Comparative Paracrine Factor Expression in ASCs vs BMSCs vs UCB-MSCs: Implications for Cell-Based Therapies
This article systematically compares the paracrine factor expression profiles of mesenchymal stem cells derived from adipose tissue (ASCs), bone marrow (BMSCs), and umbilical cord blood (UCB-MSCs).
Comparative Paracrine Factor Expression in ASCs vs BMSCs vs UCB-MSCs: Implications for Cell-Based Therapies
Abstract
This article systematically compares the paracrine factor expression profiles of mesenchymal stem cells derived from adipose tissue (ASCs), bone marrow (BMSCs), and umbilical cord blood (UCB-MSCs). For researchers and drug development professionals, we explore foundational biological differences, methodological approaches for characterization, troubleshooting for experimental and clinical translation, and validation through functional outcomes. The analysis synthesizes current evidence to guide source selection for specific therapeutic applications, particularly in cardiovascular repair, wound healing, and tissue engineering, where paracrine-mediated effects including angiogenesis, cytoprotection, and immunomodulation are critical.
Defining the Secretome: Fundamental Biological Differences in MSC Paracrine Factor Expression
Core Principles of the Paracrine Hypothesis in Stem Cell Therapy
The field of regenerative medicine has undergone a fundamental paradigm shift with the emergence and validation of the paracrine hypothesis. This concept proposes that the therapeutic benefits of stem cells, particularly Mesenchymal Stem Cells (MSCs), are mediated primarily through the secretion of bioactive molecules rather than direct cell replacement [1]. These molecules—including growth factors, cytokines, and chemokines—act in a paracrine fashion on resident cells, influencing processes such as cell survival, angiogenesis, immunomodulation, and tissue repair in a temporal and spatial manner [1].
Initially, the therapeutic potential of stem cells was attributed to their ability to engraft into damaged tissues and differentiate into functional target cells to replace lost or damaged ones [1]. However, numerous studies revealed that injected adult stem cells suffered from poor survivability and low long-term engraftment rates, yet significant functional improvements were still observed [1] [2]. This paradox led researchers to investigate alternative mechanisms. Seminal experiments demonstrated that administering conditioned media from stem cell cultures—containing the secreted factors but no cells—was sufficient to recapitulate most of the therapeutic benefits observed with whole cell therapy [1] [2]. This critical finding established the foundation for the paracrine hypothesis, which has since become a central principle in stem cell research and therapy development.
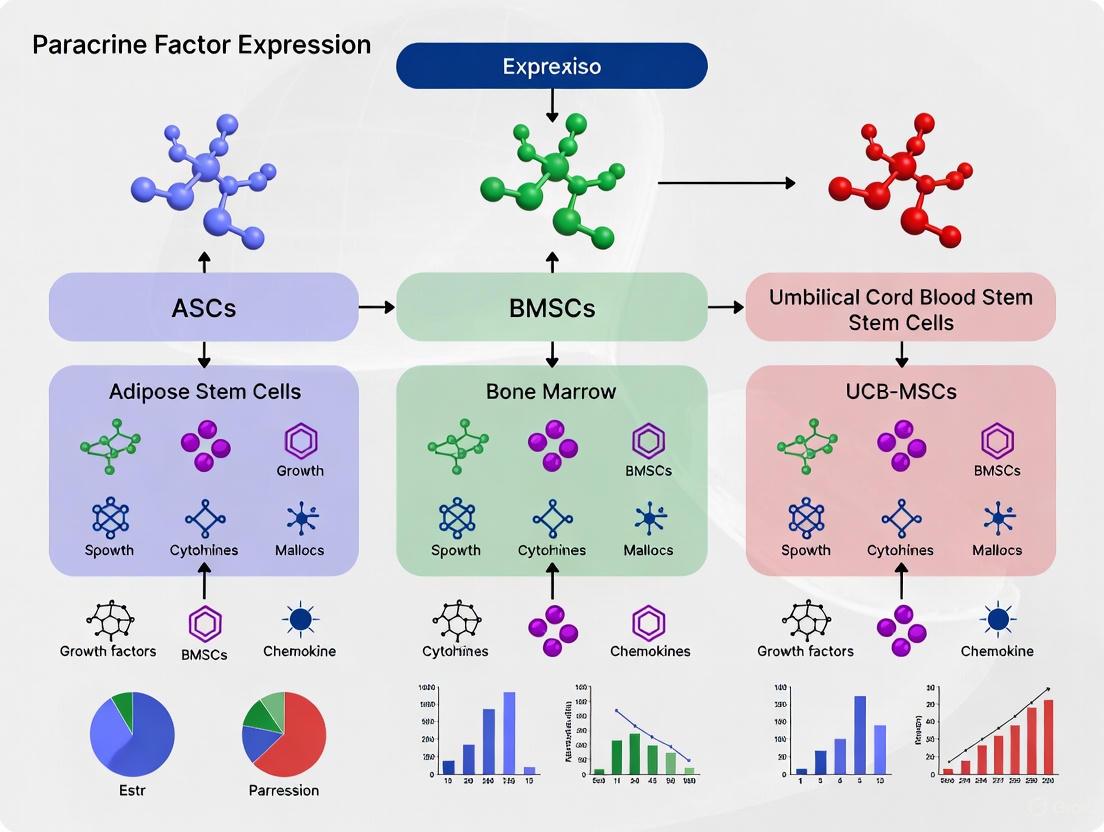
Comparative Paracrine Factor Expression in ASCs vs BMSCs vs UCB-MSCs
The paracrine activity of MSCs varies significantly depending on their tissue origin. Understanding these differences is crucial for selecting the optimal cell source for specific therapeutic applications. The following analysis compares the paracrine profiles of Adipose-derived Stem Cells (ASCs), Bone Marrow-derived MSCs (BMSCs), and Umbilical Cord Blood-derived MSCs (UCB-MSCs).
Table 1: Comparative Expression of Key Paracrine Factors Across Different MSC Sources
| Paracrine Factor | Function | ASCs | BMSCs | UCB-MSCs | References |
|---|---|---|---|---|---|
| VEGF-A | Angiogenesis, endothelial cell survival | Comparable | Comparable | Higher (vs. BMSCs) | [3] [4] |
| VEGF-D | Angiogenesis, lymphangiogenesis | Higher | Lower | Not Specified | [3] |
| IGF-1 | Cell survival, proliferation, metabolism | Higher | Lower | Not Specified | [3] |
| IL-8 | Angiogenesis, chemotaxis | Higher | Lower | Not Specified | [3] |
| Angiogenin | Angiogenesis, ribonuclease activity | Comparable | Comparable | Not Specified | [3] |
| bFGF (FGF2) | Angiogenesis, cell proliferation | Comparable | Comparable | Not Specified | [3] [2] |
| HGF | Angiogenesis, anti-fibrosis, mitogenesis | Conflicting Data | Conflicting Data | Higher (vs. BMSCs) | [4] [2] |
| Neurotrophic Factors | Neuroprotection, neurogenesis | Not Specified | Not Specified | Higher | [4] |
Functional Implications of Expression Differences
The distinct paracrine signatures of different MSC populations translate to varied functional capabilities. A direct comparative analysis of angiogenic potential revealed that incubation of endothelial cells with conditioned media from ASCs resulted in increased tubulogenic efficiency compared to media from other MSC populations [3]. Furthermore, this study identified VEGF-A and VEGF-D as major growth factors secreted by ASCs that directly supported endothelial tubulogenesis [3].
Beyond angiogenesis, the immunomodulatory properties of MSCs are also heavily influenced by their secretome. All MSC sources release factors like TGFβ, HGF, PGE2, and IL-6, which collectively inhibit T-cell proliferation, prevent dendritic cell maturation, and modulate B-cell and Natural Killer (NK) cell function [1]. However, the potency and specific profile of these immunomodulatory secretions can vary with tissue origin, influencing their effectiveness in different inflammatory disease contexts.
Experimental Protocols for Evaluating Paracrine Activity
To generate the comparative data presented, rigorous experimental methodologies are employed. The following section outlines the key protocols used to isolate MSCs from different sources and evaluate their paracrine activity.
Cell Isolation and Culture Protocols
Primary Culture of Human Adipose-derived Stem Cells (ASCs): ASCs are isolated from subcutaneous adipose tissue via collagenase digestion. The digested tissue is centrifuged to separate stromal cells from adipocytes. The cell pellet is resuspended, filtered through a mesh, and plated in culture flasks. Non-adherent cells are removed after overnight incubation [3].
Primary Culture of Human Bone Marrow-derived Stem Cells (BMSCs): While the exact protocol from the search results is truncated, commercial BMSCs are widely used and are typically isolated from bone marrow aspirates by density gradient centrifugation to obtain mononuclear cells, which are then plated. The adherent fraction is expanded and characterized based on surface marker expression [3].
Primary Culture of Human Umbilical Cord Blood-derived MSCs (UCB-MSCs): UCB-MSCs are isolated from umbilical cord blood collected after birth. The mononuclear cell fraction is separated using density gradient centrifugation and plated in specialized media to select for the adherent MSC population [4].
Paracrine Factor Analysis Protocols
Conditioned Media (CM) Collection: MSCs are cultured until they reach 70-80% confluence. The culture medium is then replaced with a serum-free medium to avoid contamination from serum proteins. After 24-72 hours, the conditioned media is collected and centrifuged to remove cells and debris. The supernatant (CM) is concentrated if necessary and stored for analysis [3] [5].
mRNA Expression Analysis (qRT-PCR): Total RNA is extracted from MSCs, reverse transcribed into cDNA, and analyzed using quantitative real-time PCR (qRT-PCR) with gene-specific primers for target paracrine factors (e.g., VEGF-A, IGF-1, HGF). Expression levels are normalized to housekeeping genes and compared across different MSC populations [3].
Protein Analysis (ELISA and Mass Spectrometry): The concentration of specific proteins in the CM is quantified using Enzyme-Linked Immunosorbent Assay (ELISA) kits. For a broader, unbiased analysis, liquid chromatography coupled with tandem mass spectrometry (LC/MS/MS) is used to identify and quantify hundreds of proteins in the secretome [3] [5].
Functional Tubulogenesis Assay: To assess angiogenic paracrine activity, endothelial cells are seeded on a basement membrane matrix (e.g., Matrigel) and incubated with MSC-conditioned media. The formation of capillary-like tube structures by endothelial cells is visualized and quantified by measuring total tube length or number of branches, providing a functional readout of angiogenic potential [3].
Signaling Pathways in Paracrine-Mediated Repair
The paracrine factors released by MSCs activate complex signaling pathways in recipient cells to promote repair and regeneration. The diagram below illustrates the key pathways involved in the core paracrine mechanisms of cytoprotection, angiogenesis, and immunomodulation.
Diagram 1: Core paracrine signaling pathways activated by MSC-secreted factors, leading to key therapeutic outcomes.
Key Pathway Interactions
- Cytoprotection: Factors like IGF-1 and the novel protein HASF activate the Akt/PI3K pathway, inhibiting caspase activity and mitochondrial pore opening to prevent apoptosis. Sfrp2 protects cells by binding to Wnt3a and attenuating pro-apoptotic Wnt/β-catenin signaling [1].
- Angiogenesis: VEGF and bFGF bind to their respective receptors on endothelial cells, activating MAPK/ERK pathways to promote cell proliferation and tube formation, crucial for building new blood vessels [3] [2].
- Immunomodulation: MSC-secreted PGE2, TGFβ, and TSG-6 modulate NF-κB signaling and other pathways in immune cells, shifting the balance from pro-inflammatory (M1) to anti-inflammatory (M2) macrophage phenotypes and suppressing T-cell proliferation [1].
The Scientist's Toolkit: Essential Research Reagents
Research into the paracrine hypothesis relies on a specific set of reagents and tools. The following table details key solutions used in the experiments cited within this guide.
Table 2: Key Research Reagent Solutions for Paracrine Mechanism Studies
| Reagent / Tool | Function / Application | Example from Context |
|---|---|---|
| Type I Collagenase | Enzymatic digestion of tissues for cell isolation. | Used to dissociate human adipose tissue to release ASCs from the extracellular matrix [3]. |
| Dulbecco’s Modified Eagle’s Medium (DMEM) | Basal cell culture medium for MSC expansion. | Used as the base medium for culturing ASCs, BMSCs, DPCs, and DSCs under identical conditions [3]. |
| Fetal Calf Serum (FCS) | Provides essential nutrients and growth factors for cell growth. | Added at 10% concentration to DMEM to support the growth of various MSC types [3]. |
| Neutralizing Antibodies | Functionally blocks specific proteins or pathways. | Used to identify VEGF-A and VEGF-D as major contributors to ASC-mediated endothelial tubulogenesis [3]. |
| Enzyme-Linked Immunosorbent Assay (ELISA) Kits | Quantifies specific protein concentrations in solution. | Used to confirm the protein levels of angiogenin and VEGF-A in MSC-conditioned media [3]. |
| Basement Membrane Matrix (e.g., Matrigel) | Provides a substrate for in vitro tubulogenesis assays. | Used as a surface for endothelial cells to form capillary-like tubes when stimulated with MSC-conditioned media [3]. |
| Fura-2 AM | Ratiometric fluorescent calcium indicator for live-cell imaging. | Used to load human lens epithelial cells to visualize and record mechanically induced Ca²⁺ waves in paracrine communication studies [6]. |
| Carbenoxolone (CBX) & Apyrase | Pharmacological inhibitors for pathway dissection. | CBX (gap junction blocker) and Apyrase (ATP-hydrolyzing enzyme) used to dissect the role of different signaling mechanisms in Ca²⁺ wave propagation [6]. |
The core principles of the paracrine hypothesis have redefined our understanding of stem cell therapy, shifting the focus from cell replacement to cell-based drug delivery. The comparative analysis reveals that the tissue origin of MSCs—ASCs, BMSCs, or UCB-MSCs—critically shapes their secretome and thus their functional therapeutic profile. ASCs demonstrate a particularly strong angiogenic signature, characterized by elevated expression of VEGF-D, IGF-1, and IL-8, which translates to superior pro-angiogenic effects in functional assays [3].
This mechanistic understanding opens the door to more sophisticated and effective regenerative strategies. Future research is moving beyond whole cell therapy towards the use of defined factor cocktails, engineered exosomes, and primed MSCs whose therapeutic secretomes can be optimized for specific clinical applications. By harnessing the precise signaling pathways and paracrine factors identified in comparative studies like this one, the next generation of regenerative therapies will be more controlled, targeted, and potent.
The therapeutic potential of mesenchymal stem cells (MSCs) in regenerative medicine is increasingly attributed to their paracrine activity rather than their direct differentiation capacity. MSCs isolated from different anatomical niches exhibit distinct biological properties and secretory profiles, influenced by their unique tissue-specific microenvironments. This review provides a comparative analysis of the paracrine factor expression in MSCs derived from adipose tissue (ASCs), bone marrow (BMSCs), and dermal tissue (DSCs/DPCs), synthesizing experimental data to guide researchers in selecting appropriate cell sources for specific therapeutic applications.
Anatomical Niches and Their Microenvironmental Determinants
Stem cell behavior is governed by specialized microenvironments, or niches, that integrate structural, biochemical, and mechanical cues to regulate cellular functions [7]. These niches comprise immediate stromal neighbors, extracellular matrix scaffolds, and tissue-specific architectural variants that collectively influence stem cell fate decisions [7].
Table 1: Characteristics of MSC Anatomical Niches
| Tissue Source | Key Niches | Cellular Constituents | ECM Components | Architectural Features |
|---|---|---|---|---|
| Adipose Tissue | Subcutaneous adipose tissue [8] | Adipocytes, preadipocytes, lymphocytes, leukocytes, erythrocytes [8] | Collagen, laminin, fibronectin [7] | Lipid-rich vacuoles, stromal vascular fraction [8] |
| Bone Marrow | Endosteal niche, perivascular niche [7] | Osteoblasts, sinusoids, HSCs, vascular cells [7] | Laminin, collagen, fibronectin, proteoglycans [7] | Trabecular networks, CXCL12-rich sinusoids [7] |
| Dermal Tissue | Hair follicle bulge, follicle neck, dermal papilla [9] | Dermal sheath cells, dermal papilla cells, keratinocytes [3] [10] | Collagen, elastin fibers, basement membrane [8] | Follicular units, continuous with epidermal basal layer [9] |
The anatomical location dictates functional specialization, with bone marrow niches supporting hematopoiesis, adipose niches optimized for energy storage and endocrine function, and dermal niches coordinating epithelial maintenance and hair cycling [7]. These microenvironmental differences imprint distinct identities on resident MSC populations that persist even after in vitro expansion.
Comparative Paracrine Factor Expression Profiles
Quantitative Analysis of Secretory Molecules
Table 2: Comparative Paracrine Factor Expression in MSC Populations
| Paracrine Factor | ASCs | BMSCs | Dermal MSCs (DPCs/DSCs) | Functional Significance |
|---|---|---|---|---|
| IGF-1 | ↑ Higher [3] [10] | Lower | Lower | Promotes cell survival, proliferation, metabolism |
| VEGF-D | ↑ Higher [3] [10] | Lower | Lower | Lymphangiogenesis, endothelial cell activation |
| IL-8 | ↑ Higher [3] [10] | Lower | Lower | Neutrophil chemotaxis, angiogenesis |
| VEGF-A | Comparable [3] [10] | Comparable | Comparable | Angiogenesis, endothelial cell proliferation |
| Angiogenin | Comparable [3] [10] | Comparable | Comparable | Angiogenesis, RNA cleavage |
| bFGF | Comparable [3] [10] | Comparable | Comparable | Fibroblast proliferation, tissue repair |
| NGF | Comparable [3] [10] | Comparable | Comparable | Neuronal survival, differentiation |
| Leptin | Lower | Lower | ↑ Higher [3] [10] | Appetite regulation, metabolism |
Protein analysis of conditioned media confirms that VEGF-A and angiogenin secretion are comparable among all MSC populations, while dermal MSCs produce significantly higher concentrations of leptin [3]. Functional validation through endothelial tubulogenesis assays demonstrates that ASC-conditioned media results in increased tubulogenic efficiency compared to DPC-conditioned media, with VEGF-A and VEGF-D identified as major mediators of this enhanced angiogenic response [3] [10].
Secretome Composition Beyond Conventional Factors
Recent investigations reveal that MSC secretomes comprise not only soluble proteins but also extracellular vesicles (EVs), including exosomes and microvesicles, which carry proteins, lipids, and nucleic acids [11] [8]. The composition of these secretomes shows significant variations among MSC lines from different tissues and even within populations obtained with different extraction methods [11].
ASC and dental pulp MSC (DPSC) secretomes contain specific sets of microRNAs, either free or enclosed in EVs, that impact diverse cellular processes [11]. microRNAs more highly expressed in DPSCs are mainly involved in oxidative stress and apoptosis pathways, while those of ASCs play regulatory roles in cell cycle and proliferation [11].
Experimental Methodologies for Secretory Profile Analysis
Cell Isolation and Culture Protocols
Adipose-Derived Stem Cell Isolation
- Tissue Processing: Adipose tissue (∼200 mL) is minced into 1 mm³ pieces, washed extensively with PBS, and digested with 0.075% type I collagenase at 37°C for 60 minutes [3]
- Cell Recovery: Cells are collected by centrifugation at 300g for 10 minutes, resuspended in growth medium, and filtered through a 100μm nylon mesh [3]
- Erythrocyte Removal: Cell pellets are treated with 0.16M NH4Cl for 5 minutes at room temperature for red blood cell lysis [3]
- Culture Conditions: Cells are plated in Dulbecco's Modified Eagle Medium low-glucose medium supplemented with 10% fetal calf serum and 1% antibiotic-antimycotic solution [3]
Bone Marrow-Derived Stem Cell Culture
- Commercial BMSCs are cultured according to supplier specifications in specialized media formulations [3]
- Cells between passages 3-6 are typically used for experiments to maintain phenotypic stability [3]
Dermal MSC Isolation
- Tissue Harvesting: Hair follicles are microdissected from scalp specimens, gripping at the supra-bulbar region [3]
- Dermal Sheath Cell Isolation: Whole hair follicles are explanted, allowing DSCs to migrate from the mesenchymal layer over 7 days [3]
- Dermal Papilla Cell Isolation: Dermal papillae are released from hair follicle bulbs by microdissection and anchored to culture dishes with a fine needle to break the basal lamina [3]
Secretome Collection and Analysis
Conditioned Media Preparation
- Cells are cultured to 70-80% confluence before media exchange with serum-free basal media [3] [11]
- Conditioned media is typically collected after 24-48 hours of incubation [11]
- Media is centrifuged to remove cellular debris and concentrated using centrifugal filter devices [11]
Analytical Techniques
- mRNA Expression Analysis: Quantitative RT-PCR for paracrine factor genes [3]
- Protein Analysis: Enzyme-linked immunosorbent assays (ELISA) for specific factors in conditioned media [3]
- Functional Assays: Endothelial tubulogenesis assays using human umbilical vein endothelial cells (HUVECs) on Matrigel substrates [3]
- Extracellular Vesicle Characterization: Nanoparticle tracking analysis for size distribution and concentration measurements [11]
- microRNA Profiling: Next-generation sequencing of EV-associated and free-circulating microRNAs [11]
Technological Advances in Niche Characterization
Emerging computational approaches like NicheCompass enable quantitative characterization of cell niches in spatially resolved omics data by modeling cellular communication to learn interpretable cell embeddings that encode signaling events [12]. This graph deep-learning method identifies niches based on communication pathways and consistently outperforms alternative approaches that group cells based solely on histology or spatial gene expression without considering underlying cellular interactions [12].
Therapeutic Implications and Clinical Translation
The variation in paracrine factors across different MSC populations contributes to functionally distinct levels of angiogenic activity, suggesting that ASCs may be preferred over other MSC populations for augmenting therapeutic approaches dependent upon angiogenesis [3] [10]. The secretory profile of ASCs, rich in VEGF-D, IGF-1, and IL-8, makes them particularly suitable for applications requiring enhanced vascularization, such as wound healing and tissue engineering of volumetric constructs [3] [8].
For clinical translation, the updated International Society for Cellular Therapy (ISCT) recommendations emphasize that tissue origin significantly influences MSC surface marker profiles and functional properties, highlighting the importance of adopting more precise and source-dependent criteria for MSC characterization [8]. Furthermore, the development of cell-free therapies utilizing MSC secretomes offers a potentially safer and more effective alternative to whole-cell therapies, circumventing issues related to cell survival, engraftment, and potential malignant transformation [8].
The anatomical niche imposes distinct identities on resident MSC populations that manifest as unique secretory profiles with specific functional capabilities. ASCs demonstrate enhanced expression of key angiogenic factors including IGF-1, VEGF-D, and IL-8, correlating with superior pro-angiogenic activity in functional assays. BMSCs and dermal MSCs exhibit their own unique secretory signatures, suggesting preferential applications for different therapeutic contexts. Researchers should carefully consider these source-dependent variations when designing cell-based therapies or selecting MSC sources for specific regenerative applications. Future work should focus on standardizing isolation and characterization protocols to better elucidate the fundamental relationships between niche-specific identities and secretory profiles, potentially enabling engineering of optimized secretomes for targeted clinical applications.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for MSC Secretome Studies
| Reagent/Category | Specific Examples | Research Application |
|---|---|---|
| Digestive Enzymes | Type I collagenase, collagenase 1A [3] [11] | Tissue dissociation for cell isolation |
| Cell Culture Media | Dulbecco's Modified Eagle Medium (DMEM), αMEM [3] [11] | MSC expansion and maintenance |
| Serum Supplements | Fetal bovine serum (FBS), fetal calf serum [3] [11] | Cell growth and proliferation support |
| Characterization Antibodies | CD73, CD90, CD105, CD11b, CD14, CD19, CD45, HLA-DR [8] | MSC immunophenotyping by flow cytometry |
| Extracellular Vesicle Isolation | Differential centrifugation kits, size-exclusion chromatography [11] | Secretome fractionation and EV purification |
| Angiogenesis Assays | Matrigel, HUVECs, tubulogenesis scoring systems [3] | Functional validation of pro-angiogenic activity |
| Cytokine Detection | ELISA kits for VEGF, IGF-1, IL-8, angiogenin [3] | Quantification of secreted factors |
Niche Influence on MSC Secretory Profiles
Secretory Profile Analysis Workflow
The therapeutic application of Mesenchymal Stem Cells (MSCs) has undergone a significant paradigm shift. While initially valued for their differentiation potential, research now indicates that their primary mechanism of action lies in the secretion of bioactive factors, a concept known as the "paracrine hypothesis" [13]. These factors—including growth factors, cytokines, and chemokines—collectively termed the "secretome," modulate the immune system, promote cell survival, and stimulate angiogenesis and tissue repair [14] [15]. The composition of this secretome is not uniform; it is profoundly influenced by the MSC's tissue of origin. This guide provides a detailed, data-driven comparison of the expression levels of key growth factors—VEGF, HGF, FGF2, and IGF-1—across the most clinically relevant MSC sources: Adipose-Derived Stromal Cells (ASCs), Bone Marrow-MSCs (BM-MSCs), and Umbilical Cord Blood-MSCs (UCB-MSCs), providing researchers with a foundation for making informed, evidence-based decisions for their therapeutic strategies.
Quantitative Comparison of Key Growth Factor Expression
The therapeutic potential of MSCs is largely dictated by their paracrine "fingerprint." The following tables consolidate experimental data on the expression and secretion of critical growth factors from different MSC sources, highlighting their unique strengths.
Table 1: Growth Factor Expression Profile Across MSC Sources
| MSC Source | VEGF | HGF | FGF2 | IGF-1 | Key Supporting Evidence |
|---|---|---|---|---|---|
| Adipose-Derived (ASCs) | Moderate to High [16] [17] | Moderate [17] | Very High [16] | High [15] | ASCs expressed significantly higher FGF-2 levels compared to Stromal Vascular Fraction (SVF) cells [16]. |
| Bone Marrow (BM-MSCs) | Very High [17] | Moderate [13] | Moderate [13] | Moderate [18] | BM-MSCs secreted the highest level of VEGF among compared sources and showed strong tubulogenesis [17]. |
| Umbilical Cord Blood (UCB-MSCs) | Information Limited | Information Limited | Information Limited | Information Limited | UCB-MSCs are a component of the broader secretome studies, though direct quantitative comparisons for these specific factors in the provided literature are less emphasized [13] [15]. |
Table 2: Functional Angiogenic Potential of Different MSC Sources
| MSC Source | Proangiogenic Bioactivity | Key Angiogenic Factors Identified |
|---|---|---|
| ASCs | Promotes tissue growth and angiogenesis in vivo [19]. Secretome shows high tubulogenic efficiency [15]. | FGF-2, VEGF-A, VEGF-D, IGF-1, IL-8 [15] [16] [19] |
| BM-MSCs | Conditioned medium significantly promotes endothelial cell tube formation [17]. | VEGF, HGF, FGF2, IGF-1, EMMPRIN [13] [18] [15] |
| UCB-MSCs | Exhibits proangiogenic effects, though comparative potency may vary against other sources [15]. | VEGF, HGF, FGF2, IGF-1 (Specific expression levels relative to other sources require further direct comparison) [13] [15] |
Detailed Experimental Protocols for Factor Analysis
To ensure the reproducibility of data and facilitate future research, this section outlines standard experimental methodologies used to generate the comparative findings discussed in this guide.
Protocol for Quantitative Real-Time PCR (qRT-PCR)
Objective: To quantify and compare the mRNA expression levels of angiogenic genes (e.g., VEGF-A, HGF, FGF-2, IGF-1) across different MSC sources [16] [17].
- Cell Culture: Culture MSCs from different sources (ASC, BM-MSC, UCB-MSC) under identical standard conditions (e.g., DMEM/F12 with 10% FBS) to minimize environmental variability.
- RNA Extraction: At approximately 80% confluence, harvest cells and extract total RNA using a commercial kit (e.g., E.Z.N.A. Total RNA Kit I).
- cDNA Synthesis: Reverse transcribe equal amounts of RNA into complementary DNA (cDNA) using a reverse transcription kit (e.g., MLV RT Kit).
- qRT-PCR Amplification: Perform real-time PCR using a system (e.g., Applied Biosystem 7900/7300) with SYBR Green detection chemistry. Run each sample in triplicate for statistical robustness.
- Data Analysis: Calculate relative gene expression using the 2–ΔΔCT method, normalizing to a housekeeping gene (e.g., GAPDH). Compare fold-change differences between MSC types [17].
Protocol for Enzyme-Linked Immunosorbent Assay (ELISA)
Objective: To quantitatively measure the concentration of specific growth factor proteins (e.g., VEGF, HGF) secreted into the conditioned medium (CM) [17].
- Conditioned Medium Collection: Culture MSCs from different sources until sub-confluent. Replace growth medium with a serum-free basal medium (e.g., EBM-2). After 24-48 hours, collect the CM.
- Sample Preparation: Centrifuge the CM to remove cell debris and filter it through a 0.2 μm filter. Store aliquots at -80°C until analysis.
- ELISA Procedure: Use commercial, human-specific ELISA kits for each target protein. Follow the manufacturer's instructions to load samples and standards, incubate, wash, and develop the assay.
- Quantification: Measure the absorbance and determine the protein concentration in each sample by interpolating from the standard curve. Normalize data to cell number or total protein content [17].
Protocol for In Vitro Tube Formation Assay
Objective: To functionally assess the proangiogenic capacity of MSC-derived conditioned medium by measuring its ability to induce endothelial cell network formation [16] [17].
- Matrigel Preparation: Thaw Growth Factor Reduced Matrigel on ice. Coat wells of a 96-well plate with a thin, even layer of Matrigel and incubate at 37°C for 30-60 minutes to polymerize.
- Endothelial Cell Seeding: Harvest human umbilical vein endothelial cells (HUVECs) and resuspend them in the conditioned media collected from the different MSC types. Seed the HUVECs onto the polymerized Matrigel.
- Incubation and Imaging: Incubate the plate at 37°C for 4-18 hours. Under a microscope, endothelial cells will form capillary-like tube structures.
- Quantification: Capture images from multiple random fields per well. Use image analysis software to quantify key parameters such as total tube length, number of branching points, and number of closed meshes.
Signaling Pathways and Experimental Workflows
The following diagrams, generated using Graphviz DOT language, illustrate the core signaling pathways influenced by MSC-derived factors and a standard experimental workflow for comparative secretome analysis.
Key Angiogenic Signaling Pathways
This diagram visualizes the synergistic interplay between the key growth factors secreted by MSCs and their signaling pathways in endothelial cells, leading to angiogenesis.
Experimental Workflow for Secretome Comparison
This flowchart outlines a standardized experimental methodology for comparing the proangiogenic potential of different MSC sources, from cell culture to functional analysis.
The Scientist's Toolkit: Essential Research Reagents
This section details key reagents and materials required to perform the experiments described in this comparison guide.
Table 3: Essential Research Reagents for MSC Secretome Analysis
| Reagent / Material | Function / Application | Example from Literature |
|---|---|---|
| DMEM/F12 or DMEM Basal Medium | Standard culture medium for expanding and maintaining MSCs from various sources. | Used as the base complete culture medium for BMSCs, AMSCs, UMSCs, and PMSCs [17]. |
| Endothelial Basal Medium (EBM-2) | Serum-free medium used for collecting conditioned medium (CM) to avoid interference from serum-derived factors. | Used for conditioning MSC secretome prior to collection for functional assays [17]. |
| Fetal Bovine Serum (FBS) | Essential supplement for cell growth, providing hormones, growth factors, and other nutrients. | Typically used at 10% concentration in standard MSC culture medium [17]. |
| Growth Factor-Reduced Matrigel | A basement membrane matrix extracted from mice. Used for in vitro tube formation assays to simulate a vascularization environment. | Used to coat plates for the endothelial tube formation assay with HUVECs and MSC-CM [16] [17]. |
| Human Umbilical Vein Endothelial Cells (HUVECs) | Primary endothelial cells used as target cells to functionally test the proangiogenic effects of MSC-CM. | Cultured in EGM-2MV and used at passages 3-5 for tube formation and migration assays [15] [17]. |
| qRT-PCR Kits & Primers | For quantifying mRNA expression levels of specific growth factors and cytokines in different MSCs. | Used with specific primers to analyze gene expression of VEGF, FGF-2, HGF, etc. [16] [17]. |
| Human Cytokine ELISA Kits | For quantifying the concentration of specific secreted proteins (e.g., VEGF, HGF) in the MSC-CM. | Used to confirm protein-level secretion of factors like VEGF and HGF from different MSCs [17]. |
| Specific Growth Factors (VEGF, bFGF, IGF-1) | Used for positive controls in functional assays or for preconditioning MSCs to enhance their secretome. | Used at 20-50 ng/mL concentrations for in vitro experiments and MSC preconditioning [18] [17]. |
The therapeutic efficacy of mesenchymal stem cells (MSCs) is increasingly attributed to their paracrine activity rather than their direct differentiation potential. These cells secrete a complex array of immunomodulatory factors—including both anti-inflammatory and pro-inflammatory mediators—that collectively shape immune responses in damaged or diseased tissues. This paracrine signature varies significantly across MSC sources, creating distinct immunomodulatory profiles that influence their therapeutic application. Understanding these variations is crucial for researchers and drug development professionals selecting optimal cell sources for specific disease contexts. This guide provides a systematic comparison of the immunomodulatory factor secretion from three prominent MSC sources: Adipose-Derived Stem Cells (ASCs), Bone Marrow-Mesenchymal Stem Cells (BM-MSCs), and Umbilical Cord Blood-Mesenchymal Stem Cells (UCB-MSCs).
The immunomodulatory potency of MSCs is not uniform; it is intrinsically linked to their tissue of origin. The following analysis compares the defining secretory characteristics and functional outputs of ASCs, BM-MSCs, and UCB-MSCs.
Table 1: Key Immunomodulatory Characteristics by MSC Source
| Feature | ASCs (Adipose-Derived) | BM-MSCs (Bone Marrow) | UCB-MSCs (Umbilical Cord Blood) |
|---|---|---|---|
| Key Anti-Inflammatory Factors | IL-10, PGE2, TGF-β [20] | IL-10, PGE2, TGF-β, TSG-6, HGF [20] | Not Specified in Search Results |
| Key Pro-Inflammatory Associations | Distinct cytokine profile during differentiation [21] | Higher secretion of IL-6, IL-8, TNF-α compared to other sources [22] | Not Specified in Search Results |
| Mechanism of Action | Paracrine secretion of trophic factors and exosomes [23] | Paracrine immunomodulation; exosomes inhibit inflammation via specific signaling axes [20] | Robust paracrine signals, growth factors, cytokines, and EVs [24] |
| Primary Immunomodulatory Effects | Immunomodulation and tissue repair [25] | Inhibits pro-inflammatory immune cells; promotes Treg expansion; polarizes macrophages to M2 phenotype [25] [20] | Tenogenic differentiation; immunomodulation [24] |
| Therapeutic Context | Neurodegenerative, cardiovascular, and autoimmune diseases [22] | Osteoarthritis, Rheumatoid Arthritis, tissue repair [25] [20] | Tendon repair (e.g., rotator cuff), regenerative applications [24] [26] |
Quantitative Analysis of Secreted Mediators
The qualitative differences in secretory profiles are underpinned by quantifiable variations in the expression of specific factors. The tables below summarize key experimental data from comparative studies.
Table 2: Anti-Inflammatory Factor Expression
| Factor | Function | ASCs | BM-MSCs | UCB-MSCs | Experimental Context |
|---|---|---|---|---|---|
| TSG-6 | Inhibits NF-κB pathway; reduces inflammatory factor release [20] | Not Reported | Significant Upregulation | Not Reported | BM-MSC transplantation in OA models [20] |
| IL-10 | Anti-inflammatory cytokine; inhibits p38 MAPK pathway [20] | Secreted [20] | Secreted [20] | Not Reported | In vitro paracrine profiling [20] |
| PGE2 | Inhibits NF-κB nuclear translocation [20] | Secreted [20] | Secreted [20] | Not Reported | In vitro paracrine profiling [20] |
| IDO | Immunomodulatory enzyme [25] | Not Reported | Induces M2 macrophage polarization [25] | Not Reported | In vitro co-culture studies [25] |
Table 3: Pro-Inflammatory & Lineage-Specific Factor Expression
| Factor / Marker | Function / Association | ASCs | BM-MSCs | UCB-MSCs | Experimental Context |
|---|---|---|---|---|---|
| Senescence Markers (p16, p21) | Cellular aging [22] | Lower | Higher | Lowest | Gene expression analysis in early passages [22] |
| Osteogenic Markers | Bone formation tendency [27] | Lower | Higher | Lower | Gene expression (e.g., ALP, OCN) [27] |
| Tenogenic Genes (SCX, MKX) | Tendon lineage commitment [24] [26] | Not Reported | Lower | Highest (3.12- & 5.92-fold upregulation vs BM-MSC) [24] [26] | In vitro tenogenic differentiation in T-3D constructs [24] [26] |
| Pro-inflammatory Secretion | Inflammatory milieu [22] | Lower | Higher (IL-1α, IL-6, IL-8, TNF-α) [22] | Not Reported | In vitro cytokine profiling [22] |
Detailed Experimental Protocols for Key Findings
To ensure the reproducibility of critical comparative studies, this section outlines the essential methodological details for key experiments cited in this guide.
Protocol 1: In Vitro Tenogenic Differentiation Comparison
This protocol is derived from a head-to-head comparison of BM-, UCB-, and UC-MSCs for tendon regeneration [24] [26].
- Cell Culture & Differentiation: MSCs from all three sources were cultured in tensioned three-dimensional (T-3D) constructs to drive tenogenic differentiation.
- Gene Expression Analysis: After a defined period in T-3D culture, cells were analyzed using quantitative reverse transcription polymerase chain reaction (qRT-PCR). Key tenogenic markers assessed included:
- Scleraxis (SCX)
- Mohawk (MKX)
- Type I Collagen (COL1)
- Tenascin-C (TNC)
- Histological Analysis: The formation of a tendon-like extracellular matrix was evaluated histologically, with a specific focus on organized, parallel collagen-I fibers.
- Key Outcome: UC-MSCs consistently showed the strongest upregulation of tenogenic genes and produced the most organized collagen-I matrix compared to BM-MSCs and UCB-MSCs [24] [26].
Protocol 2: Cytokine Profiling During ASC Differentiation
This protocol outlines the methodology for mapping cytokine expression during ASC differentiation, revealing a distinct inflammatory profile associated with lineage commitment [21].
- Cell Culture & Differentiation: Human ASCs were isolated from subcutaneous adipose tissue and cultured in either osteogenic differentiation medium (ODM) or adipogenic differentiation medium (ADM).
- Time Points: Cells were collected for analysis on days 7, 14, and 21 of differentiation.
- Staining and Quantification:
- Osteogenic Cultures: Fixed and stained with Alizarin Red to visualize calcium deposition. Stain was quantified after extraction with cetylpyridinium chloride (CPC).
- Adipogenic Cultures: Fixed and stained with Oil Red O to visualize neutral lipids. Stain was quantified after extraction with isopropanol.
- Gene Expression Analysis: Total RNA was extracted using an RNeasy Mini Kit. After DNase I digestion and cDNA synthesis, the expression of a panel of pro- and anti-inflammatory cytokines was assessed using quantitative RT-PCR (qRT-PCR) [21].
Protocol 3: Assessing BM-MSC Paracrine Effects in Osteoarthritis
This protocol describes the methodology used to investigate the paracrine-mediated immunomodulation of BM-MSCs in an osteoarthritis context [20].
- In Vivo Model: BM-MSCs were transplanted into animal models of osteoarthritis (OA).
- Histological & Molecular Analysis:
- Joint tissues were analyzed for histological changes in cartilage degradation and inflammation.
- Levels of key pro-inflammatory factors (TNF-α, IL-1β, IL-6) and components of the NF-κB pathway (p50, p65) were measured in the OA joint tissue, typically via ELISA or immunohistochemistry.
- Mechanistic Investigation: The role of specific paracrine factors like PGE2 and TSG-6 was probed, confirming their action in inhibiting NF-κB nuclear translocation and its subsequent transcriptional activity on inflammatory genes [20].
Signaling Pathways in MSC-Mediated Immunomodulation
The following diagrams, generated using Graphviz DOT language, illustrate the key signaling pathways by which paracrine factors from MSCs, particularly BM-MSCs, exert their immunomodulatory effects.
BM-MSC Paracrine Action in Osteoarthritis
Experimental Workflow for Tenogenic Comparison
The Scientist's Toolkit: Essential Research Reagents
The following table details key reagents and materials required to perform the types of comparative immunomodulatory studies described in this guide.
Table 4: Essential Research Reagents and Kits
| Reagent / Kit | Function / Application | Specific Example / Target |
|---|---|---|
| qRT-PCR Assays | Quantification of gene expression for cytokines, lineage markers, and transcription factors. | Tenogenic genes (SCX, MKX, COL1, TNC); Senescence markers (p16, p21); Osteogenic markers (ALP, OCN) [24] [21] [27]. |
| Cell Differentiation Media | Directing stem cell differentiation into specific lineages for subsequent secretory profiling. | Osteogenic Differentiation Medium (ODM); Adipogenic Differentiation Medium (ADM); Tenogenic conditions (T-3D culture) [24] [21]. |
| Flow Cytometry Antibodies | Characterization of MSC surface markers to confirm cell identity and purity prior to experiments. | CD73, CD90, CD105 (positive); CD14, CD34, CD45 (negative) [28] [22]. |
| Cytokine Detection Kits | Measuring concentrations of secreted immunomodulatory factors in cell culture supernatants. | ELISA kits for PGE2, TSG-6, IL-10, TNF-α, IL-1β, IL-6 [20]. |
| Histological Stains | Visualizing and quantifying differentiation outcomes and matrix formation. | Alizarin Red (calcium/mineralization); Oil Red O (lipid droplets) [21]. |
| RNA Extraction Kit | High-quality RNA isolation for downstream gene expression analysis. | RNeasy Mini Kit [21]. |
This guide provides an objective comparison of transcriptional (mRNA expression) and translational (protein secretion) analysis methodologies within the context of mesenchymal stem cell (MSC) research. Focusing on comparative paracrine factor expression in adipose-derived stem cells (ASCs), bone marrow-derived stem cells (BMSCs), and umbilical cord blood-derived MSCs (UCB-MSCs), we present structured experimental data, detailed protocols, and analytical frameworks to inform research and drug development efforts. The data underscore that mRNA abundance alone is an insufficient predictor of protein secretion levels, necessitating integrated multi-omic approaches for accurate functional characterization of MSC paracrine activities.
Gene expression is a two-step process: transcription, where DNA is copied into messenger RNA (mRNA), and translation, where mRNA is decoded by ribosomes to synthesize proteins [29]. In molecular research, transcriptional analysis quantifies mRNA levels, providing insights into the initial, regulatory stage of gene expression. Translational analysis, conversely, directly assesses the functional output—proteins—including their synthesis rates, modifications, and secretion [30].
For MSCs, whose therapeutic potential is heavily reliant on secreted paracrine factors [3] [31], distinguishing between mRNA expression and protein secretion is critical. A high mRNA level does not guarantee commensurate protein production due to extensive post-transcriptional regulation and translational control [32] [33]. This guide systematically compares these two analytical dimensions, leveraging data from comparative studies of ASCs, BMSCs, and other MSCs to highlight the technical and biological considerations essential for robust experimental design and data interpretation.
Comparative Paracrine Factor Expression in MSCs
The paracrine activity of MSCs, vital for tissue engineering and regenerative medicine, varies significantly depending on the tissue source. The following tables summarize comparative data on mRNA expression and protein secretion of key paracrine factors from ASCs, BMSCs, and Dermal Sheath Cells (DSCs).
Table 1: Comparative mRNA Expression of Paracrine Factors in Human MSCs
| Paracrine Factor | Function | ASCs | BMSCs | DSCs |
|---|---|---|---|---|
| IGF-1 | Promotes cell growth and survival | Higher | Lower | Lower [3] |
| VEGF-D | Lymphangiogenesis and angiogenesis | Higher | Lower | Lower [3] |
| IL-8 | Chemotactic and angiogenic factor | Higher | Lower | Lower [3] |
| VEGF-A | Key regulator of angiogenesis | Comparable | Comparable | Comparable [3] |
| Angiogenin | Induces blood vessel formation | Comparable | Comparable | Comparable [3] |
| bFGF | Broad mitogenic activity | Comparable | Comparable | Comparable [3] |
| NGF | Supports neuron growth and survival | Comparable | Comparable | Comparable [3] |
Table 2: Comparative Protein Secretion and Functional Output
| Analysis Type | Target | ASCs | BMSCs | DSCs/DPCs |
|---|---|---|---|---|
| Protein Secretion | VEGF-A | Comparable | Comparable | Comparable [3] |
| Protein Secretion | Angiogenin | Comparable | Comparable | Comparable [3] |
| Protein Secretion | Leptin | Lower | Lower | Higher [3] |
| Functional Assay | In vitro Endothelial Tubulogenesis | Increased Efficiency | Intermediate | Reduced Efficiency [3] |
The data reveals a complex relationship between transcriptional and translational outputs. For instance, while ASCs express significantly higher mRNA levels for IGF-1, VEGF-D, and IL-8 [3], the protein secretion of broadly expressed factors like VEGF-A and angiogenin is comparable across MSC types. This discrepancy highlights that transcript abundance does not always linearly correlate with secreted protein levels. Furthermore, DSCs and Dermal Papilla Cells (DPCs) secrete significantly higher levels of leptin protein, a factor not necessarily predicted by mRNA data alone [3].
Functionally, the distinct paracrine profiles translate into different biological activities. Conditioned media from ASCs (ASC-CM) demonstrated superior ability to promote endothelial tubulogenesis compared to that from DPCs (DPC-CM) [3]. Neutralization experiments identified VEGF-A and VEGF-D as major contributors to this enhanced angiogenic capacity of ASCs [3].
Methodologies for Transcriptional and Translational Analysis
Transcriptional Analysis via RNA Sequencing
Workflow Overview: Cell culture → RNA extraction → library preparation (e.g., stranded total RNA with Ribo-Zero) → High-throughput sequencing (e.g., Illumina NovaSeq) → Bioinformatic analysis [32] [34].
Detailed Protocol:
- Cell Culture and Harvesting: Grow MSCs from different sources (ASCs, BMSCs, UCB-MSCs) under identical, standardized culture conditions to minimize artifactual variation. Upon reaching ~80% confluency, wash cells with PBS and lyse directly in a denaturing guanidinium thiocyanate-containing buffer to immediately stabilize RNA [3] [34].
- RNA Extraction: Isolate total RNA using spin column-based kits or phenol-chloroform extraction. Assess RNA integrity and purity using spectrophotometry (A260/A280 ratio) and an instrument such as a Bioanalyzer (RNA Integrity Number, RIN > 9.0 is optimal) [34].
- Library Preparation and Sequencing: For mRNA-seq, use kits that selectively enrich for polyadenylated RNA or deplete ribosomal RNA. Convert RNA into a strand-specific cDNA library, add platform-specific adapters, and amplify. Sequence on a platform such as an Illumina NovaSeq 6000 to a sufficient depth (e.g., 25-50 million paired-end reads per sample) [32] [34].
- Data Analysis: Process raw sequencing data through a bioinformatic pipeline:
- Quality Control: Use FastQC to assess read quality.
- Alignment: Map reads to a reference genome (e.g., GRCh38 for human) using splice-aware aligners like STAR.
- Quantification: Count reads aligning to genes using featureCounts or HTSeq.
- Differential Expression: Identify significantly differentially expressed genes between MSC populations using packages like DESeq2, with a false discovery rate (FDR) adjusted p-value < 0.05 and |log2(fold change)| > 1 as common thresholds [34].
Translational Analysis via Proteomic and Functional Assays
Workflow Overview: Cell culture → Conditioned media collection → Protein quantification (ELISA/MS) → Functional validation (e.g., tubulogenesis assay).
Detailed Protocol:
- Conditioned Media (CM) Collection: Culture ASCs, BMSCs, and UCB-MSCs to 70-80% confluency. Replace growth medium with a serum-free basal medium to avoid serum protein interference. After 24-48 hours, collect the CM and centrifuge to remove cells and debris. Concentrate CM using centrifugal filters (e.g., 3 kDa cutoff) and store at -80°C [3].
- Protein Quantification - ELISA:
- Coat a 96-well plate with a capture antibody specific to the target protein (e.g., VEGF-A).
- Block nonspecific binding sites with a protein blocker (e.g., BSA).
- Add concentrated CM and a series of diluted protein standards to the plate.
- After incubation and washing, add a detection antibody, followed by an enzyme-conjugated secondary antibody.
- Develop the assay with a substrate and measure the absorbance. Interpolate protein concentrations in the CM from the standard curve [3].
- Protein Quantification - Mass Spectrometry:
- Precipitate proteins from CM and reconstitute in a denaturing buffer.
- Reduce disulfide bonds with TCEP and alkylate with iodoacetamide.
- Digest proteins into peptides with trypsin.
- Desalt peptides and analyze by LC-MS/MS (Liquid Chromatography with Tandem Mass Spectrometry).
- Identify and quantify proteins by searching fragment spectra against a protein database (e.g., Swiss-Prot) [30].
- Functional Validation - Endothelial Tubulogenesis Assay:
- Thaw Matrigel on ice and coat the wells of a 96-well plate. Polymerize at 37°C for 30 minutes.
- Seed human umbilical vein endothelial cells (HUVECs) onto the Matrigel surface in the different MSC-conditioned media.
- Incubate for 6-16 hours and capture images of the formed tubular networks under a microscope.
- Quantify the total tube length, number of branches, or number of meshes using image analysis software (e.g., ImageJ with Angiogenesis Analyzer plugin) [3].
The Scientist's Toolkit: Essential Research Reagents
Successful transcriptional and translational analysis requires a suite of reliable reagents and tools. The following table details essential solutions for the featured experiments.
Table 3: Key Research Reagent Solutions for MSC Paracrine Analysis
| Reagent / Solution | Function / Application | Example Use Case |
|---|---|---|
| DMEM-low glucose Medium | Basal cell culture medium for MSC expansion | Standardized growth of ASCs, BMSCs, and UCB-MSCs [3]. |
| Fetal Calf Serum (FCS) | Provides essential growth factors and nutrients for cell growth | Standard serum supplement for MSC culture [3]. |
| Type I Collagenase | Enzymatic digestion of tissues for cell isolation | Liberation of ASCs from adipose tissue [3]. |
| Ribo-Zero Plus Kit | Depletion of ribosomal RNA during RNA-seq library prep | Ensures high-quality sequencing data focused on mRNA and lncRNAs [34]. |
| Differential Expression Analysis Software (DESeq2) | Identifies statistically significant changes in gene expression | Comparing mRNA levels between ASCs and BMSCs [34]. |
| VEGF-A & VEGF-D Neutralizing Antibodies | Block specific protein activity in functional assays | Validating the contribution of specific factors to angiogenic paracrine effects [3]. |
| Matrigel | Basement membrane extract for 3D cell culture | Substrate for in vitro endothelial tubulogenesis assays [3]. |
Signaling Pathways in Paracrine Communication
The functional outcome of MSC-secreted factors is mediated through complex signaling pathways that can be inferred from gene expression data. A prime example is the interferon signaling cascade, which exemplifies how extracellular communication leads to gene induction.
This pathway illustrates the multi-step process from extracellular signal to functional protein output. Inferred from ligand-receptor pair expression [35], this cascade shows how a cytokine like IFN-β or a growth factor like VEGF secreted by one cell is received by another, triggering a signaling pathway that culminates in the transcriptional activation of specific genes (e.g., DDX58) and their subsequent translation into effector proteins [36]. This framework is directly applicable to understanding how paracrine factors from ASCs or BMSCs, such as VEGF-A, initiate signaling in endothelial cells to promote processes like tubulogenesis [3].
From Bench to Bedside: Methodologies for Characterization and Therapeutic Applications
Standardized Isolation and Culture Protocols for Cross-Study Comparisons
The field of regenerative medicine extensively investigates Mesenchymal Stromal Cells (MSCs) for their multipotent differentiation capacity, immunomodulatory properties, and paracrine activity. However, inconsistent methodological approaches across studies present a significant challenge for comparative analysis, particularly in evaluating the therapeutic potential of MSCs from different tissue sources. Researchers isolating MSCs from adipose tissue (ASCs), bone marrow (BMSCs), or umbilical cord blood (UCB-MSCs) employ varied protocols for isolation, expansion, and characterization, leading to data heterogeneity and challenges in reproducibility [37] [38].
This guide objectively compares standard operating procedures for MSC isolation and culture, focusing on the context of comparative paracrine factor expression. We present structured experimental data and detailed methodologies to facilitate the adoption of more uniform protocols, thereby enhancing cross-study comparability and accelerating the translation of MSC-based therapies from bench to bedside.
Comparative Analysis of MSC Isolation Techniques
The initial isolation of MSCs from tissue sources is a critical step that significantly impacts cell yield, purity, and subsequent functionality. The two primary approaches are enzymatic digestion and explant culture, each with distinct advantages and limitations [39].
Table 1: Comparison of Primary MSC Isolation Techniques
| Technique | Principle | Speed to Confluence | Cell Viability & Integrity | Cost & Complexity | Common Tissue Sources |
|---|---|---|---|---|---|
| Enzymatic Digestion | Uses enzymes (e.g., collagenase) to break down tissue and release cells [37] [3]. | ~7 days [39] | Risk of over-digestion; may compromise viability [39]. | Higher (cost of enzymes, growth factors) [39]. | Adipose tissue [3], Bone Marrow (from MNCs) [40], Umbilical Cord [37]. |
| Explant Culture | Tissue fragments are plated, allowing cells to migrate out spontaneously [37] [39]. | ~15 days [39] | Superior preservation of cell integrity and genetic stability [39]. | Lower (avoids enzymes and supplements) [39]. | Umbilical Cord [37], Dental Pulp [37], Amniotic Membrane [38]. |
For bone marrow aspirates, a common starting point for BMSCs, density gradient centrifugation with media like Ficoll-Paque PLUS is standard for isolating mononuclear cells (MNCs), which include the MSC population [40]. This process can be performed either manually or using automated systems like the Sepax S-100. A recent comparative study found that while the automated Sepax system demonstrated slightly higher MNC yields, no significant differences were observed in the subsequent number of MSC colony-forming units (CFUs) or their differentiation potential [40]. This indicates that for research settings, the manual method is a cost-effective and reliable option.
Essential Research Reagent Solutions
Successful isolation and culture of MSCs depend on a standardized set of reagents and materials. The following table outlines key components of the MSC research toolkit.
Table 2: Essential Research Reagent Toolkit for MSC Isolation and Culture
| Reagent/Material | Function/Application | Examples & Notes |
|---|---|---|
| Lymphocyte Separation Medium | Density gradient centrifugation to isolate MNCs from bone marrow or UCB [40] [41]. | Ficoll-Paque PLUS (ρ=1.077 g/l) [41]. |
| Digestive Enzymes | Breaks down extracellular matrix in tissue sources for enzymatic isolation. | Type I Collagenase [3]. |
| Basal Culture Media | Provides essential nutrients for MSC growth and expansion. | α-MEM, DMEM (low glucose), DMEM/F12 [42] [41]. α-MEM is frequently indicated as suitable [42]. |
| Serum/Sup.lements | Provides critical growth factors and adhesion proteins. | Fetal Bovine Serum (FBS), typically 10-20% [40] [43]. Human platelet lysate is a xeno-free alternative [42]. |
| Cell Culture Flasks | Surface for plastic-adherent MSC growth. | Standard tissue culture-treated polystyrene flasks [40] [41]. |
| Dissociation Agent | Detaches adherent MSCs for sub-culturing (passaging). | Trypsin/EDTA solution [40]. |
Impact of Culture Conditions on MSC Phenotype and Function
The culture environment post-isolation is a major source of variation. Parameters such as basal medium formulation, glucose concentration, and oxygen tension can profoundly influence MSC physiology and paracrine output.
Basal Media Selection: Studies comparing basal media have shown that α-MEM often generates high yields of both bone marrow and adipose-derived MSCs [42]. However, the "optimal" medium can be cell-source dependent; one study reported that a specific DMEM/HG formulation yielded higher proliferation rates for BM-MSCs at early passages [42]. Researchers must balance proliferation with the preservation of intrinsic MSC properties.
Glucose Concentration: Media formulations contain varying glucose levels, from low (1,000 mg/L) to high (4,500-10,000 mg/L). High glucose conditions (e.g., 5,000 mg/L) have been shown to suppress proliferation, reduce colony-forming ability, induce cellular senescence, and alter morphology in BM-MSCs compared to low glucose conditions [42]. This is a critical consideration for studies modeling metabolic disease or seeking to maintain a robust MSC population.
Physiologic Culture Conditions: Standard normoxic (20-21% O₂) culture does not mimic the physiological hypoxic niche (1-5% O₂) where MSCs reside in vivo. Culturing MSCs under physiologic hypoxia has been demonstrated to better preserve their stemness, improve proliferation, and enhance angiogenic potential [42]. Moving towards these more physiologic culture approaches is essential for generating clinically relevant data.
Experimental Protocols for Paracrine Factor Analysis
A key application of standardized protocols is the reliable comparison of paracrine factor expression across different MSC types. Below is a detailed methodology for quantifying and comparing the secretory profile of ASCs, BMSCs, and UCB-MSCs.
Protocol: Comparative Analysis of Paracrine Factor Expression
This protocol is adapted from established methods for evaluating the angiogenic paracrine activity of different MSC populations [3].
MSC Culture and Conditioned Media Collection
- Cell Culture: Culture ASCs, BMSCs, and UCB-MSCs under identical, standardized conditions (e.g., in α-MEM with 10% FBS, 37°C, 5% CO₂). Use cells at equivalent early passages (e.g., P3-P6) [3].
- Serum Starvation: When cells reach 70-80% confluence, wash with PBS and switch to a basal medium without serum or growth factors.
- Collection: Collect conditioned media (CM) after 24-48 hours. Centrifuge CM at 2,000 × g for 10 minutes to remove cell debris. Aliquot and store the supernatant (CM) at -80°C.
mRNA Expression Analysis (qRT-PCR)
- RNA Extraction: Isolate total RNA from each MSC type using a standard kit (e.g., TRIzol).
- cDNA Synthesis: Synthesize cDNA using a reverse transcription kit.
- Quantitative PCR: Perform qPCR using primers for target paracrine factors (e.g., VEGF-A, VEGF-D, IGF-1, IL-8, angiogenin, bFGF) and housekeeping genes (e.g., GAPDH). Calculate relative gene expression using the 2^(-ΔΔCt) method.
Protein Level Analysis (ELISA)
- Quantification: Use commercial Enzyme-Linked Immunosorbent Assay (ELISA) kits to quantify the concentration of specific proteins (e.g., VEGF-A, angiogenin, leptin) in the CM from each MSC type [3].
- Normalization: Normalize protein concentrations to the total cell number or total cellular protein of the source culture.
Functional Tubulogenesis Assay
- Endothelial Cell Culture: Seed human umbilical vein endothelial cells (HUVECs) on a basement membrane matrix (e.g., Matrigel).
- Treatment: Incubate HUVECs with CM from ASCs, BMSCs, or UCB-MSCs. Use basal medium as a negative control.
- Analysis: After 6-18 hours, image the formed tubular structures. Quantify parameters such as total tube length, number of branches, and number of meshes per field.
- Neutralization Studies: To identify key functional factors, repeat the assay with CM pre-incubated with neutralizing antibodies against specific factors (e.g., anti-VEGF-A, anti-VEGF-D) [3].
Representative Data and Comparative Findings
Table 3: Exemplar Paracrine Factor Expression Data Across MSC Types
| Analysis Method | Target | ASCs | BMSCs | UCB-MSCs | Notes & Experimental Context |
|---|---|---|---|---|---|
| mRNA Expression | IGF-1, VEGF-D, IL-8 | Higher [3] | Lower [3] | Not Specified | Comparative analysis of ASCs vs. BMSCs and dermal cells [3]. |
| mRNA Expression | VEGF-A, Angiogenin, bFGF | Comparable [3] | Comparable [3] | Not Specified | No significant difference between ASCs and BMSCs observed [3]. |
| Protein Secretion (ELISA) | VEGF-A, Angiogenin | Comparable [3] | Comparable [3] | Not Specified | Secreted levels correlated with mRNA findings [3]. |
| Protein Secretion (ELISA) | Leptin | Lower [3] | Lower [3] | Not Specified | Dermal-derived MSCs produced significantly higher leptin [3]. |
| Functional Assay | Endothelial Tubulogenesis | Increased tubulogenesis [3] | Intermediate/Lower activity [3] | Not Specified | ASC-CM promoted greater tube formation than other MSC types; mediated by VEGF-A and VEGF-D [3]. |
The data in Table 3, derived from a comparative study, highlights that ASCs may possess a superior angiogenic paracrine profile compared to BMSCs, primarily driven by higher expression of specific factors like IGF-1, VEGF-D, and IL-8, and confirmed by functional tubulogenesis assays [3]. This underscores the importance of source selection for therapies dependent on angiogenesis.
Visualizing the Experimental Workflow
The following diagram illustrates the logical workflow for the comparative isolation, culture, and paracrine analysis of MSCs from different sources, as described in the protocols above.
The pursuit of reliable cross-study comparisons in MSC research hinges on the adoption of standardized, well-documented protocols for isolation and culture. Evidence indicates that the biological source of MSCs (ASC, BMSC, UCB-MSC) inherently influences their paracrine signature, a effect that can only be accurately discerned against a backdrop of methodological consistency. By implementing the detailed protocols, reagent standards, and comparative frameworks outlined in this guide, researchers can significantly reduce technical variability. This will pave the way for more robust, reproducible, and clinically relevant insights into the therapeutic potential of different MSC populations, ultimately advancing the field of regenerative medicine.
The therapeutic potential of mesenchymal stem cells (MSCs) is largely attributed to their secretome—the complex mixture of proteins, extracellular vesicles, and other factors they secrete [14]. This paracrine activity enables MSCs to modulate immune responses, promote tissue repair, and support regeneration without requiring long-term engraftment [14] [44]. However, the composition of the secretome varies significantly between MSC sources, creating a critical need for rigorous analytical comparison. For researchers investigating the comparative paracrine factor expression in adipose-derived stem cells (ASCs) versus bone marrow-derived MSCs (BMSCs) versus umbilical cord blood-derived MSCs (UCB-MSCs), selecting appropriate analytical techniques is paramount. This guide objectively compares the performance of three cornerstone technologies—proteomics, ELISA, and RNA analysis—for secretome characterization, providing experimental data and methodologies to inform study design in preclinical and therapeutic development contexts.
Core Analytical Techniques: Principles and Applications
Proteomics by Mass Spectrometry
Principles: Mass spectrometry (MS)-based proteomics enables the unbiased identification and quantification of hundreds to thousands of proteins in a secretome sample simultaneously. The most common approach uses liquid chromatography-tandem mass spectrometry (LC-MS/MS), where peptides from digested secretome proteins are separated by liquid chromatography and then ionized and fragmented in the mass spectrometer to generate identification data [45].
Key Applications in MSC Research:
- Discovery Profiling: Identifying the full spectrum of proteins secreted by different MSC types under varying conditions (e.g., resting vs. licensed states) [46].
- Comparative Analysis: Detecting quantitative differences in secretome composition between ASCs, BMSCs, and UCB-MSCs.
- Biomarker Identification: Finding specific secretory signatures indicative of MSC potency or source.
Experimental Protocol for Secretome Proteomics:
- Sample Preparation: Isolate MSCs from adipose tissue, bone marrow, or umbilical cord. Culture in serum-free media for 24-48 hours to collect conditioned media (CM). Centrifuge and filter CM to remove cells and debris [45].
- Protein Processing: Concentrate CM proteins using ultrafiltration. Digest proteins into peptides using trypsin.
- LC-MS/MS Analysis: Separate peptides by liquid chromatography. Analyze eluted peptides using a tandem mass spectrometer.
- Data Analysis: Identify proteins by searching fragmentation spectra against protein databases. Perform quantitative analysis using label-free or isotopic labeling methods [45].
Enzyme-Linked Immunosorbent Assay (ELISA)
Principles: ELISA is a targeted, antibody-based technique that detects and quantifies specific, known proteins with high sensitivity and specificity. In the common sandwich ELISA format, a capture antibody immobilized on a plate binds the target protein from the secretome sample, which is then detected using a second, enzyme-linked antibody, generating a measurable signal [47].
Key Applications in MSC Research:
- Targeted Quantification: Precisely measuring specific, clinically relevant factors (e.g., IDO, VEGF, IL-6) in MSC-CM [46].
- Validation: Verifying discoveries from proteomic screens.
- Potency Assay Development: Creating robust, quantitative assays for clinical lot release.
Experimental Protocol for Sandwich ELISA:
- Coating: Adsorb a capture antibody specific to the target protein onto a polystyrene microplate.
- Blocking: Add an irrelevant protein (e.g., BSA) to cover any remaining protein-binding sites.
- Sample Incubation: Add MSC-conditioned media or standards to the wells, allowing the target antigen to bind the capture antibody.
- Detection Antibody Incubation: Add an enzyme-conjugated detection antibody that binds a different epitope on the target protein.
- Signal Development: Add an enzyme substrate to produce a colorimetric, fluorescent, or chemiluminescent signal.
- Quantification: Measure the signal intensity and interpolate concentrations from a standard curve [47].
RNA Expression Analysis
Principles: RNA analysis techniques, such as microarrays and quantitative real-time PCR (qRT-PCR), measure the mRNA transcript levels of genes, providing an indirect assessment of the secretome's potential.
Key Applications in MSC Research:
- High-Throughput Screening: Using microarrays to assess the expression of thousands of genes encoding secreted factors [48].
- Validation of Specific Transcripts: Using qRT-PCR to accurately measure the expression levels of a limited number of genes of interest.
- Correlation Studies: Investigating the relationship between mRNA levels and actual protein secretion.
Experimental Protocol for Microarray-Based RNA Analysis:
- RNA Extraction: Isolate total RNA from ASCs, BMSCs, or UCB-MSCs using a method that preserves RNA quality.
- Labeling and Hybridization: Convert RNA to cDNA, then to labeled cRNA, and hybridize to a gene chip microarray (e.g., Affymetrix).
- Scanning and Data Acquisition: Scan the microarray to quantify the intensity of hybridization for each gene.
- Bioinformatic Analysis: Normalize data and perform statistical analysis to identify differentially expressed genes [48].
Comparative Performance Analysis
Technical Capabilities and Limitations
Table 1: Comparison of Key Technical Aspects of Secretome Profiling Techniques
| Feature | Proteomics (LC-MS/MS) | ELISA | RNA Analysis (qRT-PCR/Microarray) |
|---|---|---|---|
| Scope of Analysis | Broad, untargeted (entire secretome) | Narrow, targeted (single protein) | Targeted to broad (single to thousands of transcripts) |
| Quantification | Semi-quantitative to quantitative | Highly quantitative | Highly quantitative for RNA |
| Throughput | Medium | High | High |
| Sensitivity | Moderate (ng range) | High (pg-pg range) | High (can detect few copies) |
| Specificity | Moderate (based on peptide sequences) | High (based on antibody affinity) | High (based on primer/probe sequence) |
| Primary Output | List of identified proteins with abundances | Concentration of a specific protein | mRNA expression level of genes |
| Key Limitation | Cannot distinguish functional from inactive proteins; complex data analysis | Requires specific antibodies; limited to known targets | mRNA level may not correlate with protein secretion [48] |
Application to MSC Source Comparison
The choice of technique directly influences the insights gained into the differences between ASC, BMSC, and UCB-MSC secretomes.
- Proteomics has revealed that licensed (inflammatory primed) secretomes across all MSC sources are enriched with immunomodulatory proteins like chemotactic factors, while resting secretomes are defined by extracellular matrix (ECM) and pro-regenerative proteins [46]. It can also identify source-specific signatures, such as the presence of proteins related to proliferative potential and telomere maintenance in UCB-MSCs and iMSCs, compared to a higher abundance of fibrotic and ECM-related proteins in adult tissue-derived MSCs like ASCs and BMSCs [46].
- ELISA provides the precise data needed to validate these discoveries. For instance, it can be used to confirm the significant upregulation of Indoleamine 2,3-dioxygenase (IDO)—a key immunomodulatory factor—in the secretome of inflammatory-licensed MSCs compared to their resting state, a finding consistent across different MSC sources [46].
- RNA Analysis offers a high-throughput method to screen for differences in gene expression. However, a study comparing microarray-based RNA expression with ELISA-based protein determination highlighted a critical limitation: while a strong correlation was found for some markers like HER2 and uPA, the correlation for PAI-1 was poor (r=0.27) [48]. This underscores that mRNA levels are not always reliable proxies for secreted protein levels, necessitating direct protein measurement for conclusive secretome characterization.
Experimental Data from Comparative MSC Studies
Quantitative Secretome Data
Table 2: Exemplary Secretome Data from Comparative Studies of Different MSC Types
| Analyte | Technique | ASC Secretome | BMSC Secretome | UCB-MSC Secretome | Experimental Context |
|---|---|---|---|---|---|
| IDO | ELISA | >10-fold increase [46] | >10-fold increase [46] | >10-fold increase [46] | Upon inflammatory licensing (IFN-γ & TNF-α) |
| General Protein Secretion | Cytokine Array | "More prominent" profile [49] | Information Missing | "More prominent" profile [49] | Comparative side-by-side study |
| Pro-regenerative Factors (e.g., VEGF, HGF, FGF) | Proteomics / Antibody Array | Present [14] | Present [14] | Present [14] | CM from hypoxic preconditioned MSCs |
| PAI-1 Protein vs RNA | ELISA vs Microarray | Poor correlation (r=0.27) [48] | Poor correlation (r=0.27) [48] | Information Missing | Direct comparison in breast cancer tissue |
Correlating Technique with Biological Outcome
The functional consequences of secretome differences can be traced back to the analytical data. For example, the poor correlation between PAI-1 mRNA and protein levels [48] suggests that post-transcriptional regulation is important and that RNA data alone is insufficient to predict biological activity. Furthermore, proteomic and ELISA data revealing a strong inflammatory licensing response across all MSC sources [46] provides a mechanistic explanation for the enhanced immunomodulatory efficacy of primed MSCs in models of inflammatory disease. The identification of a distinct protein signature in natal vs. adult MSC secretomes via proteomics [46] offers a potential molecular basis for their reported differences in proliferative capacity and therapeutic performance.
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for Secretome Profiling
| Reagent / Material | Function in Analysis | Application Notes |
|---|---|---|
| Serum-Free Media | Used for conditioning during secretome collection to avoid contamination from serum proteins. | Essential for clean MS and ELISA analysis; different formulations can influence MSC secretome [45]. |
| Antibody Pairs (Matched) | Critical for the capture and detection steps in a sandwich ELISA. | Specificity and affinity determine assay performance; must be validated for each target [50]. |
| Trypsin | Protease used to digest secretome proteins into peptides for LC-MS/MS analysis. | Standard enzyme for bottom-up proteomics; requires high purity (sequencing grade) [45]. |
| CD73, CD90, CD105 Antibodies | Surface marker antibodies for characterizing MSC populations by flow cytometry prior to secretome analysis. | Part of ISCT minimal criteria for defining MSCs; ensures cell quality [46]. |
| IFN-γ & TNF-α | Cytokines used to induce inflammatory licensing (MSC2 phenotype) prior to secretome collection. | Standardizes the activation state; typically used at 10-20 ng/mL for 24-48 hours [46]. |
| Protein Binding ELISA Plates | Solid phase for immobilizing capture antibodies or antigens in ELISA. | Clear plates for colorimetric detection; white/black for chemiluminescent/fluorescent detection [47]. |
| LC-MS/MS System | Instrumentation for separating and analyzing peptides in proteomics. | High-resolution systems provide greater accuracy and proteome coverage [45] [46]. |
Experimental Workflow and Signaling Pathways
Secretome Profiling Workflow
The following diagram illustrates the integrated experimental workflow for the comparative analysis of MSC secretomes using the discussed techniques.
MSC Inflammatory Licensing Pathway
A key preconditioning step that profoundly affects the secretome is inflammatory licensing. The following diagram outlines the core signaling pathway involved in this process.
Functional in vitro assays are indispensable for evaluating the therapeutic potential of different Mesenchymal Stem Cell (MSC) populations. When framed within the context of comparative paracrine factor expression in Adipose-derived Stem Cells (ASCs), Bone Marrow Mesenchymal Stem Cells (BMSCs), and Umbilical Cord Blood Mesenchymal Stem Cells (UCB-MSCs), these assays transition from simple quality controls to powerful tools for elucidating mechanistic pathways. The secretome of a cell—the repertoire of growth factors, cytokines, and other signaling molecules it secretes—directly influences key processes such as the formation of new vascular tubes (tubulogenesis), cellular survival, and directed movement (migration). By employing standardized functional assays, researchers can move beyond cataloging expressed factors to understanding their functional consequences, thereby identifying the most suitable MSC source for specific regenerative applications [3] [51].
Tubulogenesis Assays
Matrigel Tubulogenesis Assay
The Matrigel assay is a cornerstone in vitro method for modeling the critical process of tubulogenesis, where endothelial cells form capillary-like structures. This process is fundamental to angiogenesis and is strongly influenced by paracrine signals from MSCs [52] [51].
- Principle: When plated on a basement membrane extract like Matrigel, endothelial cells undergo rapid morphological changes, including attachment, migration, and organization into interconnected networks of tube-like structures that mimic early-stage capillary formation [52].
- Protocol Summary:
- Coating: Coat wells of a 96-well plate with 50 µL of growth factor-reduced Matrigel and allow it to polymerize.
- Cell Seeding: Trypsinize and resuspend endothelial cells (e.g., HUVEC or ECFC-derived cells) at a density of 1.5 x 10⁴ cells/well in the appropriate medium. The medium can be supplemented with MSC-conditioned media to test paracrine effects or with direct angiogenic inhibitors/inducers.
- Incubation: Incubate the plates for 6–20 hours at 37°C in a humidified atmosphere with 5% CO₂.
- Imaging and Fixation: After incubation, acquire phase-contrast images using an inverted microscope (e.g., 4x magnification). Subsequently, remove media, wash the cells, and fix with 100% ice-cold methanol for downstream analysis [52].
- Quantification Methods: Analysis can be performed using automated image analysis systems which provide robust and reproducible quantification. Key parameters include [52]:
- Total Tubule Length: The combined length of all capillary-like structures.
- Number of Junctions: Branch points within the tubular network.
- Mesh Area: The area enclosed by the tubular structures.
Comparative Data and Relevance to MSC Paracrine Activity
Functional tubulogenesis data correlates directly with the paracrine profiles of different MSC populations. Research has shown that the conditioned medium from ASCs leads to a significantly increased tubulogenic efficiency in endothelial cells compared to conditioned media from other MSC types like dermal papilla cells (DPCs) [3]. Furthermore, this pro-angiogenic effect has been mechanistically linked to specific factors within the ASC secretome. For instance, the use of neutralizing antibodies has identified VEGF-A and VEGF-D as two of the major pro-angiogenic factors secreted by ASCs that are responsible for supporting endothelial tubulogenesis [3]. This functional evidence aligns with mRNA expression data showing that ASCs express higher levels of potent angiogenic factors such as IGF-1, VEGF-D, and IL-8 compared to BMSCs and other MSC populations, which may account for their superior performance in this assay [3].
Table 1: Key Paracrine Factors from Different MSC Sources Influencing Tubulogenesis
| MSC Source | Key Pro-Angiogenic Factors | Functional Effect on Tubulogenesis |
|---|---|---|
| ASCs | VEGF-A, VEGF-D, IGF-1, IL-8 [3] | Conditioned media results in increased tubulogenic efficiency of endothelial cells [3]. |
| BMSCs | VEGF-A, Angiogenin, bFGF, NGF (comparable levels) [3] | Supports tubulogenesis, though may be less potent than ASC-conditioned media in comparative studies. |
| UCB-MSCs | Information not specifically covered in search results | Further research needed for direct comparison. |
Cell Survival and Proliferation Assays
Metabolic Activity Assay (MTT Assay)
Cell survival and proliferation are fundamental to the regenerative capacity of MSCs. The MTT assay is a widely used colorimetric method to assess metabolic activity, which serves as a proxy for cell viability and proliferation.
- Principle: Metabolically active cells reduce the yellow tetrazolium salt MTT to insoluble purple formazan crystals. The quantity of formazan produced, measured spectrophotometrically after solubilization, is directly proportional to the number of viable cells [53].
- Protocol Summary:
- Cell Seeding: Seed ASCs or BMSCs at a density of 3 x 10³ cells/cm² in 96-well plates and culture for set time points (e.g., 3, 7, 14, 21 days).
- MTT Incubation: At each time point, add MTT solution to the wells (typically a 1:4 dilution in culture medium) and incubate for 4 hours at 37°C.
- Solubilization: Remove the MTT solution and add a solvent (e.g., isopropanol) to dissolve the formed formazan crystals.
- Quantification: Measure the absorbance at a specific wavelength (e.g., 570 nm) using a microplate reader [53].
- Considerations: It is crucial to perform proliferation assays on low-passage cells (passages 3-6) and ensure sub-confluence (typically <70%) to avoid contact inhibition, which can diminish proliferative activity. Serum starvation may be used to induce a quiescent state before stimulating with test substances [51].
Comparative Proliferation and Survival Data
Donor-matched comparative studies reveal intrinsic biological differences between MSC sources. In general, ASCs demonstrate a significantly higher proliferation rate compared to BMSCs when cultured in vitro [53]. This enhanced proliferative capacity is a significant practical advantage for generating sufficient cell numbers for therapy. Furthermore, the survival and function of MSCs can be influenced by their differentiation commitment. For instance, BMSCs undergoing osteogenic commitment exhibit different biological behaviors compared to those directed toward an adipogenic lineage, which can indirectly affect their persistence and function in a therapeutic context [54].
Table 2: Comparison of Functional Characteristics Across MSC Sources
| Functional Assay | ASCs | BMSCs | UCB-MSCs |
|---|---|---|---|
| Proliferation | Significantly higher proliferation rate [53] | Lower proliferation rate compared to ASCs [53] | Information not specifically covered in search results |
| Adipogenic Capacity | High lipid vesicle formation, strong adipogenic gene expression [53] | Lower adipogenic capacity [53] | Less prominent and efficient adipogenesis compared to ASCs [49] |
| Osteogenic Capacity | Lower osteogenic capacity, less ALP activity and calcium deposition [53] | Higher osteogenic and chondrogenic capacity, strong ALP activity [53] | No significant difference in osteogenesis vs. ASCs in some studies [49] |
Cell Migration and Invasion Assays
Scratch Wound Healing Assay
The scratch wound assay is a simple and common method to study two-dimensional (2D) cell migration, a process critical for homing to injury sites and tissue repair.
- Principle: A scratch is created in a confluent cell monolayer, and the migration of cells into the wound area is monitored over time, providing a measure of collective cell migration [55] [54].
- Protocol Summary:
- Create a Confluent Layer: Seed BMSCs or other cells at high density (e.g., 1x10⁶ cells/well in a 12-well plate) and allow them to form a confluent monolayer.
- Create Scratch: Use a sterile pipette tip to create a straight, uniform "scratch" through the cell layer.
- Wash and Add Medium: Wash away detached cells with PBS and add fresh medium (e.g., growth, osteogenic, or adipogenic differentiation medium). To investigate specific pathways, inhibitors (e.g., AMD3100 for CXCR4) can be added.
- Image and Quantify: Capture images of the scratch at defined intervals (0, 6, 12, 24 hours). Quantify migration by measuring the change in the width of the wound area over time [54].
Transwell Migration and Invasion Assay
The Transwell (or Boyden chamber) assay is a robust method for quantifying directed cell migration and invasion through a porous membrane, with the key difference being the presence of an ECM matrix for invasion.
- Principle for Migration: Cells are placed in an upper chamber separated from a lower chamber containing a chemoattractant (e.g., serum or specific cytokines) by a porous membrane. Cells that migrate through the pores to the underside of the membrane are stained and counted [55] [54].
- Principle for Invasion: The membrane is coated with a layer of ECM (e.g., Matrigel or VitroGel) to create a barrier that simulates the extracellular matrix. Invasive cells must degrade and move through this matrix to reach the other side [55] [56].
- Protocol Summary:
- Prepare Chambers: For invasion assays, coat the Transwell membrane with an ECM matrix and allow it to gel.
- Seed Cells: Seed cells in serum-free medium into the upper chamber. Place medium with a chemoattractant (e.g., 10% FBS) in the lower chamber.
- Incubate: Incubate for a set time (e.g., 12-24 hours) to allow migration/invasion.
- Quantify: Remove non-migrated cells from the top of the membrane with a cotton swab. Fix and stain cells that have migrated to the lower side. Count these cells manually or by using imaging software [54] [56].
Advanced 3D Invasion Models
More physiologically relevant 3D models are gaining traction. These include 3D spheroid invasion assays, where cell spheroids are embedded in or placed on top of a hydrogel matrix, and cells invade the surrounding matrix radially. These models better preserve cell-cell interactions and mimic the in vivo microenvironment [56]. Hydrogel systems like VitroGel offer a tunable, xeno-free alternative to traditional Matrigel, allowing researchers to study the effects of matrix stiffness, degradability, and biochemical composition on cell invasion [56].
Comparative Migration Data and Signaling Pathways
Migration is not a uniform property and can vary based on MSC source and lineage commitment. A critical finding is that BMSCs undergoing osteogenic commitment demonstrate a higher migratory capacity compared to those directed toward adipogenic commitment [54]. This differential migration appears to be regulated by specific molecular pathways. Research points to the Sdf1/Cxcr4 axis as a key regulator, where inhibition of the Cxcr4 receptor with AMD3100 significantly reduces the enhanced migration of osteogenically-committed BMSCs [54]. Furthermore, differences in the expression of integrins, such as Itgα1 and Itgα5, have been implicated in this process, highlighting the complex interplay between adhesion receptors and migratory behavior [54].
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagents and Materials for Functional In Vitro Assays
| Item | Function/Description | Example Use Case |
|---|---|---|
| Growth Factor-Reduced Matrigel | A basement membrane extract used to stimulate and support the formation of capillary-like tubule structures by endothelial cells. | Tubulogenesis Assay [52] |
| Transwell Inserts | Cell culture inserts with a porous membrane that separates two chambers, enabling the study of directed cell migration and invasion. | Transwell Migration/Invasion Assay [55] [54] |
| VitroGel Hydrogel System | A tunable, xeno-free hydrogel that can be used as a defined ECM barrier in invasion assays or for 3D spheroid culture. | 3D Invasion Assay [56] |
| ROCK Inhibitor (Y-27632) | A small molecule inhibitor of Rho-associated kinase. It enhances cell survival after passaging and promotes stemness in 3D spheroid cultures. | Preventing Anoikis in Stem Cell Culture [57] |
| CXCR4 Inhibitor (AMD3100) | A selective antagonist of the CXCR4 receptor. Used to probe the role of the Sdf1/Cxcr4 signaling axis in directed cell migration. | Mechanistic Migration Studies [54] |
| Ultra-Low Attachment (ULA) Plates | Culture plates with a covalently bound hydrogel layer that minimizes cell attachment, forcing cells to aggregate and form 3D spheroids. | 3D Spheroid Formation [57] |
The integration of standardized functional assays—tubulogenesis, survival, and migration—provides a powerful, multi-parametric framework for comparing ASCs, BMSCs, and UCB-MSCs. The experimental data gleaned from these assays directly links the distinct paracrine factor expression profiles of each MSC type to tangible, quantifiable biological outcomes. This approach moves beyond molecular cataloging to functional validation, enabling researchers to make informed, evidence-based decisions when selecting an MSC source for specific therapeutic goals, such as promoting vascularization in ischemic tissues (favoring ASCs) or enhancing bone regeneration (favoring BMSCs). As the field advances, the adoption of more physiologically relevant 3D models and defined matrices will further enhance the predictive power of these in vitro tools, accelerating the translation of MSC-based therapies from the bench to the clinic.
The therapeutic potential of mesenchymal stem cells (MSCs) is now largely attributed to their secretome—the complex mixture of proteins, extracellular vesicles, cytokines, and growth factors they secrete—rather than to the cells themselves. [58] [59] This paradigm shift towards cell-free therapies offers significant advantages, including reduced risks of immune rejection and tumorigenicity, and simpler manufacturing and storage. [58] However, the composition and potency of the secretome are highly influenced by the MSC's tissue of origin, creating a critical need to understand how source-dependent variations impact therapeutic efficacy in preclinical models. [59] [46]
This guide provides a comparative analysis of the secretomes from three prominent MSC sources: Adipose-Derived Stem Cells (ASCs), Bone Marrow-Derived MSCs (BMSCs), and Umbilical Cord-Derived MSCs (UC-MSCs), focusing on their paracrine factor expression and functional outcomes in animal studies.
Comparative Analysis of MSC Secretome Profiles
The secretory profile of MSCs is not uniform. The tissue source dictates the concentration of key paracrine factors, which in turn influences the mechanism and effectiveness of tissue repair. [49] [46]
Table 1: Key Paracrine Factors by MSC Source
| Paracrine Factor | ASC Expression | BMSC Expression | UC-MSC Expression | Primary Function |
|---|---|---|---|---|
| IGF-1 [3] [10] | Higher | Lower | Not Specified | Proangiogenic, cell survival |
| VEGF-A [3] [10] | High | High | High | Angiogenesis |
| VEGF-D [3] [10] | Higher | Lower | Not Specified | Angiogenesis |
| IL-8 [3] [10] | Higher | Lower | Not Specified | Chemotaxis, angiogenesis |
| Angiogenin [3] [10] | High | High | High | Angiogenesis |
| bFGF [3] [10] | High | High | High | Angiogenesis, tissue repair |
| HGF [58] | Not Specified | Not Specified | High | Anti-apoptotic, proangiogenic |
| TSG-6 [58] | Not Specified | Not Specified | High | Anti-inflammatory |
| Cytokine Secretion Profile [49] | Less Prominent | Not Specified | More Prominent | Immunomodulation |
Table 2: Functional Correlations in Preclinical Models
| Therapeutic Application | ASC Secretome Efficacy | UC-MSC Secretome Efficacy | BMSC Secretome Efficacy | Supporting Evidence |
|---|---|---|---|---|
| Angiogenesis | Enhanced tubulogenesis; superior perfusion recovery in limb ischemia [3] | Effective in reducing lung injury (BPD models) [58] | Proangiogenic, but may be less potent than ASCs in some models [3] | In vitro endothelial tubulogenesis; murine hindlimb ischemia [3] |
| Anti-inflammatory & Tissue Repair | Effective in osteoarthritis models, reducing inflammation and promoting cartilage regeneration [60] | Potent anti-inflammatory effects; high levels of protective molecules (e.g., TSG-6, IL-10) [58] | Effective in osteoarthritis models [60] | Rodent models of experimentally induced osteoarthritis [60] |
| Overall Secretome Potency | Strong proangiogenic focus | High proliferative capacity; robust immunomodulation; preferred for neonatal applications [58] [46] | Gold standard, but donor-age-related functional decline [58] | Comparative profiling and animal studies [58] [3] [46] |
Experimental Protocols for Secretome Analysis
Standardized methodologies are crucial for the reproducible production, collection, and analysis of MSC secretomes. [61]
Secretome Production and Collection
- Cell Culture: MSCs are typically expanded in standard 2D culture. To enhance secretome potency, 3D culture systems (spheroids) or hypoxic conditioning (1-10% O₂) are employed. Hypoxia upregulates hypoxia-inducible factor 1-α (HIF-1α), boosting secretion of proangiogenic factors like VEGF. [61]
- Inflammatory Licensing: To direct MSCs toward an immunomodulatory phenotype (MSC2), cells are stimulated with cytokines such as interferon-gamma (IFN-γ) and tumor necrosis factor-alpha (TNF-α). Successful licensing is validated by measuring upregulated surface markers (HLA-ABC, HLA-DR) and increased secretion of immunomodulatory factors like indoleamine 2,3-dioxygenase (IDO). [46]
- Collection: To avoid contamination with serum proteins, MSCs are switched to a serum-free medium for 24-48 hours. The conditioned medium (CM) is then collected and subjected to a series of processing steps. [61]
Characterization and Functional Assays
- Proteomic Profiling: High-resolution techniques like liquid chromatography-tandem mass spectrometry (LC-MS/MS) are used to comprehensively identify and quantify proteins in the secretome. This reveals differences between resting and licensed states and between different MSC sources. [46]
- Functional In Vitro Assays:
- Endothelial Tubulogenesis Assay: The proangiogenic potential of a secretome is tested by applying CM to endothelial cells cultured on a basement membrane matrix. The formation of tube-like structures is quantified and compared to controls. [3] [10]
- Neutralizing Antibodies: To confirm the role of specific factors, functional assays are repeated with CM that has been pre-incubated with neutralizing antibodies (e.g., against VEGF-A). [3] [10]
- In Vivo Validation: Promising secretomes are tested in translationally relevant animal models, such as rodent models of osteoarthritis, limb ischemia, or neonatal conditions like bronchopulmonary dysplasia (BPD). Outcomes are measured through histological analysis, functional recovery tests, and biomarker assessment. [58] [60] [59]
Correlating Secretome Signature to In Vivo Efficacy
Understanding how a secretome's molecular signature translates to therapeutic action in a whole organism is key to predicting clinical success.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Secretome Research
| Research Reagent / Tool | Function in Secretome Studies |
|---|---|
| Collagenase Type I | Enzymatic digestion of tissue (e.g., adipose, umbilical cord) for initial isolation of MSCs. [49] |
| CD73, CD90, CD105 Antibodies | Flow cytometry antibodies used to confirm the identity of isolated MSCs per ISCT criteria. [46] |
| IFN-γ and TNF-α Cytokines | Used for in vitro inflammatory licensing of MSCs to induce an immunosuppressive (MSC2) phenotype. [46] |
| Serum-Free Media | Essential for producing conditioned medium (CM) free of contaminating proteins from fetal bovine serum. [61] |
| Ultracentrifugation / TFF System | Equipment for concentrating and purifying secretome components, particularly extracellular vesicles. [58] [61] |
| Anti-VEGF Neutralizing Antibody | Functional tool to block specific factors in CM and confirm their role in assays like tubulogenesis. [3] [10] |
| ELISA Kits (e.g., for IDO) | Used to quantify the levels of specific immunomodulatory factors in the conditioned medium. [46] |
| Matrigel / Basement Membrane Matrix | Substrate for in vitro endothelial tubulogenesis assays to assess proangiogenic potential. [3] |
| "Humanized" and "Naturalized" Mouse Models | Translationally relevant animal models with human immune components or diverse microbiomes for testing secretome efficacy. [62] |
The correlation between MSC secretome profiles and therapeutic efficacy in animal models is unequivocally influenced by the cellular source. ASCs demonstrate a strong proangiogenic signature, UC-MSCs exhibit potent immunomodulatory and proliferative capacities, and BMSCs, while effective, may show age-related limitations. [58] [3] [46]
The future of secretome-based therapeutics lies in moving beyond naive secretomes. Research is increasingly focusing on manipulating MSCs through priming (e.g., 3D culture, hypoxia, licensing) and genetic engineering to create tailored, potent, and consistent secretome products for specific clinical applications. [61] [59] Standardizing production and analytical protocols will be the critical next step in translating these promising preclinical outcomes into effective human therapies. [58] [61]
The therapeutic application of Mesenchymal Stem Cells (MSCs) has evolved from a generic approach to a precision-based paradigm that recognizes the profound impact of tissue origin on functional efficacy. While MSCs from various sources share fundamental characteristics—adherence to plastic, expression of specific surface markers, and multilineage differentiation potential—their secretome, differentiation bias, and functional capabilities exhibit remarkable diversity [63]. This biological heterogeneity stems from distinct developmental origins and tissue-specific microenvironmental imprinting, resulting in specialized therapeutic profiles that make different MSC sources uniquely suited for specific clinical indications [64] [3].
The foundation of precision medicine in MSC therapy rests on matching the inherent strengths of each MSC type with the pathophysiological requirements of the target condition. For cardiac repair, this might prioritize cardiomyogenic differentiation potential and robust angiogenic activity [65] [64], while wound healing may demand potent immunomodulation and epithelial regeneration capabilities. Understanding these source-specific therapeutic profiles enables researchers to strategically select MSC sources based on mechanistic alignment with disease pathology rather than mere availability.
This review comprehensively analyzes the comparative therapeutic profiles of MSCs derived from adipose tissue (ASCs), bone marrow (BMSCs), and umbilical cord blood (UCB-MSCs) within the context of their paracrine signatures, with a specific focus on matching these cellular attributes to two distinct clinical indications: cardiac repair and wound healing.
Anatomical Origin and Biological Characteristics
The tissue niche from which MSCs are isolated fundamentally shapes their biological properties and functional capabilities. Adipose-derived MSCs (ASCs) are typically isolated from lipoaspirate or adipose tissue fragments through collagenase digestion and centrifugation methods [3]. These cells demonstrate particularly high yield and proliferation rates compared to other sources [66]. Bone marrow-derived MSCs (BMSCs) are traditionally isolated from bone marrow aspirates via density gradient centrifugation and plastic adherence [37]. As the most extensively studied population, BMSCs represent the gold standard against which other MSCs are often compared, though their numbers decline with donor age [66]. Umbilical cord blood-derived MSCs (UCB-MSCs) are obtained from cord blood collections through density gradient separation and exploit the non-invasive accessibility and neonatal character of this source [37]. While isolation success rates for UCB-MSCs are lower (approximately 63%) compared to ASCs and BMSCs (near 100%), they exhibit superior expansion potential and longer culture periods [66].
Paracrine Factor Expression Profiles
The therapeutic benefits of MSCs are increasingly attributed to their paracrine activity rather than direct differentiation and engraftment. Comparative analysis of MSC secretomes reveals distinct expression patterns of angiogenic, immunomodulatory, and trophic factors across different tissue sources.
Table 1: Comparative Paracrine Factor Expression Across MSC Sources
| Paracrine Factor | ASCs | BMSCs | UCB-MSCs | Functional Significance |
|---|---|---|---|---|
| VEGF-A | High [3] | High [3] | Moderate | Angiogenesis, endothelial survival |
| VEGF-D | High [3] [10] | Low [3] [10] | Not reported | Lymphangiogenesis, endothelial tubulogenesis |
| IGF-1 | High [3] [10] | Low [3] [10] | Not reported | Cardiomyocyte survival, hypertrophy |
| IL-8 | High [3] [10] | Low [3] [10] | Not reported | Neutrophil chemotaxis, angiogenesis |
| Angiogenin | Comparable [3] | Comparable [3] | Not reported | Ribonucleolytic activity, angiogenesis |
| bFGF | Comparable [3] | Comparable [3] | Not reported | Fibroblast proliferation, angiogenesis |
| HGF | Moderate-High | Moderate | Moderate | Anti-fibrotic, mitogenic, morphogenic |
| IL-6 | High (after priming) | Moderate | Moderate | Immunomodulation, inflammation resolution |
ASCs demonstrate a particularly robust angiogenic profile, with significantly higher expression of VEGF-D, IGF-1, and IL-8 compared to BMSCs and dermal-derived MSCs [3] [10]. Functional assays confirm that ASC-conditioned media induces superior endothelial tubulogenesis, which is significantly mediated by VEGF-A and VEGF-D [3]. BMSCs express comparable levels of VEGF-A and angiogenin but lower levels of other angiogenic factors, while UCB-MSCs show variable expression with generally lower angiogenic potential but strong immunomodulatory capacity.
Differentiation Potential and Lineage Bias
The differentiation capacity of MSCs varies considerably based on tissue origin, reflecting a form of "lineage memory" or predisposition toward their native tissue environment. ASCs demonstrate superior adipogenic differentiation potential, consistent with their tissue origin, while BMSCs show enhanced osteogenic capacity [66]. Most strikingly, UCB-MSCs show limited adipogenic differentiation capacity compared to ASCs and BMSCs [66]. Regarding cardiomyogenic differentiation, ASCs from specific anatomical locations demonstrate varying efficiencies; peri-ovarian ASCs exhibit broader metabolic reprogramming with engagement of glycolysis, fructose metabolism, glycerolipid metabolism, and TCA cycle during cardiomyocyte differentiation, suggesting enhanced metabolic flexibility compared to peri-renal ASCs [65].
MSC Source Selection for Cardiac Repair
Pathophysiological Considerations in Myocardial Infarction
Myocardial infarction (MI) triggers a complex pathological cascade involving cardiomyocyte death, impaired angiogenesis, excessive inflammation, and maladaptive remodeling [64] [67]. Effective therapeutic interventions must address multiple aspects: replenishing lost cardiomyocytes, promoting vascularization to restore perfusion, modulating inflammatory responses, and preventing adverse fibrosis [67]. The limited regenerative capacity of adult cardiomyocytes, with turnover rates declining from 1% at age 25 to 0.45% by age 75, underscores the need for exogenous cell-based interventions to achieve meaningful tissue regeneration [67].
Different MSC sources demonstrate variable therapeutic efficacy in cardiac repair based on their unique functional profiles:
ASCs for Cardiac Repair: ASCs demonstrate particularly strong potential for cardiac repair due to their robust angiogenic paracrine signature and cardiomyogenic differentiation capacity [65] [64]. In a comparative study targeting angiogenesis in a mouse MI model, AD-MSCs exerted better cardioprotective function than UC-MSCs with stronger anti-apoptotic effects on residual cardiomyocytes [64]. The protection of residual cells survival played a more prominent role than angiogenesis in MSC-based therapy for acute MI, highlighting the therapeutic value of ASCs [64]. Additionally, the anatomical depot of adipose tissue influences cardiac efficacy; peri-ovarian ASCs exhibit greater metabolic adaptability during cardiomyocyte differentiation through increased engagement of glycolysis, fructose metabolism, glycerolipid metabolism, and TCA cycle, potentially favoring their differentiation capacity for cardiac regenerative applications [65].
UCB-MSCs for Cardiac Repair: UCB-MSCs show superior expansion potential and immunomodulatory capacity but variable cardiac efficacy. In comparative studies, UCMSCs presented greater pro-angiogenesis activity than ADMSCs in vitro and in vivo, but ADMSCs ultimately provided better functional improvement in MI models, suggesting that angiogenic potential alone does not predict overall therapeutic efficacy for cardiac repair [64].
BMSCs for Cardiac Repair: As the most extensively studied MSC population, BMSCs have demonstrated safety and moderate efficacy in clinical trials such as POSEIDON and PROMETHEUS, with improved cardiac functionality and absence of arrythmia in treated patients [67]. However, their declining numbers with donor age and moderately invasive harvest procedure present limitations for widespread clinical application [66].
Table 2: Matching MSC Sources to Cardiac Repair Mechanisms
| Therapeutic Mechanism | Recommended MSC Source | Experimental Evidence |
|---|---|---|
| Angiogenesis | ASCs (particularly for VEGF-D mediated tubulogenesis) | ASC-CM increased endothelial tubulogenesis; neutralization of VEGF-A and VEGF-D inhibited this effect [3] |
| Cardiomyocyte Survival/Anti-apoptosis | ASCs | AD-MSCs exerted stronger anti-apoptotic effects on residual cardiomyocytes than UC-MSCs in MI model [64] |
| Cardiomyocyte Differentiation | Peri-ovarian ASCs | Demonstrated enhanced metabolic flexibility during cardiomyocyte differentiation [65] |
| Inflammation Modulation | UCB-MSCs | Superior expansion potential and immunomodulatory capacity; reduced pro-inflammatory cytokine secretion [66] |
| Metabolic Reprogramming | Peri-ovarian ASCs | Broad metabolic reprogramming with engagement of glycolysis, fructose metabolism, glycerolipid metabolism, TCA cycle [65] |
Emerging Alternatives for Cardiac Repair
Skeletal muscle-derived MSCs (mdMSCs) represent a promising alternative for cardiac repair, combining autologous origin with minimally invasive harvest procedure [68]. When embedded in a collagen bioprinted patch and delivered epicardially in a rat model of left ventricular dysfunction, mdMSC patches significantly improved LV ejection fraction compared to cell-free patches and sham-operated controls [68]. Transcriptomic analysis revealed that this benefit was accompanied by downregulation of fibrosis-, apoptosis-, and inflammation-related genes [68].
MSC Source Selection for Wound Healing
Pathophysiological Considerations in Wound Healing
Wound healing involves a coordinated sequence of events including inflammation, proliferation, and remodeling phases. Effective therapeutic interventions must modulate immune responses, promote angiogenesis, stimulate fibroblast proliferation and migration, and facilitate extracellular matrix reconstruction. The inflammatory phase immediately following injury involves hematoma formation and immune cell infiltration releasing cytokines that recruit MSCs to the site of injury [69]. This sets the stage for subsequent proliferative phases where MSCs differentiate into various cell types and secrete factors that promote tissue regeneration.
While the search results provided limited direct comparative studies on MSC sources for wound healing, extrapolation from their paracrine profiles and differentiation capacities allows for therapeutic recommendations:
ASCs for Wound Healing: ASCs demonstrate superior pro-angiogenic activity through their robust secretion of VEGF-A, VEGF-D, and other angiogenic factors that are crucial for the neovascularization required during wound healing [3]. Their high expression of IGF-1 promotes fibroblast proliferation and keratinocyte migration, while IL-8 facilitates neutrophil recruitment and microbial clearance in early wound phases.
UCB-MSCs for Wound Healing: UCB-MSCs offer potent immunomodulatory capabilities that can help regulate the excessive inflammation often present in chronic wounds [66]. Their neonatal character may provide enhanced plasticity and responsiveness to wound microenvironmental cues.
BMSCs for Wound Healing: BMSCs have demonstrated efficacy in bone-related wound healing through their strong osteogenic differentiation capacity and secretion of bone morphogenetic proteins [69]. Their ability to differentiate into osteoblasts makes them particularly suitable for bone fracture healing applications.
Experimental Methodologies for MSC Characterization
Standardized Isolation and Culture Protocols
Proper isolation and characterization methodologies are essential for obtaining reproducible MSC populations for research and clinical applications. The International Society for Cell & Gene Therapy (ISCT) has established minimum criteria for defining MSCs: (1) adherence to plastic under standard culture conditions; (2) expression of CD73, CD90, and CD105 (≥95%) while lacking expression of CD34, CD45, CD14 or CD11b, CD79α or CD19, and HLA-DR (≤2%); and (3) ability to differentiate into osteoblasts, adipocytes, and chondrocytes under standard in vitro conditions [63] [37].
ASCs Isolation Protocol: ASCs are typically isolated from adipose tissue samples through collagenase digestion followed by centrifugation to separate the stromal vascular fraction from adipocytes [3]. The stromal vascular fraction is then plated in culture vessels, where ASCs adhere and expand. Specific characterization should include flow cytometry for standard MSC markers plus additional adipose-related markers.
UCB-MSCs Isolation Protocol: UCB-MSCs are isolated from umbilical cord blood collections through density gradient centrifugation (Ficoll-Paque) to isolate mononuclear cells, which are then plated in specialized media formulations [37]. Due to lower MSC frequency in cord blood, optimization of plating density and media supplements is critical for successful isolation.
BMSCs Isolation Protocol: BMSCs are obtained from bone marrow aspirates through density gradient centrifugation to isolate mononuclear cells, followed by plastic adherence and expansion in complete media [37]. Donor age significantly affects cell yield and proliferation capacity, with younger donors preferable.
Paracrine Factor Analysis Methodologies
Comprehensive characterization of MSC secretomes is essential for understanding their therapeutic mechanisms and predicting efficacy for specific indications:
Conditioned Media Collection: MSCs are cultured to 70-80% confluence, washed thoroughly, and incubated with serum-free medium for 24-48 hours. The conditioned media is then collected, centrifuged to remove cells and debris, and concentrated using centrifugal filter devices [3].
Protein-Level Analysis: Multiplex immunoassays (Luminex) enable simultaneous quantification of multiple angiogenic and inflammatory factors in MSC-conditioned media [68]. ELISA assays provide quantitative measurement of specific factors like VEGF, HGF, and IGF-1.
mRNA Expression Analysis: RNA sequencing and RT-qPCR with specific primers for paracrine factors allow comprehensive profiling of gene expression patterns [64]. For example, RNA sequencing analysis revealed differences in gene expression related to angiogenesis and apoptosis pathways between UCMSCs and ADMSCs [64].
Functional Angiogenesis Assays:
- Endothelial Tube Formation Assay: Human umbilical vein endothelial cells (HUVECs) are seeded on Matrigel-coated plates with MSC-conditioned media. Tube formation is quantified by measuring total tube length, number of nodes, and number of junctions after 4-8 hours [64].
- Matrigel Plug Assay: MSCs are mixed with liquid Matrigel and injected subcutaneously into immunodeficient mice. Plugs are harvested after 14 days and assessed for vascular invasion through hemoglobin content measurement and immunohistological analysis of endothelial markers [64].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Research Reagents for MSC Characterization
| Reagent/Category | Specific Examples | Research Application | Function |
|---|---|---|---|
| Isolation Reagents | Collagenase Type I, Ficoll-Paque, Percoll | MSC isolation from tissue sources | Tissue digestion, density-based cell separation |
| Culture Media | DMEM/low-glucose, α-MEM, Fetal Bovine Serum (FBS) | MSC expansion and maintenance | Provide nutrients, growth factors for cell growth |
| Characterization Antibodies | CD73, CD90, CD105, CD34, CD45, HLA-DR | Flow cytometry immunophenotyping | Confirm MSC identity per ISCT criteria |
| Differentiation Kits | Adipogenic, Osteogenic, Chondrogenic | Multilineage differentiation assessment | Induce lineage-specific differentiation |
| Angiogenesis Assay Reagents | Matrigel, HUVECs, Endothelial Cell Media | Endothelial tube formation assay | Assess pro-angiogenic potential of MSC secretome |
| Molecular Biology Tools | RNA extraction kits, cDNA synthesis kits, qPCR primers | Gene expression analysis | Quantify paracrine factor expression |
| Protein Analysis Tools | Multiplex immunoassay kits, ELISA kits | Secretome profiling | Quantify secreted angiogenic/immunomodulatory factors |
| Animal Models | Immunodeficient mice, Myocardial infarction models | In vivo efficacy testing | Evaluate therapeutic potential in disease models |
Signaling Pathways in MSC-Mediated Cardiac Repair
The therapeutic benefits of MSCs in cardiac repair are mediated through complex signaling pathways that regulate angiogenesis, cell survival, and inflammation. The diagram below illustrates the key signaling mechanisms involved in MSC-mediated cardiac repair, particularly highlighting the enhanced angiogenic pathway activation characteristic of ASCs.
Diagram 1: Signaling pathways in MSC-mediated cardiac repair. ASCs demonstrate enhanced secretion of key paracrine factors (VEGF, IGF-1, HGF, IL-8) that activate specific signaling pathways promoting angiogenesis, cell survival, and immunomodulation, ultimately contributing to cardiac repair. The PI3K/AKT pathway is particularly important for IGF-1-mediated cardiomyocyte survival, while VEGFR2 signaling drives VEGF-induced angiogenesis.
The paradigm of MSC therapeutics is shifting from a one-size-fits-all approach to precision matching of MSC source with specific clinical indications. For cardiac repair, ASCs—particularly from specific anatomical depots—demonstrate superior therapeutic potential due to their robust angiogenic secretome, strong anti-apoptotic effects on cardiomyocytes, and enhanced metabolic flexibility during cardiomyocyte differentiation [65] [64]. For wound healing applications, ASCs again offer advantages through their potent angiogenic capabilities, while UCB-MSCs may be preferable for immunomodulation-dominant applications.
Future research directions should focus on standardized comparative studies using clinically relevant models, development of potency assays that predict in vivo efficacy, and exploration of combination therapies that leverage the unique strengths of different MSC sources. As the field advances, precision matching of MSC source to disease pathology will maximize therapeutic outcomes and accelerate the clinical translation of MSC-based therapies for regenerative medicine applications.
Navigating Challenges: Isolation Efficiency, Donor Variability, and Protocol Standardization
Within the field of regenerative medicine, mesenchymal stromal cells (MSCs) have emerged as a cornerstone for therapeutic development due to their multipotent differentiation potential and potent immunomodulatory properties. The therapeutic efficacy of MSCs is largely mediated through their paracrine activity, whereby they secrete a diverse array of bioactive factors that influence the local microenvironment, promote tissue repair, and modulate immune responses. However, the expression profiles of these paracrine factors are not uniform across MSCs derived from different tissue sources. This variability is intrinsically linked to the efficiency with which MSCs can be isolated from their source tissues and the resulting cell yields, critical parameters that determine their feasibility for research and clinical applications. This guide provides a objective comparison of the isolation success rates and cell yields of three prominent MSC sources: adipose-derived stromal cells (ASCs), bone marrow-derived mesenchymal stromal cells (BMSCs), and umbilical cord blood-derived mesenchymal stromal cells (UCB-MSCs), framing this comparison within the broader context of their paracrine factor expression.
The selection of an MSC source involves balancing factors such as accessibility, isolation efficiency, and functional properties. The table below provides a comparative overview of ASCs, BMSCs, and UCB-MSCs based on key parameters.
Table 1: Comprehensive Comparison of MSC Sources
| Parameter | Adipose-Derived Stromal Cells (ASCs) | Bone Marrow-Derived MSCs (BMSCs) | Umbilical Cord Blood-Derived MSCs (UCB-MSCs) |
|---|---|---|---|
| Tissue Accessibility | High; obtained from lipoaspirate, minimally invasive | Moderate; invasive, painful aspiration procedure | High; from medical waste post-delivery, non-invasive |
| Isolation Success Rate | Consistently high | Variable | Generally low; often reported not to exceed 60% [70] |
| Primary Isolation Method | Enzymatic digestion (e.g., Collagenase) [49] [3] | Density gradient centrifugation [70] [71] | Density gradient centrifugation [70] |
| Typical Cell Yield | High yield of stromal vascular fraction (SVF) | Low frequency of MSCs in aspirate (~0.001%-0.01%) | Variable and often low cell number [70] |
| Proliferation Capacity | High | Decreases with donor age [49] | High; considered more primitive [70] |
| Key Paracrine Factors | Higher expression of IGF-1, VEGF-D, and IL-8 [3] | Expression of VEGF-A, angiogenin, bFGF, NGF [3] | Distinct cytokine secretion profile [49] |
| Donor Variability | Subject to donor age and BMI | Subject to significant donor age-related decline | High donor-dependent heterogeneity [72] |
Detailed Isolation Methodologies and Experimental Protocols
The isolation protocol is a critical determinant of the initial cell population's quality, quantity, and subsequent behavior. Below are detailed standardized protocols for isolating MSCs from each source, which are essential for ensuring reproducibility in research.
Isolation of Adipose-derived Stromal Cells (ASCs)
The isolation of ASCs relies on enzymatic digestion of adipose tissue to release the stromal vascular fraction (SVF), which contains the ASC population [49] [3].
- Tissue Processing: Subcutaneous adipose tissue (e.g., ~200 mL from lipoaspiration) is washed extensively with phosphate-buffered saline (PBS) to remove blood contaminants and minced into small pieces of approximately 1 mm³ [3].
- Enzymatic Digestion: The minced tissue is digested with 0.075% collagenase type I solution at 37°C for 45-60 minutes with continuous agitation [49] [3].
- Termination and Centrifugation: The digestion is halted by adding a culture medium containing serum. The suspension is centrifuged (e.g., 300-1200 g for 5-10 minutes) to separate the mature adipocytes (floating layer) from the stromal vascular fraction (SVF) pellet [49] [3].
- Red Blood Cell Lysis: The SVF pellet is resuspended in a red blood cell lysis buffer (e.g., 0.16 M NH₄Cl) for 5 minutes at room temperature [3].
- Filtration and Plating: The cell suspension is filtered through a 100 μm mesh filter to remove debris, then resuspended in complete culture medium (e.g., DMEM with 10% FBS) and plated in culture flasks [49] [3].
- Culture and Expansion: After 24-48 hours, non-adherent cells are removed by changing the medium. The adherent cells are cultured and expanded, with the medium changed every 2-3 days [49].
Isolation of Bone Marrow-derived MSCs (BMSCs)
BMSCs are typically isolated from bone marrow aspirates using density gradient centrifugation to separate mononuclear cells, followed by adherence selection [70] [71].
- Sample Dilution: The bone marrow aspirate is diluted with an equal volume of PBS or a balanced salt solution [71].
- Density Gradient Centrifugation: The diluted sample is carefully layered over a density gradient medium such as Ficoll-Paque and centrifuged at 400-800 g for 20-30 minutes at room temperature (with brakes off) [71].
- Mononuclear Cell Collection: The mononuclear cell layer (buffy coat) at the interface is carefully aspirated and transferred to a new tube [71].
- Washing: The collected cells are washed with PBS and centrifuged to remove residual gradient medium and platelets [71].
- Plating and Culture: The cell pellet is resuspended in complete medium (e.g., DMEM with 10% FBS) and seeded in culture flasks. After 48-72 hours, non-adherent cells (mainly hematopoietic cells) are removed by medium change. The adherent MSCs are then cultured and expanded [71].
Isolation of Umbilical Cord Blood-derived MSCs (UCB-MSCs)
The protocol for UCB-MSCs is similar to that for BMSCs but faces the challenge of low MSC frequency in cord blood [70].
- Volume Reduction: UCB is processed to reduce volume, often by centrifugation and removal of plasma and platelets.
- Density Gradient Centrifugation: The concentrated blood is layered over Ficoll-Paque and centrifuged under conditions similar to BMSC isolation [70].
- Mononuclear Cell Collection and Washing: The mononuclear cell layer is collected and washed with PBS [70].
- Plating and Adherence: The cells are resuspended in complete medium and plated. Due to the low frequency of MSCs in UCB, a key challenge is that MSCs are isolated by their adherence to plastic in primary culture, and the success rate is limited [70].
Diagram 1: Experimental workflow for MSC isolation from different sources. SVF: Stromal Vascular Fraction; MNC: Mononuclear Cell; RBC: Red Blood Cell.
Paracrine Factor Expression and Functional Implications
The therapeutic potential of MSCs is heavily influenced by their secretome. Comparative analyses have revealed significant differences in the expression of key paracrine factors among MSC sources, which directly correlate with their functional performance in areas like angiogenesis.
Table 2: Comparative Paracrine Factor Expression and Functional Impact
| Paracrine Factor | ASCs | BMSCs | UCB-MSCs | Primary Functional Role |
|---|---|---|---|---|
| IGF-1 | ↑ Higher expression [3] | Comparable | Not specifically quantified | Promotes cell survival, proliferation, and metabolism |
| VEGF-D | ↑ Higher expression [3] | Comparable | Not specifically quantified | Stimulates angiogenesis and lymphangiogenesis |
| VEGF-A | Comparable [3] | Comparable [3] | Not specifically quantified | Potent stimulator of angiogenesis |
| IL-8 | ↑ Higher expression [3] | Comparable | Not specifically quantified | Chemoattractant, promotes angiogenesis |
| Angiogenin | Comparable [3] | Comparable [3] | Not specifically quantified | Induces blood vessel formation |
| bFGF | Comparable [3] | Comparable [3] | Not specifically quantified | Broad mitogenic activity, supports angiogenesis |
| NGF | Comparable [3] | Comparable [3] | Not specifically quantified | Supports neuronal growth and survival |
| HGF | Not specifically quantified | Not specifically quantified | Distinct profile [49] | Immunomodulation and tissue repair |
These expression differences translate to functional superiority in specific assays. For instance, conditioned media from ASCs (ASC-CM) demonstrated enhanced in vitro angiogenic paracrine activity, leading to increased endothelial tubulogenesis compared to other MSC populations. This pro-angiogenic effect was largely attributed to the higher secretion of VEGF-A and VEGF-D by ASCs [3].
Diagram 2: Relationship between MSC source, paracrine factor expression, and functional outcome.
The Scientist's Toolkit: Essential Research Reagents
Successful isolation and characterization of MSCs require a suite of specialized reagents and materials. The table below lists key solutions used in the protocols cited in this guide.
Table 3: Key Research Reagent Solutions for MSC Isolation and Culture
| Reagent/Material | Function | Application Notes |
|---|---|---|
| Collagenase Type I | Enzymatic digestion of extracellular matrix in adipose tissue. | Critical for liberating ASCs in the Stromal Vascular Fraction (SVF) [49] [3]. |
| Ficoll-Paque | Density gradient medium for separation of mononuclear cells from whole blood/bone marrow. | Core reagent for isolating BMSCs and UCB-MSCs via density gradient centrifugation [70] [71]. |
| Fetal Bovine Serum (FBS) | Standard supplement for cell culture media, providing growth factors and attachment factors. | Widely used but faces regulatory concerns; potential replacement with Human Platelet Lysate (HPL) for clinical applications [70] [71]. |
| Human Platelet Lysate (HPL) | Xeno-free supplement for cell culture, rich in human growth factors. | Powerful and clinically-compliant alternative to FBS; can enhance MSC proliferation rates [70]. |
| Dulbecco's Modified Eagle's Medium (DMEM) | Basal nutrient medium for cell growth and expansion. | The foundational component of culture media for all three MSC types, supplemented with serum or HPL [71] [3]. |
| Flow Cytometry Antibodies (CD73, CD90, CD105, CD34, CD45, CD14, HLA-DR) | Immunophenotypic characterization of isolated cells. | Essential for confirming MSC identity according to ISCT criteria (positive for CD73, CD90, CD105; negative for hematopoietic markers) [71] [37]. |
| Recombinant Trypsin | Proteolytic enzyme for detaching adherent cells during subculturing. | Used for passaging cells while maintaining high viability [72]. |
Impact of Donor Age, Health Status, and Culture Conditions on Secretome Stability
The therapeutic application of the mesenchymal stromal cell (MSC) secretome—the complex mixture of proteins, growth factors, cytokines, and extracellular vesicles secreted by these cells—represents a promising cell-free paradigm in regenerative medicine [73]. The stability and composition of this secretome are critical determinants of its therapeutic efficacy, yet these properties are not fixed. They are significantly influenced by donor-specific factors such as age and tissue source, as well as by external culture conditions used during production [74] [14]. This variability presents a substantial challenge for the standardization and clinical translation of secretome-based therapies. Within the broader context of comparative paracrine factor expression research, this guide objectively analyzes experimental data on how these factors impact the stability of the secretome from adipose-derived stem cells (ASCs), bone marrow-derived MSCs (BMSCs), and umbilical cord blood-derived MSCs (UCB-MSCs). A systematic understanding of these influences is essential for researchers and drug development professionals to develop potent, consistent, and clinically viable secretome products.
Donor Age and Its Impact on Secretome Composition
Donor age is a major biological variable suspected to alter the regenerative capacity of MSCs and their secretome. While the effects are complex and can be source-dependent, proteomic analyses have identified specific age-related alterations in protein secretion.
Key Experimental Findings on Age-Related Changes
Recent studies provide a nuanced picture of how donor age influences the MSC secretome. Quantitative proteomic profiling reveals that while the global secretome profile may be stable, the abundance of specific, functionally critical proteins can be significantly modulated by age.
Table 1: Impact of Donor Age on Key Secretome Components
| MSC Source | Analyzed Factor | Change with Donor Age | Experimental Model | Citation |
|---|---|---|---|---|
| Equine BMSC/ASC | CTHRC1 & LOX | Decrease (Age-related decrease in abundance) | In vitro (LC-MS/MS) | [74] |
| Human ASC | VEGF-A | Decrease (Lower in EC/co-cultures with old ASC) | In vitro co-culture sprouting assay | [75] |
| Human ASC | FGF-2, G-CSF, HGF, IL-8, VEGF-C | No Significant Change (No significant modulation with age) | In vitro multiplex assay | [75] |
| Human ASC | Pro-angiogenic Function | No Significant Change (No impairment in vascular sprouting induction) | In vitro sprouting assay | [75] |
Experimental Protocol for Assessing Age-Related Effects
To investigate the effect of donor age on the secretome, researchers typically employ a standardized workflow involving cell isolation, culture, secretome collection, and advanced proteomic analysis.
Detailed Methodology:
- Cell Sourcing and Grouping: MSCs are isolated from healthy donors of pre-defined age groups (e.g., Young: <30 years; Old: >50 years). Tissues used include subcutaneous adipose tissue (for ASCs) [75] or bone marrow aspirates (for BMSCs) [74].
- Cell Culture Standardization: Cells from all donors are cultured under identical, standardized conditions (e.g., medium formulation, oxygen tension, seeding density, and passage number) to isolate the effect of age from culture artefacts [74] [75].
- Secretome Collection (Conditioned Media): At a predetermined passage (e.g., passage 2-3), cells are grown to 70-80% confluence. The culture medium is then replaced with a serum-free medium to avoid contamination from serum proteins. After an incubation period (typically 24-48 hours), the Conditioned Medium (CM) is collected [74] [61].
- CM Processing: The collected CM is centrifuged (e.g., 800g for 10 minutes) to remove cell debris and then concentrated using ultrafiltration devices with specific molecular weight cut-offs [61].
- Proteomic Analysis:
- LC-MS/MS (Liquid Chromatography-Tandem Mass Spectrometry): For an untargeted proteomic profile, proteins in the CM are digested into peptides, separated by liquid chromatography, and identified and quantified by tandem mass spectrometry. This approach identified the age-related decrease in CTHRC1 and LOX in equine MSCs [74].
- Multiplex Immunoassays (Luminex): For targeted quantification of specific growth factors and cytokines, multiplex bead-based arrays are used. This allows for the simultaneous measurement of dozens of pre-selected analytes (e.g., VEGF, FGF-2, HGF, IL-6, IL-8) [75].
- Functional Validation: The functional consequence of age-related secretome changes is often tested using in vitro bioassays. A common method is a vascular sprouting assay, where endothelial cells are co-cultured with MSC-derived CM or with MSCs themselves, and the total length of the formed capillary-like structures is quantified to assess pro-angiogenic potency [75].
The following workflow diagram visualizes this experimental process and the logical relationships between donor factors, processing, and analysis:
Influence of Tissue Source on Paracrine Factor Expression
The tissue of origin is a primary determinant of the MSC secretome's compositional and functional profile. Comparative analyses consistently show that ASCs, BMSCs, and UCB-MSCs exhibit distinct paracrine "signatures," which may make them uniquely suited for different therapeutic applications.
Table 2: Paracrine Factor Expression Across Different MSC Tissue Sources
| MSC Source | Key Strengths and Characteristic Factors | Supporting Evidence |
|---|---|---|
| Adipose-derived (ASC) | High Pro-angiogenic Potential. Significantly higher expression of IGF-1, VEGF-D, and IL-8. Enhanced tubulogenesis in endothelial cell assays, primarily mediated by VEGF-A and VEGF-D [3] [10]. | [3] [10] |
| Bone Marrow (BMSC) | Broad Regenerative Profile. Produces key factors like VEGF-A, Angiogenin, bFGF, and NGF at levels comparable to ASCs. Considered a standard source with well-characterized secretome [3]. | [3] |
| Umbilical Cord (UCB-MSC / WJ-MSC) | Potent Neurotrophic & Proliferative Capacity. Higher reported amounts of HGF, FGF-2, BDNF, and bNGF compared to adult sources. Excellent proliferation capacity, offering an ethically non-controversial source [76]. | [76] |
Effects of Culture Conditions and Preconditioning Strategies
The MSC secretome is highly dynamic and can be deliberately modulated through specific culture conditions and preconditioning strategies. This "priming" allows researchers to steer the secretome toward a desired therapeutic profile.
Key Preconditioning Strategies:
- Hypoxic Culture: Culturing MSCs under low oxygen tension (1-5% O₂) instead of atmospheric 21% more closely mimics their physiological niche and enhances the secretion of pro-angiogenic and regenerative factors. This is largely mediated by the stabilization of Hypoxia-Inducible Factor 1-alpha (HIF-1α), which upregulates genes like VEGF and Angiopoietin [74] [61] [14].
- 3D Culture Systems: Growing MSCs as spheroids or in hydrogels, as opposed to traditional 2D monolayers, better mimics the in vivo cellular microenvironment. This 3D format can create nutrient and oxygen gradients, leading to a secretome with enhanced anti-inflammatory and tissue-regenerative properties [61].
- Inflammatory Priming (Licensing): Exposing MSCs to low doses of inflammatory cytokines such as IFN-γ and TNF-α potently enhances their immunomodulatory function. This "licensing" boosts the secretion of anti-inflammatory factors like Prostaglandin E2 (PGE2), IL-6, TGF-β, and HGF, which promote M2 macrophage activation and suppress detrimental immune responses [61] [14].
The diagram below illustrates how these preconditioning stimuli are sensed by the MSC and translated into a modified secretome through key intracellular signaling pathways:
Storage Conditions and Long-Term Stability of Secretome
For clinical translation, the long-term stability of the secretome product is paramount. Lyophilization (freeze-drying) is a key method for preserving the secretome, but the storage conditions post-lyophilization critically impact the stability of its bioactive components.
Quantitative Data on Storage Stability
Research indicates that storage temperature and the use of stabilizing agents are crucial factors in maintaining the integrity of the lyophilized secretome over time.
Table 3: Stability of Lyophilized MSC Secretome Components Under Different Storage Conditions
| Storage Duration | Storage Temperature | Key Changes in Secretome Composition | Effect of Trehalose Supplementation | Citation |
|---|---|---|---|---|
| 3 Months | -80°C | Excellent preservation: >80% of all evaluated components retained. | Not Required | [76] |
| -20°C | Significant decrease in BDNF and bNGF. | Not Reported | [76] | |
| +4°C / RT | Pronounced decrease in BDNF, bNGF, and sVCAM-1. | Improved outcome, stabilizing components at 4°C and RT. | [76] | |
| 30 Months | -80°C | Good preservation: >70% of components retained; only viable option. | Not Required | [76] |
| -20°C | Significant decrease in BDNF, bNGF, and VEGF-A. | Not Reported | [76] | |
| +4°C / RT | Severe degradation of BDNF, bNGF, VEGF-A, IL-6, and sVCAM-1. | Partially protective, but degradation still significant. | [76] |
The Scientist's Toolkit: Essential Reagents and Materials
Standardizing secretome production and analysis requires a suite of specific research tools and reagents. The following table details key solutions used in the featured experiments.
Table 4: Research Reagent Solutions for Secretome Studies
| Reagent / Material | Function in Secretome Research | Example Application |
|---|---|---|
| Collagenase Type I/II | Enzymatic digestion of tissues to isolate MSCs (ASCs, BMSCs). | Isolation of ASCs from lipoaspirate or adipose tissue [3] [75]. |
| Serum-Free Medium | Production of conditioned medium free of confounding serum proteins. | Essential step during secretome collection to ensure all analyzed proteins are cell-derived [61] [77]. |
| Ultrafiltration Devices | Concentration of conditioned media and buffer exchange. | Preparing protein-rich secretome samples for proteomic analysis [61]. |
| Trehalose | Stabilizing agent (cryoprotectant/lyoprotectant) for biomolecules. | Added to secretome prior to lyophilization to protect protein structure and stability during storage [76]. |
| Luminex Multiplex Assays | High-throughput, simultaneous quantification of multiple cytokines/growth factors. | Targeted quantification of dozens of pre-selected analytes in secretome samples [76] [75]. |
| LC-MS/MS System | Untargeted identification and quantification of proteins in a complex mixture. | Global proteomic profiling of the secretome to discover novel components and differences [74]. |
The stability and compositional profile of the MSC secretome are not intrinsic properties but are dynamically shaped by a confluence of factors. Donor age induces specific, quantifiable changes in proteins critical for tissue repair, such as CTHRC1 and LOX, though the functional impact on processes like angiogenesis may be variable or source-dependent [74] [75]. The tissue source itself is a primary driver of variability, with ASCs exhibiting a strong pro-angiogenic profile, while UCB-MSCs offer a potent neurotrophic signature [3] [76]. Furthermore, strategic preconditioning through hypoxia, 3D culture, or inflammatory priming provides a powerful means to intentionally steer the secretome toward a desired therapeutic outcome [61] [14]. Finally, the long-term stability of these carefully crafted secretomes is critically dependent on storage conditions, with -80°C being essential for preserving lyophilized products over extended periods [76]. For researchers, this underscores the necessity of rigorous donor screening, standardized and optimized culture protocols, and controlled storage logistics to ensure the consistent production of a potent and reliable secretome-based therapeutic product.
The transition of mesenchymal stem cells (MSCs) from laboratory research to large-scale clinical production hinges on overcoming two critical, and often interconnected, challenges: maintaining robust proliferation capacity and managing replicative senescence. Cellular senescence, a state of irreversible cell cycle arrest, poses a significant barrier to producing the vast quantities of cells required for therapeutic applications [78]. This process is characterized not only by proliferation cessation but also by altered morphology, increased granularity, and a pro-inflammatory secretory phenotype known as the Senescence-Associated Secretory Phenotype (SASP) [78]. The selection of optimal MSC sources is therefore paramount for developing efficient and economically viable bioprocesses. This guide provides a objective comparison of the proliferation and senescence characteristics of three prominent MSC types—Adipose-Derived Stem Cells (ASCs), Bone Marrow-MSCs (BM-MSCs), and Umbilical Cord Blood-MSCs (UCB-MSCs)—within the broader context of comparative paracrine factor expression research.
The biological characteristics of MSCs are profoundly influenced by their tissue of origin. The table below provides a consolidated comparison of ASCs, BM-MSCs, and UCB-MSCs based on key parameters critical for large-scale production.
Table 1: Biological Characteristics of Different MSC Sources
| Parameter | Adipose-Derived Stem Cells (ASCs) | Bone Marrow-MSCs (BM-MSCs) | Umbilical Cord Blood-MSCs (UCB-MSCs) |
|---|---|---|---|
| Tissue Accessibility | High; minimally invasive harvest, abundant tissue [22] | Low; highly invasive, painful harvest [22] | Moderate; non-invasive, but dependent on donor availability [49] |
| Initial Cell Frequency | High (~2% of stromal vascular fraction) [22] | Very Low (~0.002% of nucleated cells) [22] | Low frequency in cord blood [22] |
| Proliferation Rate | High [49] | Lower; decreases significantly with age and passage [22] | Highest reported; superior clonogenicity [22] |
| Senescence Markers | — | High expression of p53, p21, p16 at later passages [22] | Lower expression of p53, p21, p16; more primitive state [22] |
| Key Paracrine Factors | ↑ IGF-1, VEGF-D, IL-8 [3] [10] | Inflammatory profile (IL-1α, IL-6, IL-8, TNF-α, TGF-β1) [22] | Broad spectrum of growth factors/cytokines [22] |
Experimental Assessment of Proliferation and Senescence
Standardized experimental protocols are essential for generating comparable data on MSC expansion potential and senescence onset. The following methodologies are routinely employed in the field.
Assessing Proliferation Capacity
Protocol: Growth Curve Analysis using Cell Counting Kit-8 (CCK-8) [49]
- Cell Seeding: Harvest MSC populations (ASCs, BM-MSCs, UCB-MSCs) at passage 3 by trypsinization. Adjust cell concentration to 2.0 × 10⁴ cells/mL in complete culture medium. Seed 100 µL of this suspension into each well of a 96-well plate.
- Incubation and Daily Measurement: Incubate the seeded plate under standard conditions (37°C, 5% CO₂). For nine consecutive days, select five experimental wells and one control well (medium only) daily.
- Viability Staining: Add 10 µL of CCK-8 solution to each selected well. Incubate the plate for 2 hours to allow for the formation of formazan dye by viable, metabolically active cells.
- Absorbance Measurement: Measure the absorbance of each well at 450 nm using an enzyme immunoassay analyzer. The absorbance value is directly proportional to the number of viable cells.
- Data Analysis: Calculate the mean absorbance for the five wells each day. Plot the mean absorbance against time to generate a growth curve for each MSC type, allowing for the comparison of population doubling times and saturation densities.
Assessing Senescence Status
Protocol: Flow Cytometric Analysis of Apoptosis (Annexin V/PI Staining) [49]
This protocol assesses the antiapoptotic ability of MSCs, which can be altered in senescent cultures.
- Cell Preparation and Treatment: Harvest MSCs at passage 3 and seed them in 6-well plates at a density of 3 × 10⁵ cells/mL. Incubate until 80% confluency is reached.
- Senescence Induction: To induce stress, add the apoptosis-inducing agent dexamethasone to the culture medium at a final concentration of 1 × 10⁻⁶ mol/L. Incubate the cells for 48 hours.
- Cell Staining: Collect the cells by trypsinization and resuspend them in 500 µL of a specific binding buffer. Add 5 µL of Annexin V-FITC and 5 µL of Propidium Iodide (PI) to the cell suspension. Mix gently and incubate in the dark at room temperature for 5-15 minutes.
- Flow Cytometry: Analyze the stained cells using a flow cytometer (e.g., BD FACSAria). The analysis distinguishes between:
- Viable cells: Annexin V⁻/PI⁻
- Early apoptotic cells: Annexin V⁺/PI⁻
- Late apoptotic/necrotic cells: Annexin V⁺/PI⁺ A lower percentage of apoptotic cells in a population indicates a stronger antiapoptotic ability, a feature that can be associated with senescence evasion.
Signaling Pathways in Senescence
The entry into senescence is primarily regulated by two key tumor suppressor pathways. The diagram below illustrates the core components and their interactions in response to cellular stressors.
Figure 1: Core Senescence Signaling Pathways. The p53/p21 and p16/Rb pathways are activated by cellular stressors, leading to cell cycle arrest and the Senescence-Associated Secretory Phenotype (SASP) [78].
The Scientist's Toolkit: Essential Research Reagents
Successful experimentation in this field relies on a suite of well-defined reagents and tools. The following table details key solutions used in the featured protocols and broader senescence research.
Table 2: Essential Research Reagents for MSC Proliferation and Senescence Studies
| Reagent / Kit | Primary Function | Experimental Context |
|---|---|---|
| Collagenase Type I | Tissue digestion and isolation of primary MSCs from adipose tissue or umbilical cord Wharton's Jelly [49]. | Initial cell isolation and culture establishment. |
| Cell Counting Kit-8 (CCK-8) | Colorimetric assay to quantify cell viability and proliferation based on metabolic activity [49]. | Generating growth curves for proliferation analysis. |
| Annexin V-FITC / PI Apoptosis Kit | Fluorescent staining to distinguish between viable, early apoptotic, and late apoptotic/necrotic cells via flow cytometry [49]. | Assessing antiapoptotic ability and cell death in senescence studies. |
| SA-β-Gal Staining Kit | Histochemical detection of β-galactosidase activity at pH 6.0, a common biomarker for senescent cells [78] [79]. | Identifying and quantifying senescent cells in a culture. |
| Dexamethasone | A synthetic glucocorticoid used as an inducer of cellular stress and apoptosis in experimental models [49]. | Studying cellular response to stress and evaluating antiapoptotic pathways. |
| CD105, CD73, CD90 Antibodies | Positive immunophenotypic surface markers used to identify MSCs as per International Society for Cellular Therapy (ISCT) criteria [80] [22]. | Confirming MSC identity and purity through flow cytometry. |
The choice of MSC source presents a critical trade-off for process development in large-scale clinical production. ASCs offer a compelling combination of high accessibility, favorable initial yield, and a strong pro-angiogenic paracrine profile, making them a robust candidate for processes requiring large cell quantities [3] [22]. In contrast, UCB-MSCs demonstrate superior proliferation capacity and lower senescence marker expression, suggesting an advantage for achieving the highest possible population doublings, though their sourcing can be more limited [22]. BM-MSCs, while the historical gold standard, are hampered by invasive harvesting and a pronounced tendency for age- and passage-dependent senescence, posing significant challenges for scalable manufacturing [22]. A comprehensive understanding of the proliferation, senescence, and paracrine factor expression profiles of each MSC type is indispensable for designing optimized bioreactor processes, media formulations, and quality control checks. This ensures the consistent production of high-quality, potent MSC therapies for clinical application.
Mesenchymal stromal cell (MSC)-based therapies represent a rapidly advancing frontier in regenerative medicine and drug development. However, their clinical translation has been hampered by significant challenges in standardization, primarily stemming from the inherent heterogeneity of MSC populations and variations in culture methods. This heterogeneity manifests across multiple dimensions: tissue sources (adipose tissue, bone marrow, umbilical cord), donor-specific factors (age, health status), and manufacturing protocols (culture supplements, expansion techniques). Such variability directly impacts the therapeutic efficacy and consistency of MSC products, making comparative analysis of their functional capacities an essential pursuit for the field [81] [82].
A paradigm shift in understanding MSC mechanisms has redirected focus from cellular differentiation and engraftment toward paracrine signaling as the primary driver of their therapeutic effects. The MSC "secretome" – comprising growth factors, cytokines, chemokines, and extracellular vesicles – mediates most observed benefits in preclinical and clinical studies [83]. Consequently, understanding and comparing the paracrine factor expression profiles across different MSC sources provides critical insights for product standardization and appropriate source selection for specific clinical applications.
Tissue-Source Variations
MSCs can be isolated from numerous tissues, each imparting distinct functional characteristics. Bone marrow-derived MSCs (BM-MSCs) represent the most extensively studied type, while adipose-derived MSCs (ASCs) and umbilical cord-derived MSCs (UC-MSCs) have gained prominence for their accessibility and robust proliferation [81]. Single-cell transcriptomic analyses reveal that these source-dependent differences extend to fundamental cellular identity, with MSCs lacking expression of core stemness genes (SOX2, NANOG, POU5F1) found in pluripotent stem cells, instead expressing distinct functional genes like TMEM119 and FBLN5 [84].
Research indicates that UC-MSCs often exhibit enhanced proliferation capacity and lower immunogenicity compared to adult tissue-derived MSCs [81] [63]. ASCs demonstrate superior yields from isolation procedures and have shown particularly strong angiogenic paracrine activity [3]. A transcriptome comparison of giant panda MSCs revealed that UC-MSCs and BM-MSCs exhibit distinct expression patterns, with UC-MSCs showing upregulated pathways related to proliferation and immunomodulation [85].
Donor-Associated Heterogeneity
Even within the same tissue source, significant inter-individual variation exists. Donor age represents a critical factor, with MSCs from neonatal tissues (umbilical cord, placenta) demonstrating longer telomeres, enhanced proliferation capacity, and reduced senescence compared to those from adult donors [81] [82]. Aging correlates with MSC functional decline, including reduced differentiation potential and altered secretome profiles [82]. Additional donor factors including sex, body mass index, and underlying health conditions further contribute to heterogeneity by influencing MSC phenotype, morphology, and functional capabilities [81].
Culture Method-Induced Heterogeneity
Manufacturing protocols introduce substantial variability through several technical parameters. Culture medium composition – particularly the type of serum supplement (fetal bovine serum versus human platelet lysate versus serum/xeno-free formulations – significantly impacts MSC characteristics and secretome composition [86]. Studies demonstrate that while BMSCs expanded in different media (FBS, platelet lysate, or serum/xeno-free conditions) share core molecular fingerprints, their extracellular vesicle cargo and anti-inflammatory potency exhibit subtle but potentially important differences [86].
Other manufacturing variables including cell seeding density, passage number, oxygen tension, and harvesting techniques further contribute to product heterogeneity. Even MSC populations originating from single-cell clones develop functional heterogeneity over time in culture [81].
Comparative Analysis of Paracrine Factor Expression
Experimental Methodology for Secretome Analysis
Cell Sourcing and Culture Conditions: In comparative studies, MSCs are typically isolated from adipose tissue (via collagenase digestion and centrifugation), bone marrow (through density gradient separation and plastic adherence), and umbilical cord (using explant culture or enzymatic digestion) [3]. To ensure valid comparisons, cells from different sources are cultured under identical conditions – same basal medium, serum lot, oxygen tension (typically 5% CO2 at 37°C), and passage range (usually P3-P6) [3].
Secretome Collection: Upon reaching 70-80% confluence, cells are thoroughly washed to remove serum contaminants and incubated in serum-free medium for 24-48 hours. Conditioned media (CM) is then collected and sequentially centrifuged (300-4000 × g) to remove cellular debris, followed by filtration (0.22 μm) and storage at -80°C until analysis [3] [86].
Analytical Techniques:
- mRNA Analysis: Quantitative RT-PCR assesses gene expression of paracrine factors.
- Protein quantification: ELISA and multiplex immunoassays measure secreted protein levels.
- Functional assays: Endothelial tube formation assays assess angiogenic potential; lymphocyte proliferation assays evaluate immunomodulatory activity.
- Proteomic profiling: LC-MS/MS provides comprehensive protein identification and quantification.
- Extracellular vesicle analysis: Nanoparticle tracking analysis characterizes vesicle size and concentration, while miRNA sequencing profiles cargo content [3] [86] [83].
Quantitative Comparison of Key Paracrine Factors
Table 1: Comparative Expression of Paracrine Factors Across MSC Sources
| Paracrine Factor | Function | ASCs | BM-MSCs | UC-MSCs | Assessment Method |
|---|---|---|---|---|---|
| VEGF-A | Angiogenesis | +++ | +++ | +++ | mRNA, CM protein [3] |
| VEGF-D | Angiogenesis, lymphangiogenesis | +++ | + | ++ | mRNA [3] |
| IGF-1 | Cell survival, proliferation | +++ | + | ++ | mRNA [3] |
| IL-8 | Angiogenesis, chemotaxis | +++ | + | ++ | mRNA [3] |
| Angiogenin | Angiogenesis | +++ | +++ | +++ | CM protein [3] |
| bFGF | Angiogenesis, tissue repair | +++ | +++ | +++ | mRNA [3] |
| NGF | Neural growth, survival | +++ | +++ | +++ | mRNA [3] |
| Leptin | Metabolism, angiogenesis | + | + | ++ | CM protein [3] |
| HGF | Angiogenesis, antifibrosis | ++ | ++ | ++ | Literature report [83] |
| TGF-β1 | Immunomodulation, fibrosis | ++ | ++ | ++ | Literature report [83] |
| SDF-1 | Stem cell homing, angiogenesis | ++ | ++ | ++ | Literature report [83] |
Expression levels are summarized as relative comparisons between MSC sources based on experimental data. CM = Conditioned Media.
Table 2: miRNA Cargo Profile in MSC-Derived Extracellular Vesicles
| miRNA | Function | ASCs | BM-MSCs | UC-MSCs | Therapeutic Association |
|---|---|---|---|---|---|
| miR-21 | Angiogenesis, immunomodulation | ++ | ++ | ++ | Enhanced tube formation [83] |
| miR-23a | Angiogenesis | ++ | + | ++ | Vascular development [83] |
| miR-125b | Antifibrosis | ++ | ++ | + | Attenuates fibrosis [83] |
| miR-29 | Antifibrosis | ++ | ++ | ++ | Collagen repression [83] |
| miR-146a | Immunomodulation | ++ | ++ | ++ | Anti-inflammatory [83] |
Functional Correlations of Secretome Profiles
The distinct molecular signatures across MSC sources translate to functionally different therapeutic potentials. ASCs demonstrate superior pro-angiogenic capabilities, with their conditioned media inducing significantly greater endothelial tube formation compared to other MSC sources [3]. Neutralization experiments identified VEGF-A and VEGF-D as major contributors to this enhanced angiogenic activity [3].
BM-MSCs exhibit robust immunomodulatory properties, characterized by strong expression of factors like indoleamine 2,3-dioxygenase (IDO) and TNF-α-stimulated gene 6 (TSG-6) [83] [63]. UC-MSCs often display an intermediate profile with strong proliferation capacity and balanced paracrine activity, making them attractive for allogeneic applications [81] [63].
Signaling Pathways Governing Paracrine Expression
The molecular mechanisms underlying paracrine factor regulation in MSCs involve several key signaling pathways that respond to environmental cues and intrinsic programming.
Diagram 1: Key Signaling Pathways Regulating MSC Paracrine Expression
The PI3K/Akt pathway emerges as a central regulator of MSC survival, proliferation, and paracrine function, particularly in response to growth factors and hypoxic conditions [85] [83]. HIF-1α activation under low oxygen tension upregulates angiogenic factors like VEGF, SDF-1, and IL-8 [83]. NF-κB signaling mediates inflammatory priming, enhancing production of immunomodulatory molecules including IDO, TSG-6, and PGE2 when MSCs encounter IFN-γ and TNF-α [87]. Additionally, mechanotransduction pathways like YAP/TAZ respond to matrix stiffness and regulate ECM remodeling components that influence secretory profiles [83].
Strategies to Overcome Heterogeneity Challenges
Manufacturing Standardization Approaches
Addressing MSC heterogeneity requires integrated strategies across the manufacturing pipeline. Donor screening and selection criteria represent the first critical control point, with careful attention to age, health status, and tissue source selection based on intended therapeutic application [81] [82].
Culture protocol harmonization can significantly reduce technical variability. Defined culture media formulations – particularly serum/xeno-free alternatives – enhance reproducibility and safety profiles compared to traditional FBS-containing media [86]. Functional potency assays that measure specific secretome components or biological activities (e.g., angiogenic potential, immunomodulatory capacity) provide more relevant quality metrics than surface marker expression alone [81] [3].
Advanced approaches include MSC pooling strategies that combine cells from multiple donors to create more consistent products with averaged biological properties [81]. Inflammatory priming with IFN-γ and/or TNF-α can enhance immunomodulatory potency while reducing heterogeneity in MSC responses to inflammatory environments [87].
Analytical Framework for MSC Characterization
A comprehensive analytical framework should integrate multiple assessment modalities:
- Surface marker profiling (CD73, CD90, CD105 positive; CD45, CD34, HLA-DR negative)
- Trilineage differentiation potential (adiopogenic, osteogenic, chondrogenic)
- Secretome analysis (growth factors, cytokines, extracellular vesicles)
- Functional potency assays (immunomodulation, angiogenesis, tissue repair)
- Molecular profiling (transcriptomics, proteomics) to identify batch-to-batch variations [81] [82] [84]
Single-cell RNA sequencing technologies now enable unprecedented resolution in characterizing MSC heterogeneity and identifying distinct functional subpopulations [84] [87].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for MSC Secretome Studies
| Reagent/Category | Specific Examples | Research Function | Considerations |
|---|---|---|---|
| Culture Media | DMEM/F12, MEM Alpha, StemPro MSC SFM XenoFree | Cell expansion and maintenance | Serum/xeno-free formulations enhance reproducibility [86] |
| Serum Supplements | Fetal Bovine Serum (FBS), Human Platelet Lysate | Provides growth factors and adhesion proteins | Platelet lysate reduces xenogenic risks [86] |
| Dissociation Reagents | TrypLE Express, Collagenase | Cell passaging and tissue digestion | Enzyme selection affects viability and function [3] [84] |
| Characterization Antibodies | CD73, CD90, CD105, CD45, CD34, HLA-DR | Surface marker verification by flow cytometry | Essential for ISCT criteria confirmation [86] [82] |
| Priming Cytokines | IFN-γ, TNF-α | Enhance immunomodulatory potency | Concentration and timing require optimization [87] |
| Secretome Analysis Tools | ELISA kits, Multiplex arrays, LC-MS/MS, miRNA sequencing | Quantify paracrine factors and EV cargo | Multi-platform approach recommended [3] [86] [83] |
| Functional Assay Kits | Endothelial tube formation, Lymphocyte proliferation | Assess biological potency | Correlate molecular data with function [3] |
The journey toward standardized MSC-based therapies requires acknowledging and systematically addressing the inherent heterogeneity of these complex cellular products. The comparative analysis of paracrine factor expression across ASCs, BM-MSCs, and UC-MSCs reveals both challenges and opportunities – while consistency remains elusive, the distinct secretory profiles of different MSC sources may be strategically matched to specific clinical applications. ASCs demonstrate particular promise for angiogenic applications, while BM-MSCs and UC-MSCs may be preferred for immunomodulatory indications.
Future standardization efforts should integrate advanced analytical technologies, particularly single-cell omics and functional potency assays, with manufacturing strategies like donor pooling and cytokine priming. Such approaches will ultimately enhance the consistency, efficacy, and predictability of MSC-based therapies, fulfilling their potential to transform treatment paradigms across regenerative medicine, immunology, and drug development.
The therapeutic potential of mesenchymal stem cells (MSCs) is largely attributed to their paracrine activity—the secretion of bioactive factors that promote tissue repair, modulate immune responses, and enhance angiogenesis [88] [63]. However, the clinical efficacy of native MSCs can be limited by their poor survival after transplantation and insufficient production of these therapeutic factors in diseased microenvironments [89] [90]. To overcome these limitations, researchers have developed two primary strategies for enhancing MSC potency: hypoxic preconditioning, a non-genetic approach that primes cells by exposing them to sub-lethal oxygen deprivation, and genetic modification, which involves directly altering the MSC genome to enhance the expression of desirable factors [91] [92] [90].
The comparative effectiveness of these strategies is particularly evident when examined through the lens of paracrine factor expression across different MSC sources. Adipose-derived stem cells (ASCs), bone marrow-derived MSCs (BMSCs), and umbilical cord blood-derived MSCs (UCB-MSCs) each possess distinct inherent paracrine profiles that influence their responsiveness to potency enhancement strategies [3] [10]. This guide provides a comparative analysis of hypoxic preconditioning and genetic modification, offering experimental data, methodologies, and practical tools for researchers seeking to optimize MSC therapeutic efficacy.
Different MSC sources exhibit distinct inherent paracrine expression profiles, which form the baseline upon which enhancement strategies operate. Understanding these inherent differences is crucial for selecting the appropriate MSC source for specific therapeutic applications.
Table 1: Comparative Basal Paracrine Factor Expression Across MSC Sources
| Paracrine Factor | ASCs | BMSCs | UCB-MSCs | Functional Significance |
|---|---|---|---|---|
| VEGF-A | Comparable | Comparable | Comparable | Angiogenesis, cell survival |
| VEGF-D | Higher | Lower | Not specified | Angiogenesis, lymphangiogenesis |
| Angiogenin | Comparable | Comparable | Not specified | Angiogenesis, ribonuclease activity |
| IGF-1 | Higher | Lower | Not specified | Cell growth, proliferation |
| IL-8 | Higher | Lower | Not specified | Neutrophil chemotaxis, angiogenesis |
| bFGF | Comparable | Comparable | Not specified | Mitogenesis, angiogenesis |
| NGF | Comparable | Comparable | Not specified | Neuronal growth, survival |
| HGF | Not specified | Not specified | Higher [93] | Anti-fibrotic, mitogenic |
mRNA expression analysis identified IGF-1, VEGF-D, and IL-8 at higher levels in ASCs compared to other MSC populations, while VEGF-A, angiogenin, bFGF, and NGF were expressed at comparable levels [3] [10]. Functional assays demonstrated that ASC-conditioned media resulted in increased tubulogenic efficiency compared to other MSC populations, establishing ASCs as particularly suitable for angiogenesis-dependent applications [3].
Hypoxic Preconditioning: Methodology and Outcomes
Hypoxic preconditioning involves exposing MSCs to controlled, sub-lethal oxygen deprivation before transplantation. This stress triggers adaptive cellular responses, primarily mediated by hypoxia-inducible factors (HIFs), which enhance the cells' survival and therapeutic potential upon encountering the harsh conditions of damaged tissues [93] [89].
Experimental Protocols for Hypoxic Preconditioning
Standardized protocols are essential for achieving reproducible and effective hypoxic preconditioning:
- Equipment Setup: Utilize specialized incubators capable of maintaining precise low-oxygen environments. The Heraeus HERAcell 150 tri-gas humidified incubator can maintain 1% O₂, while systems like the GENbox Jar (BioMerieux) or Anaero Pouch–Anaero (Mitsubishi Gas Chemical Company Inc.) can achieve severe hypoxia (<0.1% O₂) [91] [89].
- Cell Culture Conditions: Seed MSCs at a density of 5×10³ cells per cm² and culture until 80% confluent. Replace growth medium with serum-free medium immediately before hypoxia exposure to synchronize cell responses [91].
- Preconditioning Parameters: Expose cells to hypoxia for varying durations (typically 12-72 hours) at different oxygen concentrations (0.1%-5% O₂). Optimal parameters depend on MSC source and application, with 24 hours at <0.1% O₂ effectively enhancing angiogenic paracrine activity in ASCs [91].
- Conditioned Media Collection: Following preconditioning, collect media and centrifuge at 875×g for 10 minutes to remove cell debris. Filter through a 0.2-μm filter and concentrate using centrifugal filter columns with 3-kDa molecular weight cutoff (e.g., Amicon Ultra-15) [91].
Signaling Pathways in Hypoxic Preconditioning
The following diagram illustrates the key molecular pathways activated during hypoxic preconditioning that lead to enhanced MSC survival and paracrine function:
Quantitative Outcomes of Hypoxic Preconditioning
Hypoxic preconditioning consistently enhances the production of key paracrine factors across MSC types, though the magnitude of enhancement varies by cell source and specific factor.
Table 2: Paracrine Factor Enhancement Through Hypoxic Preconditioning
| Paracrine Factor | MSC Source | Hypoxic Condition | Fold Increase | Experimental Model |
|---|---|---|---|---|
| VEGF-A | ASCs | <0.1% O₂, 24h | Significant (protein) [91] | In vitro ELISA |
| VEGF-A | ASCs | <0.1% O₂, 24h | ~2.5x (mRNA) [91] | In vitro qPCR |
| Angiogenin | ASCs | <0.1% O₂, 24h | Significant (protein) [91] | In vitro Western blot |
| Angiogenin | ASCs | <0.1% O₂, 24h | ~3x (mRNA) [91] | In vitro qPCR |
| SDF-1a | ASCs | <0.1% O₂, 24h | Significant (protein) [89] | In vitro ELISA |
| Angiogenic Effect | ASCs | <0.1% O₂, 24h | 6.0% vs 4.1% vascular volume [91] | In vivo mouse sponge implant |
| Cellular Viability | ASCs | <0.1% O₂, 24h preconditioning | Significant enhancement [89] | Post-OGD exposure |
Hypoxic preconditioning of ASCs (<0.1% O₂, 24 hours) significantly increased angiogenesis in vivo, with vascular volume of 6.0%±0.5% compared to 4.1%±0.7% in normoxic controls [91]. Antibody neutralization studies confirmed that VEGF-A and angiogenin were critical mediators of this enhanced angiogenic response [91].
Genetic Modification: Approaches and Efficacy
Genetic modification encompasses various techniques to alter the MSC genome, enhancing their inherent therapeutic properties by enabling sustained overexpression of specific beneficial factors.
Genetic Engineering Methods for MSCs
Multiple vector systems are employed for MSC genetic modification, each with distinct advantages and limitations:
- Viral Vector Systems: Lentiviral and retroviral vectors are most prevalent due to high transduction efficiency (~90%) and stable long-term transgene expression [92] [90]. Adenoviral vectors offer high efficiency but generate transient expression and higher immunogenicity. Adeno-associated viruses (AAV) provide low immunogenicity but reduced consistency in proliferating cells [90].
- Non-Viral Methods: These include physical methods (electroporation, nucleofection, sonoporation) and chemical methods (lipidic agents, polymers, inorganic nanoparticles). While offering better safety profiles and large-scale manufacturability, non-viral methods typically show lower efficiency and transient transgene expression [90].
- Gene Editing Technologies: Emerging tools like CRISPR/Cas9, ZFNs, and TALENs enable site-specific gene integration or knockout. These systems improve safety by directing integration to specific genomic locations but require further optimization for clinical MSC applications [90].
Genetic Modification Workflow
The following diagram outlines the key decision points and processes in the genetic modification of MSCs:
Therapeutic Applications of Genetically Modified MSCs
Genetic engineering enables customized MSC therapies for specific disease targets by selectively overexpressing appropriate therapeutic factors.
Table 3: Genetic Modification Strategies and Therapeutic Outcomes
| Transgene | MSC Source | Vector | Disease Model | Therapeutic Outcome |
|---|---|---|---|---|
| VEGF-A | BMSCs | Adenovirus | Myocardial infarction | Improved cardiac function, increased vascular density [91] |
| Angiopoietin-1 | BMSCs | Retrovirus | Myocardial infarction | Improved cardiac function, enhanced angiogenesis [91] |
| BDNF | UCB-MSCs | Lentivirus | Intraventricular hemorrhage | Reduced hydrocephalus, improved myelination [88] |
| VEGF-A | ASCs | Not specified | Hyperoxic lung injury | Attenuated impaired alveolarization and angiogenesis [88] |
| BMP-2 | BMSCs | Adenovirus | Bone defect | Enhanced bone regeneration [92] |
| IFN-β | ASCs | Retrovirus | Melanoma, glioma | Inhibition of tumor growth [92] |
A systematic review of 85 studies revealed that genetically engineered MSCs showed significant therapeutic enhancement in 39 studies, moderate effects in 33 studies, and no effect in one human trial [92]. Research has predominantly focused on cancer treatment (51 studies), with the remainder addressing various non-tumor conditions including bone defects, neurological disorders, and cardiovascular diseases [92].
Direct Comparison: Hypoxic Preconditioning vs. Genetic Modification
When selecting a potency enhancement strategy, researchers must consider multiple factors including efficacy, safety, technical complexity, and regulatory pathway.
Table 4: Strategic Comparison of Enhancement Approaches
| Parameter | Hypoxic Preconditioning | Genetic Modification |
|---|---|---|
| Mechanism | Physiological activation of endogenous cellular response pathways | Introduction of exogenous genes for sustained factor expression |
| Technical Complexity | Low to moderate (requires controlled environment equipment) | High (vector design, production, safety testing) |
| Regulatory Path | More straightforward (non-genetic modification) | Complex (gene therapy regulations) |
| Safety Profile | Favorable (natural adaptive responses) | Higher risk (insertional mutagenesis, immune reactions) |
| Therapeutic Persistence | Transient effect (days to weeks) | Long-term, potentially permanent |
| Factor Specificity | Broad spectrum (multiple pathways activated) | Highly specific (targeted factor expression) |
| Clinical Translation | Faster pathway (multiple clinical trials ongoing) | Slower pathway (safety concerns to address) |
| Cost Considerations | Lower implementation cost | High development and production costs |
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of MSC enhancement strategies requires specific laboratory reagents and systems. The following table details essential research solutions for conducting experiments in this field.
Table 5: Essential Research Reagents for MSC Potency Enhancement Studies
| Reagent/Category | Specific Examples | Research Function |
|---|---|---|
| Hypoxia Systems | HERAcell tri-gas incubator, GENbox Jar, Anaero Pouch | Create controlled low-oxygen environments for preconditioning |
| Cell Culture Media | DMEM-low glucose, fetal calf serum, antibiotic-antimycotic | MSC expansion and maintenance under defined conditions |
| Analysis Kits | Live/Dead Viability/Cytotoxicity Kit, TaqMan Assay-On-Demand primers | Assess cell viability and quantitative gene expression |
| Protein Assays | Quantikine ELISA kits (VEGF-A), bicinchoninic acid (BCA) protein assay | Quantify secreted paracrine factors in conditioned media |
| Neutralizing Antibodies | Anti-VEGF-A, anti-angiogenin, anti-SDF-1a | Functionally validate specific paracrine factor contributions |
| Viral Vectors | Lentiviral, retroviral, adenoviral constructs | Deliver genetic material for sustained transgene expression |
| Non-Viral Transfection | Electroporation systems, lipid nanoparticles | Introduce genetic material without viral vectors |
| Angiogenesis Assays | Matrigel plug assay, endothelial tube formation | Evaluate functional outcomes of enhanced paracrine secretion |
Both hypoxic preconditioning and genetic modification offer effective pathways to enhance the therapeutic potency of MSCs through distinct mechanisms. Hypoxic preconditioning provides a physiologically relevant, safer approach that enhances multiple paracrine factors simultaneously, making it particularly suitable for applications requiring complex, multifaceted healing responses. Genetic modification enables highly specific, potent enhancement of selected factors, offering advantages for conditions requiring sustained, high-level expression of particular therapeutic proteins.
The selection between these strategies should be guided by the specific therapeutic application, regulatory considerations, and risk-benefit analysis. For many research and clinical applications, hypoxic preconditioning offers a more immediately translatable approach, while genetic modification holds promise for conditions requiring highly targeted, potent interventions. Future research may explore sequential or combined approaches, leveraging the advantages of both strategies to further advance the field of regenerative medicine.
Functional Validation and Direct Comparisons: Angiogenic Potential, Cytoprotection, and Immunomodulation
Within the evolving field of regenerative medicine, the therapeutic potential of mesenchymal stem cells (MSCs) is increasingly attributed to their paracrine activity—the secretion of a complex mixture of growth factors, cytokines, and extracellular vesicles that orchestrate tissue repair and angiogenesis [88]. While MSCs can be isolated from numerous tissues, those from adipose tissue (ASCs), bone marrow (BMSCs), and umbilical cord blood (UCB-MSCs) are among the most extensively studied. The central thesis of comparative paracrine factor expression research posits that the tissue-specific origin of MSCs dictates a unique secretory signature, which in turn leads to significant functional differences in angiogenic potency. This guide provides a direct, data-driven comparison of these cell types, synthesizing evidence from key in vitro and in vivo studies to inform the decisions of researchers and therapy developers.
In Vitro Paracrine Factor Secretion Profiles
A direct head-to-head comparison of conditioned media from different human MSC types revealed distinct secretomes. Quantitative analysis via ELISA shows notable differences in the concentration of key angiogenic growth factors produced under standardized culture conditions [94].
Table 1: Quantitative Comparison of Key Angiogenic Factor Secretion by Different MSC Types (in vitro)
| Growth Factor | ASCs | BMSCs | Cardiosphere-Derived Cells (CDCs) | BM-MNCs |
|---|---|---|---|---|
| Vascular Endothelial Growth Factor (VEGF) | High | Moderate | Highest | Low |
| Hepatocyte Growth Factor (HGF) | High | High | Highest | Low |
| Insulin-like Growth Factor-1 (IGF-1) | High [3] | Moderate | High | Low |
| Fibroblast Growth Factor-2 (FGF-2) | High | High | High | Low |
| Angiopoietin-2 | Data Incomplete | Data Incomplete | Highest | Data Incomplete |
| Overall Angiogenic Potential | High | Moderate | Highest | Low |
Beyond individual factors, the functional consequence of these secretomes was assessed using an in vitro tube formation assay, a standard test for angiogenic potency. When cultured on an extracellular matrix, CDCs demonstrated the greatest capacity to form tubular networks, followed by ASCs and then BMSCs. BM-MNCs showed negligible activity in this assay [94]. This functional data correlates with the quantitative factor analysis, suggesting that the balanced, high-level production of multiple angiogenic factors by CDCs and ASCs translates to superior pro-angiogenic activity in a controlled environment.
Detailed Experimental Protocol: In Vitro Tube Formation Assay
Objective: To quantitatively compare the angiogenic potency of conditioned media from different MSC types. Method Summary: [94]
- Cell Culture & Conditioned Media Collection: ASCs, BMSCs, CDCs, and BM-MNCs are cultured in serum-free media for a standardized period (e.g., 72 hours). The conditioned media is collected and centrifuged to remove cells and debris.
- ECMatrix Coating: A 96-well plate is coated with a solidified ECMatrix gel, which provides a basement membrane substitute.
- Seeding and Incubation: Human Umbilical Vein Endothelial Cells (HUVECs) are seeded onto the gel in the respective conditioned media. Positive controls use HUVECs in optimal growth media, while negative controls use basal media.
- Imaging and Quantification: After 6-18 hours of incubation, the formed tubular structures are imaged under a microscope. The total tube length, number of branches, and number of meshes are quantified using image analysis software (e.g., Image-Pro Plus).
In Vivo Functional Angiogenic Outcomes
Translating in vitro findings to functional outcomes in living organisms is critical. A head-to-head study in a severe combined immunodeficiency (SCID) mouse model of myocardial infarction provided direct comparative data. Cells were injected into the infarct border zone, and cardiac function was assessed by echocardiography after three weeks [94].
Table 2: In Vivo Functional Outcomes in a Mouse Myocardial Infarction Model
| Parameter | ASCs | BMSCs | CDCs | BM-MNCs | Control (PBS) |
|---|---|---|---|---|---|
| Improvement in LV Ejection Fraction | Moderate | Moderate | Greatest | Minimal | Deterioration |
| Cell Engraftment Rate | Moderate | Moderate | Highest | Low | N/A |
| Reduction in Apoptotic Cells | Moderate | Moderate | Greatest | Minimal | Baseline |
| Myogenic Differentiation | Low | Low | Highest | Low | N/A |
Hearts treated with CDCs showed the most significant improvement in left ventricular ejection fraction (LVEF), the least adverse remodeling, and the highest number of engrafted cells. ASCs and BMSCs provided a moderate, comparable functional benefit, which was superior to that of BM-MNCs [94]. This suggests that while the paracrine activity of ASCs and BMSCs is potent, the integrated paracrine and direct differentiation potential of CDCs may offer superior therapeutic effects in ischemic contexts.
Another study specifically investigating the secretome of ASCs in an endothelial tubulogenesis assay confirmed their high potency. Using neutralizing antibodies, researchers identified that VEGF-A and VEGF-D were the major growth factors secreted by ASCs responsible for supporting the formation of endothelial tubes. This study concluded that the variation in paracrine factors across MSC populations contributes to different levels of angiogenic activity, and that ASCs may be preferred for therapeutic approaches dependent on angiogenesis [3].
Detailed Experimental Protocol: Mouse Model of Myocardial Infarction
Objective: To directly compare the functional regenerative capacity of different stem cell types in an in vivo model of ischemic injury. Method Summary: [94]
- Infarction Induction: SCID-beige mice (10-12 weeks old) are anesthetized. The left anterior descending coronary artery (LAD) is permanently ligated with a suture to induce a myocardial infarction.
- Cell Implantation: Immediately after ligation, the experimental groups receive intramyocardial injections at the infarct border zone of either:
- Phosphate-buffered saline (PBS, control)
- 1×10^5 CDCs
- 1×10^5 ASCs
- 1×10^5 BMSCs
- 1×10^6 BM-MNCs (a higher dose due to smaller cell size)
- Functional Assessment: At baseline (3 hours post-surgery) and 3 weeks post-treatment, mice undergo transthoracic echocardiography under light anesthesia. LV end-diastolic volume, LV end-systolic volume, and LV ejection fraction are calculated from 2D long-axis views.
- Histological Analysis: After the final echocardiography, hearts are explanted for histological assessment of engraftment, apoptosis (via TUNEL assay), and morphology.
Signaling Pathways in MSC-Mediated Angiogenesis
The paracrine factors secreted by MSCs activate a complex signaling network in endothelial cells to promote angiogenesis. The following diagram synthesizes the key pathways and their interactions as elucidated from the cited literature.
MSC Angiogenic Signaling Pathways
This network illustrates how factors like VEGF, FGF-2, and HGF bind to their respective receptors on endothelial cells, activating intracellular pathways such as PI3K/Akt and MAPK/ERK. These signals converge to promote endothelial cell survival, proliferation, and migration—the fundamental processes required for new blood vessel formation [95] [94]. The diagram provides a logical framework for understanding the mechanistic basis of the functional data presented in the previous sections.
The Scientist's Toolkit: Key Research Reagent Solutions
To conduct rigorous, reproducible comparisons of MSC angiogenic potency, specific reagents and tools are essential. The following table details key solutions referenced in the studies cited in this guide.
Table 3: Essential Research Reagents for MSC Angiogenesis Studies
| Reagent / Kit | Primary Function | Example Use Case |
|---|---|---|
| Human ELISA Kits (VEGF, HGF, IGF-1, etc.) | Quantification of specific growth factor concentrations in conditioned media. | Measuring paracrine factor secretion profiles of ASCs vs. BMSCs [94]. |
| In Vitro Angiogenesis Assay Kit (e.g., ECMatrix) | Provides a standardized basement membrane substitute for tube formation assays. | Evaluating the functional angiogenic potential of MSC-conditioned media on HUVECs [94]. |
| Cell Isolation Kits (for ASCs, BMSCs, etc.) | Isolation and purification of specific MSC populations from tissue samples. | Obtaining primary ASCs from adipose tissue or BMSCs from bone marrow [3]. |
| FACS Antibodies (CD105, CD90, CD73, CD45, CD34) | Characterization of MSC surface marker expression via flow cytometry. | Confirming the immunophenotype of isolated MSCs according to ISCT criteria [80] [94]. |
| Small Interfering RNA (siRNA) | Gene knockdown to determine the functional role of specific secreted factors. | Validating the critical role of VEGF in ASC-mediated tubulogenesis [3] [88]. |
| HUVECs & Endothelial Cell Culture Media | Provide a standardized cellular system for testing the effects of MSC paracrine factors. | Serving as target cells in tube formation and migration assays [94]. |
Direct head-to-head functional comparisons reveal a hierarchy of angiogenic potency among common MSC types. The evidence from both in vitro secretome analysis and in vivo disease models consistently indicates that CDCs exhibit the most robust pro-angiogenic profile, followed closely by ASCs, which show superior activity in certain contexts, particularly through VEGF-dependent mechanisms. BMSCs demonstrate a more moderate but significant potency, while BM-MNCs are the least potent. The selection of a cell source for therapeutic angiogenesis should therefore be guided by the specific clinical need and the desired paracrine factor profile. Future research optimizing culture conditions and leveraging engineered MSCs will further enhance the translational potential of these findings.
Abstract This guide provides a direct comparison of the cytoprotective efficacy of Mesenchymal Stem Cells (MSCs) from three prominent sources—Adipose-derived Stem Cells (ASCs), Bone Marrow-derived Mesenchymal Stem Cells (BMSCs), and Umbilical Cord-derived Mesenchymal Stem Cells (UCMSCs). Focusing on ischemic injury models, we objectively analyze their paracrine signatures, functional outcomes in reducing apoptosis and infarct size, and subsequent implications for cardiac function recovery. The data, derived from head-to-head experimental studies, are synthesized to inform preclinical research and therapeutic development.
The therapeutic potential of MSCs in ischemic injuries, such as myocardial infarction (MI) and stroke, is increasingly attributed to their paracrine activity rather than direct differentiation. These cells secrete a plethora of bioactive factors—including growth factors, cytokines, and chemokines—that collectively modulate the hostile ischemic microenvironment. This paracrine secretome can inhibit apoptotic pathways, promote neovascularization, and dampen detrimental inflammation, leading to a significant reduction in overall cell death and infarct volume. However, MSCs are not a uniform therapeutic entity. Significant functional heterogeneity exists among MSCs derived from different tissue sources, influenced by their native microenvironment [64] [96]. This biological variation directly impacts their secretory profile and, consequently, their efficacy as cytoprotective agents. This guide performs a donor-matched and cross-comparative analysis of ASCs, BMSCs, and UCMSCs to delineate their distinct cytoprotective strengths and provide a data-driven foundation for selecting the optimal cell source for specific therapeutic applications.
Comparative Analysis of Paracrine Factor Expression
The foundational mechanism behind the cytoprotective efficacy of MSCs lies in their secretome. A comparative analysis of mRNA and protein expression reveals distinct profiles that predispose different MSC types toward specific therapeutic actions.
2.1 Growth Factor and Cytokine Expression A direct comparison of human adult MSCs derived from bone marrow, adipose, and dermal tissue highlighted key differences in the expression of critical paracrine factors. While certain factors like VEGF-A, angiogenin, and nerve growth factor (NGF) were expressed at comparable levels, others showed significant variation [3] [10]. Specifically, ASCs demonstrated higher mRNA expression levels of Insulin-like Growth Factor-1 (IGF-1), Vascular Endothelial Growth Factor-D (VEGF-D), and Interleukin-8 (IL-8) compared to other MSC populations [10]. Functional assays further confirmed that the potent pro-angiogenic activity of ASCs is largely mediated by their secretion of VEGF-A and VEGF-D [3]. This robust angiogenic and growth factor expression profile suggests ASCs are potent inducers of new blood vessel formation, a critical process for salvaging ischemic tissue.
2.2 Divergent Expression in Disease-Specific Contexts More recent research in a myocardial infarction model provides a functional context to these expression differences. Transcriptome sequencing (RNA-Seq) analysis of UCMSCs and ADMSCs (a subset of ASCs) confirmed that their gene expression profiles diverge significantly, particularly in pathways related to angiogenesis and apoptosis [64]. This molecular heterogeneity translates into observable functional differences in their secretome's activity.
Table 1: Comparative Paracrine Factor Expression Profiles of MSCs
| Paracrine Factor | ASCs/ADMSCs | BMSCs | UCMSCs | Primary Functional Role |
|---|---|---|---|---|
| VEGF-A | Comparable/High [3] | Comparable [3] | Data Incomplete | Angiogenesis, Endothelial Cell Survival |
| VEGF-D | ↑ Higher [10] | Lower | Data Incomplete | Angiogenesis, Lymphangiogenesis |
| IGF-1 | ↑ Higher [10] | Lower | Data Incomplete | Cell Survival, Proliferation, Anti-apoptosis |
| IL-8 | ↑ Higher [10] | Lower | Data Incomplete | Neutrophil Chemoattractant, Angiogenesis |
| bFGF | Comparable [3] | Comparable [3] | Data Incomplete | Mitogenesis, Angiogenesis |
| Angiogenin | Comparable [3] | Comparable [3] | Data Incomplete | Angiogenesis, rRNA Transcription |
| HGF | Data Incomplete | Data Incomplete | ↑ Higher (vs. ADMSCs) [64] | Anti-apoptosis, Mitogenesis, Motogenesis |
Quantitative Efficacy in Ischemic Injury Models
Controlled in vivo studies are crucial for translating in vitro paracrine profiles into measurable cytoprotective outcomes. The following data, primarily from a donor-matched comparative study on acute myocardial infarction, provides a clear, quantitative comparison of functional efficacy.
3.1 Myocardial Infarction Model In a mouse model of acute MI, both UCMSCs and ADMSCs were administered via intramyocardial injection post-surgery. Cardiac function was assessed by echocardiography at day 28, and histological analyses were performed to quantify infarction size, apoptosis, and angiogenesis [64]. The results demonstrated a clear divergence in therapeutic emphasis.
Table 2: Quantitative Functional Outcomes in a Mouse Myocardial Infarction Model [64]
| Functional Parameter | UCMSCs | ADMSCs | Control (MI-only) |
|---|---|---|---|
| Cardiac Function (Ejection Fraction) | Significant Improvement | Significant Improvement | Baseline Impairment |
| Infarction Size Reduction | Significant Reduction | Significant Reduction | No Reduction |
| Angiogenesis (Capillary Density) | ↑↑ Greater Increase | ↑ Increase | No Change/Low Level |
| Anti-apoptotic Effect (on Cardiomyocytes) | Significant Effect | ↑↑ Stronger Effect | No Effect/High Apoptosis |
| Proposed Primary Mechanism | Enhanced Angiogenesis | Enhanced Cell Survival & Anti-apoptosis | N/A |
The study concluded that while both cell types improved overall outcomes, ADMSCs exerted a stronger cardioprotective function, primarily attributed to their superior anti-apoptotic effect on residual cardiomyocytes. In contrast, UCMSCs presented greater pro-angiogenesis activity both in vitro and in vivo [64]. This suggests that for acute cytoprotection where salvaging viable cardiomyocytes is the priority, ADMSCs might be superior, whereas UCMSCs could be more effective in long-term reperfusion and repair via vascularization.
3.2 Bone Regeneration as a Corollary Model While not a model of ischemia per se, bone regeneration studies provide additional evidence of the inherent functional differences between MSC sources. A donor-matched comparison of ASCs and BMSCs found that BMSCs demonstrated superior osteogenic and chondrogenic capacity compared to ASCs [53]. This was evidenced by BMSCs having earlier and higher alkaline phosphatase (ALP) activity, greater calcium deposition, and higher expression of osteogenesis-related genes and proteins [53]. This lineage predisposition, rooted in their tissue of origin, reinforces the concept that the "fittest" MSC source depends on the target tissue and desired therapeutic outcome. Proteomic analysis further identified the integrin expression profile, particularly the strong up-regulation of Integrin 11a in BMSCs, as a key distinctive feature underlying their superior osteogenic capacity [97].
Experimental Protocols for Key assays
To ensure the reproducibility and rigorous comparison of cytoprotective efficacy, the following details the core experimental protocols used to generate the data cited in this guide.
4.1 In Vivo Myocardial Infarction and Cell Transplantation
- Animal Model: Male C57BL/6 mice (7-8 weeks old) are commonly used.
- MI Induction: Myocardial infarction is surgically induced by permanent ligation of the left anterior descending (LAD) coronary artery.
- Cell Administration: Immediately or shortly after MI induction, MSCs (e.g., 1×10^5 cells in PBS) are delivered via intramyocardial injection directly into the border zone of the infarct. The control group receives an injection of the vehicle alone.
- Functional Assessment: Echocardiography is performed at predetermined endpoints (e.g., day 28 post-MI) under light anesthesia to measure left ventricular dimensions and calculate ejection fraction and fractional shortening.
- Histological Analysis: At sacrifice, hearts are harvested, sectioned, and stained.
- Infarct Size: Staining with 2,3,5-Triphenyltetrazolium Chloride (TTC) is used to quantify the area of necrosis. Viable tissue stains red, while the infarct area appears pale.
- Apoptosis: TUNEL (Terminal deoxynucleotidyl transferase dUTP Nick End Labeling) assay is performed on heart sections to detect DNA fragmentation in apoptotic cells. The number of TUNEL-positive cardiomyocytes is counted.
- Angiogenesis: Immunohistochemical staining for CD31 or α-SMA is used to identify endothelial cells and quantify capillary density within the infarct border zone [64].
4.2 In Vitro Tube Formation Assay (Angiogenic Potential)
- Matrigel Preparation: A 96-well plate is coated with a thin layer of Basement Membrane Matrigel (e.g., 50 µL/well) and allowed to polymerize at 37°C for 1 hour.
- Cell Preparation: Human Umbilical Vein Endothelial Cells (HUVECs) are suspended in Conditioned Media (CM) collected from the MSC cultures of interest (ASCs, BMSCs, UCMSCs).
- Assay Execution: The HUVEC suspension is seeded onto the polymerized Matrigel layer at a density of 20,000 cells/well.
- Quantification: After 4-8 hours of incubation, capillary-like tube structures form. Multiple images per well are taken randomly. The total tube length, number of nodes (branch points), and number of junctions are quantified using image analysis software like ImageJ [64] [3].
Signaling Pathways in MSC-Mediated Cytoprotection
The cytoprotective effects of MSCs are mediated through the modulation of complex signaling pathways in recipient cells. The following diagram synthesizes the key pathways implicated in reducing apoptosis and promoting survival, as evidenced by the comparative studies.
The Scientist's Toolkit: Essential Research Reagents
The following table catalogues key reagents and materials essential for conducting the experiments described in this comparison guide.
Table 3: Essential Research Reagents for Cytoprotective Efficacy Studies
| Reagent / Material | Function / Application | Example Use Case |
|---|---|---|
| Human MSCs (ASCs, BMSCs, UCMSCs) | The primary therapeutic agents under investigation; source-specific differences are the focus of study. | Donor-matched comparisons to minimize genetic variability [98] [53]. |
| Conditioned Media (CM) | To isolate and study the paracrine effects of MSCs, independent of cell presence. | In vitro tube formation assays with HUVECs [64] [3]. |
| Matrigel | Basement membrane extract used as a substrate to simulate the in vivo environment for endothelial tube formation. | Coating plates for the in vitro angiogenesis assay [64]. |
| HUVECs (Human Umbilical Vein Endothelial Cells) | A standard cell model for assessing angiogenic potential in vitro. | Seeding on Matrigel with MSC-CM to quantify tube formation [3]. |
| TUNEL Assay Kit | To detect and quantify apoptotic cells by labeling DNA fragmentation. | Assessing anti-apoptotic effects in heart tissue sections post-MI [64]. |
| Antibodies (CD31, α-SMA) | For immunohistochemical staining to identify endothelial cells and vessels. | Quantifying capillary density in infarct border zones [64]. |
| 2,3,5-Triphenyltetrazolium Chloride (TTC) | A water-soluble dye used to distinguish between metabolically active (red) and necrotic (pale) tissue. | Staining heart sections to measure infarct size [64]. |
| Flow Cytometry Antibodies (CD73, CD90, CD105, CD34, CD45, HLA-DR) | To characterize and confirm the immunophenotype of MSCs according to ISCT criteria. | Ensuring purity and identity of MSC populations before experimentation [53]. |
The comparative data presented in this guide underscores a central principle: the choice of MSC source is not trivial and should be strategically aligned with the primary therapeutic goal. The evidence indicates that ADMSCs/ASCs demonstrate a potent capacity for enhancing cell survival and attenuating apoptosis, making them a strong candidate for acute cytoprotection in ischemic injury. In contrast, UCMSCs exhibit superior angiogenic prowess, potentially offering greater benefit in the reparative phase to re-establish perfusion. BMSCs, while not the focus of the primary MI data herein, have established efficacy in bone repair, highlighting their context-dependent utility.
For researchers and drug developers, these findings advocate for a pathology-driven, precision medicine approach to MSC therapy. The selection between ASCs, BMSCs, and UCMSCs should be informed by the dominant pathophysiological processes of the target disease—whether it is rampant apoptosis, inadequate perfusion, or a combination thereof. Future research should focus on standardizing isolation and expansion protocols to minimize donor-related heterogeneity [53] [96] and further explore combinatorial or pre-activated MSC products engineered to simultaneously exploit multiple cytoprotective pathways for maximal therapeutic benefit.
Immunophenotypic Similarities and Critical Functional Differences
Within regenerative medicine and advanced therapeutic development, Mesenchymal Stromal Cells (MSCs) represent a cornerstone for their dual capacity for tissue repair and immunomodulation. Sourced from diverse tissues, these cells are widely investigated for clinical applications. This guide provides a structured, objective comparison of three prominent MSC types: Adipose-Derived Mesenchymal Stromal Cells (ASCs), Bone Marrow-Derived Mesenchymal Stromal Cells (BM-MSCs), and Umbilical Cord Blood-Derived Mesenchymal Stromal Cells (UCB-MSCs). Framed within a broader thesis on comparative paracrine factor expression, this analysis delves beyond surface-level similarities to reveal critical functional heterogeneities. We summarize quantitative experimental data and detailed methodologies to equip researchers and drug development professionals with the evidence needed to select the optimal MSC source for specific therapeutic applications.
Immunophenotypic Similarities: A Unified Identity
Despite their different tissue origins, ASCs, BM-MSCs, and UCB-MSCs share a core immunophenotypic profile that defines their identity. This consistent marker expression forms the basis for their classification as MSCs according to the International Society for Cellular Therapy (ISCT) guidelines [80].
Table 1: Core Immunophenotypic Markers of MSCs
| Marker Category | Specific Markers | Expression Status | Functional Significance |
|---|---|---|---|
| Positive Markers | CD105, CD73, CD90 | Consistently Expressed [99] [80] | Defines standard MSC phenotype; adhesion and signaling functions. |
| Adhesion Molecules | CD29, CD44, CD54 | Highly Expressed [99] | Facilitates homing, interaction with extracellular matrix, and niche retention. |
| Negative Markers | CD34, CD45, CD14, CD19, HLA-DR | Not Expressed [99] [80] | Confirms non-hematopoietic origin and low immunogenicity. |
A key unifying characteristic is their low immunogenicity. All three cell types demonstrate low or no expression of major histocompatibility complex class II (HLA-DR) and co-stimulatory molecules (e.g., CD80, CD86) [99]. This allows them to evade host immune responses, making them attractive for allogeneic transplantation. Furthermore, UCB-MSCs have been reported to express even lower levels of HLA-ABC than BM-MSCs, potentially offering an advantage in reducing transplant rejection [99].
Critical Functional Differences: The Devil in the Details
While their surface markers are similar, the functional capabilities of ASCs, BM-MSCs, and UCB-MSCs differ significantly. These differences, driven by their unique tissue niches, are most evident in their paracrine factor secretion, proliferative capacity, and therapeutic strengths.
Proliferation and Hematopoietic Support
UCB-MSCs exhibit a superior proliferation rate, with a population doubling time of 2–3 days, significantly faster than that of BM-MSCs [99]. This high proliferative capacity, combined with their non-invasive collection, makes them a practical and abundant cell source.
In the context of hematopoietic support, UCB-MSCs excel by secreting a robust profile of cytokines. They express key factors such as Stem Cell Factor (SCF), Leukemia Inhibitory Factor (LIF), and Macrophage-Colony Stimulating Factor (M-CSF) [99]. Notably, they also produce Granulocyte Colony-Stimulating Factor (G-CSF) and Granulocyte-Macrophage Colony-Stimulating Factor (GM-CSF), which are not typically produced by BM-MSCs in these analyses, highlighting a unique supportive capacity for blood cell formation [99].
Paracrine Factor Expression and Angiogenic Potential
The paracrine "secretome" of MSCs is a primary mechanism for their therapeutic effects. A comparative analysis of conditioned media from these cells reveals distinct expression patterns critical for angiogenesis and cell survival.
Table 2: Comparative Paracrine Factor Expression and Functional Profiles
| Functional Attribute | ASC | BM-MSC | UCB-MSC | Supporting Experimental Data |
|---|---|---|---|---|
| mRNA/Protein Expression | ||||
| IGF-1, VEGF-D, IL-8 | Higher [3] | Lower | Lower | mRNA expression analysis & conditioned media protein confirmation [3] |
| VEGF-A, Angiogenin | Comparable [3] | Comparable | Comparable | mRNA expression analysis & conditioned media protein confirmation [3] |
| G-CSF, GM-CSF | Information Missing | Not Found [99] | Produced [99] | Cytokine spectroscopy studies [99] |
| Functional Outcome | ||||
| In Vitro Angiogenic Activity | Superior [3] | Lower | Information Missing | Endothelial cell tubulogenesis assay; neutralization by VEGF-A/VEGF-D antibodies [3] |
| Anti-Cancer Properties | Information Missing | Information Missing | Yes (e.g., inhibits HL-60, K562 proliferation) [99] | Co-culture with leukemia cells; activates p38MAPK, arrests cell cycle [99] |
| Proliferation Rate | High | Lower [99] | Highest [99] | Population doubling time analysis [99] |
Functional tubulogenesis assays confirm that the distinct secretome of ASCs translates into superior pro-angiogenic activity. Endothelial cells incubated in ASC-conditioned media demonstrated increased tubulogenic efficiency compared to those incubated with conditioned media from other MSC types like dermal papilla cells. This effect was largely abolished by neutralizing antibodies against VEGF-A and VEGF-D, identifying these two factors as major contributors to ASC-mediated angiogenesis [3].
Diagram 1: ASC Secretome Drives Angiogenesis. The paracrine factors highly expressed by ASCs (VEGF-A, VEGF-D, IGF-1, IL-8) bind to receptors on endothelial cells, stimulating tubulogenesis.
Immunomodulation and Therapeutic Niche
All MSC sources possess immunomodulatory properties, but their mechanistic emphasis may vary. UCB-MSCs have demonstrated a unique role in reducing the incidence and severity of Graft-versus-Host Disease (GVHD) following Hematopoietic Stem Cell Transplantation (HSCT) [99]. Their action involves inhibiting pro-inflammatory immune cells while promoting the expansion of regulatory T cells (Tregs), contributing to immune homeostasis [25].
Meanwhile, ASCs have shown significant promise in clinical applications for immune-related conditions. For instance, in a phase I/IIa study on Rheumatoid Arthritis (RA), a single intravenous infusion of autologous ASCs was found to be safe and effective in improving joint function over 52 weeks [25].
Experimental Protocols for Functional Comparison
To ensure the reproducibility of comparative studies, this section outlines standard experimental methodologies for evaluating key MSC functions.
The isolation process is specific to each tissue source, but subsequent culture conditions can be standardized for valid comparison [3].
Primary Cell Isolation:
- ASCs: Minced subcutaneous adipose tissue is digested with 0.075% type I collagenase. The stromal vascular fraction is obtained via centrifugation, plated, and washed to remove non-adherent cells [3].
- UCB-MSCs: Cells are isolated from the Wharton's Jelly or subcortical endothelium of the umbilical cord. The tissue is explanted or enzymatically digested, and the resulting cell suspension is cultured [99].
- BM-MSCs: Commercial sources or bone marrow aspirates are used, and the mononuclear cell fraction is isolated and plated. Non-adherent cells are removed after overnight incubation [3] [80].
In Vitro Culture: All isolated MSC populations are cultured under identical conditions (e.g., Dulbecco’s modified Eagle’s medium low-glucose with 10% fetal calf serum) to minimize artifacts from serum concentration or passaging methods [3].
Flow Cytometry Characterization: Cells are characterized by flow cytometry for positive (CD105, CD73, CD90) and negative (CD34, CD45, CD14, CD19, HLA-DR) markers to confirm immunophenotype [99] [80].
Diagram 2: MSC Comparison Workflow. A standardized process for isolating, culturing, and comparing MSCs from different tissues.
Protocol: Evaluating Paracrine Factor Expression and Angiogenic Potential
This protocol assesses the secretory profile and functional impact of MSC-conditioned media.
Conditioned Media (CM) Collection:
- MSCs are cultured until 70-80% confluent.
- The culture medium is replaced with a serum-free medium to avoid interference from serum proteins.
- After 24-48 hours, the CM is collected and centrifuged to remove cells and debris. The supernatant is aliquoted and stored at -80°C [3].
Analysis of Paracrine Factors:
Functional Tubulogenesis Assay:
- Endothelial cells (e.g., HUVECs) are seeded on a basement membrane matrix (e.g., Matrigel).
- The cells are incubated with CM from the different MSC types.
- After several hours, tubular structures form. The total tube length or number of meshes is quantified using image analysis software.
- To identify key factors, neutralizing antibodies against specific growth factors (e.g., VEGF-A, VEGF-D) are added to the CM to block their activity [3].
The Scientist's Toolkit: Essential Research Reagents
The following table details key reagents and their functions for conducting the experiments described in this guide.
Table 3: Essential Research Reagents for MSC Comparative Studies
| Reagent / Material | Function / Application | Example / Note |
|---|---|---|
| Type I Collagenase | Enzymatic digestion of tissues for primary cell isolation (ASCs). | Worthington Biochemical [3] |
| Dulbecco’s Modified Eagle’s Medium (DMEM) | Base culture medium for MSC expansion. | Use low-glucose formulation for standardized culture [3]. |
| Fetal Calf Serum (FCS) | Serum supplement for cell culture media to support growth. | Batch testing is recommended for optimal MSC performance [3]. |
| Flow Cytometry Antibodies | Immunophenotypic characterization of MSCs. | Anti-CD105, CD73, CD90 (positive); anti-CD34, CD45, HLA-DR (negative) [99] [80]. |
| Matrigel | Basement membrane matrix for endothelial cell tubulogenesis assays. | Used to assess the functional angiogenic potential of MSC-conditioned media [3]. |
| Neutralizing Antibodies | Functional blocking of specific factors in conditioned media. | Anti-VEGF-A, Anti-VEGF-D to confirm factor involvement in angiogenesis [3]. |
| ELISA Kits | Quantitative measurement of specific secreted proteins. | For VEGF-A, VEGF-D, IGF-1, G-CSF, etc., in conditioned media [3]. |
The decision to use ASCs, BM-MSCs, or UCB-MSCs is not one-size-fits-all. While they are united by a common immunophenotype, this analysis underscores that their critical functional differences in paracrine factor expression, proliferative capacity, and therapeutic specialization are paramount. ASCs, with their superior angiogenic secretome, are a powerful tool for vascularization. UCB-MSCs offer rapid growth and potent hematopoietic support, ideal for transplant medicine. BM-MSCs remain a well-characterized benchmark. The choice for researchers and clinicians must therefore be guided by the specific mechanistic requirements of the intended application, leveraging these functional differences to maximize therapeutic success.
The therapeutic application of mesenchymal stem cells (MSCs) hinges on two primary mechanisms: their capacity to differentiate into specific cell lineages and their secretion of paracrine factors that promote repair and regeneration. The balance between these mechanisms varies significantly across MSC types, influenced by their tissue of origin. This guide provides a direct comparison of the differentiation potential and paracrine output of Adipose-derived Stem Cells (ASCs), Bone Marrow-derived MSCs (BMSCs), and Umbilical Cord Blood-derived MSCs (UCB-MSCs), presenting key experimental data and methodologies to inform preclinical research and therapeutic development.
Mesenchymal stem cells (MSCs) are multipotent stromal cells with the capacity for self-renewal and differentiation into mesodermal lineages such as osteocytes, adipocytes, and chondrocytes [100]. Initially, the therapeutic potential of MSCs was attributed primarily to their ability to differentiate and replace damaged cells. However, a growing body of evidence demonstrates that their benefits are also mediated powerfully through paracrine actions—the secretion of a complex mixture of growth factors, cytokines, and extracellular vesicles, collectively known as the secretome [101] [102] [100]. These factors influence the local microenvironment by modulating immune responses, promoting angiogenesis, enhancing cell survival, and activating resident progenitor cells [101]. This guide objectively compares the differentiation capacity and paracrine output of three prominent MSC types—ASCs, BMSCs, and UCB-MSCs—to elucidate their relative therapeutic contributions.
Comparative Experimental Profiles
Paracrine Factor Secretion
The paracrine signature of MSCs is a critical determinant of their therapeutic efficacy. Direct comparative studies reveal both shared and unique secretion profiles.
Table 1: Comparative Paracrine Factor Expression Profiles of Different MSCs
| Paracrine Factor | ASCs | BMSCs | UCB-MSCs | Functional Role |
|---|---|---|---|---|
| IGF-1 | Higher mRNA expression [3] [10] | Lower than ASCs [3] [10] | Information Missing | Cytoprotection, cell migration, contractility [101] |
| VEGF-A | Comparable mRNA and protein levels [3] [10] | Comparable mRNA and protein levels [3] [10] | Information Missing | Angiogenesis, cytoprotection, cell proliferation [101] |
| VEGF-D | Higher mRNA expression [3] [10] | Lower than ASCs [3] [10] | Information Missing | Angiogenesis (supports endothelial tubulogenesis) [3] |
| HGF | Information Missing | Information Missing | Information Missing | Cytoprotection, angiogenesis, cell migration [101] |
| bFGF (FGF-2) | Comparable mRNA levels [3] [10] | Comparable mRNA levels [3] [10] | Information Missing | Cell proliferation and migration, angiogenesis [101] |
| Angiogenin | Comparable mRNA and protein levels [3] [10] | Comparable mRNA and protein levels [3] [10] | Information Missing | Angiogenesis, cell proliferation [101] |
| IL-8 | Higher mRNA expression [3] | Lower than ASCs [3] | Information Missing | Pro-angiogenic, neutrophil chemotaxis |
| SDF-1 | Information Missing | Information Missing | Information Missing | Progenitor cell homing [101] |
Table 2: Functional Potency in In Vitro and In Vivo Models
| Model / Assay | ASCs | BMSCs | UCB-MSCs | Notes |
|---|---|---|---|---|
| In Vitro Tubulogenesis | Increased tubulogenic efficiency [3] | Information Missing | Information Missing | ASC-conditioned media was superior to DPC-conditioned media [3] |
| In Vivo Cardiac Repair | Functional benefit observed [94] | Functional benefit observed [94] | Information Missing | In a head-to-head study, CDCs (cardiac stem cells) showed superior benefit; BMSCs and ASCs also showed efficacy [94] |
| In Vivo Bone Formation | Information Missing | Higher clonogenic & osteogenic capacity [100] | Information Missing | CD271+ BMSCs show enhanced osteogenesis [100] |
| Immunomodulation | Effective [100] | Effective [100] | Strongest potential to suppress T-cell proliferation [100] | UCB-MSCs induce cell-cycle arrest and apoptosis in T-cells [100] |
Lineage-Specific Differentiation Capacity
While all MSCs are multipotent, their propensity to differentiate down specific lineages varies, a phenomenon influenced by their tissue-specific origin and microenvironment.
Table 3: Comparative Differentiation Potential of MSCs
| Differentiation Lineage | ASCs | BMSCs | UCB-MSCs | Supporting Evidence |
|---|---|---|---|---|
| Adipogenic | High (Preferential) [3] | Moderate [3] | Information Missing | ASCs have a natural propensity for adipogenesis [3] |
| Osteogenic | Moderate [3] | High (Preferential) [3] [100] | Stable trilineage potential [100] | BMSCs are more efficiently directed toward bone [3] |
| Chondrogenic | Moderate [3] | High (Preferential) [3] | Stable trilineage potential [100] | BMSCs are more efficiently directed toward cartilage [3] |
| Cardiomyogenic | Demonstrated [94] | Demonstrated [94] | Information Missing | In a direct comparison, CDCs showed the greatest myogenic potency [94] |
Diagram 1: MSC Source Dictates Therapeutic Strengths
Detailed Experimental Protocols for Comparison
To ensure reproducible and valid comparisons, standardized experimental workflows are essential. Below are detailed protocols for key assays used to generate the data in this guide.
Protocol: Isolation and Culture of MSCs from Different Tissues
Primary Objective: To isolate and expand human MSCs from adipose tissue, bone marrow, and umbilical cord blood under comparable in vitro conditions to minimize experimental artifacts [3].
ASCs Isolation (from Adipose Tissue):
- Tissue Digestion: Minced adipose tissue is digested with 0.075% type I collagenase at 37°C for 60 minutes with shaking [3].
- Cell Recovery: The digest is centrifuged to remove adipocytes and the pellet is resuspended in DMEM-low glucose with 10% FCS [3].
- Filtration & Lysis: The cell suspension is filtered through a 100μm mesh, centrifuged, and red blood cells are lysed with 0.16 M NH₄Cl [3].
- Plating & Expansion: Cells are plated in culture flasks. Non-adherent cells are removed after 24 hours. ASCs between passages 3-6 are used for experiments [3].
BMSCs Culture:
UCB-MSCs Isolation:
- Collection: Umbilical cord blood is collected with informed consent.
- Separation: Mononuclear cells are isolated via density gradient centrifugation (e.g., using Ficoll-Paque).
- Plating & Expansion: Cells are plated in MSC-specific media (e.g., MesenPRO RS Medium). Adherent cells are expanded and characterized based on ISCT criteria [100].
Protocol: Evaluating Paracrine Factor Expression
Primary Objective: To quantitatively compare the secretion levels of key paracrine factors from different MSC populations [3] [94].
Conditioned Media (CM) Collection:
Analysis of Secreted Factors:
Functional Tubulogenesis Assay:
- Use a commercial tube formation assay kit (e.g., from Chemicon) [94].
- Seed endothelial cells (e.g., HUVECs) on ECMatrix-coated plates.
- Incubate the endothelial cells with CM from the different MSCs.
- After 6-12 hours, image the formed tube networks and quantify total tube length using image analysis software (e.g., Image-Pro Plus) [3] [94].
Protocol: Assessing Differentiation Potential
Primary Objective: To directly compare the tri-lineage differentiation capacity of ASCs, BMSCs, and UCB-MSCs [100].
Osteogenic Differentiation:
- Culture MSCs to near confluence.
- Switch to osteogenic induction medium: DMEM supplemented with 10% FBS, 0.1µM dexamethasone, 50µM ascorbic acid, and 2-20mM β-glycerophosphate [103].
- Maintain cultures for 2-4 weeks, refreshing the medium twice weekly.
- Staining: Assess mineralization by Alizarin Red S staining of calcium deposits.
Adipogenic Differentiation:
- Culture MSCs to full confluence.
- Switch to adipogenic induction medium: DMEM with 10% FBS, 1µM dexamethasone, 0.5mM isobutylmethylxanthine, 50µM indomethacin, and 10µg/ml insulin.
- Maintain cultures for 2-3 weeks, refreshing the medium twice weekly.
- Staining: Visualize lipid vacuoles by Oil Red O staining.
Chondrogenic Differentiation:
- Pellet 2.5×10⁵ MSCs in a conical tube.
- Culture the pellet in chondrogenic induction medium: DMEM-high glucose with 1% ITS+ premix, 50µM ascorbic acid, 0.1µM dexamethasone, and 10ng/ml TGF-β3.
- Maintain pellets for 3-4 weeks.
- Analysis: Assess cartilage matrix production (proteoglycans) by Safranin O staining of histological sections.
Diagram 2: Experimental Workflow for MSC Comparison
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Reagents for MSC Differentiation and Paracrine Studies
| Reagent / Kit | Function / Application | Example Use in Context |
|---|---|---|
| Type I Collagenase | Tissue digestion for primary cell isolation. | Isolation of ASCs from adipose tissue [3]. |
| MesenPRO RS Medium | Specialized medium for MSC culture. | Expansion of rat and human MSCs with reduced serum [103]. |
| Fetal Bovine Serum (FBS) | Critical supplement for cell culture media. | Standard component (typically 10%) in basal media for MSC expansion [3] [103]. |
| ELISA Kits (VEGF, HGF, IGF-1) | Quantitative protein measurement in conditioned media. | Direct comparison of paracrine factor secretion from different MSCs [94]. |
| In Vitro Angiogenesis Assay Kit | Measure tube formation of endothelial cells. | Functional assessment of the pro-angiogenic activity of MSC-conditioned media [94]. |
| Osteogenic Induction Supplements | Direct differentiation towards bone lineage. | Contains dexamethasone, ascorbic acid, and β-glycerophosphate [103]. |
| Adipogenic Induction Supplements | Direct differentiation towards fat lineage. | Contains dexamethasone, IBMX, indomethacin, and insulin. |
| Alizarin Red S | Stains calcium deposits in differentiated osteocytes. | Quantification of in vitro mineralization after osteogenic induction [103]. |
| Oil Red O | Stains lipid droplets in differentiated adipocytes. | Quantification of lipid accumulation after adipogenic induction. |
The comparative analysis of ASCs, BMSCs, and UCB-MSCs reveals a clear trade-off: no single MSC source is superior across all therapeutic dimensions. ASCs demonstrate a robust paracrine signature, particularly for angiogenic applications, while BMSCs retain a strong, perhaps more intrinsic, capacity for osteogenic and chondrogenic differentiation. UCB-MSCs emerge as potent immunomodulators.
The future of MSC therapeutics appears to be shifting toward a "cell-free" paradigm that leverages the paracrine secretome, potentially overcoming challenges associated with whole-cell transplantation, such as low engraftment and safety concerns [103] [100]. Furthermore, strategies to engineer or "prime" MSCs—for instance, through genetic manipulation (e.g., Smurf1 silencing) [103] or nanoparticle-modulation (e.g., with zinc oxide) [104]—are being actively explored to enhance their inherent secretory or differentiation profiles. This evolving landscape underscores the importance of a targeted, mechanism-driven selection of MSC types—or their refined products—for specific clinical indications in regenerative medicine.
This guide objectively compares the paracrine factor expression profiles of mesenchymal stem cells (MSCs) derived from adipose tissue (ASCs), bone marrow (BMSCs), and dermal tissues (DSCs/DPCs), consolidating experimental data within the broader research context of their therapeutic potential.
Quantitative Comparison of Paracrine Factor Expression
The comparative potential of different MSC populations is largely defined by their secretome. The tables below summarize key experimental findings from direct comparative studies.
Table 1: mRNA Expression Profile of Angiogenic Paracrine Factors in MSCs
| Paracrine Factor | ASCs | BMSCs | Dermal-derived MSCs (DSCs/DPCs) |
|---|---|---|---|
| IGF-1 | Higher [3] [10] | Lower [3] [10] | Lower [3] [10] |
| VEGF-D | Higher [3] [10] | Lower [3] [10] | Lower [3] [10] |
| IL-8 | Higher [3] [10] | Lower [3] [10] | Lower [3] [10] |
| VEGF-A | Comparable [3] [10] | Comparable [3] [10] | Comparable [3] [10] |
| Angiogenin | Comparable [3] [10] | Comparable [3] [10] | Comparable [3] [10] |
| bFGF | Comparable [3] [10] | Comparable [3] [10] | Comparable [3] [10] |
| NGF | Comparable [3] [10] | Comparable [3] [10] | Comparable [3] [10] |
Table 2: Protein Secretion and Functional Activity in MSC Conditioned Media
| Analysis Type | ASCs | BMSCs | Dermal-derived MSCs (DSCs/DPCs) |
|---|---|---|---|
| VEGF-A Protein | Comparable [3] [10] | Comparable [3] [10] | Comparable [3] [10] |
| Angiogenin Protein | Comparable [3] [10] | Comparable [3] [10] | Comparable [3] [10] |
| Leptin Protein | Lower [3] [10] | Lower [3] [10] | Significantly Higher [3] [10] |
| In Vitro Tubulogenesis | Increased tubulogenic efficiency [3] [10] | Not Specified | Reduced tubulogenic efficiency [3] [10] |
| Key Functional Factors | VEGF-A, VEGF-D identified as major contributors [3] [10] | Not Specified | Not Specified |
Detailed Experimental Methodologies
A systematic review is a "protocol-driven comprehensive review and synthesis of data" [105]. The methodologies from key comparative studies are detailed below to ensure transparency and reproducibility.
Primary Cell Isolation and Culture
All studies emphasized culturing different MSC populations under identical conditions to minimize artifacts from serum concentration or passaging methods [3].
- ASCs: Isolated from human abdominal subcutaneous adipose tissue digested with 0.075% type I collagenase. Cells were filtered, centrifuged, and plated in Dulbecco's modified Eagle's medium low-glucose (DMEM-lg) with 10% fetal calf serum (FCS) [3].
- DSCs and DPCs: Isolated via microdissection of human scalp hair follicles. DSCs were cultured via explant migration, while DPCs were anchored to a culture dish after release from the follicle bulb, both using DMEM-lg with 10% FCS [3].
- BMSCs: Commercially sourced human BMSCs were cultured in the same DMEM-lg medium to maintain consistency [3].
Paracrine Factor Expression Analysis
- mRNA Expression: Analysis was performed to identify and compare the transcriptional levels of various angiogenic and cytoprotective factors across the MSC populations [3] [10].
- Protein Analysis: Conditioned media (CM) from each MSC population was collected and analyzed to confirm the secretion of identified factors at the protein level, using methods such as enzyme-linked immunosorbent assay (ELISA) [3] [10].
Functional Tubulogenesis Assay
- Method: Endothelial cells were incubated in conditioned media from the different MSC populations (ASCs, DPCs, etc.) [3] [10].
- Measurement: The "tubulogenic efficiency" of endothelial cells—their ability to form tube-like structures mimicking early vascular development—was quantified and compared between groups [3] [10].
- Neutralization: To identify key functional factors, neutralizing antibodies against specific proteins like VEGF-A and VEGF-D were used in the ASC-conditioned media to confirm their role in the observed pro-angiogenic effect [3] [10].
Experimental Workflow
The following diagram illustrates the key stages of a comparative experimental study, from sample collection to data synthesis, as derived from the described methodologies.
The Scientist's Toolkit: Essential Research Reagents
The following table details key reagents and their functions essential for conducting research in this field, based on the methodologies cited.
Table 3: Key Research Reagent Solutions for MSC Paracrine Factor Studies
| Reagent / Tool | Function in Research | Application in Featured Studies |
|---|---|---|
| Type I Collagenase | Digests collagen in tissues to isolate individual cells. | Used for digestion of human adipose tissue to isolate ASCs [3]. |
| Dulbecco’s Modified Eagle’s Medium (DMEM-lg) | A low-glucose cell culture medium that provides nutrients for cell growth and maintenance. | Served as the base culture medium for all MSC populations (ASCs, BMSCs, DSCs, DPCs) under identical conditions [3]. |
| Fetal Calf Serum (FCS) | Provides essential growth factors, hormones, and proteins to support cell proliferation in vitro. | Added to DMEM-lg at 10% concentration to support the growth of all MSC types [3]. |
| Neutralizing Antibodies | Bind to specific proteins and block their biological activity. | Used to inhibit VEGF-A and VEGF-D in ASC-conditioned media, confirming their critical role in endothelial tubulogenesis [3] [10]. |
| Protocol and Systematic Review Guidelines (e.g., PRISMA) | Provide a structured framework for designing, conducting, and reporting reviews to ensure transparency and minimize bias. | Essential for planning a robust systematic review, defining key questions, and reporting findings [105] [106] [107]. |
Interpretation and Research Implications
The consistent finding of elevated IGF-1, VEGF-D, and IL-8 in ASCs, coupled with their superior functional performance in tubulogenesis assays, suggests a molecular basis for their enhanced pro-angiogenic profile [3] [10]. This evidence supports the preference for ASCs over BMSCs or dermal MSCs in therapeutic contexts where rapid vascularization is critical, such as in engineering thick tissue constructs or treating ischemic conditions [3].
Future research and systematic reviews in this area must address widespread challenges in preclinical research design, including lack of randomization, inadequate blinding, and pseudo-replication, to enhance the validity and reproducibility of findings [108] [109]. Adherence to robust reporting standards like the PRISMA guidelines is crucial for generating reliable evidence that effectively informs clinical translation in personalised medicine [106] [109].
Conclusion
The comparative analysis reveals that ASCs, BMSCs, and UCB-MSCs possess distinct paracrine factor expression profiles that significantly influence their therapeutic potential. ASCs frequently demonstrate superior angiogenic capacity, UCB-MSCs exhibit high proliferative potential, while BMSCs remain a well-characterized benchmark. These differences are not merely quantitative but functional, impacting their efficacy in specific disease contexts like cardiac repair, wound healing, and immunomodulation. Future research should focus on standardizing secretome characterization, developing potency assays based on paracrine factor profiles, and exploring combinatorial approaches or engineered vesicles to harness the full therapeutic potential of each MSC source, ultimately paving the way for precision regenerative medicine.
References
- 1. Emerging Concepts in Paracrine Mechanisms in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4874329/]
- 2. A Systematic Review of Pre-clinical Studies | Stem Cell ... [https://link.springer.com/article/10.1007/s12015-022-10429-6]
- 3. Comparative Analysis of Paracrine Factor Expression ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3411362/]
- 4. A Brief Overview of Global Trends in MSC-Based Cell ... [https://link.springer.com/article/10.1007/s12015-022-10369-1]
- 5. Secreted spermidine synthase reveals a paracrine role for ... [https://www.nature.com/articles/s41419-025-07639-4]
- 6. Gap Junctional Versus Paracrine Signaling in the Human ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12637001/]
- 7. Stem-Cell Niches in Health and Disease [https://www.mdpi.com/2073-4409/14/13/981]
- 8. Therapeutic and Clinical Potential of Adipose-Derived ... [https://www.mdpi.com/2073-4409/14/21/1727]
- 9. Beyond the Niche: Tissue-Level Coordination of Stem Cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3897713/]
- 10. Comparative analysis of paracrine factor expression in ... [https://pubmed.ncbi.nlm.nih.gov/22188562/]
- 11. Comparative analysis of human Mesenchymal Stromal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12616983/]
- 12. Quantitative characterization of cell niches in spatially ... [https://www.nature.com/articles/s41588-025-02120-6]
- 13. Paracrine Factors Released by Stem Cells of Mesenchymal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9622561/]
- 14. Mesenchymal Stromal Cell Secretome: Influencing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6288292/]
- 15. Paracrine Mechanisms of Mesenchymal Stromal Cells in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7085399/]
- 16. ASC and SVF Cells Synergistically Induce ... [https://www.mdpi.com/1422-0067/23/1/185]
- 17. Heterogeneity of proangiogenic features in mesenchymal ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-016-0418-9]
- 18. Molecular Imaging for Comparison of Different Growth ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4932172/]
- 19. Adipose-derived stem cells promote angiogenesis and tissue ... [https://www.omicsdi.org/dataset/biostudies-literature/S-EPMC3638514]
- 20. Frontiers | Immunoregulatory paracrine effect of mesenchymal ... stem [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2024.1411507/full]
- 21. and Anti-Inflammatory Cytokines Secreted by Adult Stem ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4537750/]
- 22. Regenerative Capacity of Adipose Derived Stem Cells ... [https://www.mdpi.com/1422-0067/20/10/2523]
- 23. -derived exosomes: a new therapeutic... Mesenchymal stem cell [https://pubmed.ncbi.nlm.nih.gov/31753036/]
- 24. UC-MSC vs. Bone-Marrow & Cord-Blood ... [https://vegastemcell.com/articles/uc-msc-vs-bone-marrow-cord-blood-msc-stem-cell-for-tendon-repair-2023-evidence-key-takeaways-seo-guide/]
- 25. Review: progression and heterogeneity of multiple sources of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12239375/]
- 26. Comparison of mesenchymal stem cells from bone marrow, ... [https://www.sciencedirect.com/science/article/pii/S2405580823000675]
- 27. Comparison of infant bone marrow- and umbilical cord ... [https://www.sciencedirect.com/science/article/pii/S235232042400172X]
- 28. Comparison of the chondrogenic potential of eBMSCs and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11831820/]
- 29. Translation: DNA to mRNA to Protein [https://www.nature.com/scitable/topicpage/translation-dna-to-mrna-to-protein-393/]
- 30. Multi-omic assessment of mRNA translation dynamics in ... [https://www.nature.com/articles/s41597-025-05861-5]
- 31. Understanding the molecular basis of mesenchymal stem ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12583528/]
- 32. The landscape of transcriptional and translational changes ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9645810/]
- 33. Overview of Protein Expression Systems [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-protein-expression-systems.html]
- 34. Comprehensive analysis of mRNA and lncRNA expression ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12119550/]
- 35. Deciphering cell–cell interactions and communication from ... [https://www.nature.com/articles/s41576-020-00292-x]
- 36. Noise propagation through extracellular signaling leads to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3906959/]
- 37. Validated methods for isolation and qualification of ... [https://translational-medicine.biomedcentral.com/articles/10.1186/s12967-025-06972-8]
- 38. A comparison of isolation and culture protocols for human ... [https://pubmed.ncbi.nlm.nih.gov/35412950/]
- 39. Comparing MSC Isolation Techniques [https://www.cellandgene.com/doc/comparing-msc-isolation-techniques-0001]
- 40. Comparison between manual and automated methods for ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2025.1556697/full]
- 41. Molecular Medicine Reports [https://www.spandidos-publications.com/10.3892/mmr.2019.10767]
- 42. Towards Physiologic Culture Approaches to Improve ... [https://www.mdpi.com/2073-4409/10/4/886]
- 43. An improved protocol for isolation and culture of mesenchymal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5982388/]
- 44. Challenges in Clinical Development of Mesenchymal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6811694/]
- 45. Secretome Proteomic Approaches for Biomarker Discovery [https://pmc.ncbi.nlm.nih.gov/articles/PMC7559921/]
- 46. Proteomic profiling of iPSC and tissue-derived MSC ... [https://www.nature.com/articles/s41536-024-00382-y]
- 47. ELISA Assay Technique | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-elisa.html]
- 48. Comparison of microarray-based RNA expression with ELISA-based protein determination of HER2, uPA and PAI-1 in tumour tissue of patients with breast cancer and relation to outcome - PubMed [https://pubmed.ncbi.nlm.nih.gov/20204407/]
- 49. Side-by-Side Comparison of the Biological Characteristics of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3722850/]
- 50. A new enzyme-linked immunosorbent assay (ELISA) for human free and bound kallikrein 9 | Clinical Proteomics | Full Text [https://clinicalproteomicsjournal.biomedcentral.com/articles/10.1186/s12014-017-9140-6]
- 51. A critical analysis of current in vitro and in vivo angiogenesis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2697546/]
- 52. A Comparison of Methods for Quantifying Angiogenesis in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3162381/]
- 53. Adipose-derived and bone marrow mesenchymal stem cells [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-018-0914-1]
- 54. Comparison of Differences in Cell Migration during the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12183369/]
- 55. In vitro cell migration and invasion assays [https://www.sciencedirect.com/science/article/abs/pii/S1383574212000464]
- 56. Consistent Cell Invasion and Migration with VitroGel® [https://www.thewellbio.com/applications/functional-assay/cell-invasion-assay/]
- 57. Standardized Methodology of Scaffold-Free ... - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC12566299/]
- 58. Exploring the therapeutic potential of MSC-derived secretomes in neonatal care: focus on BPD and NEC | Stem Cell Research & Therapy | Full Text [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-025-04616-8]
- 59. Frontiers | Translational Animal Models Provide Insight Into Mesenchymal Stromal Cell (MSC) Secretome Therapy [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2021.654885/full]
- 60. The Efficacy of Stem Cells Secretome Application in Osteoarthritis: A Systematic Review of In Vivo Studies - PubMed [https://pubmed.ncbi.nlm.nih.gov/32556944/]
- 61. Method standardization of secretome production, collection ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12423684/]
- 62. Modern animal models for biomedical research ... - STAT News [https://www.statnews.com/2025/11/07/animal-research-mice-models-invaluable-biomedical-discovery/]
- 63. Mesenchymal stem cells in treating human diseases [https://www.nature.com/articles/s41392-025-02313-9]
- 64. The therapeutic efficacy comparison of MSCs derived ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12077008/]
- 65. Comparative cardiomyocyte differentiation potential of rat ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2025.1604605/full]
- 66. Comparative Analysis of Mesenchymal Stem Cells from ... [https://www.academia.edu/21835471/Comparative_Analysis_of_Mesenchymal_Stem_Cells_from_Bone_Marrow_Umbilical_Cord_Blood_or_Adipose_Tissue?uc-g-sw=91028944]
- 67. Cardiac repair and regeneration: cell therapy, in vivo ... [https://www.nature.com/articles/s12276-025-01549-3]
- 68. Repair of infarcted myocardium by skeletal muscle-derived ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12326654/]
- 69. Challenges and future perspectives in using mesenchymal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12162511/]
- 70. Assessment of Umbilical Cord Mesenchymal Stem Cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11862980/]
- 71. Comparison of therapeutic effects of mesenchymal stem ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9358877/]
- 72. Pooling umbilical cord-mesenchymal stromal cells derived ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-025-04361-y]
- 73. The paradigm of stem cell secretome in tissue repair and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11711308/]
- 74. Mesenchymal Stromal Cell Secretome Is Affected by ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10631804/]
- 75. Deciphering influence of donor age on adipose-derived ... [https://www.nature.com/articles/s41598-024-73875-x]
- 76. Storage conditions affect the composition of the lyophilized ... [https://www.nature.com/articles/s41598-024-60787-z]
- 77. Mesenchymal stem cells and their secretome modulate stress ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-025-04442-y]
- 78. Vascular senescence and aging: mechanisms, clinical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12106568/]
- 79. Persistent accumulation of therapy-induced senescent cells [https://www.nature.com/articles/s41368-025-00380-w]
- 80. Exploring MSC and HSPC interactions: new frontiers in ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-025-04753-0]
- 81. The issue of heterogeneity of MSC-based advanced therapy ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11310176/]
- 82. The heterogeneity of mesenchymal stem cells: an important ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-023-03587-y]
- 83. Effects of Mesenchymal Stem Cell‐Derived Paracrine Signals ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7995150/]
- 84. Unveiling distinctions between mesenchymal stromal cells ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11872483/]
- 85. Comparison of transcriptome profiles of mesenchymal stem ... [https://www.sciencedirect.com/science/article/abs/pii/S0378111922006734]
- 86. Secretome and extracellular vesicle signatures in bone ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12367461/]
- 87. Single cell transcriptomic analysis of human mesenchymal ... [https://www.nature.com/articles/s41419-019-1583-4]
- 88. Strategies to enhance paracrine potency of transplanted ... [https://www.nature.com/articles/pr2017249]
- 89. The Roles of VEGF-A and SDF-1a in Rat Adipose Stem Cells [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2020.580131/full]
- 90. Genetic Engineering as a Strategy to Improve the ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2020.00737/full]
- 91. Hypoxic Conditioning Enhances the Angiogenic Paracrine ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3653395/]
- 92. Enhancement of the Therapeutic Capacity of Mesenchymal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7661472/]
- 93. The effect of hypoxia preconditioning on the neural and ... [https://www.sciencedirect.com/science/article/abs/pii/S1473050217300563]
- 94. Direct comparison of different stem cell types and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3292778/]
- 95. Mesenchymal Stem/Stromal Cells for Therapeutic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10486439/]
- 96. Potential pre-activation strategies for improving therapeutic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8981790/]
- 97. Comparative proteomic analysis of osteogenic ... [https://pubmed.ncbi.nlm.nih.gov/32885592/]
- 98. Comparison of bone regenerative capacity of donor- ... [https://pubmed.ncbi.nlm.nih.gov/33242173/]
- 99. Biological Characteristics of Umbilical Cord Mesenchymal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8126649/]
- 100. The secretion profile of mesenchymal stem cells and ... [https://www.nature.com/articles/s41392-022-00932-0]
- 101. Paracrine mechanisms in adult stem cell signaling and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2667788/]
- 102. Effects of microenvironment and biological behavior on the ... [https://www.sciencedirect.com/science/article/pii/S235230422300140X]
- 103. Paracrine activity of Smurf1-silenced mesenchymal stem cells ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-025-04165-0]
- 104. Enhanced stem cell-mediated therapeutic immune ... [https://www.sciencedirect.com/science/article/abs/pii/S0142961225001516]
- 105. Systematic Review Methods - NCBI - NIH [https://www.ncbi.nlm.nih.gov/books/NBK44088/]
- 106. Systematic review [https://en.wikipedia.org/wiki/Systematic_review]
- 107. What are systematic reviews? - UCL Databases [https://library-guides.ucl.ac.uk/systematic-reviews/what]
- 108. Evaluation of Published Preclinical Experimental Studies in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6885208/]
- 109. Recommendations for robust and reproducible preclinical ... [https://bmcmedicine.biomedcentral.com/articles/10.1186/s12916-022-02719-0]