose precise composition dictates its functional interactions with cellular machinery. The following diagram illustrates the key structural forms and their primary biological consequences.
Figure 1: The relationship between 5' cap structures and their core biological functions. The Cap-1 structure is critical for immune evasion, while non-natural modifications like N6-propargyladenosine (N6pAm) can modulate immune response [4] [3].
Regulation of mRNA Stability
The 5' cap protects mRNA from degradation by 5' to 3' exonucleases [1] [2]. Its chemical resemblance to the 3' end of an RNA molecule provides significant resistance to exonucleolytic degradation [1]. Furthermore, the cap structure is recognized by specific cap-binding proteins that actively block the access of decapping enzymes [1]. The cytoplasmic (re)-capping complex found in mammalian cells adds a layer of regulation, potentially reactivating stored mRNAs in P-bodies by recapping them, which allows for rapid translational responses to cellular stimuli [2].
Promotion of Translation
The cap is fundamental for efficient cap-dependent translation initiation. In the nucleus, the cap-binding complex (CBC) associates with the cap [1] [2]. After export to the cytoplasm, this complex is replaced by the translation initiation factor eIF4E, which is part of the eIF4F complex [1]. eIF4E bound to the cap interacts with eIF4G, which in turn recruits the 43S pre-initiation complex, facilitating ribosome loading and the scanning process to locate the start codon [1] [2]. This interaction also promotes 5' to 3' looping of the mRNA, bringing the cap and poly(A) tail into proximity to enhance circularization and repeated rounds of translation [3].
Modulation of Immunogenicity
The specific structure of the 5' cap is a primary mechanism for the innate immune system to distinguish self-RNA from non-self-RNA [3] [2]. The cap-1 structure (m7GpppNm) is particularly important for evading detection by cytosolic innate immune receptors such as RIG-I and IFIT1, which can recognize uncapped RNA or RNA with only a cap-0 structure as "non-self," triggering a potent type I interferon (IFN) response [4] [3]. This response can block the translation of the mRNA vaccine, undermining its efficacy [4]. Recent research also explores non-natural cap modifications; for instance, N6-propargyladenosine (N6pAm) at the transcription start nucleotide has been shown to increase the immune response of reporter and SARS-CoV-2 RBD mRNAs in human cells by approximately threefold, suggesting a strategy for tailoring mRNA for specific therapeutic applications [4].
Capping Methodologies and Quantitative Comparisons
The method used to cap in vitro transcribed (IVT) mRNA is a critical determinant of its quality and functionality. The primary methodologies are co-transcriptional capping and post-transcriptional enzymatic capping.
Co-transcriptional Capping with CleanCap
CleanCap technology represents a significant advancement in mRNA synthesis. It employs a trinucleotide cap analog (e.g., CleanCap AG) that is added directly to the IVT reaction [5] [6]. This analog base-pairs with an A-inserted T7 class III φ6.5 promoter during transcription initiation, leading to the direct incorporation of a cap-1 structure with high efficiency [6]. This one-pot strategy streamlines manufacturing, reducing production timelines and costs compared to other capping methods [5].
Post-Transcriptional Enzymatic Capping
This two-step method involves first producing uncapped RNA via IVT, followed by the sequential action of capping enzymes. The Vaccinia Capping Enzyme (VCE) and S-adenosylmethionine (SAM) are used to generate the cap-0 structure, which can then be converted to cap-1 by the mRNA Cap 2'-O-Methyltransferase enzyme [3] [7]. While this method is effective, it requires additional enzymatic steps and purification, making it more time-consuming and complex than co-transcriptional capping [3].
Table 1: Quantitative Comparison of mRNA Capping Methodologies
| Methodology | Cap Structure | Capping Efficiency | Protein Expression | Key Advantages | Key Limitations |
|---|---|---|---|---|---|
| CleanCap (Co-transcriptional) [5] [7] | Cap-1 | >95% [5] | Highest (e.g., +30% with CleanCap M6) [5] | Single-step, high yield, simplified workflow, superior efficiency | Higher cost, patent/licensing requirements [7] |
| ARCA (Co-transcriptional) [3] [7] | Cap-0 | 50-80% [7] | Moderate | No licensing requirements | Lower efficiency, requires additional step for Cap-1 [7] |
| Enzymatic (Post-transcriptional) [3] [7] | Cap-0 → Cap-1 | High (when performed sequentially) | High | High fidelity, produces natural cap structure | Multi-step process, longer workflow, higher complexity [3] |
The Scientist's Toolkit: Essential Reagents for mRNA Capping Research
Table 2: Key Research Reagent Solutions for mRNA Capping
| Item | Function/Description | Example Product/Catalog |
|---|---|---|
| CleanCap Reagent | Trinucleotide cap analog for co-transcriptional synthesis of Cap-1 mRNA. | CleanCap AG, CleanCap M6 [5] |
| Vaccinia Capping Enzyme (VCE) | Adds a 7-methylguanylate cap (Cap-0) to the 5' end of uncapped RNA. | Takara Bio #2460 [7] |
| mRNA Cap 2'-O-Methyltransferase | Converts Cap-0 to Cap-1 structure by methylating the 2'-O position of the first nucleotide. | Takara Bio #2470 [7] |
| S-Adenosylmethionine (SAM) | Methyl group donor for methylation reactions catalyzed by methyltransferase enzymes. | Included with capping enzymes [7] |
| In Vitro Transcription Kit | Provides optimized system for high-yield mRNA synthesis, compatible with cap analogs. | Takara IVTpro mRNA Synthesis System [3] |
| Linearized DNA Template | Template for IVT; requires specific transcription start site (e.g., AGG for CleanCap AG). | Cloning Kit for mRNA Template [7] |
Detailed Experimental Protocols
Protocol: High-Yield mRNA Synthesis Using CleanCap Co-Transcriptional Capping
This protocol is adapted for the Takara IVTpro mRNA Synthesis System and CleanCap Reagent AG [3] [7].
Materials:
- Linearized DNA template with transcription start site AGG [7]
- Takara IVTpro mRNA Synthesis Kit (includes T7 RNA Polymerase, NTPs, Buffer)
- CleanCap Reagent AG (e.g., TriLink BioTechnologies)
- DNase I (RNase-free)
- LiCl Precipitation Solution
Procedure:
- Reaction Setup: In a nuclease-free microcentrifuge tube, assemble the following components at room temperature to prevent precipitation of DNA templates:
Incubation: Mix gently and incubate at 37°C for 2 hours.
Template Digestion: Add 2 µL of DNase I (RNase-free) to the reaction mix. Mix gently and incubate at 37°C for 15 minutes to digest the DNA template.
mRNA Purification: Purify the mRNA using the LiCl precipitation method included in the kit or a suitable chromatography-based method. Resuspend the purified mRNA in nuclease-free water.
Quality Control: Quantify mRNA yield by UV absorbance. Analyze cap incorporation efficiency and integrity by LC-MS or analytical gel electrophoresis [8].
Protocol: Analysis of 5' Cap Integrity and Purity by LC-MS
Monitoring cap integrity is crucial, as cap degradation impurities (e.g., hydrolysis products) can significantly impact protein expression [8].
Materials:
- Capped mRNA sample
- Nuclease P1
- Snake Venom Phosphodiesterase (SVPD)
- Alkaline Phosphatase
- LC-MS system with reversed-phase column
Procedure:
- mRNA Digestion to Nucleosides:
- Dilute 2-5 µg of mRNA to a final volume of 20 µL in nuclease-free water.
- Add 2.5 µL of 100 mM ammonium acetate pH 5.3, 0.5 µL of nuclease P1 (1 U/µL), and 1 µL of SVPD (0.001 U/µL).
- Incubate at 37°C for 2 hours.
- Add 3.5 µL of 100 mM Tris-HCl pH 8.0 and 1 µL of alkaline phosphatase (1 U/µL). Incubate at 37°C for an additional 2 hours [4].
LC-MS Analysis:
- Analyze the digested sample using an LC-MS system with a C18 reversed-phase column.
- Use a gradient of methanol or acetonitrile in an aqueous mobile phase (e.g., ammonium acetate).
- Operate the mass spectrometer in negative ion mode for detection.
- Monitor for specific mass transitions corresponding to cap structures (e.g., Am, m6Am, N6pAm) and their degradation products using dynamic multiple-reaction monitoring (dMRM) for sensitive quantification [4].
Data Interpretation: Identify and quantify cap structures and impurities by comparing retention times and mass spectra to synthesized standards (e.g., Am, m6Am, N6pAm) [4]. Be aware that method-induced artifactual degradation can occur during LC-MS analysis; use reference standards and optimize methods to minimize this [8].
Advanced Applications and Future Directions
Engineering the Cap Structure to Modulate Immune Response
Beyond the natural cap-1 structure, researchers are exploring chemo-enzymatic modifications to fine-tune mRNA properties. The methyltransferase CAPAM, responsible for N6-methylation of adenosine start nucleotides (forming m6Am), has been shown to accept synthetic AdoMet analogs [4]. This allows for the installation of non-natural modifications, such as propargyl groups (N6pAm). mRNAs with this modification are efficiently translated and have shown an approximately threefold increased immune response in human cells, presenting a novel strategy for designing next-generation vaccines with enhanced adjuvant activity [4].
Cap-Independent Translation and 5' End Modification
While cap-dependent translation is the standard for most mRNA therapies, cap-independent translation via Internal Ribosome Entry Sites (IRESs) holds promise for targeting diseases like cancer and neurodegeneration, where cap-dependent translation is impaired [6]. A recent innovation involves priming IVT with an azido-functionalized dinucleotide (CleaN3), which incorporates a 5' azide moiety instead of a cap. This allows for efficient post-transcriptional modification via click chemistry (e.g., with AF647 dye), significantly enhancing the stability and protein output of cap-independently translated mRNAs without eliciting immunogenicity [6]. This approach also provides a powerful tool for studying IRES-driven translation and cellular mRNA fate.
The 5' cap is a master regulator of mRNA biology, with its specific structure dictating stability, translational efficiency, and interaction with the host immune system. The advent of co-transcriptional capping technologies like CleanCap has revolutionized the production of therapeutic mRNA, enabling the high-yield synthesis of cap-1 mRNA with >95% efficiency, which is critical for minimizing immunogenicity and maximizing protein expression [5]. As the field progresses, advanced techniques for cap analysis, the engineering of novel cap structures, and the exploration of cap-independent translation mechanisms will provide researchers with an expanding toolkit to tailor mRNA molecules for precise and potent therapeutic applications.
The 5' cap structure is an essential modification for eukaryotic messenger RNA (mRNA), influencing its stability, translational efficiency, and immunogenicity. While the basic cap-0 structure (m7GpppN...) is sufficient for some functions, the cap-1 structure (m7GpppN'm...), with a additional 2'-O-methylation on the first transcribed nucleotide, is critical for high protein expression and evading the host immune response. This application note decodes the structural and functional distinctions between cap-0 and cap-1, underscoring the transformative impact of cap-1 on mRNA therapeutic performance. We provide a detailed protocol for synthesizing cap-1 mRNA using the co-transcriptional capping method with CleanCap technology, which streamlines production and achieves superior capping efficiency exceeding 95%. Quantitative data and structured workflows are included to guide researchers in leveraging this advanced capping strategy for robust protein expression in vaccine and therapeutic development.
The 5' m7G cap is an evolutionarily conserved modification found on the 5' end of all eukaryotic mRNAs [2]. This structure is not merely a protective end; it serves as a dynamic molecular module that recruits cellular proteins to mediate critical biological functions throughout the mRNA life cycle. These functions include pre-mRNA processing, nuclear export, and the cap-dependent initiation of protein synthesis [2]. The cap structure fundamentally exists in two primary forms that are pivotal for applied mRNA research:
- Cap 0: This base structure consists of an N7-methylated guanosine (m7G) linked to the first nucleotide of the RNA via a 5' to 5' triphosphate bridge (m7GpppN) [1] [9].
- Cap 1: This enhanced structure includes an additional 2'-O-methylation on the ribose of the first transcribed nucleotide adjacent to the m7G cap (m7GpppN'm) [2] [9].
Recent research has highlighted a crucial biological role for the cap 1 structure: it acts as an identifier of "self" RNA, enabling the innate immune system to distinguish host mRNA from foreign RNA, which often lacks this modification. Recognition of non-cap 1 RNA can trigger a potent antiviral response, leading to the suppression of translation and degradation of the RNA [2]. For mRNA therapeutics, this means the cap 1 structure is indispensable for minimizing immunogenicity and ensuring high levels of target protein expression.
Cap Structures: A Comparative Analysis
The incremental structural difference between Cap 0 and Cap 1 has profound implications for the functionality and efficacy of synthetic mRNA.
Table 1: Comparative Analysis of Cap 0 and Cap 1 Structures and Their Functional Impact
| Feature | Cap 0 Structure | Cap 1 Structure |
|---|---|---|
| Chemical Structure | m7GpppN... | m7GpppN'm... |
| Methylation Status | N7-methylguanine only | N7-methylguanine + 2'-O-methylation of the +1 nucleotide |
| Immunogenicity | Higher; recognized as "non-self" by innate immune system [2] | Lower; mimics native human mRNA, evading immune detection [9] |
| Translational Efficiency | Moderate | High; promotes superior protein expression [5] |
| mRNA Stability | Standard | Enhanced |
| Typical Production Method | Enzymatic capping (e.g., VCE) or ARCA analogs | Advanced co-transcriptional capping (e.g., CleanCap) [10] |
The following diagram illustrates the key structural and functional relationships of the mRNA cap, from its basic role in translation to the critical immune evasion conferred by the Cap 1 structure.
Diagram 1: Functional impact of cap structures on translation and immunogenicity. The Cap 1 structure is essential for evading immune detection and enabling high levels of protein production.
Quantitative Impact on Protein Expression
The choice of capping strategy directly influences capping efficiency and translational capacity. Legacy methods struggle to achieve high capping efficiency, resulting in a mixed population of capped and uncapped transcripts that compromise overall protein yield. Quantitative comparisons demonstrate the superiority of modern co-transcriptional capping.
Table 2: Performance Comparison of mRNA Capping Methods
| Capping Method | Cap Structure | Typical Capping Efficiency | Relative Protein Expression | Key Characteristics |
|---|---|---|---|---|
| Enzymatic Capping (VCE + 2'-O-MTase) | Cap 1 | Variable; requires optimization | Baseline (High if efficient) | Multi-step, post-transcriptional process [9] |
| ARCA (Anti-Reverse Cap Analog) | Cap 0 | 50-80% [10] | Lower than Cap 1 | Co-transcriptional; requires additional step for Cap 1 [10] |
| CleanCap AG Analog | Cap 1 | >95% [5] | High | Single-step, co-transcriptional [5] [10] |
| CleanCap M6 Analog | Cap 1 (with m6A) | >95% [5] | Highest (≥30% higher than AG) [5] | Next-generation analog with additional modifications |
Protocol: Co-Transcriptional mRNA Synthesis with CleanCap Technology
This section provides a detailed methodology for synthesizing high-quality, cap-1 mRNA using the one-pot co-transcriptional capping approach with CleanCap reagent. This protocol prioritizes efficiency and simplicity, enabling the production of mRNA suitable for preclinical research and therapeutic development [5] [11].
Materials and Reagents
Table 3: Research Reagent Solutions for Co-Transcriptional Capping
| Item | Function | Example Product |
|---|---|---|
| DNA Template | Template for IVT; must end with AGG transcription start site [10]. | Cloning Kit for mRNA Template (Takara) |
| CleanCap Analog | Cap 1 analog incorporated during transcription [5]. | CleanCap AG or M6 (TriLink) |
| RNA Polymerase | Enzyme that catalyzes RNA synthesis from DNA template. | T7 RNA Polymerase |
| Nucleotide Triphosphates (NTPs) | Building blocks for RNA synthesis. | Modified NTPs (e.g., N1-methylpseudouridine) |
| RNase Inhibitor | Protects synthesized mRNA from degradation. | Recombinant RNase Inhibitor |
| DNase I | Degrades DNA template post-transcription. | RNase-free DNase I |
| Purification Kit/Reagents | Purifies IVT mRNA from reaction components. | LiCl precipitation or spin-column kits |
Step-by-Step Workflow
- Template Design and Preparation: Linearize a plasmid DNA template using a restriction enzyme that leaves a 3' overhang or uses a blunt-end cutter downstream of the poly(A) tail sequence. Critically, the +1, +2, and +3 nucleotides of the template must be A-G-G to be fully compatible with CleanCap AG analog [10]. For other analogs like CleanCap AU, verify the required start sequence.
- In Vitro Transcription (IVT) Reaction Setup: Assemble the following reaction at room temperature to prevent precipitation of reaction components:
- Linearized DNA template (1 µg)
- 10X IVT Buffer (2 µL)
- CleanCap AG or M6 Analog (e.g., 6 mM, 4 µL)
- ATP, CTP, UTP, GTP (e.g., 75 mM each, 10 µL total)
- T7 RNA Polymerase (e.g., 2 µL)
- RNase Inhibitor (e.g., 0.5 µL)
- Nuclease-free water to a final volume of 20 µL Mix the components gently by pipetting and incubate at 37°C for 2 hours.
- DNase I Treatment: After incubation, add 2 µL of DNase I (RNase-free) to the reaction mix. Gently mix and incubate at 37°C for 15 minutes to digest the DNA template.
- mRNA Purification: Purify the mRNA using a method such as LiCl precipitation or a silica-membrane based spin-column kit according to the manufacturer's instructions. LiCl precipitation is effective at removing proteins and unincorporated NTPs. Determine the mRNA concentration and purity by measuring absorbance at 260 nm and the A260/A280 ratio, respectively. Analyze mRNA integrity by denaturing agarose gel electrophoresis.
The following workflow diagram summarizes the key steps of this protocol, highlighting the streamlined nature of co-transcriptional capping.
Diagram 2: Co-transcriptional capping workflow for Cap 1 mRNA synthesis. This one-pot method efficiently produces high-quality mRNA in a simplified process.
Quality Control and Analysis
- Capping Efficiency: Analyze capping efficiency using reverse-phase HPLC or LC-MS. CleanCap technology routinely achieves efficiencies >95% [5].
- mRNA Purity and Integrity: Assess mRNA integrity via capillary electrophoresis (e.g., Fragment Analyzer) or agarose gel electrophoresis. A single, sharp band should be visible. Monitor for double-stranded RNA (dsRNA) contaminants, which can be highly immunogenic.
- Functional Assay: Transfert the synthesized mRNA into a relevant cell line (e.g., HEK-293) and measure target protein expression using an appropriate method (e.g., ELISA, western blot, fluorescence microscopy) 24-48 hours post-transfection.
The evolution from Cap 0 to Cap 1 represents a critical advancement in mRNA technology. The structural sophistication of the Cap 1 structure is not merely incremental; it is a functional necessity for producing therapeutic-grade mRNA that is highly translatable and minimally immunogenic [2] [9]. The adoption of co-transcriptional capping technologies, particularly CleanCap, has fundamentally streamlined mRNA manufacturing. This method eliminates the need for post-transcriptional enzymatic steps, reducing production time by days and overall manufacturing costs by an estimated 20-40% while simultaneously achieving superior capping efficiency (>95%) and the highest reported levels of protein expression [5].
For researchers and drug development professionals, the implications are clear. The commitment to a high-fidelity Cap 1 structure from the outset of mRNA synthesis is a crucial determinant of success. Integrating co-transcriptional capping into the mRNA synthesis workflow ensures the production of a high-quality product, accelerating the path from research to clinical application in vaccines, protein replacement therapies, and other emerging modalities.
The 5' cap structure is an indispensable modification for messenger RNA (mRNA), serving critical roles in stability, translation efficiency, and immunogenicity regulation. For in vitro transcribed (IVT) mRNA used in therapeutics and vaccines, the capping method is a critical quality attribute directly impacting product efficacy and safety [12]. The field has undergone significant evolution, migrating from multi-step enzymatic capping performed after transcription to streamlined co-transcriptional methods where capping occurs simultaneously with mRNA synthesis. This evolution has been largely driven by the introduction of advanced cap analogs like CleanCap technology, which offer high efficiency and simplified workflows while maintaining biological functionality [5] [13]. This application note traces this technological progression, providing detailed protocols and analytical methods for researchers developing mRNA-based vaccines and therapeutics.
Capping Methodologies: A Comparative Analysis
Post-Transcriptional Enzymatic Capping
Enzymatic capping utilizes enzymes derived from viruses, such as the Vaccinia Capping Enzyme (VCE) or Faustovirus Capping Enzyme (FCE), to add a cap structure to mRNA in a separate reaction step following IVT [14] [15]. This multi-step process biochemically mimics the natural capping pathway in eukaryotic cells.
The enzymatic process typically involves two sequential reactions:
- Cap-0 formation: The capping enzyme (VCE or FCE) catalyzes the addition of a 7-methylguanylate (m7G) cap to the 5' end of the uncapped mRNA, creating a Cap-0 structure. This reaction requires GTP and the methyl donor S-adenosylmethionine (SAM) [14].
- Cap-1 formation: The mRNA Cap 2′-O-Methyltransferase (2′-O-MTase) then adds a methyl group to the 2′-O position of the first nucleotide of the mRNA, resulting in the superior Cap-1 structure, again using SAM as a methyl donor [14] [15].
Table 1: Key Components for Post-Transcriptional Enzymatic Capping
| Component | Concentration | Function |
|---|---|---|
| Capping Enzyme (VCE or FCE) | 10-25 U/µL | Adds 7-methylguanylate cap (Cap-0) [14] |
| 10X Capping Buffer | 10X | Provides optimal reaction conditions [14] |
| GTP | 10 mM | Substrate for the capping reaction [14] |
| S-adenosylmethionine (SAM) | 32 mM | Methyl donor for cap methylation [14] |
| mRNA Cap 2′-O-Methyltransferase | 50 U/µL | Converts Cap-0 to Cap-1 structure [14] |
Co-Transcriptional Capping with Cap Analogs
Co-transcriptional capping simplifies the process by incorporating a synthetic cap analog directly into the IVT reaction mixture. The RNA polymerase then incorporates this analog at the 5' terminus of the nascent mRNA chain during transcription initiation [12]. While early analogs like the Anti-Reverse Cap Analog (ARCA) improved upon the standard mCap, the field has since advanced to trinucleotide cap analogs such as CleanCap [13].
CleanCap technology represents a significant leap forward. It uses a trinucleotide analog (e.g., CleanCap AG) that is co-transcriptionally incorporated by the RNA polymerase, resulting in a natural Cap-1 structure with reported efficiencies exceeding 95% [5] [16]. This "one-pot" strategy integrates capping and transcription into a single step, eliminating the need for additional enzymatic reactions and purifications [13].
Table 2: Comparison of mRNA Capping Methods
| Characteristic | Enzymatic Capping | Co-Transcriptional (ARCA) | Co-Transcriptional (CleanCap) |
|---|---|---|---|
| Cap Structure | Cap-0 or Cap-1 | Cap-0 | Cap-1 |
| Capping Efficiency | 80-100% [14] | Lower than newer methods [13] | >95% [5] |
| Workflow | Multi-step, post-transcriptional [14] | Single-step, co-transcriptional | Single-step, co-transcriptional [5] |
| Key Advantage | All caps in correct orientation; high control [12] | Simple, one-step process | High efficiency, natural Cap-1, streamlined manufacturing [5] [13] |
| Key Disadvantage | Time-consuming, requires extra reagents/purification [13] | Lower efficiency; Cap-0 can be immunogenic [13] | Patent-protected technology |
| Ideal For | Large-scale mRNA manufacturing [15] | Legacy applications | Therapeutic & vaccine R&D, high-yield production [5] [17] |
Detailed Experimental Protocols
Protocol A: Two-Step Enzymatic Capping for High-Quality Cap-1 mRNA
This protocol is ideal for applications requiring precise control over the cap structure and is scalable for manufacturing [15].
Materials:
- Purified, uncapped mRNA transcript
- Vaccinia Capping Enzyme (VCE) or Faustovirus Capping Enzyme (FCE)
- mRNA Cap 2′-O-Methyltransferase (2′-O-MTase)
- 10X Capping Buffer
- GTP (10 mM)
- S-adenosylmethionine (SAM, 32 mM)
- Nuclease-free water
Procedure:
- Cap-0 Reaction Setup: In a nuclease-free tube, combine the following components on ice:
- 1 µg of uncapped mRNA
- 1 µL of 10X Capping Buffer
- 0.5 µL of GTP (10 mM)
- 0.5 µL of SAM (32 mM)
- 1 µL of Capping Enzyme (VCE or FCE, 10 U/µL)
- Nuclease-free water to a final volume of 10 µL.
- Incubation for Cap-0: Mix gently and incubate the reaction at 37°C for 30-60 minutes [14].
- Cap-1 Reaction Setup: Directly to the same tube, add:
- 1 µL of mRNA Cap 2′-O-Methyltransferase (2′-O-MTase, 50 U/µL)
- 1 µL of SAM (32 mM).
- Incubation for Cap-1: Mix gently and incubate at 37°C for 30-60 minutes [14].
- Purification: Terminate the reaction and purify the capped mRNA using standard methods like LiCl precipitation or column-based purification [14].
Note: For a one-step enzymatic protocol, the capping enzyme and the 2′-O-MTase can be combined in a single reaction mixture, as they can function simultaneously [14].
Protocol B: Streamlined Co-Transcriptional Capping Using CleanCap AG Reagent
This protocol leverages modern cap analogs to produce high-quality Cap-1 mRNA in a single, simplified reaction, saving significant time and resources [5] [16].
Materials:
- Linearized DNA template (with T7 promoter)
- CleanCap AG (or AG 3'OMe) Reagent
- N1-methylpseudouridine-5’-triphosphate (or other NTPs)
- CleanScript IVT Enzyme Mix (or T7 RNA Polymerase)
- 10X IVT Buffer
- Nuclease-free water
Procedure:
- IVT Reaction Setup: Combine the following components at room temperature to avoid precipitate formation:
- 1 µg of linearized DNA template
- 2 µL of 10X IVT Buffer
- 6 µL of NTP Mix (e.g., containing 7.5 mM of ATP, CTP, UTP, and modified GTP such as N1-methylpseudouridine-5’-triphosphate)
- 4 µL of CleanCap AG Reagent (e.g., 10 mM)
- 1 µL of RNA Polymerase Mix (e.g., CleanScript IVT Enzyme)
- Nuclease-free water to a final volume of 20 µL [17].
- Incubation: Mix the components gently and incubate the reaction at 37°C for 2-4 hours.
- DNase Treatment: After incubation, add 2 µL of DNase I (if not included in the enzyme mix) and incubate for another 15 minutes at 37°C to digest the DNA template.
- Purification: Purify the mRNA using LiCl precipitation or affinity-based methods to remove enzymes, free NTPs, and cap analogs [14].
Analytical Techniques for Capping Efficiency Assessment
Accurately determining capping efficiency is a critical quality control step. Several advanced methods are employed.
Table 3: Methods for Assessing mRNA Capping Efficiency
| Method | Principle | Key Advantage |
|---|---|---|
| LC-MS/MS | Enzymatic cleavage of mRNA followed by LC-MS analysis of 5' end fragments to identify and quantify capped vs. uncapped species [18] [19]. | High confidence identification and relative quantification of different cap structures (Cap-0, Cap-1) [19]. |
| ELISA | Use of cap-specific antibodies (e.g., against cap-0 or cap-1) to bind and quantify capped mRNA [18] [20]. | High-throughput, amenable to quantifying cap structures without complex instrumentation [20]. |
| qRT-PCR | Uses specialized primers that only anneal to uncapped 5' ends, allowing quantification of uncapped fraction relative to total mRNA [18]. | Highly sensitive, capable of detecting small proportions of uncapped RNA in a sample [18]. |
| Electrophoresis | RNase H cleavage to generate short 5' end fragments, followed by gel or capillary electrophoresis to separate capped and uncapped fragments [18]. | Accessible technique for labs with standard molecular biology equipment. |
Representative LC-MS/MS Workflow [19]:
- Digestion: The mRNA is enzymatically digested with RNase H or other ribonucleases to generate short 5'-end fragments (~20-40 nucleotides).
- Chromatography: The fragments are separated by reversed-phase liquid chromatography using ion-pairing conditions.
- Mass Spectrometry: The eluted fragments are analyzed by high-resolution mass spectrometry in negative ion mode.
- Data Analysis: The accurate mass data is reconstructed and analyzed to identify the uncapped fragment and various capped fragments (e.g., Cap-0, Cap-1). The capping efficiency is calculated as the peak area ratio of the capped species to the total of all 5'-end species.
The Scientist's Toolkit: Essential Reagents for mRNA Capping
Table 4: Key Research Reagent Solutions for mRNA Capping Workflows
| Reagent / Kit | Function | Application Note |
|---|---|---|
| Vaccinia Capping Enzyme (VCE) | Enzymatically adds Cap-0 structure post-transcriptionally [14]. | Robust, well-characterized enzyme for enzymatic capping workflows. |
| Faustovirus Capping Enzyme (FCE) | Enzymatically adds Cap-0 structure; broader temperature range & higher activity than VCE on difficult substrates [14] [15]. | Preferred for challenging sequences or flexible reaction temperatures. |
| mRNA Cap 2′-O-Methyltransferase | Converts Cap-0 to Cap-1 structure post-transcriptionally [14]. | Essential for generating the immunologically favored Cap-1 structure in enzymatic methods. |
| CleanCap AG Reagent | Trinucleotide cap analog for co-transcriptional capping to produce Cap-1 mRNA [5] [16]. | >95% capping efficiency; the industry standard for high-quality IVT mRNA. |
| CleanCap M6 Reagent | Advanced cap analog with additional m6A modification for even higher protein expression [5]. | Reported to increase protein expression by over 30% compared to previous analogs. |
| HiScribe T7 mRNA Kit with CleanCap | All-in-one kit for IVT including T7 polymerase, NTPs, buffer, and CleanCap reagent [15]. | Simplifies workflow, ensures component compatibility, and maximizes yield. |
| TriLink mRNA Synthesis Kit | Comprehensive kit featuring CleanCap AG (3'OMe), CleanScript polymerase, and modified nucleotides to reduce dsRNA [17]. | Designed for high yield (up to 2X more mRNA) and low immunogenicity. |
Workflow Visualization: The Evolution of mRNA Capping
The following diagram illustrates the conceptual and procedural shift from traditional enzymatic capping to modern co-transcriptional methods.
The evolution of mRNA capping from multi-step enzymatic methods to efficient co-transcriptional strategies represents a fundamental advancement in RNA technology. While post-transcriptional enzymatic capping remains valuable for its precise control, the emergence of high-performance cap analogs like CleanCap has set a new standard for therapeutic mRNA production. The ability to generate Cap-1 mRNA with >95% efficiency in a single, streamlined reaction directly addresses the needs of the rapidly developing mRNA vaccine and therapeutic pipeline, reducing manufacturing timelines and costs while ensuring high-quality products [5] [13]. The choice of capping method profoundly impacts downstream outcomes, including protein expression levels and immunogenic profile, making it a critical consideration for any mRNA-based research or development program.
The 5' cap structure is integral to the stability, translation efficiency, and immunogenicity of synthetic mRNA, influencing its performance in therapeutic and vaccine applications. Traditional mRNA capping methods, including enzymatic capping and earlier cap analogs like ARCA (Anti-Reverse Cap Analog), present manufacturing challenges such as multi-step processes, lower yields, and suboptimal capping efficiency. CleanCap technology (TriLink BioTechnologies) represents a significant advancement by enabling a one-pot co-transcriptional capping strategy during in vitro transcription (IVT). This approach simplifies mRNA production, reduces manufacturing timelines, and yields mRNA with an optimal Cap 1 structure and over 95% capping efficiency [5] [21] [22].
Co-transcriptional capping with CleanCap overcomes the limitations of legacy methods by incorporating a cap analog during the transcription reaction itself, eliminating the need for post-transcriptional enzymatic capping. This technology has been utilized in commercially approved RNA vaccines and is supported by reagents and kits suitable for both research-scale and GMP manufacturing, supporting applications from discovery to clinical-scale production [5] [23].
CleanCap Technology and Analog Comparison
Mechanism and Advantages
CleanCap technology employs trinucleotide cap analogs that base-pair with the T7 promoter sequence during transcription initiation. This design allows the RNA polymerase to initiate transcription directly from the cap analog, ensuring its precise incorporation at the 5' end of the mRNA strand. The result is a Cap 1 structure (m7GpppA2'-O-meG), which includes a 2'-O-methylation on the first transcribed nucleotide. This structure is critical for efficient translation and reduced immune recognition in higher eukaryotes [21] [22].
Key advantages of CleanCap technology include:
- Streamlined Workflow: Combines transcription and capping into a single reaction, reducing hands-on time and purification steps [5].
- High Efficiency: Achieves consistent capping efficiency exceeding 95%, surpassing the performance of dinucleotide analogs like ARCA and mCap [21].
- Enhanced mRNA Quality: Contributes to high mRNA yields and reduced levels of double-stranded RNA (dsRNA) impurities when used with optimized systems [23] [24].
- Economic Benefit: Can reduce overall mRNA manufacturing costs by 20-40% compared to other capping methods [5].
Comparative Analysis of CleanCap Analogs
TriLink offers a portfolio of CleanCap analogs tailored for different applications. The table below summarizes the key characteristics of four prominent analogs.
Table 1: Comparison of CleanCap Analog Properties and Applications
| Analog Name | Structure & Modifications | Application | Capping Efficiency | Protein Expression |
|---|---|---|---|---|
| CleanCap M6 | Cap 1, 3'-O-methylation on m7G, m6A modification [5] | mRNA [5] | >95% [5] | Highest (≥30% increase vs. AG 3'OMe) [24] [25] |
| CleanCap AG 3'OMe | Cap 1, 3'-O-methylation on m7G [5] | mRNA [5] | >95% [5] | Higher [5] |
| CleanCap AG | Cap 1 [5] | mRNA [5] | >95% [5] | High [5] |
| CleanCap AU | Cap 1, Alphavirus 5' cap [5] | Self-amplifying RNA [5] | >95% [5] | Durable [5] |
Among these, CleanCap M6 is the most advanced, featuring an N6-methyladenosine (m6A) modification on the first transcribed nucleotide. This modification impedes the Dcp2-mediated decapping process, enhancing mRNA stability and leading to a demonstrated increase in protein expression of over 30% compared to CleanCap AG (3' OMe) in vivo [24] [25] [26]. CleanCap AG (3' OMe) is a versatile analog known for its use in commercially approved vaccines, while CleanCap AU is specifically designed for self-amplifying RNA applications [5] [23].
Figure 1: CleanCap co-transcriptional capping mechanism. The trinucleotide analog base-pairs with the promoter during initiation, ensuring precise 5' cap incorporation.
Application Notes: Performance and Economic Data
Quantitative Performance Metrics
The implementation of CleanCap technology, particularly within optimized IVT kits, delivers significant improvements in key mRNA quality attributes. The following table summarizes experimental data from head-to-head comparisons.
Table 2: Experimental Performance Metrics of CleanCap IVT Kits
| Performance Metric | CleanCap AG (3' OMe) Kit | CleanCap M6 Kit | Legacy Methods (ARCA/mCap) |
|---|---|---|---|
| Capping Efficiency | >95% [24] | >95% [5] | ~70% (mCap), <90% (ARCA) [21] |
| mRNA Yield (per 100µL rxn) | 0.8-1 mg [24] | 0.8-1 mg (up to 10 mg/mL with pulse feed) [24] | Significantly lower [21] |
| dsRNA Reduction | Up to 85% [23] [24] | Up to 85% [5] | Not specified |
| Relative Protein Expression | High (Baseline) [5] | ≥30% Higher [24] [25] | Lower [21] |
These metrics underscore the dual benefit of CleanCap technology: enhancing the critical quality attributes of the mRNA (capping efficiency, purity) while simultaneously improving the yield and translational output. The reduction in dsRNA is a crucial factor in minimizing unwanted innate immune responses upon delivery [23].
Economic and Workflow Impact
Adopting a one-pot co-transcriptional capping strategy with CleanCap confers substantial economic and operational advantages:
- Process Acceleration: Cuts mRNA therapeutic production processes by nearly one week by eliminating the separate enzymatic capping step and reducing purification needs [5].
- Cost Reduction: Lowers overall manufacturing costs by 20-40% compared to other capping methods, primarily by reducing reagent consumption and process complexity [5].
- Simplified Operations: The all-in-one IVT kits consolidate high-performing components (cap analog, polymerase, nucleotides, buffer), simplifying researcher workflow and accelerating discovery [23].
Detailed Experimental Protocols
Standard Protocol for mRNA Synthesis Using CleanCap AG (3' OMe) IVT Kit
This protocol is designed for a standard 100 µL reaction using the CleanCap AG (3' OMe) CleanScript IVT Kit (TriLink, Cat. No. K-7413) to synthesize up to 1 mg of capped mRNA [24] [27].
Research Reagent Solutions & Materials: Table 3: Essential Materials for CleanCap IVT
| Item | Function/Description | Example/Component |
|---|---|---|
| DNA Template | Template for transcription; must be linearized and contain T7 promoter with AG start [27]. | 5 µg of linearized plasmid or purified PCR product [27]. |
| CleanCap Analog | Co-transcriptional capping reagent; provides Cap 1 structure. | CleanCap AG (3' OMe) or CleanCap M6 [24]. |
| NTPs / Modified NTPs | Nucleotide building blocks for RNA synthesis. | ATP, CTP, GTP, UTP; N1-Methylpseudouridine-5'-Triphosphate [27]. |
| RNA Polymerase Mix | Engineered T7 RNA polymerase for transcription with low dsRNA byproduct. | CleanScribe RNA Polymerase Mix [27]. |
| Reaction Buffer | Optimized buffer for co-transcriptional capping and high yield. | 10X CleanScript IVT Buffer [27]. |
| Nuclease-Free Water | Solvent to maintain RNase-free conditions. | - |
Methodology:
- Template Preparation: Use a linearized plasmid DNA template (50 µg/mL final concentration) or a purified PCR product (25 µg/mL final concentration). The T7 promoter must be followed by an AG initiation sequence (5'-AGG-3' or 5'-AGA-3') for proper CleanCap incorporation [27].
- Reaction Assembly: Thaw all kit components on ice and mix gently. Combine the following in a nuclease-free microcentrifuge tube:
- 10 µL of 10X AG CleanScript IVT Buffer
- 10 µL of ATP Solution (100 mM)
- 10 µL of CTP Solution (100 mM)
- 10 µL of GTP Solution (100 mM)
- 10 µL of Uridine Triphosphate Solution (100 mM) or N1-Methylpseudouridine-5'-Triphosphate
- 10 µL of CleanCap Reagent AG (3' OMe)
- 5 µL of AG CleanScribe RNA Polymerase Mix
- 5 µg of DNA Template (e.g., 10 µL of a 0.5 µg/µL solution)
- Add Nuclease-Free Water to a final volume of 100 µL [27].
- Incubation: Mix thoroughly and incubate the reaction at 37°C for 3 hours. Use a thermal cycler with a heated lid or a dry air incubator to prevent evaporation and condensation [27].
- DNase Treatment (Optional but Recommended): After IVT, add 2 µL of DNase I (provided in kit) per 100 µL reaction and incubate at 37°C for 15 minutes to degrade the DNA template [27].
- mRNA Purification: Purify the mRNA using a preferred method, such as lithium chloride precipitation or spin-column-based purification (e.g., RNeasy kits), to remove enzymes, free nucleotides, and salts [27].
- Quality Control: Analyze the purified mRNA by spectrophotometry (for concentration and purity), agarose gel electrophoresis (for integrity), and techniques like LC-MS or IP-RP HPLC to confirm capping efficiency [22].
Advanced Application: Utilizing CleanCap M6 for Enhanced Expression
For the highest protein expression levels, the CleanCap M6 IVT Kit is recommended. The protocol is similar to Section 4.1, but with kit-specific buffer and polymerase mix [24].
Key Considerations:
- 5' UTR Compatibility: The yield of the CleanCap M6 IVT reaction can be impacted by some 5' UTR sequences. If yield is lower than expected, consult technical support (support@trilinkbiotech.com) for sequence review and optimization [24].
- Pulse-Feed Protocol: For yields up to 10 mg/mL, a supplementary pulse-feed protocol can be used, where additional NTPs are added during the reaction to sustain IVT efficiency [24].
Figure 2: CleanCap IVT workflow. The process from template preparation to quality-controlled mRNA.
Implementing CleanCap: A Practical Guide to Analogs, Kits, and Workflow Integration
The 5' cap structure is a critical determinant of the stability, translational efficiency, and immunogenicity of in vitro-transcribed (IVT) mRNA [28]. Co-transcriptional capping with CleanCap technology represents a significant advancement over legacy capping methods, such as ARCA or enzymatic capping, by enabling the one-pot synthesis of mRNA with optimal Cap 1 structures at efficiencies exceeding 95% [5]. This guide provides a detailed comparative analysis of the prominent CleanCap analogs—AG, AG (3' OMe), M6, and AU—to empower researchers and drug development professionals in selecting the ideal analog for their specific RNA application, thereby accelerating the development of mRNA vaccines and therapeutics.
CleanCap Analog Comparison and Selection Guide
Comparative Analysis of Analog Properties
Selecting the appropriate cap analog is crucial for optimizing mRNA performance. The table below summarizes the key characteristics of four CleanCap analogs to inform your selection.
Table 1: Comparative Guide to CleanCap Analogs
| Analog Name | Recommended RNA Type | Key Structural Features | Capping Efficiency | Reported Protein Expression |
|---|---|---|---|---|
| CleanCap AG [5] | Standard mRNA | Cap 1 structure (2'-O-methylation on the first base) | >95% [5] | High [5] |
| CleanCap AG (3' OMe) [5] [23] | Standard mRNA | Cap 1 structure with 3'-O-methylation on m7G | >95% [5] | Higher [5] |
| CleanCap M6 [5] [29] | Potent mRNA Therapeutics | Cap 1, 3'-O-methylation on m7G, and m6A modification on the +1 adenosine [5] [25] | >95% [5] | Highest (Over 30% increase vs. other analogs) [5] [29] |
| CleanCap AU [5] | Self-Amplifying RNA (saRNA) | Cap 1 structure with an alphavirus-specific 5' cap sequence [5] | >95% [5] | Durable [5] |
Analog Selection and Application Workflow
The following diagram illustrates the decision-making process for selecting the most suitable CleanCap analog based on research goals and RNA type.
Experimental Protocols and Workflows
Standard mRNA Synthesis Protocol Using CleanCap Analogs
This protocol is optimized for the co-transcriptional synthesis of capped mRNA using CleanCap AG, AG (3' OMe), or M6 analogs.
Table 2: Research Reagent Solutions for mRNA Synthesis
| Reagent/Material | Function/Description | Example/Note |
|---|---|---|
| CleanCap Analog | Co-transcriptional capping reagent | Choose from AG, AG (3' OMe), M6, or AU based on application [5]. |
| CleanScript IVT Buffer | Optimized reaction buffer for IVT | Included in TriLink's IVT kits for increased yield [23]. |
| N1-methylpseudouridine-5'-triphosphate | Modified nucleoside to reduce immunogenicity | Replaces UTP to enhance mRNA performance and lower innate immune recognition [5] [23]. |
| CleanScribe RNA Polymerase | Mutant RNA polymerase for IVT | Reduces double-stranded RNA (dsRNA) byproducts [23]. |
| DNase I (RNase-free) | Degrades DNA template post-transcription | Essential for purification. |
| RNA Purification Kit | Purifies mRNA from IVT reaction | Removes proteins, free NTPs, and aborted transcripts. |
Procedure:
- Template Design: Ensure the DNA template has a T7 (or other phage) promoter and the coding sequence is optimized for translation. The first two transcribed nucleotides must be AG for CleanCap AG, AG (3' OMe), and M6 analogs [25].
- IVT Reaction Assembly: On ice, assemble the following reaction in a nuclease-free microcentrifuge tube:
- Linearized DNA template (0.2-0.5 µg/µL): 1 µL
- CleanScript IVT Buffer (10X): 2 µL
- ATP, CTP, UTP (100 mM each): 2 µL each
- N1-methylpseudouridine-5'-triphosphate (100 mM): 2 µL (or GTP for unmodified mRNA) [5] [23]
- CleanCap Analog (10 mM): 6 µL [5]
- CleanScribe RNA Polymerase: 2 µL
- Nuclease-free water: to 20 µL
- Incubation: Mix gently and incubate at 37°C for 2-4 hours.
- DNase I Treatment: Add 2 µL of DNase I (RNase-free) and incubate at 37°C for 15 minutes to degrade the DNA template.
- mRNA Purification: Purify the mRNA using a suitable RNA purification kit, following the manufacturer's instructions. Evaluate the yield and integrity by spectrophotometry and agarose gel electrophoresis.
High-Yield Pulse-Feed Protocol for CleanCap M6
For scalable manufacturing with CleanCap M6, a pulse-feed protocol can be employed to increase mRNA yields up to 2-fold without compromising quality [29].
Procedure:
- Primary Reaction: Set up the initial IVT reaction as described in Section 3.1, but scale down to 50% of the final desired volume.
- Pulse-Feed Addition: After 90 minutes of incubation, prepare an "NTP Feed Mix" containing additional NTPs, CleanCap M6 analog, and polymerase in IVT buffer.
- Secondary Incubation: Gently add the NTP Feed Mix to the primary reaction. Continue the incubation at 37°C for an additional 2-3 hours.
- Purification: Proceed with DNase I treatment and purification as in the standard protocol.
Performance Data and Key Findings
In Vitro and In Vivo Performance Comparison
The superior performance of CleanCap analogs, particularly M6, has been validated in multiple studies. The following diagram summarizes key findings from comparative experiments.
The data from these studies strongly supports the use of CleanCap M6 for applications requiring the highest level of protein expression. The enhanced performance is attributed to the m6Am modification on the first transcribed nucleotide, which hinders the cellular decapping process and thereby stabilizes the mRNA, leading to more sustained and higher levels of translation [25] [26].
The CleanCap platform provides a versatile and efficient solution for co-transcriptional mRNA capping. The choice of analog directly impacts the success of your mRNA-based research or therapeutic development. CleanCap AG serves as a robust foundation for standard mRNA, while CleanCap AG (3' OMe) offers a significant step up in expression. For the most demanding applications where maximum potency and yield are critical, CleanCap M6 is the superior choice, demonstrated to resist decapping and enhance protein expression both in vitro and in vivo. Finally, CleanCap AU is specifically designed for the burgeoning field of self-amplifying RNA vaccines. By aligning your research objectives with the specialized properties of each analog, you can optimize mRNA performance and streamline the path to clinical success.
The development of efficacious mRNA-based therapeutics and vaccines is critically dependent on the presence of a 5' cap structure, which governs transcript stability, translational efficiency, and immunogenicity. While traditional capping methods introduce manufacturing inefficiencies, contemporary co-transcriptional capping with CleanCap technology streamlines production by enabling single-reaction synthesis of Cap 1 structures with >95% efficiency. This application note provides detailed methodologies for integrating CleanCap technology into standardized in vitro transcription (IVT) workflows using commercially available kits, supported by quantitative performance data and optimized protocols to accelerate mRNA research and development.
The 5' cap is a fundamental modification essential for the biological activity and stability of synthetic mRNA. Eukaryotic mRNA naturally features a 5' cap structure with varying methylation patterns classified as Cap 0, Cap 1, and Cap 2, with Cap 1 being predominant in mammals and crucial for reducing unwanted immune recognition [21]. Historically, mRNA capping was achieved through either enzymatic post-transcriptional capping or co-transcriptional capping using analogs like mCap and ARCA. These legacy methods present significant limitations, including lower capping efficiency (~70% for mCap), reverse incorporation issues (partially resolved by ARCA), reduced overall mRNA yields due to high cap:GTP ratios, and multi-step processes that complicate manufacturing workflows [21].
CleanCap technology represents a transformative advance in mRNA synthesis by employing a co-transcriptional capping approach that utilizes proprietary trinucleotide cap analogs. This technology facilitates the direct incorporation of a natural Cap 1 structure during the IVT reaction, mirroring endogenous mRNA and delivering superior performance characteristics [5]. The system achieves >95% capping efficiency in a single-step reaction, eliminates orientation issues, and operates at optimal cap:GTP ratios that maximize full-length transcript yield while simultaneously reducing double-stranded RNA (dsRNA) byproducts by up to 85% [24] [23]. By integrating capping directly into transcription, CleanCap technology reduces production processes by nearly one week and lowers overall manufacturing costs by 20-40% compared to traditional methods, establishing a new standard for mRNA therapeutic manufacturing [5].
Commercial Kit Solutions for CleanCap Integration
Available Kits and Key Performance Metrics
Multiple commercial kits incorporate CleanCap technology to provide researchers with optimized, all-in-one solutions for high-quality mRNA production. These kits include all necessary components—cap analog, RNA polymerase, nucleotides, and optimized buffers—specifically formulated for compatibility and performance. The table below summarizes the primary options and their quantitative performance characteristics:
Table 1: Comparative Analysis of Commercial CleanCap IVT Kits
| Kit Specification | CleanCap AG (3' OMe) CleanScript IVT Kit | CleanCap M6 IVT Kit, High Yield | mMESSAGE mMACHINE T7 mRNA Kit with CleanCap Reagent AG |
|---|---|---|---|
| Cap Analog | CleanCap AG (3' OMe) | CleanCap M6 | CleanCap Reagent AG |
| Capping Efficiency | >95% [24] | >95% [24] | >95% [21] |
| Expected mRNA Yield | 0.8-1 mg from 100 µL reaction [24] | 0.8-1 mg (standard); Up to 10 mg/mL (pulse-feed) [24] | >5 mg/mL [21] |
| Protein Expression Level | Higher than original CleanCap AG [24] | ≥30% higher than CleanCap AG (3' OMe) [24] [5] | High (comparable to CleanCap AG) [21] |
| dsRNA Reduction | Up to 85% reduction [23] | Up to 85% reduction [24] | Significant reduction [21] |
| Transcription Start Site | AG [24] | AG [24] | AG [21] |
| Key Advantage | Versatility; minimal optimization required [24] | Highest protein expression; ultra-high yield potential [24] | Established workflow; high performance [21] |
Advanced Capping Analog Innovations
Beyond standard CleanCap AG, newer analogs provide enhanced biochemical properties. CleanCap AG (3' OMe) incorporates an additional 3'-O-methylation on the m7G moiety, improving protein expression beyond the original CleanCap AG analog and is utilized in commercially approved vaccines [24]. CleanCap M6, the most advanced analog, features an N6-methylated adenosine modification that impairs Dcp2-mediated decapping, resulting in at least 30% higher protein expression compared to previous analogs and enzymatic capping methods [24] [5]. This enhancement is attributed to prolonged mRNA half-life through inhibition of cellular decapping mechanisms.
Experimental Protocols and Methodologies
Standard IVT Protocol with CleanCap Technology
The following optimized protocol is adapted from commercial kit instructions for a standard 100 µL reaction volume, suitable for both research-scale mRNA production and therapeutic development applications.
Table 2: Research Reagent Solutions for CleanCap IVT
| Reagent Component | Function | Considerations |
|---|---|---|
| CleanCap Analog | Co-transcriptional capping to form natural Cap 1 structure | Choice of analog (AG, AG 3' OMe, M6) affects protein expression levels [24] |
| CleanScribe RNA Polymerase Mix | Engineered T7 RNA polymerase for high transcription efficiency & reduced dsRNA | Optimized for specific cap analogs; do not interchange between kits [24] |
| N1-methylpseudouridine | Modified nucleotide replacing UTP; enhances translational efficiency & reduces immunogenicity | Included in kits; critical for therapeutic applications [24] |
| IVT Buffer (10X) | Provides optimal pH, salts, and co-factors for transcription | Buffer formulations are kit-specific and optimized for respective polymerase [24] |
| DNA Template | Linearized plasmid or PCR product containing T7 promoter & gene of interest | Must initiate with "AG" for proper cap incorporation [24] [21] |
| ATP, CTP, GTP | Native nucleotides for RNA chain elongation | GTP concentration balanced with cap analog for optimal yield [21] |
Procedure:
- Reaction Setup: Combine template DNA (0.2-1 µg) with 10 µL 10X IVT buffer, 10 µL CleanCap analog (6-10 mM, depending on specific kit), 10 µL N1-methylpseudouridine (100 mM), 10 µL ATP (100 mM), 10 µL CTP (100 mM), 5 µL GTP (100 mM), 2 µL CleanScribe RNA Polymerase Mix, and nuclease-free water to 100 µL final volume [24].
- Incubation: Mix thoroughly by pipetting and incubate at 37°C for 2-4 hours.
- DNase Treatment: Add 2 µL of DNase I (provided in kits) and incubate at 37°C for 15 minutes to remove template DNA.
- mRNA Purification: Purify mRNA using standard methods such as lithium chloride precipitation or column-based purification. Additional purification to remove dsRNA contaminants (e.g., HPLC) may be unnecessary due to significantly reduced dsRNA levels with CleanCap systems [30].
- Quality Control: Analyze capping efficiency by LC-MS or reverse-phase HPLC; assess mRNA integrity by agarose gel electrophoresis; quantify by spectrophotometry.
High-Yield Pulse-Feed Protocol for CleanCap M6
For applications requiring maximal mRNA yield, the CleanCap M6 IVT Kit offers a supplementary pulse-feed protocol that can achieve yields up to 10 mg/mL [24]. This method addresses nucleotide depletion limitations in extended reactions.
Procedure:
- Initial Reaction: Setup standard IVT reaction as described in Section 3.1, but with reduced GTP concentration (2 mM initial).
- Pulse Solution Preparation: Prepare a nucleotide pulse solution containing ATP, CTP, N1-methylpseudouridine, and GTP at 50 mM each in nuclease-free water.
- Pulse Feeding: After 2 hours of incubation, add 10-20 µL of pulse solution directly to the reaction and mix gently.
- Extended Incubation: Continue incubation at 37°C for an additional 2-4 hours.
- Completion: Process reaction according to standard DNase treatment and purification steps.
Note: Some 5' UTR sequences may impact yield with the CleanCap M6 system. Consultation with technical support (support@trilinkbiotech.com) is recommended for sequence-specific optimization [24].
Troubleshooting and Optimization Guidelines
Successful implementation of CleanCap technology requires attention to several critical parameters that influence yield and capping efficiency.
- Template Design Considerations: The DNA template must contain the appropriate initiation sequence ("AG" start rather than traditional "GG") for efficient cap incorporation [24] [21]. The 5' UTR sequence can significantly impact yield, particularly with CleanCap M6 systems. Evaluation of UTR compatibility through screening experiments or consultation with manufacturer technical support is recommended.
- Nucleotide Ratio Optimization: Unlike traditional cap analogs that require high cap:GTP ratios (4:1) that compromise yield, CleanCap systems utilize balanced nucleotide ratios. However, the pulse-feed protocol specifically addresses nucleotide depletion in extended high-yield reactions [24].
- Component Compatibility: Kit-specific buffers and polymerase mixes are optimized for their respective cap analogs and should not be interchanged between different CleanCap kits, as this can substantially reduce performance [24].
- Yield Enhancement Strategies: For applications requiring maximal mRNA output, the CleanCap M6 system with pulse-feed protocol can increase yields up to 10 mg/mL. Modified nucleotides like N1-methylpseudouridine not only enhance translational efficiency but can also contribute to yield optimization by reducing polymerase stalling [24].
The integration of CleanCap technology into standardized IVT workflows via commercial kits represents a significant advancement in mRNA manufacturing methodology. By enabling single-reaction synthesis of properly capped mRNA with >95% efficiency, substantial yield improvements, and reduced dsRNA byproducts, these systems address critical bottlenecks in therapeutic development. The availability of multiple kit configurations with varying performance characteristics allows researchers to select solutions optimized for specific applications, from basic research to clinical development. As the mRNA therapeutic field continues to expand, streamlined synthesis approaches employing CleanCap technology will play an increasingly vital role in accelerating the development timeline and improving the cost-effectiveness of mRNA-based medicines.
The development of mRNA-based therapeutics and vaccines relies on precise optimization of its core components to enhance stability, translational efficiency, and immunogenic profile. This application note delineates a synergistic methodology integrating TriLink's CleanCap M6 co-transcriptional capping technology with engineered thermostable DNA polymerases (SFM4-3 and TGK) and the modified nucleotide N1-methylpseudouridine (m1ψ). We provide detailed protocols and quantitative data demonstrating that this combined approach significantly augments protein expression, surpasses the limitations of conventional enzymatic capping and T7 RNA polymerase-based transcription, and provides a robust platform for producing high-quality mRNA for research and therapeutic development.
The functional performance of synthetic mRNA is governed by three key structural attributes: the 5' cap, the nucleoside composition, and the integrity of the coding sequence. Traditional methods for producing modified mRNA often involve separate, sequential steps for capping and incorporation of modified nucleotides, which can be inefficient and time-consuming. This document details an integrated strategy that leverages:
- CleanCap M6 Technology: A novel cap analog for one-pot co-transcriptional capping that yields >95% Cap 1 structures [5] [29].
- Engineered Polymerases (SFM4-3 and TGK): Thermostable DNA polymerases engineered to efficiently incorporate a wide range of bulky or modified ribonucleoside triphosphates (rNXTPs) during synthesis, overcoming the limitations of T7 RNA polymerase [31].
- N1-methylpseudouridine (m1ψ): A modified nucleotide that enhances translational efficiency and reduces innate immune recognition [24] [32].
When combined, these technologies enable the streamlined production of highly potent mRNA with superior yield and purity, accelerating discovery and development timelines.
The Scientist's Toolkit: Research Reagent Solutions
The following table catalogues the essential components required to implement the described synergistic mRNA synthesis workflow.
Table 1: Key Research Reagents for Advanced mRNA Synthesis
| Reagent / Technology | Core Function | Key Characteristics & Benefits |
|---|---|---|
| CleanCap M6 Analog [5] [29] | Co-transcriptional 5' Cap 1 formation | >95% capping efficiency; contains m6A modification impairing Dcp2-mediated decapping; >30% higher protein expression than prior CleanCap analogs. |
| Engineered Polymerases (SFM4-3, TGK) [31] | Enzymatic synthesis of base-modified RNA | High tolerance for bulky, base-modified rNXTPs; enables site-specific or hypermodified RNA synthesis; superior to T7 RNAP for modified substrates. |
| N1-methylpseudouridine (m1ψ) [24] [32] | Modified nucleotide incorporated during IVT | Enhances protein expression; reduces innate immune activation by limiting sensor recognition (e.g., TLRs, RIG-I). |
| CleanCap M6 IVT Kit [24] | All-in-one mRNA synthesis solution | Includes CleanCap M6, CleanScribe RNA Polymerase Mix, m1ψ, and optimized buffer; increases yield and reduces dsRNA by up to 85%. |
| Base-Modified rNXTPs [31] | Introduction of functional groups/signals | Includes clickable (e.g., ethynyl), hydrophobic (e.g., phenyl), fluorescent (e.g., mBdp, Cy3/Cy5), and affinity (e.g., biotin) tags for labeling and tracking. |
Quantitative Performance Data
The synergistic effect of combining CleanCap capping with nucleotide modifications and optimized polymerases is demonstrated by direct comparisons of protein expression output and synthesis efficiency.
Table 2: Comparative Performance of Capping and Synthesis Technologies
| Technology Component | Performance Metric | Comparative Result |
|---|---|---|
| CleanCap M6 vs. CleanCap AG (3' OMe) [5] [29] | Protein Expression | >30% increase in protein expression demonstrated with Firefly Luciferase (FLuc) mRNA. |
| CleanCap M6 vs. Enzymatic Capping [29] | Protein Expression & Manufacturing | Significantly higher protein translation; 20-40% lower overall manufacturing costs [5]. |
| m1ψ-Modified vs. Unmodified mRNA [32] | Protein Expression in Cells | Significantly higher target protein expression in primary human myoblasts and dendritic cells. |
| Engineered Polymerases vs. T7 RNAP [31] | Synthesis of RNA with Multiple Modified Nucleotides | SFM4-3/TGK successfully synthesized full-length product with 3 different modified rNXTPs, while T7 RNAP substantially failed. |
| CleanCap IVT Kits vs. Standard IVT [24] [33] | mRNA Yield & dsRNA Byproduct | Up to 2x higher mRNA yield; up to 85% reduction in double-stranded RNA (dsRNA) formation. |
Experimental Protocols
Protocol: High-Yield mRNA Synthesis Using CleanCap M6 IVT Kit
This protocol is optimized for the synthesis of capped, m1ψ-modified mRNA in a single reaction [24].
- Reagent Setup: Thaw the provided IVT kit components (CleanCap M6 Reagent, M6 IVT Buffer, M6 CleanScribe RNA Polymerase Mix, and NTPs including m1ψ) on ice. Combine in a nuclease-free tube:
- 1 µg of linearized DNA template (with a 5'-AG transcription start site)
- 10 µL of M6 IVT Buffer
- 10 µL of CleanCap M6 Reagent
- 10 µL of m1ψ NTP Solution (e.g., 100 mM total NTPs)
- 2 µL of M6 CleanScribe RNA Polymerase Mix
- Nuclease-free water to a final volume of 100 µL.
- Incubation: Mix thoroughly by pipetting and incubate at 37°C for 2-3 hours.
- Pulse-Feed for Very High Yield (Optional): For yields up to 10 mg/mL, follow the supplementary pulse-feed protocol, which involves a second addition of NTPs and polymerase during the reaction [24].
- DNase Treatment: After incubation, add 2 µL of Turbo DNase, mix, and incubate at 37°C for 15 minutes.
- mRNA Purification: Purify the mRNA using a standard method such as lithium chloride precipitation or column-based purification. Determine concentration and analyze integrity by agarose gel electrophoresis.
Protocol: Site-Specific Modified RNA Synthesis via Primer Extension with Engineered Polymerases
This protocol describes the use of SFM4-3 or TGK polymerases to incorporate site-specific modified nucleotides, adapted from [31].
- Primer-Template Hybridization: In a nuclease-free tube, combine:
- 1 µL of 5'-Fluorescently-labeled RNA primer (e.g., 5'-FAM-RNA-prim15nt, 1 µM)
- 1 µL of ssDNA template (e.g., templ19ntX, 1.5 µM)
- 3 µL of nuclease-free water. Heat the mixture to 65°C for 5 minutes and slowly cool to room temperature to anneal.
- Primer Extension Reaction: To the annealed primer-template, add:
- 2 µL of 10X Thermostable Polymerase Buffer
- 1 µL of the desired base-modified rNXTP (e.g., rAETP, 10 mM)
- 4 µL of a mixture of the three natural rNTPs (each at 10 mM)
- 1 µL of engineered polymerase (SFM4-3 or TGK, 2 U/µL)
- Nuclease-free water to 20 µL.
- Incubation: Incubate the reaction at 60°C for 1 hour.
- Template Removal & Purification: Add 1 µL of TurboDNase to degrade the DNA template and incubate at 37°C for 15-30 minutes. Purify the full-length, site-specifically modified RNA using denaturing polyacrylamide gel electrophoresis (dPAGE) or a suitable cleanup method. Confirm product identity by mass spectrometry (e.g., MS-MALDI).
Technology Integration Workflow
The following diagram illustrates the logical workflow and synergistic relationship between the key technologies described in this application note.
The successful development of messenger RNA (mRNA) vaccines and therapeutics represents a transformative advancement in modern medicine. A critical component in the production of high-quality mRNA is the 5' cap structure, which is integral to mRNA stability, translational efficiency, and reduced immunogenicity [5]. Co-transcriptional capping with CleanCap technology has emerged as a superior approach, overcoming limitations of legacy capping methods by enabling a one-pot synthesis of mRNA with optimal Cap 1 structure and over 95% capping efficiency [5]. This application note provides detailed protocols and analytical methods for implementing CleanCap technology across research and Good Manufacturing Practice (GMP) scales, supporting a streamlined path from discovery to clinical application.
Cap Structures and Biological Significance
The 5' cap is a hallmark of eukaryotic mRNA, with different methylation states conferring distinct functional properties. Cap 0 structures (m7GpppN) provide basic protection from exonuclease degradation and facilitate translation initiation. However, CleanCap technology generates a natural Cap 1 structure (m7GpppNm), which includes 2'-O-methylation of the first transcribed nucleotide. This Cap 1 structure is crucial for reducing recognition by pattern recognition receptors of the innate immune system, thereby minimizing unwanted immunogenicity and enhancing protein expression in vivo [5] [34].
Comparative Capping Methods
Traditional mRNA capping approaches present significant limitations for scalable manufacturing:
- Enzymatic Capping: Post-transcriptional capping requires multiple purification steps and additional enzymes, increasing process complexity and costs [21].
- Early Cap Analogs: mCAP and Anti-Reverse Cap Analog (ARCA) suffer from orientation issues and capping efficiencies of only 70-80%, requiring high cap:GTP ratios that reduce overall mRNA yield [21].
CleanCap technology utilizes trinucleotide cap analogs that initiate transcription with higher specificity, resulting in over 95% capping efficiency without reducing transcription yield [5] [21]. This co-transcriptional approach streamlines manufacturing by combining transcription and capping in a single reaction, significantly reducing production timelines and costs compared to legacy methods [5].
Table 1: Comparison of mRNA Capping Methods
| Capping Method | Capping Efficiency | Cap Structure | Orientation Specificity | Process Steps |
|---|---|---|---|---|
| mCAP | ~70% | Cap 0 | ~50% correct | Co-transcriptional |
| ARCA | ~80% | Cap 0 | 100% correct | Co-transcriptional |
| Enzymatic | >90% | Cap 0 or Cap 1 | 100% correct | Post-transcriptional |
| CleanCap | >95% | Cap 1 | 100% correct | Co-transcriptional |
Research-Scale Implementation
IVT Kit Components and Preparation
For research-scale mRNA production, CleanCap technology is available in convenient in vitro transcription (IVT) kits that include all essential components:
- CleanCap Analog: Typically CleanCap AG (3' OMe) for high capping efficiency and protein expression
- CleanScript RNA Polymerase: Engineered for reduced double-stranded RNA (dsRNA) formation
- Modified Nucleotides: Including N1-methylpseudouridine for enhanced mRNA performance and reduced immunogenicity
- CleanScript IVT Buffer: Optimized for high yield transcription with capping [23]
These kits simplify researcher workflow by providing integrated components that deliver up to 2X more mRNA yield and up to 85% lower dsRNA compared to standard kits, enabling rapid production of high-quality mRNA for preclinical studies [23].
Research-Scale Protocol
Materials Required:
- CleanCap IVT Kit (TriLink BioTechnologies)
- DNA template with appropriate promoter (T7, A-inserted φ6.5 preferred)
- Nuclease-free water
- Thermal cycler or incubator
Procedure:
- Template Preparation: Dilute DNA template to 0.1-0.5 μg/μL in nuclease-free water. Ensure template encodes AG as the first two nucleotides for optimal CleanCap incorporation [21].
Reaction Setup:
- Thaw all IVT kit components and mix by gentle vortexing
- Assemble the reaction at room temperature:
- 10 μL CleanScript IVT Buffer (2X concentration)
- 2 μL ATP Solution (100 mM)
- 2 μL CTP Solution (100 mM)
- 2 μL GTP Solution (100 mM)
- 2 μL UTP Solution (100 mM)
- 2 μL CleanCap AG (3' OMe) (40 mM)
- 2 μL CleanScript RNA Polymerase
- 2 μL DNA Template (0.1-0.5 μg/μL)
- 6 μL Nuclease-free water
- Total reaction volume: 30 μL
Incubation:
- Incubate at 37°C for 2-4 hours
- For increased yield, incubation can be extended up to 6 hours
mRNA Purification:
Expected Outcomes:
- Typical yield: >5 mg/mL of mRNA
- Capping efficiency: >95%
- Reduced dsRNA contaminants: Up to 85% reduction compared to standard methods [23]
Scalability and GMP Manufacturing
Process Development and Scale-Up
Transitioning from research to GMP manufacturing requires careful process development and optimization. TriLink's platform provides a seamless pathway through:
- Construct-Specific Optimization: Feasibility studies and engineering runs to ensure successful GMP manufacturing
- Dedicated Manufacturing Suites: mRNA programs receive dedicated cleanroom suites for the duration of GMP manufacturing
- Robust Quality Systems: Phase-appropriate Quality Management Systems with cGMP compliance [35]
The platform approach to mRNA manufacturing enables efficient scale-up, with processes that are similar for different products, varying primarily in the antigen-encoding mRNA sequence while maintaining the same backbone elements and lipid nanoparticle technology [36].
GMP Manufacturing Facilities and Capabilities
Modern GMP facilities for mRNA manufacturing are specifically designed to support clinical-stage production:
- Facility Specifications: 32,000 ft² cGMP facility with three ISO 7 cleanrooms (1,000 ft² per suite)
- Production Capacity: Scalable from 1g to >100g per batch
- Infrastructure Features: Single-use consumables, buffer prep areas, aseptic bulk aliquoting, and on-site Quality Control testing [35]
Table 2: GMP Manufacturing Scale Capabilities
| Parameter | Laboratory Scale | Pilot Scale | Commercial Scale |
|---|---|---|---|
| Batch Size | 1-10 mg | 10 mg - 1 g | 1 g - 100 g+ |
| Facility Class | Research lab | GMP-like | cGMP |
| Quality Controls | Basic characterization | Extended panel | Full validation |
| Documentation | Research records | Development reports | Regulatory submission |
Analytical Methods and Quality Control
Comprehensive analytical methods are essential for characterizing mRNA critical quality attributes (CQAs):
- Identity: Sequence confirmation by mass spectrometry or sequencing
- Integrity: Size distribution and fragmentation analysis by capillary electrophoresis
- Potency: In vitro translation assays or cell-based expression studies
- Capping Efficiency: LC-MS methods to quantify cap structure and efficiency
- Impurities: Detection of dsRNA, protein, and DNA contaminants [35]
The platform approach to mRNA product development enables leveraging prior knowledge for regulatory submissions, potentially streamlining the path to clinical trials [36].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for CleanCap mRNA Synthesis
| Reagent | Function | Application Notes |
|---|---|---|
| CleanCap AG | Co-transcriptional capping | Generates Cap 1 structure with >95% efficiency; requires AG initiation sequence |
| CleanCap M6 | Advanced capping analog | Includes m6A modification; increases protein expression by >30% vs. earlier analogs |
| CleanCap AU | Self-amplifying RNA capping | Optimized for saRNA applications with alphavirus 5' cap compatibility |
| N1-methylpseudouridine | Modified nucleotide | Enhances mRNA performance and reduces immunogenicity |
| CleanScript RNA Polymerase | IVT enzyme | Engineered for reduced dsRNA formation during transcription |
| CleanScript IVT Buffer | Reaction buffer | Optimized for high-yield transcription with CleanCap analogs |
Regulatory Considerations and Platform Technology
mRNA-LNP products are increasingly recognized as a platform technology, which the U.S. FDA defines as "a well-understood and reproducible technology that can be adapted for more than one drug or biological product" [37]. This designation has significant implications for regulatory strategy:
- Chemistry, Manufacturing, and Controls (CMC): Platform approaches enable leveraging prior knowledge for regulatory submissions
- Preclinical Data: Biodistribution and toxicology studies may be adapted for related products
- Clinical Development: Safety and reactogenicity data from platform products can inform new product development [36]
The European Medicines Agency has acknowledged that using a platform approach is acceptable for approving strain changes in mRNA vaccines, with approvals potentially based on manufacturing/quality and non-clinical data only when the platform demonstrates predictability of clinical performance [36].
CleanCap co-transcriptional capping technology provides an efficient, scalable solution for mRNA manufacturing from research to GMP production. The technology's ability to generate Cap 1 structures with >95% efficiency in a one-pot reaction significantly streamlines manufacturing processes, reducing production timelines and costs while enhancing mRNA quality and performance. Implementation of the protocols and methodologies described in this application note supports a seamless transition from research-scale experimentation to clinical-grade manufacturing, accelerating the development of novel mRNA vaccines and therapeutics.
Maximizing Output and Minimizing Costs: Optimization and Troubleshooting for CleanCap IVT
The 5' cap structure is a cornerstone of mRNA biology, integral to the stability, translational efficiency, and immunogenicity of synthetic mRNA [5]. For researchers developing mRNA vaccines and therapeutics, achieving both high capping efficiency (>95%) and high mRNA yield is a critical determinant of success. Legacy capping methods, such as Anti-Reverse Cap Analog (ARCA) and enzymatic capping, present significant manufacturing challenges including lower efficiency (~70% for ARCA), multiple reaction steps, and increased production time and cost [38].
This application note details experimental strategies for implementing co-transcriptional capping with CleanCap technology, a approach that overcomes these limitations. We provide definitive data and optimized protocols to help you maintain peak capping performance, streamline manufacturing, and enhance the potency of your mRNA products within the context of mRNA synthesis research.
Results and Data Analysis
Performance Comparison of Capping Technologies
The following table summarizes the performance of various capping methods, highlighting the superior efficiency of CleanCap analogs.
Table 1: Comparative Analysis of mRNA Capping Technologies
| Capping Technology | Capping Efficiency | Cap Structure | Key Process Attributes | Reported Protein Expression vs. Legacy Methods |
|---|---|---|---|---|
| CleanCap M6 | >95% [5] | Cap 1 [5] | One-pot co-transcriptional [5] | >30% increase [5] [39] |
| CleanCap AG (3' OMe) | >95% [5] [23] | Cap 1 [5] | One-pot co-transcriptional [5] | Higher [5] |
| CleanCap AU | >95% [5] | Cap 1 (for saRNA) [5] | One-pot co-transcriptional [5] | Durable [5] |
| Enzymatic Capping | Variable (Method dependent) | Cap 1 | Multi-step, post-transcriptional [38] | Baseline (Varies by protocol) |
| ARCA | ~70% [38] | Cap 0 [38] | Co-transcriptional [38] | Baseline |
| mCAP | ~50% [38] | Cap 0 [38] | Co-transcriptional [38] | Lower |
Impact of Cap Structure on mRNA Functionality
Recent research elucidates the mechanistic basis for the enhanced performance of CleanCap M6. The M6 analog incorporates an N6-methyladenosine (m6A) modification at the first transcribed nucleotide, in addition to the 3'-O-methylation of the m7G [40]. This combination confers a significant functional advantage by impairing enzyme-mediated decapping.
In vitrodecapping assays with recombinant human Dcp2 enzyme demonstrated that the M6 3'OMe (CleanCap M6) structure exhibits an additive resistance to decapping compared to analogs with only one of these modifications [40]. In a reconstituted decay system (Dcp2 + Xrn1), this resulted in an approximately 2.5-fold increase in RNA half-life [40]. This enhanced stability translated directly to superior in vivo performance, with CleanCap M6 yielding 3.1-fold more total protein output in mice compared to AG 3'OH-capped mRNA, and 1.5-fold more than AG 3'OMe [40].
Experimental Protocols
Standard IVT Protocol Using CleanCap Analogs
This protocol is designed for robust production of capped mRNA using TriLink's CleanCap analogs in a single reaction tube.
- Template Design and Preparation: Use a linearized DNA template with a precise +1 nucleotide (A or G, depending on the specific CleanCap analog) following the T7 promoter sequence.
- Reaction Setup: Combine components on ice in the following order:
- Nuclease-free water to a final volume of 100 µL
- 10 µL 10X CleanScript IVT Buffer (included in kits)
- 10 µL 100 mM DTT
- 16 µL rNTP Mix (25 mM each A, U, G; 6 mM C) [39]
- 3.2 µL 100 mM CleanCap Analog (e.g., M6, AG 3'OMe)
- 4 µL 25 mM N1-methylpseudouridine-5'-triphosphate (to replace standard C [5]
- 4 µL CleanScript T7 RNA Polymerase (for high yield, low dsRNA [23]
- 1 µg linearized DNA template
- Incubation: Mix thoroughly and incubate at 37°C for 2-4 hours.
- DNase I Treatment: Add 2 µL of DNase I (provided in kits) and incubate at 37°C for 15 minutes to digest the DNA template.
- mRNA Purification: Purify the mRNA using a standard method, such as lithium chloride precipitation or column-based purification. For highest purity and to remove dsRNA impurities, consider HPLC purification [40].
Advanced Pulse-Feed Protocol for Increased Yield
To further increase mRNA yields, a pulse-feed protocol can be employed. This method addresses the depletion of critical reagents during the IVT reaction, which can limit the length and efficiency of the process [39].
- Initial Reaction Setup: Follow the standard protocol above, but initially only include half the recommended concentration of rNTPs and cap analog.
- Pulse-Feed Addition: After 60-90 minutes of incubation at 37°C, prepare a "feed" solution containing the remaining half of the rNTPs and cap analog in a small volume of the same reaction buffer.
- Second Incubation: Gently add the feed solution to the primary IVT reaction and mix thoroughly. Continue the incubation at 37°C for an additional 2-3 hours.
- Completion: Proceed with DNase I treatment and purification as described in the standard protocol. This extended, fed-batch process can lead to a significant increase in total mRNA yield per reaction [39].
The following workflow diagram illustrates the key steps and decision points in these protocols.
Diagram 1: mRNA Synthesis Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for High-Efficiency Co-transcriptional Capping
| Reagent / Material | Function in mRNA Synthesis | Key Feature / Benefit |
|---|---|---|
| CleanCap M6 Analog [5] [40] | Co-transcriptional capping agent | m6A modification provides >30% higher protein expression & resists decapping. |
| CleanCap AG (3'OMe) Analog [5] [23] | Co-transcriptional capping agent | >95% efficiency; proven in commercially approved vaccines. |
| N1-Methylpseudouridine (m1Ψ) [5] | Modified nucleoside triphosphate | Enhances mRNA performance by reducing immunogenicity. |
| CleanScript T7 RNA Polymerase [23] | Drives in vitro transcription | Engineered for high mRNA yield and low double-stranded (ds)RNA by-products. |
| CleanScript IVT Buffer [23] | Optimized reaction buffer | Formulated for use with CleanCap analogs to maximize yield and capping efficiency. |
Discussion and Mechanistic Insights
The strategic advantage of CleanCap technology is twofold. First, its one-pot co-transcriptional capping streamlines manufacturing, reducing production processes by nearly a week and cutting overall costs by 20-40% compared to other methods [5]. Second, and more critically, the technology produces a natural Cap 1 structure, which is superior to the Cap 0 structure produced by ARCA and mCAP in mitigating cellular immune recognition [5] [38].
The enhanced performance of the CleanCap M6 analog can be visualized through its unique mechanism of action, which modulates key post-transcriptional events as outlined below.
Diagram 2: M6 Mechanism: Reduced Decapping
In conclusion, adopting the CleanCap platform, particularly the latest M6 analog, and implementing the optimized protocols described herein provides a robust strategy for achieving and maintaining greater than 95% capping efficiency alongside high mRNA yield. This approach directly addresses the core challenges in mRNA manufacturing, enabling the production of more potent and stable mRNA therapeutics and vaccines.
The production of messenger RNA (mRNA) through in vitro transcription (IVT) is a cornerstone technology for vaccines, protein replacement therapies, and other emerging modalities. However, a significant challenge in manufacturing high-quality mRNA is the formation of double-stranded RNA (dsRNA) contaminants during the IVT process. These dsRNA byproducts are potent inducers of the innate immune response, triggering inflammation and suppressing protein translation, which can severely compromise the efficacy and safety of the mRNA product [41] [42] [43]. Within the broader context of optimizing mRNA synthesis via co-transcriptional capping with CleanCap, managing dsRNA is not merely a downstream purification concern but a critical quality attribute that must be addressed throughout the production workflow. This application note details established and emerging techniques for reducing dsRNA contaminants, providing researchers with structured data and actionable protocols to ensure the production of high-purity mRNA.
The dsRNA Challenge: Generation and Consequences
During IVT, phage RNA polymerases like T7 RNAP can generate dsRNA through several mechanisms. These include RNA-dependent RNA polymerase activity, where the enzyme uses the primary transcript as a template to synthesize a complementary strand, and the formation of abortive initiation fragments that can prime complementary RNA synthesis [41] [42]. The resulting dsRNA impurities are recognized by intracellular sensors such as RIG-I and MDA5, as well as endosomal Toll-like receptor 3 (TLR3), leading to the activation of interferon and pro-inflammatory cytokine pathways [42] [43]. This not only inhibits translation of the therapeutic protein but can also cause undesirable side effects, from localized redness to systemic reactions [42]. Consequently, regulatory guidelines mandate that dsRNA levels be controlled to below 0.01% in mRNA drug substances [43].
The following diagram illustrates the cellular signaling pathways activated by dsRNA contaminants:
Techniques for dsRNA Reduction
Strategies for mitigating dsRNA contamination can be broadly categorized into two approaches: preventive methods that minimize dsRNA formation during IVT and separation methods that remove dsRNA from the final mRNA product.
Preventive Methods
a. Co-transcriptional Capping with CleanCap
The CleanCap technology is a premier example of a preventive strategy integrated into the IVT reaction itself. This one-pot co-transcriptional capping method not only simplifies production but also enhances the quality of the mRNA product.
- Principle: CleanCap analogs are synthetic cap analogs that are added directly to the IVT reaction. The RNA polymerase incorporates them at the 5' end of the nascent mRNA chain, resulting in a Cap 1 structure with high efficiency (>95%) [22] [5]. This process outcompetes spurious transcription initiation events that can lead to dsRNA byproducts.
- Advantages: Compared to legacy enzymatic capping, CleanCap reduces manufacturing steps and costs by 20-40%. mRNA produced with CleanCap, particularly the newer M6 analog, demonstrates >30% higher protein expression due to reduced immunogenicity and improved cap function [5].
b. Engineered IVT Conditions with Chaotropic Agents
A simple yet effective preventive method involves modifying the IVT buffer conditions.
- Principle: The addition of optimized concentrations of chaotropic agents creates a mildly denaturing environment during IVT. This prevents the intermolecular or intramolecular base-pairing that facilitates RNA-templated dsRNA synthesis by the RNA polymerase [44].
- Advantages: This scalable and cost-effective method can significantly reduce dsRNA formation without requiring reduced magnesium concentration, elevated temperature, or custom reagents. It results in mRNA with much lower immunostimulation and more efficient protein expression [44].
c. Improved RNA Polymerases
Using engineered RNA polymerases is another preventive strategy to reduce byproduct generation.
- Principle: Specialized polymerases, such as the Codex HiCap T7 RNA Polymerase, are engineered to have reduced RNA-dependent RNA polymerase activity, thereby decreasing the primary mechanism of dsRNA formation [45].
- Advantage: This method directly targets the root cause of the contaminant and can be seamlessly integrated into standard IVT protocols.
Separation Methods
a. Cellulose-Based Purification
This is a well-established, bench-top method for purifying IVT mRNA.
- Principle: In an ethanol-containing buffer (e.g., 16% ethanol), dsRNA selectively binds to microcrystalline cellulose, while the desired single-stranded mRNA remains in the flow-through. The bound dsRNA can be discarded, and the purified mRNA is recovered from the unbound fraction [41].
- Advantages: It is a simple, fast, and cost-effective technique that requires only standard laboratory equipment. It achieves >90% removal of dsRNA contaminants with a good recovery rate of >65%, and is scalable from microgram to milligram quantities [41] [45]. The workflow for this method is straightforward, as shown below:
b. Reverse-Phase Chromatography
This is a highly effective, chromatography-based polishing step.
- Principle: Ion-pair reverse-phase high-performance liquid chromatography (IP-RP HPLC) or chromatography on monolithic styrene-divinylbenzene (SDVB) supports separates nucleic acid species based on hydrophobicity. dsRNA, being more structured, has a different retention profile than single-stranded mRNA, allowing for its physical separation [46].
- Advantages: This method is highly effective at removing not only dsRNA but also hybridized RNA fragments and residual DNA template. mRNA purified this way shows significantly increased transgene expression and reduced innate immune activation [46]. Recent advancements have simplified this technology with spin columns, making it more accessible for clinical manufacturing [46].
Comparative Analysis of dsRNA Reduction Techniques
The table below provides a quantitative comparison of the key techniques discussed.
Table 1: Comparison of dsRNA Reduction Methods
| Technique | Principle | dsRNA Reduction Efficiency | Key Advantages | Limitations |
|---|---|---|---|---|
| CleanCap Capping [22] [5] | Co-transcriptional capping to prevent spurious transcription | Prevents formation; >95% capping efficiency | Streamlined process, high-quality Cap 1, boosts protein expression | Requires specialized cap analog |
| Chaotropic Agents [44] | Denaturing IVT conditions to prevent base-pairing | "Significantly less" dsRNA formed | Simple, scalable, cost-effective, no custom reagents | Requires optimization of agent concentration |
| Cellulose-Based Purification [41] | Selective binding of dsRNA to cellulose in ethanol buffer | >90% removal | Uses standard lab equipment, scalable, good recovery (≥65%) | Not all short dsRNA (<30 bp) removed |
| Reverse-Phase Chromatography [46] | Chromatographic separation based on hydrophobicity | Highly effective removal | Also removes RNA fragments & DNA template, high purity | Requires specialized equipment, higher cost |
Detection of dsRNA Contaminants
Accurate detection and quantification are essential for process control. The field is shifting from semi-quantitative immunoblot (dot-blot) methods toward more sensitive and quantitative assays.
- Immunoblot (e.g., J2 antibody): A traditional method where dsRNA is detected on a membrane using a dsRNA-specific antibody. It is simple but has limited sensitivity, typically detecting dsRNA only above the 0.01% threshold, and provides semi-quantitative data [41] [43].
- Enzyme-Linked Immunosorbent Assay (ELISA): Now the preferred standard for its superior sensitivity and robustness. Modern ELISA methods can accurately quantify dsRNA down to nanogram-per-milliliter levels. For instance, Samsung Biologics has developed a specialized sandwich ELISA using J2/K2 antibodies that achieves recovery rates of 95-110% with low coefficients of variation, providing the precision needed for process development and quality control [43]. Recent research has also led to the development of novel antibody pairs (e.g., M2 and M5) with nanomolar affinity for dsRNA, further improving the accuracy of ELISA-based detection [42].
Table 2: Key Reagents for dsRNA Analysis and Purification
| Reagent / Material | Function | Example / Note |
|---|---|---|
| Anti-dsRNA Antibodies | Detection and quantification of dsRNA impurities. | J2, K2 (commercial); M2, M5 (novel high-affinity pairs) [42] [43]. |
| Microcrystalline Cellulose | Solid-phase matrix for purifying IVT mRNA. | Binds dsRNA in ethanol-containing buffer [41]. |
| CleanCap Analog | Co-transcriptional capping agent. | Yields >95% Cap 1 mRNA; available in RUO and GMP grades [5]. |
| Codex HiCap T7 RNAP | Engineered RNA polymerase. | Reduces generation of dsRNA during IVT [45]. |
| Chaotropic Agents | IVT additive. | Creates a denaturing environment to minimize dsRNA formation; specific agents and concentrations may be proprietary [44]. |
The success of mRNA therapeutics and vaccines is inextricably linked to the purity of the final product. As shown, dsRNA contaminants are a critical quality attribute that can be managed through a combination of preventive and separation strategies. Integrating co-transcriptional capping with CleanCap represents a foundational step in a holistic quality-by-design approach, minimizing the initial generation of byproducts. This can be effectively complemented by downstream polishing steps such as cellulose-based purification or reverse-phase chromatography, depending on the scale and purity requirements. Employing modern, quantitative ELISA methods for detection ensures that dsRNA levels are accurately monitored and controlled throughout the development and manufacturing process. By adopting these techniques, researchers can consistently produce high-purity mRNA with optimal translational capacity and minimized immunogenicity, accelerating the development of safe and effective mRNA-based drugs.
The production of messenger RNA (mRNA) for vaccines and therapeutics relies heavily on the in vitro transcription (IVT) reaction. Transitioning from traditional batch to fed-batch IVT operations presents a significant opportunity to enhance productivity and cost-efficiency. This approach involves the gradual addition of reagents to the reaction, allowing for the reuse of expensive catalysts like RNA polymerase (RNAP) and DNA template, thereby maximizing RNA output from a single reaction setup [47] [48]. However, the fed-batch process introduces complexity, with numerous interacting variables influencing the final yield and critical quality attributes, such as the 5'-cap incorporation efficiency.
When combined with advanced capping technologies like CleanCap, which enables co-transcriptional capping with efficiencies exceeding 95%, fed-batch IVT requires precise control to maintain high cap analog incorporation while minimizing reagent consumption [5] [23]. This application note details optimized, model-based fed-batch IVT strategies designed to maximize RNA yield, control costs, and maintain superior product quality for research and drug development.
Key Challenges in Fed-Batch IVT and the Role of Model-Based Optimization
The decline in reaction rate over time is a major challenge in fed-batch IVT, primarily caused by the accumulation of salts, dilution of catalysts, and a drop in pH [47]. Furthermore, the dynamic nature of the reaction makes it difficult to maintain optimal concentrations of nucleoside triphosphates (NTPs) and magnesium (Mg²⁺), which are critical for polymerase activity. Without sophisticated control, these factors can lead to suboptimal yields and inconsistent product quality.
Model-based optimization addresses these challenges by using mechanistic models that incorporate enzyme kinetics and solution thermodynamics. These models can predict the complex interplay of variables, enabling the design of feeding protocols that maintain the reaction within an optimal operating window, prevent undesirable by-products like magnesium phosphate crystals, and ultimately double the RNA yield compared to heuristic approaches [47] [49] [50].
Quantitative Optimization of Reaction Parameters
Optimizing the initial reaction conditions is foundational to a successful fed-batch process. The tables below summarize key parameters and their optimized values for maximizing mRNA productivity.
Table 1: Optimal Initial Concentrations for Key IVT Reagents
| Reagent | Function | Optimal Initial Concentration | Impact on Reaction |
|---|---|---|---|
| Mg²⁺ | RNA Polymerase co-factor | 38 - 60 mM [48] | Essential for enzymatic activity; excess can inhibit reaction [47]. |
| NTPs | RNA synthesis substrates | 7.5 mM [48] | High initial concentrations can cause pH drop, slowing the reaction [48]. |
| Spermidine | Enzyme activity enhancer | 2 mM [48] | Optimizes transcription rate; higher concentrations can be inhibitory. |
| Dithiothreitol (DTT) | Reducing agent | 20 mM [48] | Maintains enzyme stability and activity. |
Table 2: Optimized Fed-Batch Feeding Strategy for High Yield
| Reaction Phase | NTP Concentration | Mg²⁺ Concentration | Objective |
|---|---|---|---|
| Initial Batch | 7.5 mM | 38 mM | Establish high initial reaction rate [48]. |
| First Feed | 10 mM | 44 mM | Replenish consumed substrates to maintain rate. |
| Second Feed | 15 mM | 50 mM | Counteract catalyst dilution and salt inhibition. |
| Third Feed | 20 mM | 56 mM | Push reaction to maximum conversion [48]. |
Experimental Protocol: Model-Optimized Fed-Batch IVT with Integrated Capping
This protocol provides a step-by-step methodology for implementing a high-yield fed-batch IVT process, incorporating co-transcriptional capping using CleanCap analog.
Research Reagent Solutions
Table 3: Essential Materials and Reagents
| Item | Function/Description |
|---|---|
| DNA Template | Linearized plasmid containing T7 promoter and gene of interest. |
| T7 RNA Polymerase | Enzyme for RNA synthesis. CleanScribe polymerase can reduce dsRNA byproduct [23]. |
| NTPs (ATP, CTP, GTP, UTP) | Building blocks for RNA synthesis. Modified NTPs (e.g., N1-methylpseudouridine) enhance mRNA stability and reduce immunogenicity [23] [51]. |
| CleanCap AG Analog | Cap analog for one-pot co-transcriptional capping, yielding >95% Cap 1 structures [5] [23]. |
| MgCl₂ Solution | Critical co-factor for T7 RNAP activity. |
| Reaction Buffer | Typically Tris-HCl, pH ~7.9-8.0, provides optimal pH for polymerase activity [47]. |
Step-by-Step Procedure
Initial Reaction Setup:
- Combine the following components in a nuclease-free microcentrifuge tube on ice:
- DNA Template: 5-10 µg/mL (final concentration)
- T7 RNA Polymerase: As per manufacturer's recommendation (e.g., 125 U/µL)
- NTPs: 7.5 mM each (final concentration)
- CleanCap AG Analog: Molar ratio as recommended for >95% capping efficiency (e.g., 4:1 Cap:GTP) [5]
- MgCl₂: 38 mM (final concentration)
- Reaction Buffer, DTT, Spermidine: As per optimized conditions in Table 1.
- Adjust the total volume with nuclease-free water.
- Mix gently and centrifuge briefly.
- Combine the following components in a nuclease-free microcentrifuge tube on ice:
Incubation and Feeding:
- Transfer the reaction tube to a thermomixer and incubate at 37°C with constant agitation.
- Monitor pH: Use a micro-pH probe if available. A linear decrease in pH correlates with mRNA synthesis [52].
- Execute Pre-optimized Feeds: Based on the model-predicted protocol (Table 2), add the specified volumes of concentrated NTP and Mg²⁺ feed stock at predetermined timepoints (e.g., at 60, 120, and 180 minutes). The feed stock concentrations should be calculated to achieve the target final concentrations upon addition.
- Mix thoroughly after each feed.
Reaction Termination:
- After the final feed and a total reaction time of 4-6 hours, stop the reaction by adding EDTA (to chelate Mg²⁺) or by heating to 70°C for 10 minutes.
mRNA Purification and Analysis:
- Purify the mRNA using standard methods such as Amicon filters or LiCl precipitation [51].
- Analyze the yield via spectrophotometry (e.g., Nanodrop), integrity by bioanalyzer, and capping efficiency by LC-MS or other analytical techniques.
Workflow Diagram
The following diagram illustrates the logical flow and interactions of the key components in the optimized fed-batch IVT process.
Scaling Up and Technology Transfer
Optimized fed-batch conditions developed in micro-scale systems (e.g., the Beckman Coulter BioLector XT Microbioreactor) can be successfully scaled to clinically relevant volumes. The Cytiva ReadyToProcess WAVE 25 rocking bioreactor, a closed and GMP-ready system, supports IVT scaling from 20 mL to 20 L, demonstrating the industrial viability of these strategies [52]. This seamless translation from micro-scale optimization to manufacturing-scale production is crucial for accelerating the development of mRNA therapeutics and vaccines.
The integration of model-based optimization with fed-batch IVT strategies represents a powerful approach to overcoming the key challenges in mRNA synthesis. By systematically controlling reagent feeding, this method significantly enhances mRNA yield—potentially doubling output—while ensuring high co-transcriptional capping efficiency with technologies like CleanCap. The provided protocols and data offer researchers a clear pathway to implement these advanced techniques, enabling more efficient, cost-effective, and scalable production of high-quality mRNA for therapeutic applications.
The adoption of co-transcriptional capping using CleanCap technology represents a significant advancement in mRNA manufacturing, offering substantial economic and temporal benefits over legacy capping methods. This application note provides a detailed quantitative analysis of these advantages, contextualized within mRNA synthesis research. For researchers and drug development professionals, the shift from enzymatic capping and early cap analogs to sophisticated co-transcriptional methods translates to reduced production timelines, lower manufacturing costs, and enhanced mRNA quality. We present verified data on cost reductions of 20-40% and timeline savings of nearly one week per production run, alongside detailed protocols for implementing this technology in research and development settings [5].
The economic imperative for efficient mRNA manufacturing has intensified with the expansion of mRNA applications beyond COVID-19 to include investigational vaccines for influenza, rabies, and oncology therapeutics. Legacy capping methods, including Anti-Reverse Cap Analog (ARCA) and enzymatic capping, present manufacturing challenges including multiple reaction steps, inefficient capping, and suboptimal cap structures that necessitate complex purification workflows. CleanCap technology addresses these limitations through a one-pot co-transcriptional capping strategy that yields mRNA with an optimal Cap 1 structure at >95% efficiency, directly translating to economic benefits without compromising quality [5] [38].
Quantitative Economic Analysis
Comprehensive Cost and Efficiency Comparison
The economic advantage of CleanCap technology becomes evident when comparing key performance metrics against traditional capping methods. The data demonstrates superior capping efficiency, reduced production steps, and significant cost savings.
Table 1: Performance and Economic Comparison of mRNA Capping Technologies
| Capping Method | Capping Efficiency | Cap Structure | Reaction Steps | Relative Cost | Production Time |
|---|---|---|---|---|---|
| mCAP Analog | ~50% | Cap 0 | Single-step | Moderate | 3-4 days |
| ARCA | ~70% | Cap 0 | Single-step | Moderate | 3-4 days |
| Enzymatic Capping | >95% | Cap 1 | Multiple | High | 5-7 days |
| CleanCap Technology | >95% | Cap 1 | Single-step | 20-40% lower | 3-4 days |
Data compiled from TriLink Biotechnologies documentation and market analysis [5] [38]. Cost comparison is relative to enzymatic capping methods.
The implementation of CleanCap technology reduces overall manufacturing costs by 20-40% compared to other capping methods while simultaneously cutting mRNA therapeutic production processes by nearly one week [5]. This economic advantage stems from multiple factors: the single-reaction approach reduces reagent consumption and hands-on time, the high capping efficiency minimizes product loss, and the simplified workflow decreases purification requirements. For research institutions and commercial manufacturers, these savings accelerate development timelines and reduce resource constraints, particularly crucial for programs targeting emerging pathogens or competitive therapeutic areas.
Capping Efficiency and mRNA Quality Metrics
The superior capping efficiency of CleanCap technology (>95%) compared to legacy methods (50-70%) directly impacts both economic and functional outcomes [5] [38]. Higher capping efficiency correlates with enhanced translational capacity, reduced immunogenicity, and improved mRNA stability, ultimately yielding more consistent experimental results and potentially reducing the dose required for efficacy. The Cap 1 structure generated by CleanCap analogs is recognized as the optimal configuration for minimizing detection by innate immune sensors while maximizing translation efficiency, addressing a critical quality consideration in therapeutic mRNA development [5].
Table 2: CleanCap Analog Portfolio for Research Applications
| Analog | Key Features | Capping Efficiency | Protein Expression | Optimal Application |
|---|---|---|---|---|
| CleanCap M6 | m6A modification, 3’-O-methylation on m7G, Cap 1 | >95% | Highest (≥30% increase vs. AG 3'OMe) | Therapeutic mRNA requiring maximum expression |
| CleanCap AG 3'OMe | 3’-O-methylation on m7G, Cap 1 | >95% | Higher | mRNA vaccines, balanced performance |
| CleanCap AG | Cap 1 structure | >95% | High | General research mRNA |
| CleanCap AU | Alphavirus 5' cap | >95% | Durable | Self-amplifying RNA applications |
Adapted from TriLink Biotechnologies product portfolio [5]. Protein expression comparison is relative to previous generation analogs.
The selection of appropriate CleanCap analogs enables researchers to tailor capping strategies to specific applications. CleanCap M6, the newest analog, demonstrates particularly impressive performance with protein expression increases exceeding 30% compared to previous analogs, representing a significant advancement for applications requiring high protein yield [5]. This enhanced performance directly translates to economic benefits through reduced mRNA requirements for equivalent biological activity.
Experimental Protocols
Standardized CleanCap IVT Workflow
The following protocol details the optimized procedure for mRNA synthesis using CleanCap AG (3' OMe) CleanScript IVT Kit (K-7413), representative of the co-transcriptional capping approach [27]. This protocol is designed to yield 0.8-1 mg of capped mRNA per 100 μL reaction with >95% capping efficiency.
Figure 1: mRNA synthesis workflow using CleanCap co-transcriptional capping. The process integrates capping with transcription, eliminating post-transcriptional enzymatic steps required by legacy methods.
Template Preparation
- Linearized DNA Template: Use plasmid DNA (50 μg/mL) linearized with appropriate restriction enzymes or purified PCR products (25 μg/mL) containing T7 promoter followed by 5'-AGG-3' or 5'-AGA-3' initiation sequence [27].
- Template Purification: Purify linearized template using silica-membrane columns or lithium chloride precipitation to remove enzymes and contaminants. Verify complete linearization by agarose gel electrophoresis to prevent extended RNA transcripts.
- Control Template: Include TriLink's FLuc control plasmid (provided in kit) at 5 μg per 100 μL reaction for process verification [27].
Reaction Assembly and Incubation
- Thaw Components: Thaw all kit components on ice except enzymes, which should remain at -20°C until immediately before use [27].
- Master Mix Preparation: Prepare reaction master mix according to Table 3 in nuclease-free tubes to minimize variation.
Table 3: Reaction Setup for 100 μL IVT with CleanCap
| Component | Volume per Reaction | Final Concentration |
|---|---|---|
| 10X AG CleanScript IVT Buffer | 10 μL | 1X |
| ATP (100 mM) | 7.5 μL | 7.5 mM |
| CTP (100 mM) | 7.5 μL | 7.5 mM |
| GTP (100 mM) | 7.5 μL | 7.5 mM |
| Uridine-5'-Triphosphate (100 mM) | 1.88 μL | 1.88 mM |
| N1-Methyl-Pseudouridine-5'-Triphosphate (100 mM) | 5.62 μL | 5.62 mM |
| CleanCap Reagent AG (3' OMe) (10 mM) | 30 μL | 3 mM |
| AG CleanScribe RNA Polymerase Mix | 5 μL | - |
| Linearized DNA Template (0.5 μg/μL) | 10 μL | 25 μg/mL |
| Nuclease-free Water | 15 μL | - |
| Total Volume | 100 μL |
Adapted from CleanCap AG (3' OMe) CleanScript IVT Kit protocol [27]. Note: UTP is partially replaced with N1-methylpseudouridine to reduce immunogenicity.
- Incubation Conditions: Mix thoroughly by pipetting and incubate at 37°C for 3 hours using a thermal cycler with heated lid or dry air incubator to prevent evaporation [27].
Post-IVT Processing and Purification
- DNase Treatment: Add 2 μL of DNase I (optional but recommended) and incubate at 37°C for 15 minutes to remove template DNA [27].
- mRNA Purification: Purify mRNA using:
- Lithium Chloride Precipitation: Add 100 μL of lithium chloride solution (7.5 M), incubate at -20°C for 30 minutes, centrifuge at 13,000 × g for 15 minutes, wash pellet with 70% ethanol, and resuspend in nuclease-free water.
- Silica-Membrane Columns: Use commercial kits (e.g., RNeasy) following manufacturer's instructions for higher purity.
- Oligo(dT) Purification: Recommended for mRNAs containing poly(A) tails to remove truncated transcripts.
- Quality Assessment: Quantify mRNA by UV spectrophotometry, assess integrity by agarose gel electrophoresis, and verify capping efficiency by LC-MS or enzymatic methods.
Process Optimization and Troubleshooting
Yield Enhancement Protocol
For applications requiring maximum mRNA yield, implement the pulse-feed modification to the standard protocol:
- Standard Protocol: Typically yields 0.4-0.5 mg of RNA per 100 μL reaction [53].
- Pulse-Feed Enhancement: After 2 hours of initial incubation, supplement reaction with additional 10 μL of NTP mix (25 mM each NTP) and 2.5 μL of CleanScribe RNA Polymerase Mix [53].
- Extended Incubation: Continue incubation for additional 2 hours at 37°C, increasing total yield to 0.8-1 mg per 100 μL reaction [53].
Template Design Considerations
- Promoter Requirements: Ensure template contains canonical T7 promoter sequence (5'-TAATACGACTCACTATA-3') followed immediately by 5'-AGG-3' or 5'-AGA-3' transcription start for optimal CleanCap incorporation [27].
- Sequence Optimization: Avoid extreme GC content (>80% or <30%) in 5' UTR that may reduce yield, particularly with CleanCap M6 analog [53].
- Template Quality: Verify complete linearization and purity, as residual circular plasmid produces aberrant RNA products that reduce yield and complicate purification.
Research Reagent Solutions
Successful implementation of co-transcriptional capping requires specific reagents optimized for the technology. The following essential materials represent the core components of a robust mRNA synthesis system.
Table 4: Essential Reagents for CleanCap mRNA Synthesis
| Reagent Category | Specific Examples | Function | Optimized Selection |
|---|---|---|---|
| Cap Analogs | CleanCap M6, CleanCap AG (3'OMe), CleanCap AU | Co-transcriptional capping to form Cap 1 structure | Select based on application: M6 for highest expression, AU for saRNA [5] |
| Nucleotides | N1-Methylpseudouridine-5'-Triphosphate, 5-Methylcytidine triphosphate | mRNA building blocks with modified bases to reduce immunogenicity | Replace 100% of UTP with N1-methylpseudouridine for enhanced performance [27] |
| Enzymes | CleanScribe RNA Polymerase, DNase I | Catalyzes transcription, reduces dsRNA byproducts; removes DNA template | CleanScribe reduces dsRNA by up to 85% vs. wild-type T7 polymerase [27] |
| Template DNA | Linearized plasmid DNA, PCR products | Encodes mRNA sequence with T7 promoter and desired gene | Must contain T7 promoter followed by AGG or AGA start codon [27] |
| Buffers & Solutions | 10X CleanScript IVT Buffer, Nuclease-free water | Maintain reaction stability and optimal enzyme activity | Proprietary formulation increases yield and reduces dsRNA [27] |
| Purification Materials | Lithium chloride, Silica columns, Oligo(dT) beads | Remove impurities, enzymes, and truncated RNAs | Method selection depends on scale and downstream application [27] |
Economic Impact Analysis
Manufacturing Process Economics
The implementation of CleanCap technology creates value throughout the mRNA production workflow, with particularly significant impacts in time savings and resource utilization. The co-transcriptional approach eliminates multiple steps required by enzymatic capping methods, including separate capping reactions, purification between steps, and buffer exchanges, directly reducing labor and material costs [5].
Figure 2: Economic impact comparison between legacy enzymatic capping and CleanCap technology. Co-transcriptional capping reduces both production time and overall costs significantly [5].
The streamlined manufacturing process using CleanCap technology cuts production timelines by nearly one week compared to enzymatic capping methods, accelerating critical development milestones for research programs and commercial applications [5]. This temporal advantage is particularly valuable for pandemic response, where rapid vaccine development is crucial, and for competitive therapeutic areas where time-to-market significantly impacts commercial success.
Return on Investment Calculation
The economic value proposition of CleanCap technology extends beyond direct cost savings to include enhanced research productivity and development efficiency. Key factors contributing to return on investment include:
- Reduced Processing Time: Laboratory personnel requirements decrease approximately 30-40% due to simplified workflows and reduced hands-on time [5].
- Enhanced Success Rates: Higher capping efficiency (>95% vs. 50-70% for legacy methods) reduces batch failures and repeat experiments, conserving valuable research resources [5] [38].
- Downstream Efficiency: Superior Cap 1 structure with proper 2'-O-methylation enhances translational efficiency and reduces immune activation, potentially decreasing the required dose for in vivo efficacy [5].
- Scalability Advantage: The one-pot co-transcriptional method scales more efficiently than multi-step enzymatic capping, with GMP-grade materials already validated in commercial vaccines exceeding 3 billion doses [38].
The quantitative economic analysis presented in this application note demonstrates that CleanCap co-transcriptional capping technology offers substantial advantages over legacy capping methods. The documented 20-40% reduction in manufacturing costs coupled with timeline savings of nearly one week per production run provides compelling economic justification for adoption across research and development environments [5]. These benefits, combined with quality improvements in mRNA performance including >95% capping efficiency and enhanced protein expression, position CleanCap technology as the optimal choice for modern mRNA synthesis applications.
For the research community, these economic advantages translate to accelerated discovery timelines and more efficient resource utilization. For drug development professionals, the technology offers a validated, scalable path from research to clinical application, with GMP-grade materials already employed in commercially approved products [5]. As mRNA applications expand into new therapeutic areas including oncology, infectious diseases, and genetic disorders, the economic and temporal efficiencies of CleanCap technology will play an increasingly vital role in bringing innovative treatments to market faster and more cost-effectively.
Proof of Performance: Validating CleanCap Efficacy Through Data and Case Studies
Within messenger RNA (mRNA) synthesis research, the 5' cap structure is a critical determinant of success, directly influencing the stability, translational efficiency, and immunogenicity of the final transcript [5] [21]. The choice of capping strategy is therefore paramount, influencing experimental outcomes and the therapeutic potential of the mRNA. This application note provides a structured, data-driven comparison of legacy dinucleotide cap analogs (mCap and ARCA) against the modern trinucleotide CleanCap technology, contextualized within the broader thesis that co-transcriptional capping with CleanCap represents a superior methodology for robust and efficient mRNA synthesis in research and drug development.
Performance Benchmarking: A Quantitative Comparison
A comprehensive analysis of key performance metrics reveals significant advantages of CleanCap analogs over legacy dinucleotide caps. The data below, synthesized from comparative studies, provides a clear quantitative foundation for cap selection.
Table 1: Head-to-Head Performance Comparison of mRNA Cap Analogs
| Performance Metric | mCap (Cap 0) | ARCA (Cap 0) | CleanCap (Cap 1) |
|---|---|---|---|
| Capping Efficiency | ~50% [38] | ~70% [38] | >95% [5] [33] |
| Cap Structure | Cap 0 [21] | Cap 0 [21] | Cap 1 [5] [54] |
| Incorporation Orientation | 50% reverse [21] | Correct orientation only [21] | Correct orientation only [54] |
| Typical mRNA Yield | Lower (due to high cap:GTP ratio) [21] | Lower (due to high cap:GTP ratio) [21] | >5 mg/mL [21] [33] |
| Key Differentiator | First-generation analog | Prevents reverse incorporation; better than mCap | Superior efficiency, yield, and natural Cap 1 structure in a one-pot reaction [5] |
Table 2: Functional and Economic Impact of Capping Choice
| Impact Area | mCap / ARCA | CleanCap |
|---|---|---|
| Translational Efficiency | Moderate (ARCA) to Low (mCap) | High to Highest (Varies by analog; CleanCap M6 offers >30% increase in protein expression) [5] |
| Immunogenicity Profile | Higher risk with Cap 0 and dsRNA impurities [55] | Reduced (Cap 1 mimics endogenous mRNA, and kits can reduce dsRNA by up to 85%) [54] [33] |
| Manufacturing Process | Multi-step (especially enzymatic) or inefficient co-transcriptional | Streamlined, one-pot co-transcriptional capping [5] |
| Reported Cost Impact | Higher manufacturing costs | 20-40% lower overall manufacturing costs [5] |
| Production Timeline | Longer (up to 1 week added for enzymatic capping) [5] | Faster (cuts production by nearly one week) [5] |
Experimental Protocols for Performance Analysis
To ensure the benchmarking data is actionable, the following protocols outline standardized methods for generating and evaluating capped mRNA.
Protocol: Standardized mRNA Synthesis and Capping for Comparative Studies
This protocol is designed for the direct comparison of different cap analogs in a controlled in vitro transcription (IVT) reaction.
Template Design:
IVT Reaction Setup:
- Set up separate IVT reactions for each cap analog (mCap, ARCA, CleanCap AG).
- For mCap/ARCA reactions: Use a high cap analog-to-GTP ratio (e.g., 4:1) as recommended for these analogs [21].
- For CleanCap reactions: Use a lower, optimized cap analog-to-GTP ratio (e.g., 1:1), which maintains high yield and capping efficiency [21].
- Include modified nucleosides like N1-methylpseudouridine to enhance mRNA performance and reduce immunogenicity [33].
- Incubate the reactions at 37°C for 2-4 hours.
mRNA Purification:
- Purity the synthesized mRNA using methods such as spin columns or, for highest purity and reduced immunogenicity, HPLC purification [55]. HPLC purification is critical for removing immunogenic by-products like double-stranded RNA (dsRNA).
Protocol: Assessing Capping Efficiency and Functional Output
Once mRNA is synthesized, these methods quantify the success of the capping reaction.
Liquid Chromatography-Mass Spectrometry (LC-MS):
- Use LC-MS to analyze the molecular weight of the intact mRNA or 5'-end oligonucleotides.
- The presence of a Cap 1 structure (from CleanCap) versus a Cap 0 structure (from mCap/ARCA) results in a distinct mass shift, allowing for precise quantification of capping efficiency.
In Vitro Transfection and Protein Expression Analysis:
- Transfert a cell line (e.g., HEK-293) with equimolar amounts of mRNA capped with the different analogs. Use a reporter gene like firefly luciferase.
- For transfection reagent, Lipofectamine 2000 has been shown to outperform alternatives like TransIT in some cell systems [55].
- Measure luciferase activity (bioluminescence) at multiple time points (e.g., 4, 8, 12, and 24 hours) to generate a protein expression kinetic profile [55]. The slope and peak of this curve are direct indicators of the functional superiority of the cap analog.
The Scientist's Toolkit: Essential Research Reagent Solutions
The following reagents and kits are fundamental for implementing the co-transcriptional capping workflows described in this note.
Table 3: Key Reagents for mRNA Synthesis with Co-Transcriptional Capping
| Reagent / Kit | Function & Key Feature | Application Note |
|---|---|---|
| CleanCap AG (3'OMe) | Cap analog for one-pot synthesis of Cap 1 mRNA; >95% efficiency and improved protein expression [33]. | The modified version of CleanCap AG; available in TriLink's all-in-one IVT kit [33]. |
| CleanCap M6 | Advanced cap analog with m6A modification; increases protein expression by >30% vs. other CleanCap analogs [5]. | Ideal for applications demanding the highest possible protein yield. |
| N1-methylpseudouridine (m1Ψ) | Modified nucleotide; incorporated into mRNA during IVT to reduce immunogenicity and enhance translational efficiency [5] [54]. | A standard modification for therapeutic mRNA applications. |
| CleanScribe RNA Polymerase | Engineered RNA polymerase; reduces formation of dsRNA byproducts during IVT by up to 85% [33]. | Critical for lowering innate immune response and improving mRNA performance. |
| HiScribe T7 mRNA Kit with CleanCap Reagent AG (NEB #E2080) | All-in-one kit for high-yield mRNA synthesis; optimized for co-transcriptional capping with CleanCap AG [56]. | Simplifies workflow and ensures reproducible, high-quality mRNA production. |
Technical Diagrams: Mechanisms and Workflows
The following diagrams illustrate the core molecular mechanisms and experimental workflows central to understanding cap analog performance.
Cap-Dependent Translation Initiation
This diagram illustrates the critical role of the 5' cap in initiating protein synthesis. The cap structure is specifically recognized by the initiation factor eIF4E, which, together with eIF4G and eIF4A, forms the eIF4F complex. This complex is essential for recruiting the 43S ribosomal complex and initiating translation [21]. A properly incorporated cap is therefore fundamental to high protein expression.
Experimental mRNA Synthesis & Analysis Workflow
This workflow outlines the key steps for synthesizing and benchmarking capped mRNA, from template design with specific initiation sequences to downstream quality control and functional analysis. Adhering to a standardized protocol is vital for generating comparable data.
The quantitative data and protocols presented herein unequivocally demonstrate that CleanCap technology outperforms legacy mCap and ARCA analogs across every critical performance metric: capping efficiency, mRNA yield, translational output, and immunogenicity profile. The fundamental advantage of CleanCap lies in its trinucleotide design, which enables the one-step, co-transcriptional synthesis of a natural Cap 1 structure with >95% efficiency [5] [54]. This contrasts sharply with the inefficient and error-prone incorporation of dinucleotide analogs.
The broader implication for mRNA synthesis research is clear: adopting CleanCap for co-transcriptional capping streamlines workflows, reduces costs and timelines [5], and delivers a more potent and biologically faithful mRNA product. This technology, proven in billions of vaccine doses [38] and a growing number of preclinical and clinical applications [16] [54], represents the current state-of-the-art and provides a robust foundation for the next generation of mRNA therapeutics and vaccines.
Application Note: Advancing mRNA Technology with Co-Transcriptional Capping
The development of messenger RNA (mRNA) as a therapeutic modality represents a paradigm shift in vaccinology, protein replacement, and gene therapy. The 5' cap structure is a critical determinant of mRNA stability, translational efficiency, and immunogenicity [5]. This application note details how co-transcriptional capping with CleanCap technology addresses key manufacturing challenges while enhancing therapeutic outcomes across diverse biomedical applications. Unlike legacy capping methods that involve multiple post-transcriptional enzymatic steps, CleanCap enables single-pot synthesis of mRNA with optimal Cap 1 structure and >95% capping efficiency, significantly streamlining production workflows [5]. The technology's integration into therapeutic development pipelines has accelerated timelines while improving protein expression characteristics, with the newest analog, CleanCap M6, increasing protein expression by over 30% compared to previous analogs [5].
Quantitative Analysis of CleanCap Analogs
Table 1: Performance Characteristics of CleanCap Analogs in mRNA Synthesis
| Analog Name | Application | Cap Modification(s) | Capping Efficiency | Protein Expression |
|---|---|---|---|---|
| CleanCap M6 | mRNA | 2' O-methylation (Cap 1), 3' O-methylation on m7G, m6A modification | >95% | Highest |
| CleanCap AG 3' OMe | mRNA | 2' O-methylation (Cap 1), 3' O-methylation on m7G | >95% | Higher |
| CleanCap AG | mRNA | 2' O-methylation (Cap 1) | >95% | High |
| CleanCap AU | Self-amplifying RNA | 2' O-methylation (Cap 1), Alphavirus 5' cap | >95% | Durable |
Research Reagent Solutions for mRNA Synthesis
Table 2: Essential Research Reagents for mRNA-Based Therapeutic Development
| Reagent / Technology | Function | Application in Therapeutic Development |
|---|---|---|
| CleanCap Cap Analogs | Co-transcriptional capping enabling Cap 1 structure formation | All mRNA-based vaccines and therapeutics; enhances stability and translation efficiency |
| CleanScribe RNA Polymerase | Reduced dsRNA byproducts during in vitro transcription | Improves mRNA purity and reduces immunogenicity |
| N1-methylpseudouridine | Modified nucleotide incorporated into mRNA | Decreases innate immune recognition, enhances mRNA stability and protein expression |
| Lipid Nanoparticles (LNPs) | Delivery vehicle for mRNA cargo | Enables cellular uptake and endosomal release of mRNA; liver-targeting properties advantageous for many applications |
| CRISPR-Cas9 Components | Gene-editing machinery | Corrects genetic mutations in vivo when delivered with LNPs |
Protocol: mRNA Synthesis Using CleanCap Technology
Materials and Equipment
- DNA Template: Linearized plasmid or PCR product encoding gene of interest with appropriate promoter (e.g., A-inserted φ6.5 T7 promoter)
- CleanCap Analog (selected from Table 1 based on application requirements)
- Nucleotides: ATP, CTP, GTP, UTP, preferably with modified nucleotides (e.g., N1-methylpseudouridine-5'-triphosphate)
- CleanScript IVT Buffer: Optimized for high-yield in vitro transcription
- CleanScribe RNA Polymerase: T7 RNA polymerase with reduced dsRNA formation
- DNase I: For DNA template digestion post-transcription
- Purification Materials: LiCl precipitation reagents or chromatographic purification columns
- Nuclease-free Water and Microcentrifuge Tubes
Step-by-Step Procedure
Reaction Setup:
- Thaw all IVT components on ice and prepare reaction mix in the following order:
- Nuclease-free water (to final volume of 20 μL)
- CleanScript IVT Buffer (1X final concentration)
- Nucleotides (each at 7.5 mM final concentration)
- DNA template (50-100 ng/μL final concentration)
- CleanCap analog (4-8 mM final concentration, optimized for specific analog)
- CleanScribe RNA Polymerase (final concentration as manufacturer recommends)
- Thaw all IVT components on ice and prepare reaction mix in the following order:
Incubation:
- Mix components gently by pipetting and centrifuge briefly.
- Incubate at 37°C for 2-4 hours, depending on transcript length and yield requirements.
DNase Treatment:
- Add 2 μL of DNase I (1 U/μL) to the reaction mixture.
- Incubate at 37°C for 15 minutes to digest DNA template.
mRNA Purification:
- Purify mRNA using LiCl precipitation or column-based purification systems.
- For LiCl precipitation: add 0.1 volume of 5M LiCl, incubate at -20°C for 30 minutes, centrifuge at 13,000 × g for 15 minutes, wash pellet with 70% ethanol, and resuspend in nuclease-free water.
- Quantify mRNA concentration by spectrophotometry and assess integrity by agarose gel electrophoresis.
Quality Control:
- Determine capping efficiency by LC-MS analysis or differential enzymatic digestion.
- Assess dsRNA contamination by ELISA or immunoblotting.
- Verify functionality by in vitro translation assay.
Diagram 1: mRNA synthesis workflow with co-transcriptional capping.
Case Study 1: Rapid Response Vaccine Development
Background and Therapeutic Challenge
The COVID-19 pandemic demonstrated the critical need for rapid vaccine development platforms. Traditional vaccine approaches require lengthy development timelines, making them suboptimal for pandemic response. mRNA vaccines emerged as a transformative technology capable of rapid adaptation to evolving pathogens [57].
Experimental Approach and Implementation
The successful deployment of mRNA vaccines against SARS-CoV-2 leveraged the unique advantages of the mRNA platform:
Sequence-Based Design: Following publication of the SARS-CoV-2 genetic sequence, researchers designed mRNA constructs encoding the viral spike protein.
Co-Transcriptional Capping: CleanCap technology enabled single-step synthesis of properly capped mRNA, significantly reducing manufacturing time compared to enzymatic capping methods [5].
LNP Formulation: The mRNA was encapsulated in lipid nanoparticles for efficient delivery to human cells.
Rapid Production: The entire process from sequence to clinical lot production was completed in weeks rather than years.
Results and Therapeutic Outcomes
- Efficacy: mRNA vaccines demonstrated 95% efficacy in preventing COVID-19 in phase III clinical trials [57].
- Speed: Development and authorization timelines were reduced from years to months.
- Manufacturing Efficiency: Co-transcriptional capping reduced overall manufacturing costs by 20-40% compared to other capping methods [5].
- Adaptability: The platform technology allowed rapid development of updated boosters targeting emerging variants.
Case Study 2: Gene Therapy for Rare Genetic Disease
Background and Therapeutic Challenge
Carbamoyl phosphate synthetase 1 (CPS1) deficiency is a rare, life-threatening genetic disorder characterized by the inability to break down ammonia. Conventional management requires strict protein restriction and eventual liver transplantation, with high mortality risk during intercurrent illnesses [58].
Experimental Approach and Implementation
A research team developed a personalized CRISPR-based therapy for an infant with CPS1 deficiency:
Target Identification: Identified the specific mutation in the CPS1 gene causing ammonia metabolism dysfunction.
CRISPR-Cas9 Formulation: Designed guide RNA targeting the mutated sequence and packaged with Cas9 mRNA in liver-tropic lipid nanoparticles.
Dosing Strategy: Employed a multi-dosing regimen made possible by LNP delivery:
- Initial low dose (0.1 mg/kg) to establish safety
- Subsequent higher doses (0.3 mg/kg and 1.0 mg/kg) to increase editing efficiency
Monitoring: Tracked ammonia levels, protein tolerance, and clinical status.
Diagram 2: In vivo gene therapy workflow for CPS1 deficiency.
Results and Therapeutic Outcomes
- Rapid Development: The personalized therapy was developed, approved, and administered within six months of diagnosis [58].
- Clinical Improvement: The infant demonstrated increased protein tolerance and reduced medication requirements.
- Infection Resilience: Successfully weathered intercurrent infections without dangerous ammonia spikes.
- Safety: No serious adverse effects were observed, demonstrating the feasibility of personalized in vivo gene editing.
Case Study 3: Protein Replacement for Monogenic Disorder
Background and Therapeutic Challenge
Duchenne muscular dystrophy (DMD) is a fatal X-linked disorder caused by mutations in the massive dystrophin gene (≈12 kb), which exceeds the packaging capacity of conventional AAV vectors. Previous gene therapy approaches could only deliver miniaturized dystrophin versions (micro-dystrophin) representing just one-third of the full protein [59].
Experimental Approach and Implementation
Researchers implemented a novel protein trans-splicing strategy to achieve full-length dystrophin expression:
Split Intein Design: Divided the dystrophin gene into three fragments, each fused to split intein sequences that facilitate precise protein ligation.
AAV Delivery: Packaged each fragment into separate AAVMYO1 vectors with high muscle tropism.
In Vivo Assembly: Upon co-infection of muscle cells, the dystrophin fragments were expressed and the intein sequences mediated precise excision and ligation to form full-length, functional dystrophin.
Testing: Evaluated therapy in both young (pre-symptomatic) and old (advanced disease) mdx4cv mouse models.
Results and Therapeutic Outcomes
- Full-Length Protein Expression: Successfully detected full-length dystrophin in tibialis anterior, diaphragm, and heart muscles.
- Histological Improvement: Significant amelioration of dystrophic pathology, including improved myofiber size and reduced collagen content.
- Functional Recovery: Muscle force was significantly improved in treated animals.
- Rescue in Advanced Disease: Even 17-month-old mice with established disease showed substantial functional improvement in key tissues including diaphragm and heart [59].
Comparative Analysis and Future Directions
The case studies presented demonstrate the versatility of mRNA and gene editing technologies across therapeutic domains. CleanCap co-transcriptional capping technology provides a foundation for efficient mRNA synthesis that underpins multiple therapeutic approaches, from conventional vaccines to sophisticated gene editing applications.
Quantitative Therapeutic Outcomes
Table 3: Comparative Therapeutic Outcomes Across Case Studies
| Therapeutic Approach | Development Timeline | Key Efficacy Metric | Manufacturing Advantage |
|---|---|---|---|
| mRNA Vaccine Platform | Months (vs. years for traditional platforms) | 95% reduction in symptomatic COVID-19 | 20-40% cost reduction with co-transcriptional capping |
| Personalized Gene Therapy (CPS1) | 6 months from diagnosis to treatment | Successful ammonia metabolism during illness | LNP delivery enables re-dosing (impossible with viral vectors) |
| Protein Trans-Splicing (DMD) | Preclinical development | Full-length dystrophin expression and functional rescue | Overcomes AAV packaging limitations for large genes |
Emerging Innovations and Protocol Adaptations
Future directions in mRNA-based therapeutics include:
Cap-Independent Translation Systems: Emerging research explores internal ribosome entry sites (IRESs) and cap-independent translational enhancers (CITEs) for tissue-specific optimization, particularly valuable in cancer and neurodegenerative diseases where cap-dependent translation is impaired [6].
Manufacturing Innovations: Modular, decentralized production systems like BioNTech's BioNTainer and Quantoom's Ntensify platform enable more agile, scalable vaccine production with 60% cost reduction compared to conventional batch manufacturing [60].
Expanded Therapeutic Targets: Ongoing clinical trials are investigating mRNA and gene editing applications for hereditary transthyretin amyloidosis (hATTR), hereditary angioedema (HAE), HIV, cancer, and autoimmune diseases [61].
The integration of co-transcriptional capping with these advancing technologies positions mRNA and gene editing platforms as transformative tools across the therapeutic spectrum, enabling researchers to address previously untreatable genetic conditions while maintaining preparedness for emerging infectious threats.
The demonstration of potent neutralizing antibody (nAb) responses in validated animal models is a critical milestone in the development of prophylactic and therapeutic biologics. This application note details standardized protocols for quantifying nAb potency and breadth, and for evaluating in vivo efficacy in disease models, within the context of mRNA-based antibody delivery. The use of co-transcriptional capping with CleanCap in mRNA synthesis is foundational to this workflow, as it produces mRNA with superior translational fidelity and stability, leading to consistent and high-level in vivo expression of target antibodies [22] [62].
Summarized Quantitative Data from Recent Studies
The following tables consolidate key quantitative findings from recent in vivo and in vitro studies, highlighting the efficacy of various antibody and vaccine candidates.
Table 1: In Vivo Efficacy of Antibody and Vaccine Candidates in Disease Models
| Candidate | Target / Model | Key In Vivo Efficacy Findings | Source |
|---|---|---|---|
| PR306007 mAb | RSV in murine model | Potent antiviral activity against both lower and upper respiratory tract infections. | [63] |
| Multivalent MPXV NP Vaccine | Lethal VACV and MPXV challenge in mice | A single dose elicited cross-protective immunity against lethal challenge. | [64] |
| Trispecific Antibodies (Tri-1, Tri-2) | Authentic SARS-CoV-2 variants & SARS-CoV | Effectively neutralized all tested authentic viruses with IC50 values comparable or superior to parental antibodies. | [65] |
| mRNA Cancer Vaccine (Model Simulation) | Virtual patient cohorts (n=1,635) | 45% average tumor size reduction and 60% increase in CD8+ T cell infiltration in responsive tumors. | [66] |
Table 2: Neutralizing Antibody Responses in Clinical and Preclinical Studies
| Candidate / Cohort | Neutralization Target | Key Neutralization Findings | Source |
|---|---|---|---|
| XBB Vaccines in Older Adults | D614G, BA.5, XBB.1.5, JN.1, KP.2, KP.3 | 100% seroconversion post-booster; Trivalent XBB.1.5 vaccine showed 3.7 to 18.8-fold rise in GMTs. | [67] |
| Trispecific Antibodies (Tri-1, Tri-2) | Pseudotyped SARS-CoV, SARS-CoV-2 WT, Omicron BA.1 & XBB.1 | Demonstrated potency and breadth superior to any single parental antibody. | [65] |
| PR306007 mAb | RSV-A and RSV-B subgroups | Exhibited superior broad-spectrum neutralization against both subgroups. | [63] |
| Multivalent MPXV NP Vaccine | MPXV/VACV | Induced robust nAb responses, primarily targeting the M1 antigen. | [64] |
Detailed Experimental Protocols
Protocol: Pseudovirus-Based Neutralization Assay
This protocol is used to quantify the potency and breadth of nAbs against enveloped viruses in a BSL-2 setting.
1. Reagent Preparation:
- Pseudovirus Stock: Generate VSV-G pseudotyped or HIV-based lentiviral particles expressing the target viral glycoprotein (e.g., SARS-CoV-2 Spike, RSV F protein) and a reporter gene (e.g., luciferase, GFP).
- Serum/mAb Samples: Serially dilute test sera or purified monoclonal antibodies in cell culture medium.
- Cell Line: Culture susceptible cells (e.g., Vero E6, HEK-293T-ACE2) to 80-90% confluency.
2. Neutralization Reaction: - Incubate a standardized volume of pseudovirus with an equal volume of each serum/mAb dilution for 1 hour at 37°C.
3. Infection: - Add the pseudovirus-antibody mixture to the pre-plated cells. - Centrifuge plates at 800-1000 × g for 30-60 minutes at room temperature to enhance infection (spinoculation). - Incubate for 48-72 hours at 37°C, 5% CO₂.
4. Quantification and Analysis: - Lyse cells and measure reporter activity (e.g., luciferase luminescence). - Calculate the percentage neutralization relative to virus-only controls (no antibody). - Determine the half-maximal inhibitory concentration (IC50) or dilution (NT50) using a four-parameter logistic (4PL) regression model [67] [65].
Protocol: In Vivo Efficacy Testing in a Prophylactic RSV Model
This protocol assesses the ability of a candidate mAb or vaccine to prevent viral infection and pathogenesis in mice.
1. Animal Grouping and Administration: - Use 6-8 week old, female BALB/c mice (n=5-10 per group). - Randomize mice into groups: (1) Negative Control (PBS/isotype control), (2) Positive Control (e.g., Palivizumab), (3) Test Article(s). - Administer the test article (mAb or vaccine) via an appropriate route (e.g., intramuscular for mRNA-LNP, intraperitoneal for purified mAb) 24 hours prior to viral challenge.
2. Viral Challenge: - Anesthetize mice and intranasally inoculate with a pre-determined lethal or replication-competent dose of RSV (e.g., RSV A2 strain) in a small volume (e.g., 50 μL).
3. Sample Collection and Analysis (3-5 days post-challenge): - Necropsy: Euthanize mice and collect lung and nasal turbinate tissues. - Viral Load Quantification: - Homogenize tissues in culture medium. - Clarify homogenates by centrifugation. - Determine viral titers in the supernatant by plaque assay or quantitative PCR (qPCR). - Histopathology: Preserve lung lobes in formalin, embed in paraffin, section, and stain with H&E to evaluate inflammation and tissue damage [63].
Visualizing Workflows and Mechanisms
The following diagrams illustrate the core experimental workflow and the conceptual link between mRNA capping quality and immune efficacy.
Diagram 1: In vivo nAb validation workflow.
Diagram 2: CleanCap role in nAb efficacy.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for nAb Validation Experiments
| Item | Function / Application | Example / Note |
|---|---|---|
| CleanCap AG Reagent | Co-transcriptional capping during IVT to produce Cap-1 mRNA. | TriLink BioTechnologies; enables >94% capping efficiency, crucial for high-yield antibody expression [22] [62]. |
| Takara IVTpro System | All-in-one system for high-yield mRNA synthesis. | Includes template preparation and IVT reagents; compatible with CleanCap [62]. |
| VitalPatch Biosensor | Continuous physiologic monitoring in vivo. | Measures post-vaccination/reactogenicity changes (e.g., heart rate) for correlative studies [68]. |
| Vero E6 / BHK-21 Cells | Cell lines for pseudovirus and authentic virus neutralization assays. | ATCC C1008 / CCL-10. |
| BALB/c Mice | Common murine model for infectious disease challenge studies. | Used for RSV, MPXV, and other pathogen models [63] [64]. |
The advent of co-transcriptional capping using CleanCap technology represents a pivotal advancement in mRNA therapeutic and vaccine development. This innovative approach, which allows for the direct incorporation of a Cap 1 structure during in vitro transcription (IVT), has overcome significant limitations of legacy capping methods, enabling faster development timelines and improved efficacy of mRNA products [5]. The technology's critical importance has been demonstrated through its incorporation into commercially approved vaccines and a growing pipeline of clinical-stage investigational therapies.
This application note details the industry-wide adoption of CleanCap technology, providing quantitative data on its performance and presenting detailed protocols for its implementation in mRNA synthesis workflows. The information is structured to assist researchers, scientists, and drug development professionals in leveraging this platform for their developmental programs.
Industry Adoption and Commercial Impact
CleanCap technology has transitioned from a novel innovation to an industry standard, providing a robust foundation for both commercial products and clinical-stage development.
Commercialized Vaccines
The most significant validation of CleanCap technology comes from its use in commercially approved mRNA vaccines. TriLink's CleanCap AG (3' OMe) cap analog has been used in three commercially approved RNA vaccines, with billions of doses administered globally, demonstrating a tremendous safety profile [5] [23]. These vaccines, including the Pfizer-BioNTech (BNT162b2) COVID-19 vaccine, have played a transformative role in global public health [16] [69].
Preclinical and Clinical Pipeline
Beyond commercialized products, CleanCap reagents are being utilized in over 45 institutions and companies for programs in preclinical and clinical settings [5]. This broad adoption spans applications in vaccinology, cell therapies, gene replacement therapies, and protein replacement [5]. The technology is supported by a growing body of literature, with studies published in journals such as Molecular Therapy and The Lancet demonstrating its application in vaccines targeting Lyme disease, Respiratory Syncytial Virus (RSV), and self-amplifying RNA (saRNA) vaccines [16].
Table: Documented Applications of CleanCap Technology in Vaccine Development
| Pathogen/Target | Vaccine Type | Development Stage | Key Findings | Citation |
|---|---|---|---|---|
| SARS-CoV-2 | Non-replicating mRNA | Commercial | 95% protection; tremendous safety profile, billions of doses | [5] [16] |
| SARS-CoV-2 | Self-amplifying RNA | Phase I Trial | Well-tolerated, no serious adverse reactions | [16] |
| Lyme Disease | Non-replicating mRNA | Preclinical | Induced tick resistance, prevented transmission | [16] |
| Respiratory Syncytial Virus | Non-replicating mRNA | Phase I Trial | Well-tolerated, immune response elicited | [16] |
Quantitative Performance and Comparative Analysis
The industry adoption of CleanCap is driven by its superior performance metrics compared to legacy capping methods, including higher capping efficiency, increased protein expression, and significant process economics.
Capping Efficiency and Protein Expression
CleanCap technology consistently achieves a capping efficiency of >95%, a substantial improvement over first-generation cap analogs like ARCA (Anti-Reverse Cap Analog), which typically exhibits 50-80% efficiency [5] [70]. This high efficiency directly translates into a greater proportion of functional mRNA molecules.
Furthermore, the latest CleanCap analogs provide enhanced protein expression. The newest analog, CleanCap M6, increases protein expression by over 30% compared to previous analogs such as CleanCap AG and AG 3'OMe [5] [39].
Table: Performance Comparison of CleanCap Analogs
| Analog | Capping Efficiency | Cap Structure | Protein Expression | Key Application |
|---|---|---|---|---|
| CleanCap M6 | >95% | Cap 1 | Highest (30% higher than predecessors) | Standard mRNA |
| CleanCap AG 3'OMe | >95% | Cap 1 | Higher | Standard mRNA (used in commercial vaccines) |
| CleanCap AG | >95% | Cap 1 | High | Standard mRNA |
| CleanCap AU | >95% | Alphavirus 5' cap | Durable | Self-amplifying RNA (saRNA) |
| ARCA (Legacy Method) | 50-80% | Cap 0 | Moderate | Standard mRNA (requires additional enzymatic step for Cap 1) |
Process Economics and Development Timelines
Adopting CleanCap's one-pot co-transcriptional capping strategy streamlines mRNA manufacturing, offering tangible economic and timeline benefits:
- Reduced Production Time: Cuts mRNA therapeutic production processes by nearly one week by eliminating post-transcriptional enzymatic capping steps [5].
- Lower Manufacturing Costs: Reduces overall manufacturing costs by 20-40% compared to other capping methods [5].
- Increased mRNA Yield: Optimized protocols, such as the pulse-feed method for CleanCap M6, can further increase IVT yields, lowering the cost of goods [39].
Essential Research Reagent Solutions
Successful implementation of mRNA synthesis with CleanCap requires a suite of specialized reagents. The following table details key components and their functions.
Table: Key Reagent Solutions for mRNA Synthesis with CleanCap
| Reagent / Material | Function / Role in IVT | Example or Note |
|---|---|---|
| CleanCap Analog | Co-transcriptional capping to form Cap 1 structure. | Choose analog (e.g., M6, AG, AU) based on application [5]. |
| DNA Template | Provides the sequence to be transcribed; must start with "AGG" after T7 promoter. | Plasmid DNA or PCR fragment; requires specific initiation sequence [70] [71]. |
| RNA Polymerase | Enzymatically synthesizes RNA from DNA template. | T7 RNA Polymerase; variants like CleanScribe can reduce dsRNA [23]. |
| Nucleotide Triphosphates (NTPs) | Building blocks for RNA synthesis. | Can include modified nucleotides (e.g., N1-methylpseudouridine) to reduce immunogenicity [5] [23]. |
| DNase I | Degrades the DNA template after transcription is complete. | Essential for purifying the final mRNA product [71]. |
| Purification Kits/Reagents | Removes enzymes, salts, and aberrant RNA products (e.g., dsRNA) post-IVT. | Lithium chloride precipitation or chromatographic methods [22]. |
Detailed Experimental Protocols
Basic Protocol: Standard IVT with CleanCap for Cap 1 mRNA Synthesis
This protocol, adapted from the Current Protocols publication, describes the synthesis of Cap 1 mRNA using CleanCap AG reagent, yielding up to 5 mg/mL of mRNA with 94% capping efficiency [22].
Materials:
- Nuclease-free water
- 10X IVT Buffer (e.g., 400 mM HEPES-KOH, pH 7.5, 120 mM MgCl₂, 20 mM spermidine)
- Ribonucleotide Solution (ATP, CTP, UTP, 25 mM each; GTP, 10 mM)
- CleanCap AG Reagent (e.g., 100 mM stock)
- T7 RNA Polymerase (e.g., 50,000 U/mL)
- DNA Template (0.1-0.5 μg/μL, with AGG transcription start site)
- DNase I (RNase-free)
Procedure:
- Reaction Setup: Assemble the IVT reaction on ice in a nuclease-free microcentrifuge tube in the following order:
- Nuclease-free water (to a final volume of 20 μL)
- 10X IVT Buffer: 2 μL
- ATP, CTP, UTP (25 mM each): 4 μL of each
- GTP (10 mM): 4 μL
- CleanCap AG Reagent (100 mM): 2 μL
- DNA Template: 2 μL (0.5-1 μg total)
- T7 RNA Polymerase: 2 μL
- Final Volume: 20 μL
Incubation: Mix the reaction gently by pipetting and incubate at 37°C for 2-3 hours.
DNase I Treatment: After incubation, add 2 μL of DNase I to the reaction tube. Mix gently and incubate at 37°C for 15 minutes to digest the DNA template.
mRNA Purification: Purify the mRNA using a preferred method, such as lithium chloride (LiCl) precipitation or spin-column-based purification kits [22]. The synthesized mRNA can be analyzed for yield, purity, and capping efficiency by techniques like agarose gel electrophoresis, ion-pair reversed-phase HPLC, or LC-MS [22].
Advanced Protocol: Increasing Yield with CleanCap M6 Pulse-Feed
For applications requiring very high mRNA yields, TriLink recommends a pulse-feed protocol with the CleanCap M6 analog [39]. This method addresses substrate depletion over time in a standard IVT.
Additional Materials:
- CleanCap M6 analog
- Pulse-Feed Solution (containing additional NTPs and cap analog)
Procedure:
- Initial Reaction Setup: Prepare and run a standard IVT as in Basic Protocol 5.1, but using CleanCap M6.
Pulse-Feed Addition: After 60-90 minutes of incubation, add a small volume (e.g., 10% of the initial reaction volume) of a pulse-feed solution containing NTPs and CleanCap M6.
Continued Incubation: Mix the reaction gently and continue the incubation at 37°C for an additional 1-2 hours.
Purification and Analysis: Proceed with DNase I treatment, purification, and analysis as described in the basic protocol. This method can significantly increase the final mRNA yield compared to a single-batch reaction [39].
Workflow and Pathway Visualizations
Co-transcriptional Capping Workflow
The following diagram illustrates the streamlined workflow for synthesizing Cap 1 mRNA using the co-transcriptional capping method with CleanCap, highlighting its efficiency over traditional multi-step approaches.
Cap Structure Evolution and Immune Recognition
This diagram outlines the hierarchy of 5' cap structures, from uncapped RNA to Cap 2, and their relationship to translational efficiency and immune system evasion, which is critical for therapeutic efficacy.
Conclusion
CleanCap co-transcriptional capping represents a paradigm shift in mRNA synthesis, effectively addressing key challenges of manufacturing efficiency, product quality, and cost. By enabling a streamlined one-pot process that reliably produces mRNA with >95% Cap 1 structure efficiency, this technology directly enhances the stability, translational capacity, and therapeutic potency of mRNA products. The integration of CleanCap with complementary innovations in codon optimization, enzyme engineering, and process control is paving the way for a new generation of more potent, dose-efficient, and accessible mRNA therapeutics and vaccines. As the field advances, the continued evolution of cap analogs and synthesis strategies will be crucial for expanding the application of mRNA technology into complex disease areas like oncology, genetic disorders, and regenerative medicine.
References
- 1. Five-prime cap [https://en.wikipedia.org/wiki/Five-prime_cap]
- 2. mRNA capping: biological functions and applications - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5027499/]
- 3. 5-prime capping of mRNA [https://www.takarabio.com/learning-centers/mrna-and-cdna-synthesis/mrna-synthesis/5-prime-capping-of-mrna?srsltid=AfmBOorADTXzJX1E7vvMRYLEx1lwx2Twyf5QCx4sTxQQcGpuQkydnwpC]
- 4. Chemo‐Enzymatic Modification of the 5′ Cap Maintains ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8250829/]
- 5. CleanCap mRNA Capping Technology [https://www.trilinkbiotech.com/cleancap]
- 6. Enhancing cap-independent translation of linear mRNA [https://www.nature.com/articles/s41467-025-64257-6]
- 7. Co-transcriptional capping [https://www.takarabio.com/learning-centers/mrna-and-cdna-synthesis/mrna-synthesis/5-prime-capping-of-mrna/co-transcriptional-capping?srsltid=AfmBOor26EQTxddY63utWcwNyA8Ogb4C4bsrDJWtqDx3DPWRx1UswO44]
- 8. Cap-end impurities under forced degradation conditions [https://www.sciencedirect.com/science/article/pii/S2162253125001246]
- 9. 5-prime capping of mRNA [https://www.takarabio.com/learning-centers/mrna-and-cdna-synthesis/mrna-synthesis/5-prime-capping-of-mrna?srsltid=AfmBOoo4pYegdpqV60Oi-xjr7oR-MYvAdfY4GkFDzF5xwMmfuBoSf1sy]
- 10. Co-transcriptional capping [https://www.takarabio.com/learning-centers/mrna-and-cdna-synthesis/mrna-synthesis/5-prime-capping-of-mrna/co-transcriptional-capping?srsltid=AfmBOoqkzgjTN8RRJsD2ogcyyKwi1r8WczCL3oNUoJX-RSwAr4BWuiEI]
- 11. The design, manufacture and LNP formulation of mRNA for ... [https://pubmed.ncbi.nlm.nih.gov/40494942/]
- 12. mRNA Capping: Process, Mechanisms, Methods & Applications [https://lifesciences.danaher.com/us/en/library/mrna-capping.html]
- 13. 5 Things to Know About mRNA Capping for Next- ... [https://app.scientist.com/blog/2024/05/23/5-things-to-know-about-mrna-capping-for-next-generation-vaccines-and-therapies]
- 14. Post-transcriptional capping [https://www.takarabio.com/learning-centers/mrna-and-cdna-synthesis/mrna-synthesis/5-prime-capping-of-mrna/post-transcriptional-capping?srsltid=AfmBOopQ62zMYMyxr0mrHAcMY_RS73ySv8DCv83xK5BJdjQk4rGIXYJj]
- 15. RNA Capping [https://www.neb.com/en-us/products/rna-synthesis-and-modification/rna-capping?srsltid=AfmBOor95neFqmWLJxAcVbto8aSQw-1AsRS8unfwA-6aBHN-Kpxz_hKL]
- 16. Clean Capped mRNA: The Future of Vaccine Development [https://www.trilinkbiotech.com/cleancap-vaccine-development]
- 17. TriLink BioTechnologies® debuts its first mRNA synthesis ... [https://www.glenresearch.com/press-releases/trilink-biotechnologies-debuts-its-first-mrna-synthesis-kit-with-cleancap-capping-technology]
- 18. mRNA Capping Efficiency Assay [https://rna.bocsci.com/products-services/mrna-capping-efficiency-assay.html]
- 19. Characterization and relative quantification of mRNA 5' ... [https://sciex.com/tech-notes/biopharma/characterization-relative-quantification-mrna5capping]
- 20. Quantitative assessment for cap efficiency of messenger rna [https://patents.google.com/patent/WO2014152673A1/en]
- 21. Methods of IVT mRNA capping - Behind the Bench [https://www.thermofisher.com/blog/behindthebench/methods-of-ivt-mrna-capping/]
- 22. Cap 1 Messenger RNA Synthesis with Co-transcriptional ... [https://pubmed.ncbi.nlm.nih.gov/33524237/]
- 23. Celebrates launch with kit donations to top academic ... [https://www.trilinkbiotech.com/press-releases/trilink-biotechnologies-debuts-its-first-mrna-synthesis-kit-with-cleancap-capping-technology]
- 24. Next-level IVT, all together [https://www.trilinkbiotech.com/cleancap-ivt-kits]
- 25. Glen Report 35-21: CleanCap® M6 [https://www.glenresearch.com/reports/gr35-21]
- 26. CleanCap M6 inhibits decapping of exogenously delivered ... [https://www.sciencedirect.com/science/article/pii/S2162253125000101]
- 27. CleanCap® AG (3′ OMe) CleanScript™ IVT Kit - (K-7413) [https://shop.trilinkbiotech.com/shopping/products/ivt-and-capping-reagents/ivt-and-capping-kit/cleancap-ag-3-ome-cleanscript-ivt-kit-k-7413/]
- 28. 5-prime capping of mRNA [https://www.takarabio.com/learning-centers/mrna-and-cdna-synthesis/mrna-synthesis/5-prime-capping-of-mrna?srsltid=AfmBOoqUyK0gQMjLrNwqAZwBcysKWdqkCcFzVgNvJR951zymF6TDGcyU]
- 29. Cleancap M6 [https://trilinkbiotech.com/cleancap-m6]
- 30. Potential Unlocked! [https://www.aldevron.com/blog/enable-mrna-therapeutic-development]
- 31. Expedient production of site specifically nucleobase ... [https://www.nature.com/articles/s41467-024-47444-9]
- 32. Synergistic effect of nucleoside modification and ionizable ... [https://www.nature.com/articles/s41541-025-01263-1]
- 33. TriLink BioTechnologies® debuts its first mRNA synthesis ... [https://investors.maravai.com/news-events/press-releases/detail/112/trilink-biotechnologies-debuts-its-first-mrna-synthesis-kit-with-cleancap-capping-technology-celebrates-launch-with-kit-donations-to-top-academic-institutions]
- 34. CleanCap Technology: Leading the way in mRNA [https://www.abacusdx.com/life-science/cleancap-technology-leading-the-way-in-mrna/]
- 35. GMP mRNA Manufacturing [https://www.trilinkbiotech.com/gmp-mrna-manufacturing]
- 36. The Platform Technology Approach to mRNA Product ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11126020/]
- 37. Considerations for mRNA Product Development, ... [https://www.mdpi.com/2076-393X/13/5/473]
- 38. mRNA Capping [https://www.trilinkbiotech.com/cleancap-mrna-capping]
- 39. Increasing mRNA yields using CleanCap ® M6 pulse-feed ... [https://www.trilinkbiotech.com/cleancap-m6-pulse-feed]
- 40. CleanCap M6 inhibits decapping of exogenously delivered ... [https://www.trilinkbiotech.com/blog/cleancap-m6-inhibits-decapping-of-exogenously-delivered-ivt-mrna%20/]
- 41. A Facile Method for the Removal of dsRNA Contaminant ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6444222/]
- 42. An improved method for the detection of double-stranded ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11528662/]
- 43. Optimized Residual dsRNA Detection for mRNA ... [https://www.pharmasalmanac.com/articles/optimized-residual-dsrna-detection-for-mrna-therapeutics]
- 44. Double-stranded RNA reduction by chaotropic agents ... [https://www.sciencedirect.com/science/article/pii/S2162253122001974]
- 45. Optimize mRNA Production From the Very Start With ... [https://www.aldevron.com/download-poster-dsrna-optimize-synthesis]
- 46. Reverse-phase chromatography removes double-stranded ... [https://www.sciencedirect.com/science/article/pii/S2162253125000459]
- 47. Model‐Based Optimization of Fed‐Batch In Vitro ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12596937/]
- 48. Maximizing the mRNA productivity for in vitro transcription ... [https://www.sciencedirect.com/science/article/abs/pii/S1369703X24001992]
- 49. Model-Based Optimization of Fed-Batch In Vitro Transcription [https://chemrxiv.org/engage/chemrxiv/article-details/685c5549c1cb1ecda0146190]
- 50. Model-Based Optimization of Fed-Batch In Vitro Transcription [https://pubmed.ncbi.nlm.nih.gov/41064918/]
- 51. Optimizing Modified mRNA In Vitro Synthesis Protocol for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6722299/]
- 52. Overcoming scaling challenges in mRNA synthesis with ... [https://www.labroots.com/ms/webinar/overcoming-scaling-challenges-mrna-synthesis-fed-batch-ivt?srsltid=AfmBOor48_WgGK4SHgQr_Cq49YUyjRwITOHxxABj6hq1Cz0Q8fyYHdki]
- 53. CleanCap® IVT Kits | High-Yield, Low-dsRNA mRNA ... [https://www.tebubio.com/en_fr_eur/content/post/cleancap-ivt-kits-mrna-synthesis]
- 54. Advances in mRNA nanotherapy [https://www.trilinkbiotech.com/advances-in-mrna-nanotherapy]
- 55. Mesenchymal stem cell engineering by ARCA analog- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10425852/]
- 56. RNA Capping [https://www.neb.com/en-us/products/rna-synthesis-and-modification/rna-capping?srsltid=AfmBOoqTp8-oEp1PrPTjVAJc4nlbC16-SkIBv60-xreT9w_lCCBk_3yo]
- 57. Risks of Cuts to mRNA Vaccine Development | Johns Hopkins [https://publichealth.jhu.edu/2025/risks-of-cuts-to-mrna-vaccine-development]
- 58. Infant with rare, incurable disease is first to successfully ... [https://www.nih.gov/news-events/news-releases/infant-rare-incurable-disease-first-successfully-receive-personalized-gene-therapy-treatment]
- 59. Protein stitching to create a full-length dystrophin: a go- ... [https://www.worldmusclesociety.org/news/view/protein-stitching-to-create-a-full-length-dystrophin-a-go-to-strategy-for-large-genes]
- 60. Advancements and challenges in next-generation mRNA ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12599430/]
- 61. CRISPR Clinical Trials: A 2025 Update [https://innovativegenomics.org/news/crispr-clinical-trials-2025/]
- 62. Co-transcriptional capping [https://www.takarabio.com/learning-centers/mrna-and-cdna-synthesis/mrna-synthesis/5-prime-capping-of-mrna/co-transcriptional-capping?srsltid=AfmBOor3o_FhOe6W0i6wCjG-391xrC9Af6tn5hNENPNd1vsSo9l65NsN]
- 63. Discovery and molecular mechanism of potent neutralizing ... [https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1013674]
- 64. A multivalent nanoparticle vaccine elicits potent ... [https://www.sciencedirect.com/science/article/abs/pii/S0168365925009538]
- 65. Novel Trispecific Neutralizing Antibodies With Enhanced ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12010136/]
- 66. Biomarkers of mRNA vaccine efficacy derived from ... [https://journals.plos.org/ploscompbiol/article?id=10.1371/journal.pcbi.1013163]
- 67. Neutralizing antibody responses to three XBB protein ... [https://www.nature.com/articles/s41392-025-02132-y]
- 68. Development of a personalized digital biomarker ... [https://www.nature.com/articles/s43856-025-00840-8]
- 69. a comprehensive review of mRNA vaccine technology and ... [https://virologyj.biomedcentral.com/articles/10.1186/s12985-025-02645-6]
- 70. Co-transcriptional capping [https://www.takarabio.com/learning-centers/mrna-and-cdna-synthesis/mrna-synthesis/5-prime-capping-of-mrna/co-transcriptional-capping?srsltid=AfmBOoqFROoHayzB6flNvYvByt1HRLeVV6iYlPbxNSXNm2tnIU6ossXd]
- 71. Strategies for synthesizing in vitro transcribed (IVT) mRNA [https://www.neb.com/en-us/tools-and-resources/feature-articles/mind-your-caps-and-poly-a-tails-strategies-for-synthesizing-in-vitro-transcribed-ivt-mrna?srsltid=AfmBOooNXiNMQtX15WtSyKo3arfPQ7LlmAUkktV9m9jUc4XY7niQjpr6]