Controlled-Rate vs. Passive Freezing: A 2025 Guide for Biomedical Research and Cryopreservation
This article provides a comprehensive analysis of controlled-rate and passive freezing methodologies for researchers and drug development professionals.
Controlled-Rate vs. Passive Freezing: A 2025 Guide for Biomedical Research and Cryopreservation
Abstract
This article provides a comprehensive analysis of controlled-rate and passive freezing methodologies for researchers and drug development professionals. It covers the fundamental cryobiology of slow cooling injuries and ice crystal formation, details current industry standards and equipment for implementation, and offers troubleshooting strategies for common challenges like viability loss and scaling. Drawing on recent 2025 clinical data and industry surveys, it delivers a definitive comparison of cell viability and engraftment outcomes, empowering scientists to select and optimize cryopreservation protocols for cell and gene therapies, biobanking, and pharmaceutical development.
The Core Principles of Cryopreservation: Understanding Ice, Injury, and Cell Survival
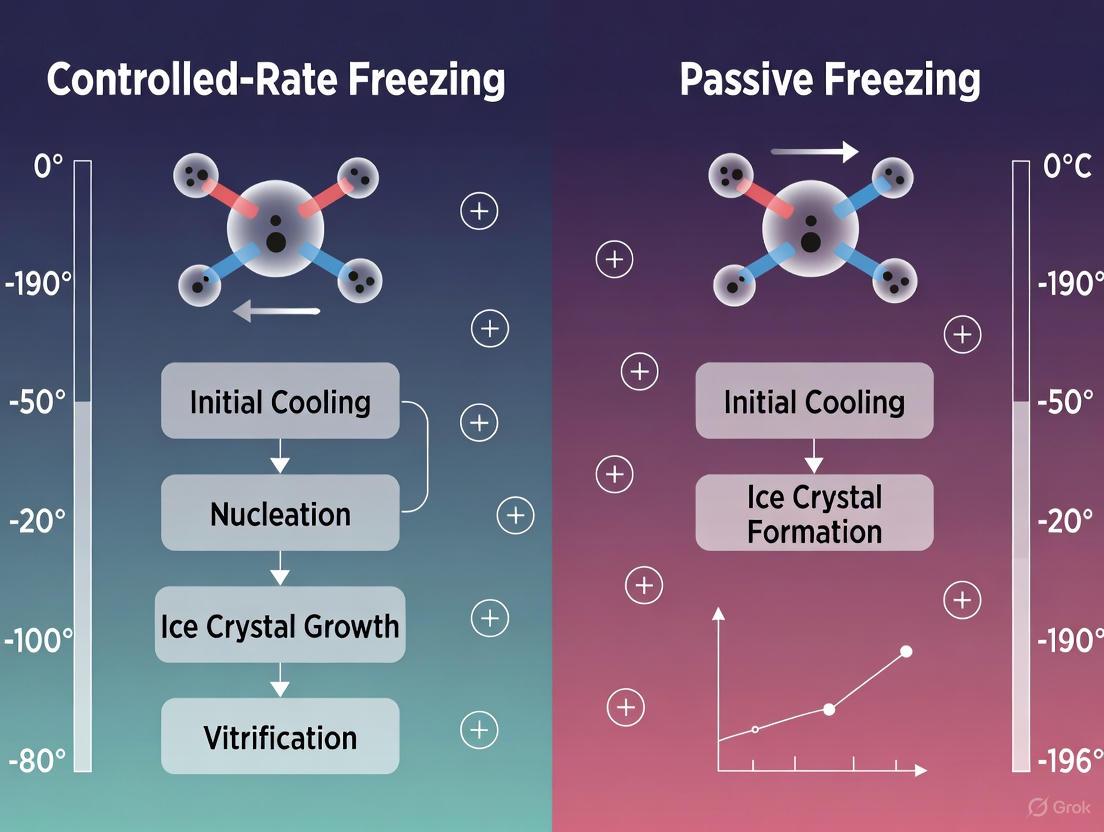
The preservation of biological materials at ultra-low temperatures is a cornerstone of modern biotechnology, regenerative medicine, and pharmaceutical development. The process of cryopreservation effectively halts cellular metabolism, enabling long-term storage of cells, tissues, and biologics while maintaining their viability and functionality upon thawing. Within this field, two principal methodological approaches have emerged: controlled-rate freezing (CRF) and passive freezing (PF). The fundamental distinction between these techniques lies in the precision of temperature reduction during the critical freezing phase. CRF employs specialized programmable equipment to maintain a predetermined, uniform cooling rate throughout the process, typically at -1°C per minute for many cell types. In contrast, PF relies on placing samples in insulated containers stored in mechanical freezers at -80°C, resulting in an uncontrolled, albeit relatively slow, cooling rate [1] [2].
The selection between these methods carries significant implications for research reproducibility, therapeutic efficacy, and commercial viability in drug development. This technical guide examines the scientific principles, practical applications, and comparative performance of CRF versus PF, providing researchers and drug development professionals with evidence-based insights for protocol selection and optimization within the broader context of cryopreservation research.
Fundamental Principles and Key Differentiators
The core challenge in cryopreservation is to transition biological materials to a vitrified or frozen state while minimizing damage from intracellular ice formation, solute concentration (cryoconcentration), and osmotic stress. The rate of cooling is a critical factor influencing cell survival, with most cells exhibiting an inverted U-shaped response where both excessively slow and rapid cooling prove detrimental [3].
Controlled-Rate Freezing operates on the principle of precise thermal management. Programmable freezers lower the temperature according to user-defined profiles, often incorporating features to manage the latent heat of fusion—the energy released when water transitions to ice—which can cause localized warming and inconsistent freezing if not properly controlled [4]. This method allows for the customization of cooling parameters for specific cell types and formulations.
Passive Freezing utilizes the insulating properties of materials like polystyrene or polyurethane to moderate the cooling rate when samples are placed in standard ultra-low temperature freezers. Devices such as the Nalgene Mr. Frosty or Corning CoolCell are designed to achieve an approximate cooling rate of -1°C per minute, though this rate is not consistently maintained throughout the entire thermal range and varies with freezer characteristics and sample volume [1] [5].
The table below summarizes the fundamental operational differences between these two approaches:
Table 1: Core Operational Characteristics of Controlled-Rate and Passive Freezing
| Parameter | Controlled-Rate Freezing | Passive Freezing |
|---|---|---|
| Cooling Rate Control | Precise, programmable, and consistent | Approximate, container-dependent, variable |
| Primary Equipment | Programmable freezer unit (e.g., CryoMed) | Insulated container placed in -80°C freezer |
| Typical Cooling Rate | User-defined, commonly -1°C/min | Approximately -1°C/min to -3°C/min |
| Latent Heat Management | Actively managed by the equipment | Passively dissipated through insulation |
| Process Documentation | Comprehensive temperature profile recording | Limited or none |
| Liquid Nitrogen Dependency | Often required for cooling | Not required |
Comparative Analysis: Performance and Outcomes
Direct comparisons between CRF and PF reveal a complex landscape where the optimal choice often depends on the specific biological material and application requirements.
Cell Viability and Engraftment
A recent retrospective study of 50 hematopoietic progenitor cell (HPC) products provides compelling clinical evidence for therapeutic applications. The research demonstrated that while mean total nucleated cell (TNC) viability post-thaw was statistically higher for the CRF group (74.2% ± 9.9%) compared to the PF group (68.4% ± 9.4%), the more critical metric of CD34+ cell viability showed no significant difference (77.1% ± 11.3% for CRF vs. 78.5% ± 8.0% for PF) [6].
Most importantly, the clinical endpoint of engraftment success revealed equivalent outcomes between methods. The time to neutrophil engraftment (12.4 ± 5.0 days for CRF vs. 15.0 ± 7.7 days for PF) and platelet engraftment (21.5 ± 9.1 days for CRF vs. 22.3 ± 22.8 days for PF) showed no statistically significant differences, leading the authors to conclude that PF is an acceptable alternative to CRF for initial cryopreservation before long-term storage [6].
Industry Adoption and Trends
According to a comprehensive survey by the ISCT Cold Chain Management & Logistics Working Group, CRF has been widely adopted in the cell and gene therapy sector, with 87% of respondents reporting its use in their current practice. Notably, among the 13% using passive freezing, the vast majority (86%) had products exclusively in early clinical development stages (up to Phase II), suggesting a potential transition to CRF as products advance toward commercialization [7].
The survey further revealed that 60% of CRF users employ default freezing profiles provided by equipment manufacturers, while the remainder invest resources in developing optimized profiles, particularly for challenging cell types including iPSCs, hepatocytes, cardiomyocytes, and certain engineered cells [7].
Table 2: Comparative Advantages and Limitations in Industrial Applications
| Consideration | Controlled-Rate Freezing | Passive Freezing |
|---|---|---|
| Process Control | High control over critical parameters | Limited control over freezing parameters |
| Regulatory Documentation | Comprehensive process data recording | Limited inherent documentation |
| Infrastructure Cost | High capital and operational expense | Low-cost, low-consumable infrastructure |
| Technical Expertise | Specialized training required | Low technical barrier to adoption |
| Scalability | Potential bottleneck for large batches | Easier to scale for multiple simultaneous batches |
| Best Applications | Late-stage clinical/commercial products; sensitive cell types | Early R&D; robust cell types; resource-limited settings |
Methodological Protocols
Standard Controlled-Rate Freezing Protocol
The following protocol outlines a generalized CRF procedure for mammalian cells, adaptable to specific cell type requirements:
- Cell Harvest and Preparation: Harvest cells during log-phase growth (typically >80% confluency) and ensure viability exceeds 90%. Centrifuge to pellet cells and carefully remove supernatant [1] [2].
- Cryoprotectant Resuspension: Resuspend cell pellet in appropriate freezing medium. For many applications, a medium containing 10% DMSO in fetal bovine serum (FBS) is used. Commercially available, serum-free, GMP-manufactured options like CryoStor CS10 are recommended for regulated applications [1] [3].
- Aliquoting: Transfer cell suspension to cryogenic vials. Use internal-threaded vials to prevent contamination during filling or storage in liquid nitrogen [1].
- Controlled-Rate Freezing: Place vials in the CRF chamber and initiate a programmed freeze cycle. A standard protocol begins at 4°C and cools at a rate of -1°C/min until reaching -40°C to -50°C, followed by a more rapid cooling to approximately -100°C before transfer to long-term storage [4].
- Long-Term Storage: Transfer cryovials to liquid nitrogen storage at ≤ -135°C (vapor phase) or -196°C (liquid phase) for long-term preservation [2].
Standard Passive Freezing Protocol
The PF protocol shares initial preparation steps with CRF but diverges in the freezing mechanism:
- Cell Harvest and Preparation: Identical to Steps 1-3 of the CRF protocol [1].
- Container-Based Freezing: Place filled cryovials into an isopropanol-based freezing container (e.g., Nalgene Mr. Frosty) or an isopropanol-free container (e.g., Corning CoolCell). Ensure the container is at room temperature before use [1].
- Freezer Incubation: Transfer the entire container to a -80°C mechanical freezer for a minimum of 4 hours, though overnight incubation is standard practice. The isopropanol or the proprietary material in the container ensures an approximate cooling rate of -1°C/min [1] [2].
- Long-Term Storage: Identical to Step 5 of the CRF protocol. Note that storage at -80°C is not recommended for long-term preservation as cell viability declines over time at this temperature [2].
Experimental Workflow for Method Comparison
The following diagram illustrates a generalized experimental workflow for comparing cryopreservation outcomes between CRF and PF methods, as exemplified in the cited research:
Essential Research Reagents and Materials
Successful cryopreservation, regardless of the freezing method, depends on the quality and appropriateness of laboratory materials and reagents. The following table details key components essential for implementing both CRF and PF protocols:
Table 3: Essential Research Reagents and Materials for Cryopreservation
| Item | Function | Examples & Notes |
|---|---|---|
| Cryoprotective Agent (CPA) | Reduces ice crystal formation; protects cellular structures from freeze damage. | DMSO (10%) is most common. Glycerol used for DMSO-sensitive cells. Toxicity and exposure time must be optimized [1] [3]. |
| Freezing Medium Base | Provides physiological environment and nutrients during freeze-thaw transition. | Fetal Bovine Serum (FBS) with CPA. For regulated work, use defined, serum-free, GMP-compliant media like CryoStor CS10 [1] [3]. |
| Cell-Specific Media | Optimized formulation for specialized cell types. | mFreSR (for human ES/iPS cells); MesenCult-ACF (for MSCs); STEMdiff Cardiomyocyte Freezing Medium [1]. |
| Cryogenic Vials | Secure, leak-proof container for sample storage. | Use internal-threaded vials (e.g., Corning) to prevent contamination in liquid nitrogen [1]. |
| Passive Freezing Device | Insulates samples to achieve ~-1°C/min in a -80°C freezer. | Nalgene Mr. Frosty (isopropanol-based); Corning CoolCell (isopropanol-free) [1] [5]. |
| Controlled-Rate Freezer | Programmable unit that ensures precise, reproducible cooling rates. | CryoMed series; Cytiva Asymptote (liquid nitrogen-free portable option) [4] [8]. |
| Long-Term Storage System | Maintains stable ultra-low temperatures (≤ -135°C). | Liquid nitrogen freezer (vapor phase recommended for safety; liquid phase for ultimate temperature) [2] [4]. |
Applications in Biopharmaceutical Development and Cell Therapy
The choice between CRF and PF extends beyond research laboratories into critical biopharmaceutical and therapeutic applications, where it impacts product quality, consistency, and regulatory compliance.
Biologics and Drug Substance Processing
In biopharmaceutical development, freezing is routinely used to preserve bulk drug substance (BDS), typically monoclonal antibodies or recombinant proteins, enabling flexible manufacturing schedules and ensuring supply chain stability. The primary challenge in this context is cryoconcentration—the phenomenon where solutes, including the protein therapeutic, are concentrated into unfrozen pockets between ice crystals during freezing. This can lead to protein aggregation, pH shifts, and loss of efficacy [9] [10].
For large-volume BDS freezing in cryovessels, active control of freezing rates can help manage the extent and pattern of cryoconcentration. While passive freezing is used in some contexts, the unpredictable and variable thermal profiles achieved in large containers make CRF the preferred method for ensuring product consistency, particularly for late-stage clinical and commercial products [9] [10].
Cell and Gene Therapies
Advanced therapeutic medicinal products (ATMPs), such as CAR-T cells, hematopoietic stem cells (HSCs), and mesenchymal stromal cells (MSCs), represent a domain where cryopreservation is indispensable for bridging manufacturing and patient treatment. The ISCT survey identified scaling as the major hurdle for cryopreservation in cell and gene therapy, with 22% of respondents citing the "Ability to process at a large scale" as the primary challenge [7].
While PF offers advantages in simplicity and cost for early-stage clinical trials, the transition to CRF is often necessary for later-phase trials and commercialization. This shift is driven by the enhanced process control and documentation provided by CRF, which are critical for regulatory filings and ensuring batch-to-batch consistency [7] [3]. The ability to document the complete temperature profile of the freeze provides crucial data for investigating any deviations in product quality.
The battle between controlled-rate and passive freezing does not yield a single universal victor. The accumulated evidence, including recent clinical studies, demonstrates that passive freezing can achieve functionally equivalent results to controlled-rate freezing for specific applications, such as hematopoietic progenitor cell engraftment [6]. This makes PF a scientifically valid and cost-effective option for basic research, early-stage clinical development, and for preserving robust cell types.
However, the superior process control, documentation, and reproducibility offered by controlled-rate freezing establish it as the gold standard for sensitive cell types, scalable manufacturing processes, and late-stage clinical and commercial therapeutics [7] [3]. The decision between these methodologies must be guided by a careful assessment of the biological system, regulatory requirements, economic constraints, and the criticality of process consistency to the final product's quality and efficacy. As cryopreservation science advances, the development of novel, standardized, and cost-effective freezing technologies will continue to reshape this fundamental battle in biopreservation.
Cryopreservation serves as a cornerstone technology in biological research and therapeutic applications, enabling the long-term storage of cells and tissues by suspending cellular metabolism at ultra-low temperatures (-80°C to -196°C) [1]. The fundamental challenge in this process lies not in the low storage temperatures themselves, but in the physics of ice formation during the freezing phase. When water within cells freezes, ice crystals can mechanically damage delicate cellular structures and create solute imbalances that compromise cell integrity [11] [1]. The process of vitrification, where water transitions into a glass-like state rather than forming crystals, represents the ideal outcome for maximizing cell viability [11].
Understanding and controlling ice crystal formation is particularly crucial when comparing controlled-rate freezing (CRF) and passive freezing (PF) methodologies. While CRF utilizes programmable equipment to precisely manage temperature decline, passive freezing relies on placing samples in insulated containers within standard -80°C mechanical freezers [11] [12]. The central thesis of contemporary cryopreservation research explores whether the technical sophistication and higher cost of CRF systems yield measurably superior outcomes for different cell types compared to optimized passive freezing protocols, especially in clinical applications where both cell viability and operational practicality must be considered.
The Physics of Ice Crystal Formation and Cellular Damage
Fundamental Mechanisms of Freezing Damage
During cryopreservation, cells face two primary mechanical threats from ice formation, both governed by the rate of temperature change:
Extracellular Ice Crystal Formation: As temperatures approach the freezing point, ice crystals typically form first in the extracellular solution. This creates an osmotic imbalance across the cell membrane, drawing water out of the cell through osmosis and causing cellular dehydration [11]. Excessive dehydration leads to irreversible damage to cellular structures and metabolic pathways.
Intracellular Ice Crystallization: At rapid cooling rates, water within the cell does not have sufficient time to exit before freezing, leading to the formation of lethal intracellular ice crystals [11] [4]. These crystals physically disrupt organelles, membranes, and the cytoskeleton, typically resulting in immediate cell death upon thawing.
The relationship between cooling rate and these damaging mechanisms is described by the "two-factor hypothesis" of freezing injury [13]. This hypothesis establishes that an optimal cooling rate exists that minimizes both intracellular ice formation (favored by rapid cooling) and solute effects/dehydration (favored by slow cooling) [13].
The Latent Heat of Fusion Challenge
A critical physical phenomenon occurring during cryopreservation is the release of latent heat of fusion [12] [4]. As the cell suspension begins to freeze, the phase change from liquid to solid releases energy in the form of heat, causing a temporary temperature spike that can disrupt controlled cooling protocols. This temperature rise can be particularly problematic for sensitive cell types, as it alters the intended cooling profile and may promote irregular ice crystal growth [4]. Controlled-rate freezers are specifically designed to counteract this effect by rapidly adjusting cooling parameters when nucleation occurs [12].
Figure 1: Pathways of ice formation and resulting cellular damage mechanisms during cryopreservation.
Quantitative Comparison: Controlled-Rate Freezing vs. Passive Freezing
Performance Metrics Across Cell Types
Research comparing controlled-rate freezing (CRF) and passive freezing (PF) has yielded nuanced results, with outcomes highly dependent on cell type, cryoprotectant formulation, and specific protocol parameters. The following table summarizes key quantitative findings from recent studies:
Table 1: Comparative performance metrics between controlled-rate and passive freezing methods
| Cell Type | Freezing Method | Post-Thaw Viability | Functional Recovery | Key Metrics | Study/Reference |
|---|---|---|---|---|---|
| Hematopoietic Progenitor Cells (HPCs) | Controlled-Rate Freezing (CRF) | 74.2% ± 9.9% (TNC) | 12.4 ± 5.0 days to neutrophil engraftment | No significant difference in CD34+ viability or engraftment times | [12] [6] |
| Hematopoietic Progenitor Cells (HPCs) | Passive Freezing (PF) | 68.4% ± 9.4% (TNC) | 15.0 ± 7.7 days to neutrophil engraftment | No significant difference in CD34+ viability or engraftment times | [12] [6] |
| Platelets (DMSO-free protocol) | Controlled-Rate Freezing (CRF) | >85% recovery | 72-82% marker expression retention | Successful maintenance of functional integrity | [14] |
| Jurkat T-cells (DMSO-containing) | Spin Freezing (Controlled) | >80% (varies with parameters) | Morphology largely preserved | Highly dependent on cooling rate and formulation | [13] |
| Jurkat T-cells (DMSO-free) | Spin Freezing (Controlled) | ~60% (with optimization) | Reduced vs. DMSO protocols | More sensitive to freezing parameters | [13] |
| Stem Cells (general) | Passive Freezing (containers) | Protocol-dependent | Varies by cell type | ~1°C/minute cooling rate achievable | [1] |
Advantages and Practical Considerations
Beyond quantitative performance metrics, each method presents distinct practical advantages and limitations that influence implementation decisions:
Table 2: Practical considerations for controlled-rate versus passive freezing systems
| Parameter | Controlled-Rate Freezing (CRF) | Passive Freezing (PF) |
|---|---|---|
| Control Over Process | Precise control of cooling rate, nucleation, and thermal profile | Limited control; dependent on insulation properties |
| Documentation | Comprehensive data logging and temperature profiling | Minimal documentation capabilities |
| Equipment Cost | High initial investment and operating costs | Low-cost equipment and minimal consumables |
| Technical Expertise | Requires specialized training and optimization | Minimal technical barrier to implementation |
| Scalability | Potential bottleneck for large batches | Easily scalable with multiple units |
| Regulatory Compliance | Extensive documentation supports validation | More challenging to validate and standardize |
| Best Applications | Sensitive cells, clinical applications, regulated environments | Research settings, robust cell types, resource-limited environments |
Experimental Protocols and Methodologies
Standardized Cryopreservation Workflow
A generalized cryopreservation protocol applicable to multiple cell types involves several critical stages where precise execution directly impacts viability outcomes [1]:
Figure 2: Standardized workflow for cell cryopreservation highlighting critical stages that impact cell viability.
Specialized Protocol for Hematopoietic Progenitor Cells
A detailed methodology for hematopoietic progenitor cell cryopreservation demonstrates the specific parameters used in comparative studies [12]:
Cell Preparation: Concentrate or dilute HPC products to achieve optimal cell concentration of 600-800 × 10⁶ total nucleated cells/mL, with a maximum of 1200 × 10⁶ TNC/mL.
Cryoprotectant Formulation: Prepare cryoprotectant solution containing 15% DMSO and 9% albumin in Plasmalyte-A. Maintain samples on a controlled-rate freezer rack or within an insulated container for passive freezing.
Controlled-Rate Freezing Protocol:
- Cool at a rate of 1°C/minute until freezing occurs
- Implement rapid cooling to counteract latent heat of fusion release
- Resume cooling at 1°C/minute until reaching desired endpoint temperature
Passive Freezing Protocol:
- Place samples in metal cassettes wrapped in absorbent pads or styrofoam insulation
- Transfer to -80°C mechanical freezer
- Achieve approximate cooling rate of 1-2°C/minute through insulation properties
Storage: Transfer all samples to liquid nitrogen freezers for long-term storage below -150°C within 48 hours of collection.
Advanced Technique: Spin Freezing for T-Cells
Recent research utilizing spin freezing technology has enabled precise separation of freezing phases to study their individual effects on Jurkat T-cell viability [13]:
Cell Culture: Maintain Jurkat T-cells in RPMI medium supplemented with 10% FBS, antibiotics, and sodium pyruvate at concentrations between 0.5-3×10⁶ cells/mL.
Cryoprotectant Formulation: Prepare both DMSO-containing (10% v/v) and DMSO-free formulations (typically sugar-based or polymer-based alternatives).
Spin Freezing Process:
- Place cell suspension in vials and spin rapidly around longitudinal axis
- Expose to cooling gas with controlled temperature and flow rate
- Utilize infrared measurements for non-invasive temperature monitoring
- Precisely control cooling rate before nucleation (0.5-10°C/minute)
- Induce nucleation at predetermined supercooling levels
- Regulate ice crystal growth phase independently (1-20°C/minute)
Parameter Optimization: Systematically vary individual freezing phases while holding others constant to isolate their effects on post-thaw viability and morphology.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful cryopreservation requires careful selection of specialized reagents and equipment tailored to specific cell types and research applications:
Table 3: Essential research reagents and equipment for cryopreservation studies
| Category | Specific Examples | Function & Application Notes |
|---|---|---|
| Cryoprotectants | Dimethyl sulfoxide (DMSO) | Penetrating cryoprotectant; reduces ice crystal formation but exhibits cytotoxicity [12] [13] |
| Cryoprotectants | Deep Eutectic Solvents (DES) | Emerging alternatives; choline chloride-glycerol shows promise for reduced toxicity [14] |
| Cryoprotectants | Sucrose, Trehalose | Non-penetrating cryoprotectants; provide extracellular protection [13] |
| Freezing Media | CryoStor CS10 | Serum-free, defined formulation for multiple cell types [1] |
| Freezing Media | mFreSR | Specialized for human ES and iPS cells [1] |
| Freezing Media | Home-made FBS/DMSO | Traditional approach; concerns about lot-to-lot variability [1] |
| Freezing Equipment | Controlled Rate Freezers (CryoMed) | Programmable cooling with documentation capabilities [12] [4] |
| Freezing Equipment | Passive Freezing Containers (Mr. Frosty, CoolCell) | Insulated devices for ~1°C/minute cooling in -80°C freezers [1] |
| Freezing Equipment | Spin Freezing Systems | Research tools for precise phase separation studies [13] |
| Storage Systems | Liquid Nitrogen Freezers | Long-term storage below -135°C required for maximum stability [4] [1] |
| Storage Systems | Mechanical -80°C Freezers | Short-term storage only; gradual viability loss occurs over time [1] |
| Quality Assessment | Flow Cytometry | Viability analysis and surface marker expression [12] [13] |
| Quality Assessment | Colony Forming Assays | Functional assessment of stem cell populations [12] [5] |
The physics of ice crystal formation presents a fundamental challenge in cryopreservation, with both controlled-rate and passive freezing methods offering distinct pathways to mitigate cellular damage. Current evidence suggests that passive freezing can achieve comparable results to controlled-rate freezing for certain cell types, particularly hematopoietic progenitor cells, when properly optimized [12] [6]. However, controlled-rate systems provide superior process control, documentation, and reproducibility advantages that remain valuable for sensitive applications and regulated environments [7].
The choice between methodologies ultimately depends on specific research requirements, cell type sensitivity, available resources, and regulatory considerations. As cryopreservation science advances, the development of novel cryoprotectants with reduced toxicity [14] [13] and improved understanding of phase-specific freezing parameters [13] will continue to enhance outcomes across both platforms. Future research directions should focus on optimizing passive freezing protocols for challenging cell types, establishing standardized validation approaches, and reducing the operational barriers associated with controlled-rate freezing systems to make high-quality cryopreservation more accessible across the research and clinical spectrum.
Cryopreservation is a fundamental technology employed in biomedical research and clinical applications to store biological materials, including cells, tissues, and therapeutic products, at extremely low temperatures, typically below -135°C [15] [16]. By reducing kinetic and molecular activity within cells, this process effectively halts biochemical processes and biological aging, enabling long-term storage and availability of biological resources on demand [15] [16]. The origins of low-temperature tissue storage research date back to the late 1800s, with significant breakthroughs occurring in the mid-20th century when the mechanisms of freezing injury began to be understood [15]. Today, cryopreservation enables critical medical applications including hepatocyte and pancreatic islet transplantation, blood transfusion, bone marrow transplantation, artificial insemination, and in vitro fertilization [15].
Without protective measures, the freezing process is typically lethal to cells. Since water constitutes approximately 80% of tissue mass, the freezing of water both intracellularly and extracellularly imposes the most significant influence over harmful biochemical and structural changes [15]. Two primary theories attempt to explain the harmful effects of freezing on cells: (1) ice crystals mechanically disrupt cellular membranes, making it impossible to obtain structurally-intact cells after thawing; and (2) lethal increases in solute concentration occur in the remaining liquid phase as ice crystals form during cooling [15]. The formation of ice crystals during freezing can compromise cellular membrane structural integrity and lead to osmotic stress from freeze-concentration of dissolved salts [17]. Cryoprotective agents (CPAs) were developed specifically to mitigate these damaging effects, allowing successful storage of cells in a solid phase at supercool temperatures without the formation of damaging ice crystals [15].
This technical guide explores the role of dimethyl sulfoxide (DMSO) and other cryoprotective agents in preventing cellular damage during cryopreservation, framed within the ongoing research discourse comparing controlled-rate freezing versus passive freezing methodologies. The efficacy of these cryoprotectants is intimately connected with the freezing protocols employed, making the understanding of both components essential for optimizing cryopreservation outcomes.
Mechanisms of Freezing Damage
To fully appreciate the protective role of cryoprotectants, one must first understand the specific mechanisms of cellular injury during freezing and thawing. When cells are exposed to temperatures below 0°C without cryoprotective intervention, several simultaneous damaging processes occur.
Intracellular and Extracellular Ice Formation
The formation of ice crystals represents one of the most significant sources of cryoinjury. Ice can form both extracellularly and intracellularly, with the latter being particularly damaging [18]. Intracellular ice crystals mechanically disrupt cellular membranes and organelles, making it impossible to obtain structurally-intact cells after thawing [15]. While early research focused on the mechanical damage from ice crystals, it was later revealed that osmotic stress is a main contributor to cell death during cryopreservation [15]. As ice forms, solutes are excluded from the developing crystal lattice and displaced to the diminishing liquid phase, effectively increasing solute concentration to lethal levels within the cell [15].
Solute Concentration and Osmotic Stress
As water freezes, the concentration of dissolved salts and other solutes in the remaining liquid phase increases dramatically, leading to a phenomenon known as freeze-concentration [17]. This creates substantial osmotic stress across cell membranes, causing water to exit cells and leading to harmful cell dehydration [15] [17]. The deadly increases in solute concentration are now understood to be a primary factor in cell injury at low cooling rates [18]. Experimental data demonstrate that the extent of damage to human red blood cells during freezing in solutions of sodium chloride/glycerol/water can be quantitatively accounted for by the increase in solute concentration [18].
Additional Mechanisms of Cryoinjury
Beyond ice formation and osmotic stress, several additional mechanisms contribute to freezing damage. Eutectic phase transformations, where mixtures of substances solidify at specific ratios, also contribute to reduced cell recovery [17]. Furthermore, during the cryopreservation of biological fluids and cellular samples, lipid peroxidation represents a major event in cellular membrane damage, serving as a trigger for other cellular alterations including oxidative stress, DNA and protein damage, abnormal lipid signaling, and activation of cell death programs [19]. The cumulative effect of these damaging mechanisms makes unprotected cooling and thawing of cells a process incompatible with life, necessitating the use of cryoprotective agents.
Cryoprotectant Classification and Mechanisms of Action
Cryoprotective agents are classified based on their ability to cross cell membranes and their specific mechanisms of action. Understanding these classifications is essential for selecting appropriate CPAs for specific cell types and applications.
Permeating Cryoprotectants
Permeating agents (PAs) are characterized by their relatively small size (typically less than 100 daltons) and amphiphilic nature, which allows them to easily penetrate cell membranes where they can exert their protective effects [15]. Common examples include dimethyl sulfoxide (DMSO), glycerol (GLY), ethylene glycol (EG), and propylene glycol (PG) [15]. These compounds are highly water soluble at low temperatures and ideally minimally toxic to cells [15]. The protective effects of permeating cryoprotectants derive primarily from their ability to hydrogen bond with water [15]. As these agents interact strongly with water through hydrogen bonding, the freezing point of water is depressed, and fewer water molecules are available to form critical nucleation sites required for crystal formation [15]. This promotes vitrification—the formation of solid water with an irregular, amorphous structure—rather than destructive ice crystallization [15]. Additionally, at specific concentrations, some PAs like DMSO increase membrane permeability by affecting membrane dynamics, potentially facilitating water replacement by cryoprotectants [15].
Non-Permeating Cryoprotectants
Non-permeating agents (NPAs) constitute the second category of cryoprotectants. These compounds are typically larger molecules or polymers that do not penetrate intracellularly and therefore exert their protective influence outside the cell [15] [19]. Commonly-used agents in this class include polyethylene glycol (PEG), polyvinylpyrrolidone (PVP), raffinose, sucrose, and trehalose [15]. Non-permeating agents induce vitrification by the same mechanism as permeating agents but operate extracellularly and to a lesser extent [15]. They increase extracellular osmolality, causing controlled cell dehydration as a stabilization mechanism before freezing [19]. Because of this function, non-permeating agents typically require slow cooling rates [19]. These agents are also added to thawing media to slow down the influx of water into the cell, thus preventing osmotic shock and lysis during rehydration [19].
Specialized Natural Cryoprotectants
In addition to synthetic compounds, several natural cryoprotectants have been identified with unique protective properties. Trehalose, a disaccharide produced by a wide variety of organisms including bacteria, fungi, insects, plants, and some invertebrates, demonstrates remarkable stability under extreme temperatures due to its acetal link structure [15]. Antifreeze proteins produced by various animals serve as non-penetrating cryoprotectants through their ability to inhibit ice recrystallization [19]. Natural deep eutectic systems (NADES) represent another class of natural cryoprotectants that have gained research interest as potential alternatives to synthetic compounds [19]. These natural agents often demonstrate reduced cellular toxicity compared to their synthetic counterparts while providing comparable or, in some cases, superior protection for specific cell types.
Table 1: Classification and Properties of Common Cryoprotectants
| Cryoprotectant | Classification | Molecular Weight | Common Concentrations | Key Mechanisms |
|---|---|---|---|---|
| Dimethyl Sulfoxide (DMSO) | Permeating | 78.13 g/mol | 5-15% [20] | Depresses freezing point, increases membrane permeability, promotes vitrification [15] |
| Glycerol (GLY) | Permeating | 92.09 g/mol | 5-15% [20] | Hydrogen bonding with water, reduces ice crystal formation [15] |
| Ethylene Glycol (EG) | Permeating | 62.07 g/mol | 4-8M (vitrification) [21] | Rapid penetration, promotes glassy state [15] |
| Propylene Glycol (PG) | Permeating | 76.09 g/mol | Varies by cell type | Low toxicity alternative for specific applications [15] |
| Trehalose | Non-Permeating | 342.3 g/mol | 0.05-0.5M [21] | Membrane stabilization, osmotic control, hydrogen bonding [15] |
| Sucrose | Non-Permeating | 342.3 g/mol | 0.1-0.5M [21] | Extracellular vitrification, osmotic buffer [15] |
| Polyethylene Glycol (PEG) | Non-Permeating | Varies by polymer | Varies by formulation | Macromolecular crowding, surface modification [19] |
| Antifreeze Proteins | Non-Permeating | Varies by protein | Low concentrations | Ice recrystallization inhibition [19] |
DMSO as the Benchmark Cryoprotectant
Dimethyl sulfoxide has emerged as the most widely utilized cryoprotectant in biomedical research and clinical applications since its discovery as a CPA, serving as the benchmark against which other cryoprotectants are measured.
Historical Context and Adoption
The cryoprotective properties of DMSO were discovered following earlier work with glycerol. In the late 1940s, researchers discovered that glycerol could increase the survivability of spermatozoa in subfreezing temperatures [15]. DMSO was subsequently identified as an effective alternative with different physicochemical properties. A commonly used cryoprotective agent currently employed is DMSO, which is added to cell media prior to the freezing process at concentrations typically around 10% [15]. DMSO's rapid penetration across cell membranes and high cryoprotective efficiency made it particularly advantageous for clinical applications where cryoprotectant removal before administration was not feasible [22].
Molecular Mechanisms of Action
DMSO exerts its cryoprotective effects through multiple molecular mechanisms. When added to cell media at standard concentrations (approximately 10% or 2M), DMSO increases the porosity of the cellular membrane, allowing water to flow more freely through the membrane [15]. Like glycerol, DMSO helps prevent the formation of water crystals by increasing intracellular solute concentration, thus aiding in the vitrification of water at low temperatures [15]. The concentration of DMSO significantly influences its effects on biological membranes. At low concentrations (5%), evidence suggests DMSO decreases membrane thickness and increases membrane permeability [15]. At commonly used concentrations (10%), water pore formation in biological membranes is induced, which can be advantageous as intracellular water can be more readily replaced by cryoprotectants that promote vitrification [15]. However, at higher, toxic concentrations (40%), lipid bilayers begin to disintegrate [15]. Recent research using updated AMBER force fields has provided more detailed understanding of DMSO-lipid interactions, showing that DMSO preferentially partitions at the hydrophobic-hydrophilic interface of lipid membranes while being partially excluded from the polar headgroup region relative to water [17].
Limitations and Toxicity Concerns
Despite its effectiveness, DMSO presents significant limitations due to its concentration-dependent toxicity to cells [17] [19]. DMSO causes mitochondrial damage to astrocytes and negatively impacts cellular membrane/cytoskeleton structure and integrity by interacting with proteins and dehydrating lipids [21]. The presence of DMSO in culture medium can induce unwanted stem cell differentiation and, with repeated use even at sub-toxic levels, can affect cellular epigenetic profile resulting in undesirable phenotypic disturbances [21]. In clinical applications, adverse reactions from cardiac, neurological, and gastrointestinal systems have been reported in patients receiving DMSO-containing cellular products [21]. These adverse reactions are dose-dependent, with guidelines recommending that the maximal daily dose of DMSO should not exceed 1 gram per kilogram of patient body weight [22]. This toxicity profile has motivated the search for DMSO-reduction strategies and DMSO-free cryopreservation protocols.
Controlled-Rate Freezing vs. Passive Freezing: The Technical Framework
The effectiveness of cryoprotectants is intimately connected with the freezing methodology employed. The debate between controlled-rate freezing and passive freezing represents a significant technical consideration in cryopreservation protocol development.
Principles of Controlled-Rate Freezing
Controlled-rate freezing (CRF) has long been considered the gold standard for cryopreservation of sensitive biological samples [6] [12]. This method utilizes specialized equipment that decreases the product temperature incrementally according to a preset program [12]. In a standard CRF protocol, the HPC product is cooled at a rate of 1°C/min until freezing occurs [12]. As the product starts to freeze, there is a release of latent heat of fusion which causes the temperature to rise, requiring the CRF program to rapidly cool the product to counteract this effect [12]. Once the product has solidified, cooling resumes at a rate of 1°C/min until the temperature reaches the desired storage temperature [12]. CRF enables precise freeze rates, contains a racking system, possesses a fully programmable controller, and provides a thermal profile of the process, offering complete documentation of the freezing parameters [12].
Methodology of Passive Freezing
Passive freezing (PF), also known as uncontrolled-rate or non-controlled rate freezing, utilizes a -80°C mechanical freezer without active temperature control programming [6] [12]. In this method, HPC products in metal cassettes may be wrapped in disposable absorbent pads or styrofoam insulation to adjust the cooling rate to the desired 1-2°C/min [12]. While nucleation is uncontrolled and cooling rates are not easily or consistently achievable with this method, it represents a simple, convenient, and cost-effective alternative to CRF [12]. Passive freezing doesn't require the presence of staff to transfer products at the end of the freeze cycle, as products can be kept in the -80°C mechanical freezer until they can be transferred to liquid nitrogen storage for long-term preservation [12].
Comparative Performance Analysis
Recent research has demonstrated comparable outcomes between controlled-rate and passive freezing methodologies. A 2025 retrospective study comparing 50 hematopoietic progenitor cell (HPC) products found that although the mean total nucleated cell (TNC) viability post-thaw was greater for HPCs frozen using CRF compared to PF (74.2% ± 9.9% vs 68.4% ± 9.4%), there was no significant difference in CD34+ cell viability post-thaw between the groups (77.1% ± 11.3% vs 78.5% ± 8.0%) [6]. Most importantly, the number of days to neutrophil engraftment (12.4 ± 5.0 vs 15.0 ± 7.7) and platelet engraftment (21.5 ± 9.1 vs 22.3 ± 22.8) were similar between the two groups, leading researchers to conclude that cryopreservation outcomes using CRF or PF are comparable [6]. These findings support PF as an acceptable alternative to CRF for initial cryopreservation before long-term storage in a liquid nitrogen freezer.
Table 2: Comparison of Controlled-Rate Freezing vs. Passive Freezing Methods
| Parameter | Controlled-Rate Freezing | Passive Freezing |
|---|---|---|
| Cooling Rate Control | Precise, programmable (~1°C/min) [12] | Uncontrolled, approximately 1-2°C/min with insulation [12] |
| Equipment Cost | High (specialized equipment) [12] | Low (standard -80°C freezer) [12] |
| Technical Complexity | High (requires trained staff) [12] | Low (simple procedure) [12] |
| Process Monitoring | Comprehensive thermal profiling [12] | Limited temperature monitoring [12] |
| Nucleation Control | Controlled | Uncontrolled [12] |
| Staff Time Requirements | High (requires transfer at cycle end) [12] | Low (can transfer next business day) [12] |
| Post-Thaw TNC Viability | 74.2% ± 9.9% [6] | 68.4% ± 9.4% [6] |
| Post-Thaw CD34+ Viability | 77.1% ± 11.3% [6] | 78.5% ± 8.0% [6] |
| Neutrophil Engraftment (days) | 12.4 ± 5.0 [6] | 15.0 ± 7.7 [6] |
| Platelet Engraftment (days) | 21.5 ± 9.1 [6] | 22.3 ± 22.8 [6] |
DMSO Reduction and Alternative Strategies
Growing concerns regarding DMSO toxicity have prompted extensive research into reduction strategies and alternative cryoprotectants, particularly for clinical applications where patient safety is paramount.
DMSO Reduction Techniques
For situations where complete DMSO elimination is not feasible, several reduction strategies have been developed. Post-thaw DMSO reduction through washing procedures has been implemented for patients at high risk of adverse reactions, such as those with chronic renal failure caused by secondary amyloidosis or cardiac amyloidosis [22]. The most widely used technique involves gradual dilution of the cell suspension with subsequent centrifugation and addition of cryoprotectant-free solution [22]. The composition of the washing medium is critical and should include components acceptable from a clinical perspective, typically saline solutions/electrolytes such as 0.9% NaCl, Normosol-R, or Plasma-Lyte 148 supplemented with dextran-40, human serum albumin, or hydroxyethyl starch [22]. Automated closed systems like the COBE 2991 Cell Processor, Sepax S-100, and Biosafe SA have been developed specifically for this purpose [22]. However, these washing procedures present their own challenges, including significant cell loss—one study reported a median decrease of 48.51% in viable CD34+ cells after DMSO reduction [22].
Vitrification Mixtures and Combination Approaches
Another effective strategy involves using vitrification mixtures that combine permeating and non-permeating agents to reduce the required concentration of any single toxic CPA [15]. Since both permeating and non-permeating agents share the same vitrification mechanism, non-permeating agents can be added to solution to allow successful cryobanking with lower concentrations of permeating agents [15]. This approach reduces PA-induced toxicity while increasing cellular viability and yields post-thaw [15]. Research by Kojayan et al. demonstrated that multi-molar combinations of reduced concentrations of ethylene glycol and DMSO could successfully cryopreserve both human and murine islet cells with reduced adverse effects [15]. Similar combination approaches have shown promise across various cell types, leveraging the synergistic effects of multiple cryoprotectants while minimizing individual compound toxicity.
Emerging DMSO-Free Cryopreservation Protocols
Complete elimination of DMSO from cryopreservation protocols represents the ultimate goal for many applications, particularly in cell therapy and regenerative medicine. Several DMSO-free alternatives have shown promising results in research settings. Natural cryoprotective agents, including antifreeze proteins, sugars, and natural deep eutectic systems, have demonstrated potential as DMSO replacements [19]. Trehalose-based cryosolutions containing ethylene glycol or glycerol have maintained high cell viability and stability in human induced pluripotent stem cells while preserving their morphology, self-renewal, pluripotency, and differentiation capacity [21]. Synthetic polymers like polyampholytes have shown remarkable cryoprotective properties, with human bone marrow-derived mesenchymal stem cells maintaining high viability and biological properties even after 24 months of cryopreservation at -80°C [21]. Commercial DMSO-free cryoprotectant solutions such as HP01 (Macopharma), CryoScarless, and CryoProtectPureSTEM have demonstrated comparable results to DMSO-preserved controls for hematopoietic stem cells, T-cells, and CD34+ cells [21].
Cell-Specific Considerations and Protocols
Optimizing cryopreservation protocols requires consideration of cell-specific characteristics, as different cell types demonstrate varying sensitivities to freezing conditions and cryoprotectants.
Cell-Type Specific Recommendations
Research has identified specific cryopreservation requirements for different cell types based on their biological properties and membrane characteristics. For example, rapid cooling is associated with better cryopreservation outcomes for oocytes, pancreatic islets, and embryonic stem cells, while slow cooling is recommended for cryopreservation of hepatocytes, hematopoietic stem cells, and mesenchymal stem cells [15]. These differences likely reflect variations in membrane lipid composition, surface area to volume ratio, and intrinsic tolerance to osmotic stress. Beyond cooling rates, yields can be further maximized by implementing additional pre-cryo steps such as pre-incubation with glucose and antioxidants, alginate encapsulation, and selecting cells within an optimal age range and functional ability [15]. The developmental origin, differentiation status, and metabolic characteristics of specific cell types all contribute to their unique cryopreservation requirements.
Standardized Cryopreservation Workflow
A generalized protocol for cell cryopreservation incorporates key steps applicable across multiple cell types with specific modifications based on cell-specific requirements. Cells should be harvested during log-phase growth at a high concentration of at least 90% viability and at as low a passage number as possible [16]. For adherent cells, gentle detachment from the tissue culture vessel following standard subculture procedures is essential to minimize damage [16]. After resuspension in complete growth medium and determination of cell concentration and viability, the cell suspension is centrifuged at approximately 100-400 × g for 5 to 10 minutes [16]. The supernatant is carefully removed, and the cell pellet is resuspended in cold freezing medium at the recommended viable cell density for the specific cell type [16]. Aliquots of the cell suspension are dispensed into sterile cryogenic storage vials, with frequent mixing to maintain a homogeneous cell suspension [16]. Finally, cells are frozen slowly by reducing the temperature at approximately 1°C per minute using a controlled rate cryo-freezer or cryo-freezing container before transfer to liquid nitrogen for long-term storage in the gas phase below -135°C [16].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Reagents and Materials for Cell Cryopreservation
| Reagent/Material | Function | Examples/Specifications |
|---|---|---|
| Cryoprotective Agents | Prevent ice crystal formation, reduce freezing point | DMSO, glycerol, ethylene glycol, trehalose, sucrose [15] [16] |
| Base Medium | Provides nutritional support during freezing | Complete growth medium with serum or serum-free formulations [16] |
| Protein Source | Protects cells from freeze-thaw stress | Fetal bovine serum, bovine serum albumin, human serum albumin, platelet lysate [16] |
| Dissociation Reagents | Detach adherent cells from culture surfaces | Trypsin, TrypLE Express, accutase [16] |
| Balanced Salt Solution | Maintain osmotic balance during processing | DPBS (without calcium, magnesium, or phenol red) [16] |
| Cryogenic Storage Vials | Secure containment for frozen samples | Sterile, leak-proof vials suitable for liquid nitrogen storage [16] |
| Controlled-Rate Freezer | Precise temperature regulation during freezing | Programmable freezer with 1°C/min cooling capability [12] |
| Passive Freezing Device | Alternative freezing method | Insulated containers (e.g., "Mr. Frosty") for -80°C freezing [16] |
| Liquid Nitrogen Storage | Long-term preservation below -135°C | Liquid nitrogen freezer with vapor phase storage capability [16] |
| Viability Assessment Tools | Pre-freeze and post-thaw quality control | Automated cell counters, hemocytometers, Trypan Blue [16] |
Cryoprotectants serve as essential shields against the multiple damaging processes that occur during freezing and thawing of biological samples. DMSO has remained the benchmark cryoprotectant for decades due to its effective penetration of cell membranes and ability to promote vitrification, but its concentration-dependent toxicity has driven the development of reduction strategies and alternative approaches. The interplay between cryoprotectant selection and freezing methodology is crucial, with recent evidence demonstrating that passive freezing can achieve comparable results to controlled-rate freezing for critical clinical applications like hematopoietic progenitor cell transplantation. As cryopreservation continues to enable advances in cellular therapies, regenerative medicine, and biomedical research, ongoing optimization of cryoprotectant formulations and freezing protocols will be essential. The ideal balance of efficacy, safety, and practicality will likely involve cell-specific solutions that may include combination approaches, novel natural cryoprotectants, and improved understanding of the fundamental mechanisms of cryoprotection at the molecular level.
In the field of cryopreservation, the successful long-term storage of biologics—from single cells to complex tissues—is paramount to advancements in drug development, cellular therapies, and regenerative medicine. The process, however, introduces two primary adversaries: slow-cooling injury and rapid-cooling injury. These distinct forms of cellular damage occur during the critical phase change of water to ice and are governed by different biophysical principles. Understanding their mechanisms is not merely an academic exercise but a practical necessity for developing robust cryopreservation protocols. This guide frames this understanding within the ongoing scientific discourse comparing controlled-rate freezing (CRF)—the traditional gold standard characterized by precise, slow cooling—and passive freezing (PF)—a simpler, uncontrolled-rate method. Recent research, including a 2025 retrospective study, confirms that PF yields comparable engraftment outcomes to CRF for hematopoietic progenitor cells (HPCs), challenging long-held assumptions and underscoring the need to optimize protocols against both cooling injuries [12] [6].
Core Injury Mechanisms: A Biophysical Perspective
The journey to sub-zero temperatures subjects cells to profound physical stresses. The two main cooling strategies inflict damage through fundamentally different pathways.
Slow-Cooling Injury ("Solution Effects" Injury)
Slow-cooling, typically defined as a controlled rate of about 1°C/min, causes water to freeze preferentially in the extracellular space. This creates a hypertonic environment, driving water out of the cell and leading to severe cellular dehydration and shrinkage [23]. The injury is not primarily from ice itself, but from the prolonged exposure to the concentrated solutes in the unfrozen fraction—the so-called "solution effects." This hypertonic stress can denature proteins, disrupt lipid membranes, and cause lethal cell shrinkage beyond a critical minimum volume [24] [23]. The damaging effects of slow cooling are most pronounced within a critical temperature range of -15°C to -60°C [24].
Rapid-Cooling Injury (Intracellular Ice Formation)
In contrast, rapid-cooling occurs at rates too fast for water to osmotically exit the cell. The intracellular solution becomes highly supercooled, leading to the nucleation and growth of ice crystals inside the cell. This intracellular ice formation (IIF) is almost universally lethal, as it mechanically disrupts organelles and the cytoskeleton, destroying the cell's internal structure [23]. The cooling rate at which IIF becomes significant varies by cell type, dictated by the cell's surface area to volume ratio and membrane water permeability [23].
The following diagram illustrates the critical decision points during cooling that determine which injury mechanism a cell will encounter.
Quantitative Comparison of Injury Profiles
The distinct mechanisms of slow and rapid-cooling injuries manifest in measurable differences in cell viability, function, and biochemical stress. The table below summarizes key comparative data, primarily from a study on umbilical cord blood mononucleated cells, which includes hematopoietic stem cells [24].
Table 1: Quantitative Comparison of Slow- vs. Rapid-Cooling Injuries in Cord Blood Mononucleated Cells
| Parameter | Slow-Cooling Injury | Rapid-Cooling Injury | P-value |
|---|---|---|---|
| Cell Viability | 75.5% | 91.9% | P = 0.003 |
| Malondialdehyde (MDA) Content | 33.25 μM | 56.45 μM | P < 0.001 |
| Apoptosis Level | 3.81% | 5.18% | P = 0.138 |
| CD34+ Cell Enumeration | 23.32 cells/μL | 2.47 cells/μL | P = 0.001 |
The data reveals a critical paradox: while rapid-cooling preserves higher general cell viability, it inflicts significantly greater oxidative stress, as indicated by the higher malondialdehyde (MDA) content, a product of lipid peroxidation [24]. Most notably, rapid-cooling was particularly detrimental to the target hematopoietic stem cells (CD34+), severely compromising their recovery [24]. This underscores that the choice of cooling rate must be optimized for the specific, clinically relevant cell population within a heterogeneous sample.
Investigating Injuries: Key Experimental Protocols
Researchers employ specific methodologies to dissect the contributions of different injury mechanisms. The two primary protocols are graded freezing and two-step freezing.
Graded Freezing (Interrupted Slow Cooling)
This protocol is designed to isolate injury from slow cooling and solution effects [25] [26].
- Objective: To identify the sub-zero temperature range where slow-cooling injury is most severe.
- Methodology:
- Cells are cooled slowly (e.g., 1°C/min) in the presence of cryoprotectants.
- Cooling is interrupted at specific target temperatures (e.g., -15°C, -30°C, -45°C).
- At each temperature, samples are either plunged directly into Liquid Nitrogen (rapid cooling) or held for a defined period before plunging.
- All samples are thawed rapidly, and viability is assessed.
- Outcome Interpretation: A drop in viability after a specific hold temperature pinpoints the range of maximal slow-cooling injury. The presence of cryoprotectants like DMSO primarily acts to reduce this form of injury [25]. This method revealed that in articular cartilage, chondrocytes in the intermediate layer are most vulnerable to slow-cooling injury [25].
Two-Step Freezing (Interrupted Rapid Cooling)
This protocol is used to study and mitigate intracellular ice formation [23] [26].
- Objective: To permit dehydration during a hold step, thereby preventing IIF during subsequent rapid cooling.
- Methodology:
- Cells are first cooled rapidly from room temperature to a predetermined, high sub-zero hold temperature (e.g., -40°C).
- Samples are held at this temperature for a specific duration (minutes to hours).
- After the hold, samples are plunged into Liquid Nitrogen for final storage.
- Outcome Interpretation: The hold step provides time for water to osmotically leave the cell, reducing supercooling and thus the risk of IIF upon plunging. Optimizing the hold temperature and duration is critical for cell survival [23].
The workflow for these core experimental protocols is visualized below.
The Scientist's Toolkit: Essential Reagents & Materials
Successful cryopreservation research requires a suite of specialized reagents and equipment to control and study cooling injuries.
Table 2: Key Research Reagents and Materials for Cryopreservation Studies
| Tool | Function & Rationale |
|---|---|
| Permeating Cryoprotectants (e.g., DMSO) | Small molecules that cross the cell membrane (e.g., DMSO, glycerol). They reduce the fraction of freezable water and dilute high salt concentrations during slow cooling, thereby mitigating "solution effects" injury [24] [12] [23]. |
| Non-Permeating Cryoprotectants (e.g., HES) | Large molecules that remain outside the cell (e.g., Hydroxyethyl Starch, sucrose). They induce protective dehydration prior to freezing and modify extracellular ice crystal structure [12] [23]. |
| Programmable Controlled-Rate Freezer (CRF) | Equipment that provides precise, user-defined cooling rates (e.g., 1°C/min). Essential for studying slow-cooling injury and developing reproducible clinical-grade protocols [12] [23]. |
| -80°C Mechanical Freezer | Used for passive freezing (PF) protocols. Provides a non-controlled cooling environment. When combined with insulation (e.g., styrofoam), it can approximate slow cooling and is a cost-effective alternative to CRF [12]. |
| Liquid Nitrogen Storage | Provides ultralow temperatures (<-150°C) for long-term storage of vitrified or frozen samples, halting all biochemical activity [24] [12]. |
| Viability & Function Assays | A panel of assays is required post-thaw. These include flow cytometry with dyes like 7-AAD for viability, CD34+ enumeration for HPCs [12], colony-forming unit (CFU) assays for functionality, and lipid peroxidation assays (e.g., MDA tests) for oxidative stress [24]. |
Implications for Controlled-Rate vs. Passive Freezing Research
The fundamental understanding of cooling injuries directly informs the debate between CRF and PF. CRF offers meticulous control, theoretically minimizing both slow-cooling injury (through optimal cooling rates) and rapid-cooling injury (by preventing uncontrolled plunges) [12]. Conversely, PF in a -80°C freezer is simpler and cheaper but introduces variability in the cooling rate, potentially exposing cells to ill-defined stresses [12].
Strikingly, a growing body of evidence demonstrates equivalence between the methods for critical clinical outcomes. A 2025 study on HPCs found that while CRF yielded slightly higher post-thaw total nucleated cell (TNC) viability (74.2% vs. 68.4%), CD34+ cell viability and, crucially, the rates of neutrophil and platelet engraftment in patients were not significantly different [12] [6]. This suggests that for some cell types, the tolerance to the less-defined stresses of PF is sufficient for therapeutic success. The choice of method, therefore, may hinge on the specific cell type, the required consistency, and practical constraints like cost and workflow, all while keeping the "enemies" of slow and rapid cooling in check. Future research will continue to refine PF protocols and define their boundaries, solidifying the role of fundamental cryobiology in applied clinical science.
In the realms of cell and gene therapy, biobanking, and drug development, the cryopreservation of biological materials is a cornerstone technology. At the heart of this process lies a fundamental thermodynamic challenge: the management of the latent heat of fusion. This phenomenon represents the energy released when water transitions from a liquid to a solid state during freezing. For researchers and scientists, navigating this exothermic event is critical, as unmanaged heat release can cause uncontrolled ice crystal formation, leading to irreversible cellular damage, reduced cell viability, and compromised therapeutic efficacy [4] [27].
The management of this phase change is a primary differentiator between two fundamental cryopreservation approaches: controlled-rate freezing (CRF) and passive freezing (PF). Controlled-rate freezing employs specialized equipment to precisely manage heat extraction during this critical phase, while passive freezing relies on simpler, uncontrolled methods. A growing body of evidence, including a recent 2025 retrospective study, demonstrates that understanding and accounting for the latent heat of fusion allows both methods to achieve comparable success in clinical outcomes such as hematopoietic progenitor cell engraftment, challenging long-held assumptions about the necessity of complex equipment [6] [12]. This technical guide explores the core principles of the latent heat of fusion and provides a detailed framework for its management in advanced research and development.
Fundamental Principles of Latent Heat of Fusion
Thermodynamic Definition and Biological Significance
In thermodynamics, the enthalpy of fusion (or latent heat of fusion) is the change in enthalpy resulting from providing energy to a specific quantity of a substance to change its state from a solid to a liquid at constant pressure. The reverse process, freezing, releases an equal amount of energy [28]. For pure water, this value is 333.55 kJ/kg at 0°C and standard pressure. This energy does not cause a temperature change but is used to break the molecular bonds of the solid phase [28].
In biological systems, the situation is more complex. Cells and tissues are aqueous solutions containing various solutes, which depress the freezing point. However, the fundamental principle remains: when the material is cooled to its freezing point, the temperature will stall or plateau until the latent heat released by the forming ice is completely removed from the system [4] [29]. This is a critical period where the risk of intracellular ice formation is highest. The size and morphology of the resulting ice crystals are directly influenced by the rate at which this heat is removed [4].
The Physics of the Freezing Process
The freezing process for a biological sample involves three distinct stages of heat removal, as illustrated in the diagram below.
Figure 1: The Three Stages of the Freezing Process. The critical Phase 2 involves the release of the latent heat of fusion, creating a temperature plateau that must be carefully managed to ensure cell viability.
- Sensible Cooling Above Freezing Point: The temperature of the sample is reduced from its initial value to its freezing point. The heat removed during this phase is termed "sensible heat" because it results in a measurable temperature change [29].
- Latent Heat Release at Freezing Point: As ice begins to form, the sample temperature remains relatively constant at its freezing point despite continued heat removal. The energy being removed is the latent heat of fusion, which facilitates the phase change from liquid water to solid ice [4] [28] [29].
- Sensible Cooling of the Frozen Solid: Once the phase change is complete, further heat removal results in a temperature drop of the now-solid sample down to the final storage temperature [29].
Controlled-Rate Freezing vs. Passive Freezing: A Technical Comparison
Core Methodologies and Heat Management
The fundamental difference between controlled-rate freezing (CRF) and passive freezing (PF) lies in their approach to managing the three stages of freezing, particularly the critical latent heat release phase.
Controlled-Rate Freezing (CRF) employs a programmable freezer designed to precisely manage the cooling rate, typically at 1°C per minute for many cell types [4]. As the sample approaches its freezing point, the release of latent heat causes a temperature rise—a phenomenon known as the "heat of fusion bump." A key feature of CRF systems is their programmed response to this event: they often initiate a rapid temperature drop to actively counteract the released heat and ensure consistent ice crystal formation throughout the sample [4] [12]. This process provides a repeatable, validated cooling profile [4].
Passive Freezing (PF), also referred to as "uncontrolled-rate" or "straight freezing," involves placing samples in an insulated container inside a -80°C mechanical freezer. The insulation, which can be disposable absorbent pads, styrofoam, or other materials, serves to slow the cooling rate to an approximate target of 1–2°C/min [12]. However, in this method, nucleation is uncontrolled, and the cooling rates are not easily or consistently achievable across all samples. The system passively absorbs the latent heat without active countermeasures, leading to less reproducible thermal profiles [12].
Comparative Experimental Data and Outcomes
Recent clinical studies have provided quantitative data comparing the outcomes of these two methods. The following table summarizes key findings from a 2025 retrospective study on hematopoietic progenitor cells (HPCs) [6] [12].
Table 1: Comparison of Cryopreservation Outcomes: Controlled-Rate vs. Passive Freezing for Hematopoietic Progenitor Cells (HPCs) [6] [12]
| Parameter | Controlled-Rate Freezing (CRF) | Passive Freezing (PF) | P-value |
|---|---|---|---|
| Total Nucleated Cell (TNC) Viability (Post-Thaw) | 74.2% ± 9.9% | 68.4% ± 9.4% | 0.038 |
| CD34+ Cell Viability (Post-Thaw) | 77.1% ± 11.3% | 78.5% ± 8.0% | 0.664 |
| Days to Neutrophil Engraftment | 12.4 ± 5.0 | 15.0 ± 7.7 | 0.324 |
| Days to Platelet Engraftment | 21.5 ± 9.1 | 22.3 ± 22.8 | 0.915 |
The data indicates that while CRF showed a statistically higher post-thaw TNC viability, this did not translate into a significant difference in the viability of the therapeutically critical CD34+ cells. Most importantly, the clinical endpoints—neutrophil and platelet engraftment times—were not significantly different between the two groups [6] [12]. This suggests that for HPCs, PF is a clinically acceptable alternative to CRF.
Further supporting this, a 2012 study on rat mesenchymal stem cells (MSCs) concluded that a " 'straight freeze' protocol is no less effective in maintaining post-thaw viability of MSC compared to controlled rate freezing methods" [30]. The workflow for such a comparative experiment is detailed below.
Figure 2: Experimental Workflow for Comparing Freezing Protocols. A standard methodology for comparing CRF and PF involves splitting a cell batch, applying different freezing methods, and conducting a multi-faceted post-thaw analysis.
Essential Protocols for Managing Latent Heat
Protocol for Controlled-Rate Freezing with Seeding
This protocol is designed for use with a programmable controlled-rate freezer and includes a "seeding" step to control ice nucleation, which helps manage the release of latent heat [4].
- Step 1: Sample Preparation. Suspend cells in a cryoprotectant solution, such as 15% DMSO and 9% albumin in Plasmalyte-A [12]. Aliquot into cryovials or bags.
- Step 2: Loading. Place samples in the controlled-rate freezer chamber, which has been pre-cooled to 4°C. Ensure temperature probes are properly positioned if required.
- Step 3: Initiate Cooling Program.
- Step 4: Seeding. Once the sample is supercooled, hold the temperature. Induce ice nucleation by briefly touching the vial with forceps cooled in liquid nitrogen or using the freezer's automated seeding function. This triggers the release of latent heat.
- Step 5: Manage Latent Heat Release. After seeding, the sample temperature will rise due to the latent heat of fusion. The CRF program should actively counteract this by holding or rapidly cooling until the temperature stabilizes, indicating the phase change is complete.
- Step 6: Final Cooling. Continue cooling at a controlled rate (e.g., 1°C/min) to a final temperature such as -80°C or -100°C [12].
- Step 7: Transfer. Immediately transfer the samples to long-term storage in the vapor phase of a liquid nitrogen freezer (<-150°C) [4].
Protocol for Passive Freezing in a -80°C Mechanical Freezer
This protocol uses insulation to approximate a slow cooling rate and is suitable for cell types where PF has been validated, such as HPCs [6] [12] and MSCs [30].
- Step 1: Sample Preparation. Suspend cells in an appropriate cryoprotectant solution. For rat MSCs, a combination of 5% DMSO and 5% Hydroxyethyl starch (HES) has been successfully used with a straight freeze protocol [30]. Aliquot into cryovials.
- Step 2: Insulation. Place the cryovials into an insulated container, such as a cardboard box, styrofoam rack, or by wrapping in disposable absorbent pads. The insulation is critical for slowing the cooling rate to the desired ~1–2°C/min [12].
- Step 3: Freezing. Place the insulated container directly into a -80°C mechanical freezer. The freezing process is uncontrolled from this point.
- Step 4: Storage Duration. Samples can be kept in the -80°C freezer for a period (e.g., overnight or until transfer is convenient) [12].
- Step 5: Long-Term Storage. For stability beyond a few months, transfer the samples to a liquid nitrogen freezer for storage at <-150°C [4] [12].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Materials and Reagents for Freezing Protocols
| Item | Function & Critical Notes |
|---|---|
| Dimethyl Sulfoxide (DMSO) | A permeating cryoprotectant that reduces ice crystal formation by penetrating cells. Its concentration can often be reduced when combined with non-permeating agents like HES, mitigating its cytotoxicity [30]. |
| Hydroxyethyl Starch (HES) | A non-permeating cryoprotectant and volume expander that acts extracellularly to dehydrate cells and modify ice crystal growth. Molecular weight (e.g., 109-609 kDa) may play a minor role in its efficacy [30]. |
| Programmable Controlled-Rate Freezer | Equipment designed to provide precise, repeatable, and validated cooling profiles. It actively counters the latent heat of fusion release, ensuring consistent process control [4]. |
| -80°C Mechanical Freezer | Standard laboratory freezer used for passive freezing protocols. Serves as the heat sink for PF, but does not provide active cooling control [6] [12]. |
| Insulated Containers (e.g., Styrofoam, Pads) | Used in PF to slow the cooling rate by providing a thermal barrier, approximating a slow cooling rate of ~1-2°C/min [12]. |
| Liquid Nitrogen Storage System | Provides long-term storage at <-150°C. Vapor phase storage is generally recommended to avoid the risk of liquid nitrogen penetration and potential contamination or vial explosion [4]. |
The precise navigation of the latent heat of fusion remains a central challenge in the cryopreservation of biological materials. While controlled-rate freezing offers a gold standard of precision with active management of this critical phase, robust passive freezing techniques have demonstrated equivalent efficacy for specific cell types, including hematopoietic progenitor cells and mesenchymal stem cells [6] [30]. The choice between these methods should be informed by a cost-benefit analysis weighing process control, regulatory requirements, scalability, and the specific sensitivity of the biological material.
Future innovation in this field is being driven by the growing market for biopharmaceuticals and cell and gene therapies [31]. Trends include increased automation, sophisticated data logging for improved process validation, and the development of more advanced cryoprotectant solutions that reduce or eliminate the need for DMSO [31] [30]. A deep understanding of the fundamental thermodynamics of freezing, particularly the latent heat of fusion, empowers researchers and drug development professionals to optimize preservation protocols, ensure product viability, and ultimately accelerate the translation of novel therapies from the bench to the clinic.
From Theory to Practice: Implementing CRF and Passive Freezing in Your Lab
In the fields of cell and gene therapy, biobanking, and pharmaceutical development, the viability of precious biological samples hinges on the cryopreservation process. Controlled-rate freezers (CRFs) are sophisticated instruments designed to cool samples at a precise, programmable rate, mitigating the cellular damage inherent in the phase change from water to ice [4]. This process stands in contrast to passive freezing (PF), a non-controlled method that relies on placing insulated samples in a -80°C mechanical freezer [12]. While PF is a low-cost and simple alternative, CRFs provide documented, repeatable, and optimized cooling profiles that are often critical for clinical applications and sensitive biological materials [5] [4]. This guide delves into the technical workings of CRFs, framed within the research context that compares their efficacy with passive freezing methods.
Core Principles: The Science of Controlled Freezing
The Challenge of Latent Heat of Fusion
The primary challenge during freezing is the management of the latent heat of fusion [4]. As a sample cools, most of its water will freeze at approximately -2°C to -5°C. The change from liquid to solid releases a significant amount of heat, which, if not actively managed, causes a temporary but detrimental rise in the sample's temperature [4]. This uncontrolled temperature spike can lead to the formation of large, destructive ice crystals both inside and outside cells, reducing post-thaw viability [12] [4]. An optimal cooling rate is one of the critical factors influencing cell survival during cryopreservation [5].
How CRFs Achieve Control
CRFs actively manage this entire process. They decrease the product temperature incrementally according to a preset program [12]. A typical protocol for hematopoietic progenitor cells (HPCs) involves cooling at a rate of 1°C/min until freezing occurs. The CRF's sensors detect the release of latent heat and rapidly counter it with a burst of cooling. Once the sample has solidified, controlled cooling resumes at a set rate (e.g., 1°C/min) until the desired final temperature (often below -100°C) is reached [12]. This precise control minimizes ice crystal formation and the associated "solution effects," such as increased solute concentration, which can damage cells [4].
Figure 1: A typical controlled-rate freezing profile, showing how the process actively manages the heat released during ice formation.
CRF Technologies and System Designs
Liquid Nitrogen-Based CRFs
Many CRFs use liquid nitrogen (LN2) as the cooling medium. These systems can be further categorized:
- Planer-type CRFs: These units inject LN2 directly into a freezing chamber. A fan distributes the cold gas evenly, and sensors provide feedback to control valves that regulate LN2 flow, creating the precise temperature profile [32] [33].
- CryoLogic-type CRFs: This design features a cryochamber that stands directly in a liquid nitrogen bath. The temperature is regulated by a heater module that counteracts the LN2's cooling, creating the desired freezing curve through a balance of heating and cooling [32].
Liquid Nitrogen-Free (Mechanical) CRFs
As an alternative to LN2-dependent systems, mechanical CRFs like the Strex CytoSensei use a built-in refrigeration system to achieve controlled cooling [34]. These devices eliminate the need for a continuous LN2 supply, reducing operational costs and hazards. They are programmable and can hold a variety of tube and bag formats, making them suitable for many research and clinical applications [34].
The "Box-in-Box" Passive Freezing Device
To bridge the gap between simple passive freezing and expensive CRF systems, researchers have developed controlled cooling devices like the "box-in-box" [5]. This device uses thermal inertia to achieve a desired cooling rate. It consists of an aluminum enclosure and a precisely calculated thickness of polyethylene foam insulation. When transferred from room temperature to a -80°C freezer, the device achieves a consistent cooling rate of -1°C to -3.5°C/min, which is optimal for HSC cryopreservation [5]. This approach provides a standardized, low-cost, and reliable method without moving parts or LN2 consumption.
Comparative Analysis: CRF vs. Passive Freezing
Technical and Performance Comparison
The table below summarizes the key differences between controlled-rate freezing and passive freezing based on the available research.
Table 1: A side-by-side comparison of Controlled-Rate Freezing (CRF) and Passive Freezing (PF) methodologies.
| Feature | Controlled-Rate Freezing (CRF) | Passive Freezing (PF) |
|---|---|---|
| Control Mechanism | Programmable, electronic control via LN2 injection or mechanical cooling [32] [12] | Uncontrolled; relies on thermal insulation (e.g., Styrofoam, alcohol chambers) [5] [12] |
| Cooling Rate | Precise and repeatable (e.g., 1°C/min) [12] | Variable and less consistent; depends on insulation, geometry, and freezer [5] |
| Documentation | Detailed temperature profile logging for validation [5] | No built-in documentation; process is not easily verified [5] |
| Initial Cost | High [5] | Very low [5] |
| Operational Cost | High (LN2 consumption or electricity) [5] | Low (standard -80°C freezer) [12] |
| Best Application | Clinical settings, sensitive cells, regulatory compliance [5] [4] | Research settings, robust cells, backup method [12] |
Experimental Outcomes: Viability and Engraftment
A pivotal 2025 retrospective study directly compared CRF and PF for hematopoietic progenitor cells (HPCs). The experimental protocol involved cryopreserving 50 HPC products using either method with a cryoprotectant solution of 15% DMSO and 9% albumin in Plasmalyte-A [6] [12]. The key findings are summarized below.
Table 2: Key outcomes from a retrospective study comparing CRF and PF for HPC cryopreservation [6] [12].
| Outcome Measure | Controlled-Rate Freezing (CRF) | Passive Freezing (PF) | P-value |
|---|---|---|---|
| Total Nucleated Cell (TNC) Viability Post-Thaw | 74.2% ± 9.9% | 68.4% ± 9.4% | 0.038 |
| CD34+ Cell Viability Post-Thaw | 77.1% ± 11.3% | 78.5% ± 8.0% | 0.664 |
| Days to Neutrophil Engraftment | 12.4 ± 5.0 | 15.0 ± 7.7 | 0.324 |
| Days to Platelet Engraftment | 21.5 ± 9.1 | 22.3 ± 22.8 | 0.915 |
The study concluded that while TNC viability was statistically higher in the CRF group, the most critical metrics—CD34+ cell viability and engraftment times—showed no significant difference [6]. This indicates that for HPCs, PF is an acceptable alternative to CRF for initial cryopreservation [6] [12].
Figure 2: Experimental workflow for comparing CRF and PF for hematopoietic progenitor cells (HPCs).
The Scientist's Toolkit: Essential Equipment and Reagents
Table 3: Key equipment and reagents used in controlled-rate freezing protocols.
| Item | Function & Importance |
|---|---|
| Controlled-Rate Freezer | The core instrument; provides precise, programmable cooling to minimize ice crystal damage and maintain cell viability [34] [4]. |
| Cryoprotectant (e.g., DMSO) | A penetrating agent like Dimethyl Sulfoxide (DMSO) reduces intracellular ice crystal formation by binding water molecules and modulating cell dehydration during freezing [12]. |
| Protein Stabilizer (e.g., Albumin) | Often used in combination with DMSO (e.g., 9% albumin), it helps protect cell membranes and mitigates the osmotic stress and toxicity of cryoprotectants [12]. |
| Cryogenic Storage Vials/Bags | Specially designed containers that can withstand extreme thermal stresses (e.g., from +25°C to -196°C) without cracking or failing [34] [35]. |
| Liquid Nitrogen Storage Tank | For long-term storage of frozen samples at temperatures below -130°C, which is necessary to ensure ultimate stability of frozen cells [4]. |
| "Box-in-Box" Device | A low-cost, passive cooling device that provides a consistent and documented cooling rate as an alternative to expensive CRF systems [5]. |
Controlled-rate freezers are engineered solutions to a complex biophysical problem: how to safely transition biological materials from a fluid to a frozen state. Their ability to precisely manage the cooling rate, particularly through the release of latent heat, makes them the gold standard for cryopreserving sensitive and high-value samples [4]. However, research demonstrates that for certain cell types, such as hematopoietic progenitor cells, simpler passive freezing methods can produce clinically equivalent results in terms of cell viability and patient engraftment [6] [12]. The choice between CRF and PF, therefore, depends on a balance of factors including the sensitivity of the biological material, regulatory requirements, available budget, and the need for process documentation and validation.
In the field of biopreservation, the technique of passive freezing using a -80°C mechanical freezer stands as a robust, cost-effective alternative to controlled-rate freezing for the cryopreservation of critical biological materials. This method is defined by an uncontrolled-rate freezing process where materials are placed directly into a -80°C mechanical freezer, allowing the cooling rate to be determined by the insulating properties of the container and the freezer's environment [6] [12]. Within the broader thesis of comparing fundamental cryopreservation methodologies, passive freezing establishes its value not by mimicking the precise engineering of controlled-rate freezers, but by providing a simpler, accessible, and empirically effective pathway for long-term cell preservation, particularly for hematopoietic progenitor cells (HPCs) [6].
The core principle involves achieving a cooling rate that is slow enough to minimize intracellular ice formation—a primary cause of cell death—yet efficient enough to be practical. While controlled-rate freezers (CRFs) are often considered the gold standard for their programmable, step-wise cooling profiles, they are complicated, expensive, and time-consuming pieces of equipment [12]. Passive freezing, in contrast, leverages the consistent low temperature of a standard -80°C freezer, often with the aid of insulating devices to modulate the cooling rate, making it a viable option for a wide range of laboratories and clinical settings [12] [36].
Fundamental Principles of Passive Freezing
The Scientific Basis of Cryopreservation
The overarching goal of cryopreservation is to stabilize cells by dramatically reducing the kinetic energy that drives biochemical reactions and enzymatic activity, thereby effectively halting biological time [36]. This is achieved by converting liquid water within and around the cell into ice. However, this process is inherently stressful. The formation of extracellular ice crystals draws water out of the cell, leading to osmotic dehydration and an increase in the intracellular concentration of solutes. If cooling is too rapid, water does not have time to exit the cell, resulting in lethal intracellular ice formation [36].
The role of a cryoprotective agent (CPA), such as dimethyl sulfoxide (DMSO), is to mitigate these stresses. DMSO penetrates the cell, reduces the amount of ice formed at any given temperature, and allows water to exit the cell in a more controlled manner during the slow cooling process [12]. The passive freezing method is designed to facilitate this slow, controlled dehydration. The insulating container placed in the -80°C freezer creates a thermal buffer, ensuring the sample cools at an optimal rate of approximately 1-2°C per minute through the critical heat release phase (around -20°C to -40°C), which is comparable to the rate often targeted by controlled-rate freezers [12].
Comparative Advantages and Limitations
The decision to implement a passive freezing setup is often driven by a balance of practical considerations against technical requirements.
Advantages:
- Cost-Effectiveness: Eliminates the need for a large capital investment in a controlled-rate freezer [12].
- Operational Simplicity: The procedure is straightforward and does not require specialized training to operate complex programming interfaces [6] [12].
- Convenience and Workflow: Samples can be placed in the freezer and left, without the need for staff to be present at the end of a freeze cycle to transfer products to long-term storage [12].
- Proven Efficacy: Multiple studies, particularly in the context of HPCs, have demonstrated that passive freezing results in engraftment outcomes equivalent to controlled-rate freezing [6] [12].
Limitations:
- Lack of Active Control: The process does not allow for real-time adjustment of the cooling rate or active compensation for the latent heat of fusion released during ice crystal formation [12].
- Potential for Variability: Without a controlled environment, factors like freezer load, sample placement, and consistency of insulation can introduce variability in the freezing rate between runs [12].
- Limited Process Monitoring: Unlike CRFs, passive freezing does not typically generate a thermal profile or record of the freezing process for quality control purposes [12].
Essential Components of the Passive Freezing Setup
Core Equipment and Research Reagent Solutions
A properly configured passive freezing setup requires specific materials to ensure reproducibility and cell viability. The table below details the essential components.
Table 1: Essential Research Reagent Solutions and Materials for Passive Freezing
| Item | Function & Technical Specification |
|---|---|
| -80°C Mechanical Freezer | Core cooling unit; provides a stable -80°C environment to facilitate the slow freezing process. Requires validation of temperature uniformity and alarm systems. |
| Passive Freezing Device (e.g., alcohol-free cell freezing container, "Mr. Frosty," "CoolCell") | Insulating container engineered to achieve a consistent cooling rate of ~1°C/min when placed in a -80°C freezer. Modern versions are often alcohol-free, eliminating flammability and contamination risks [37]. |
| Cryoprotectant (e.g., DMSO) | Penetrating cryoprotective agent; reduces intracellular ice formation by modifying ice crystal structure and cellular dehydration. Typically used at a final concentration of 10% (v/v) [12] [36]. |
| Cryopreservation Medium | Base solution containing electrolytes (e.g., Plasmalyte-A), proteins (e.g., human serum albumin), and potentially nutrients. Provides an isotonic environment and supplements the cryoprotectant [12]. |
| Cryogenic Vials | Sterile, sealable containers for holding the cell suspension during freezing and storage. Must be capable of withstanding extreme temperatures without cracking. |
| Long-Term Storage Vessel (Liquid Nitrogen Tank) | For storage at temperatures below -150°C after the initial freezing process is complete, ensuring long-term stability [6] [36]. |
Workflow and Process Integration
The passive freezing procedure is not an isolated event but a critical step within a larger, integrated workflow from cell preparation to final clinical application or experimental use. The following diagram maps this end-to-end process, highlighting the specific role of the passive freezing setup.
Diagram 1: Integrated cell cryopreservation and thawing workflow.
Experimental Protocol and Validation Data
Detailed Methodological Workflow
A standardized protocol for passive freezing, as derived from clinical studies, ensures consistent results [12]:
- Cryoprotectant Preparation: Prepare a solution containing 15% DMSO and 9% albumin in an electrolyte solution like Plasmalyte-A. This solution should be sterile and kept cold before use.
- Cell Preparation: Concentrate or dilute the HPC product to an optimal cell concentration, typically between 600 to 800 x 10^6 total nucleated cells (TNC)/mL. This step is critical to avoid excessive cell death from overcrowding or over-dilution.
- Mixing: Combine the cell suspension with the prepared cryoprotectant solution in a 1:1 ratio. This achieves a final concentration of approximately 7.5% DMSO. Mix gently but thoroughly to ensure uniform exposure.
- Aliquoting: Dispense the cell-cryoprotectant mixture into cryogenic vials or bags. It is crucial to control the fill volume to ensure consistent freezing kinetics across all samples.
- Loading into Passive Freezing Device: Place the sealed vials into the passive freezing container (e.g., an alcohol-free freezing box). Ensure the container is configured according to the manufacturer's instructions to achieve the specified cooling rate.
- Initiation of Freezing: Immediately transfer the loaded container to the -80°C mechanical freezer. The location within the freezer should be consistent and away from the door to minimize temperature fluctuations.
- Long-Term Storage: After a minimum of 24 hours (or as validated), transfer the frozen vials to a liquid nitrogen freezer for long-term storage at temperatures below -150°C [6] [12].
Performance Validation and Comparative Metrics
The efficacy of this passive freezing method is validated by comparing key cell quality metrics and, most importantly, clinical outcomes against the controlled-rate freezing gold standard. The following table summarizes quantitative data from a retrospective clinical study [6].
Table 2: Comparative Analysis of Passive Freezing vs. Controlled-Rate Freezing for HPCs
| Parameter | Controlled-Rate Freezing (CRF) | Passive Freezing (PF) in -80°C Freezer | Statistical Significance (P-value) |
|---|---|---|---|
| Total Nucleated Cell (TNC) Viability (Post-Thaw) | 74.2% ± 9.9% | 68.4% ± 9.4% | P = 0.038 |
| CD34+ Cell Viability (Post-Thaw) | 77.1% ± 11.3% | 78.5% ± 8.0% | P = 0.664 (Not Significant) |
| Days to Neutrophil Engraftment | 12.4 ± 5.0 | 15.0 ± 7.7 | P = 0.324 (Not Significant) |
| Days to Platelet Engraftment | 21.5 ± 9.1 | 22.3 ± 22.8 | P = 0.915 (Not Significant) |
The data demonstrates that while TNC viability post-thaw was statistically lower in the passive freezing group, the viability of the clinically critical CD34+ progenitor cells was equivalent [6]. Crucially, the ultimate clinical endpoints—time to neutrophil and platelet engraftment—showed no statistically significant difference, leading to the conclusion that cryopreservation outcomes using CRF or PF are comparable, and PF is an acceptable alternative [6] [12].
Within the broader research landscape comparing cryopreservation fundamentals, the -80°C mechanical freezer-based passive freezing setup firmly establishes itself not as a mere compromise, but as a validated, practical, and economically viable method for the preservation of hematopoietic progenitor cells and other cellular therapeutics. The experimental evidence confirms that despite its operational simplicity, this method supports equivalent CD34+ cell viability and engraftment potential compared to controlled-rate freezing.
The choice between passive and controlled-rate freezing ultimately depends on specific research or clinical needs, regulatory requirements, and available resources. However, for a wide array of applications demanding robustness and reliability without prohibitive cost, the passive freezing setup offers a scientifically sound and proven solution, effectively "mind the gap" between cell processing and final application [36].
Cryopreservation serves as a critical backbone for the cell and gene therapy (CGT) industry, ensuring the stability, viability, and efficacy of living cellular products from manufacturing to patient administration. As the field advances rapidly, standardized and optimized cryopreservation practices become increasingly vital for maintaining manufacturing standards and regulatory compliance. The latest survey from the ISCT Cold Chain Management and Logistics Working Group provides an authoritative snapshot of current industry practices, challenges, and trends. This whitepaper examines these key findings, framing them within the ongoing scientific discourse on controlled-rate versus passive freezing methodologies to offer researchers and drug development professionals a comprehensive technical guide.
Key Survey Findings: Industry Practices and Challenges
The 2025 ISCT survey reveals critical data on how the cell and gene therapy sector currently approaches cryopreservation.
Current Adoption of Freezing Methodologies
Table 1: Adoption Rates of Cryopreservation Methods Based on ISCT Survey Data
| Freezing Method | Overall Adoption Rate | Usage in Early Clinical Stages (Phase I/II) | Primary Use Cases |
|---|---|---|---|
| Controlled-Rate Freezing (CRF) | 87% | Widespread, but not exclusive | Late-stage and commercial products; sensitive cell types (iPSCs, CAR-T) |
| Passive Freezing (PF) | 13% | 86% of PF users | Early-stage clinical products |
The survey indicates a strong industry preference for controlled-rate freezing, particularly for late-stage and commercial products [7]. This preference is likely due to the enhanced process control and documentation capabilities of CRF, which are crucial for regulatory filings. Notably, the majority of respondents using passive freezing have products in early clinical development (up to phase II) [7]. This suggests a potential transition to controlled-rate freezing as products advance toward commercialization, possibly to avoid the significant challenge of establishing product comparability after a major process change.
Major Industry Challenges
Table 2: Key Challenges in Cryopreservation Identified by the ISCT Survey
| Challenge Area | Specific Hurdle | Reported Prevalence |
|---|---|---|
| Process Scaling | Ability to process at large scale | Identified as the biggest hurdle by 22% of respondents [7] |
| System Qualification | Lack of consensus on qualifying Controlled-Rate Freezers | Affects a majority of users; 30% rely solely on vendors [7] |
| Post-Thaw Analytics | Heavy reliance on post-thaw analytics over process data | A large number of respondents do not use freeze curves for release [7] |
| Thawing Process | Non-controlled thawing at clinical sites | Recognized as a significant risk to product quality [7] |
The survey identified scaling as the single biggest hurdle, with 22% of respondents pointing to the "Ability to process at a large scale" as the primary challenge [7]. This is compounded by a lack of consensus on how to qualify controlled-rate freezers. Nearly 30% of respondents rely entirely on vendors for system qualification, which may not represent the final user-specific conditions and container types [7]. Furthermore, the survey noted limited use of freeze curves as part of the product release process, with most facilities relying on post-thaw analytics alone.
Controlled-Rate Freezing vs. Passive Freezing: A Technical Comparison
The choice between controlled-rate freezing (CRF) and passive freezing (PF) is fundamental, with significant implications for process control, cost, and product quality.
Technical Principles and Mechanisms
Controlled-Rate Freezing employs programmable equipment to precisely lower product temperature at a defined rate (typically around -1°C/min) [1]. This allows control over critical process parameters: the cooling rate before nucleation (impacting chilling injury and cryoprotective agent toxicity), the temperature of ice nucleation (affecting osmotic stress and intracellular ice formation), and the cooling rate after nucleation (influencing dehydration and intracellular ice) [7]. This level of control aims to maximize cell viability and consistency by managing the physical stresses of the freezing process.
Passive Freezing typically involves placing product vials in an insulated container (e.g., an isopropanol-filled "Mr. Frosty") that is then placed in a -80°C mechanical freezer [1]. The container is designed to approximate a cooling rate of -1°C/min, but studies show the actual profile is often non-uniform and can vary significantly between vials located in different parts of the container [38]. This method sacrifices direct control for simplicity and lower cost.
Comparative Analysis of Advantages and Limitations
Table 3: Technical and Operational Comparison of Controlled-Rate vs. Passive Freezing
| Aspect | Controlled-Rate Freezing (CRF) | Passive Freezing (PF) |
|---|---|---|
| Process Control | High control over critical parameters (cooling rate, nucleation) | Low control; relies on passive thermal transfer |
| Documentation | Automated, detailed documentation of thermal profile | Limited to no profile documentation |
| Infrastructure Cost | High (equipment, liquid nitrogen consumption) | Low (standard freezer, consumable containers) |
| Operational Complexity | High (requires specialized expertise) | Low (simple, one-step operation) |
| Scalability | Can be a bottleneck for batch scale-up | Easier to scale for large numbers of vials |
| Cell-Type Specificity | Enables optimization for sensitive cells (iPSCs, CAR-T) | Limited adaptability; may not suit sensitive cells |
The ISCT survey found that 60% of users employ the default profiles provided with their CRF equipment [7]. However, those working with more challenging cell types—such as iPSCs, hepatocytes, cardiomyocytes, and certain immune cells—often require and invest in optimized profiles to achieve effective cryopreservation [7].
Emerging Evidence on Clinical Outcomes
Recent research adds nuance to the CRF versus PF debate. A 2025 retrospective study on Hematopoietic Progenitor Cells (HPCs) found that while total nucleated cell (TNC) viability post-thaw was higher with CRF (74.2% ± 9.9% vs. 68.4% ± 9.4%, P = 0.038), there was no significant difference in the more clinically relevant CD34+ cell viability [6] [12]. Most importantly, the study found equivalent engraftment outcomes for both neutrophil and platelet recovery, leading the authors to conclude that "PF is an acceptable alternative to CRF" for HPC cryopreservation [6] [12].
This suggests that for some robust cell types, the theoretical advantages of CRF may not translate into meaningful clinical differences, supporting the use of PF particularly in resource-constrained or high-volume settings.
Detailed Experimental Protocols and Methodologies
Protocol for Controlled-Rate Freezing
The following methodology is adapted from survey findings and standard practices for cryopreserving cell-based therapies [7] [1]:
Cell Harvest and Preparation: Harvest cells during the maximum growth phase (typically >80% confluency). Concentrate or dilute the cell product to achieve an optimal cell concentration, generally within a range of 1x10^3 to 1x10^6 cells/mL, to avoid low viability or excessive clumping [1].
Cryoprotectant Addition: Resuspend the cell pellet in a suitable freezing medium. For GMP manufacturing, this is often a defined, serum-free formulation like CryoStor CS10, containing 10% DMSO and other protective agents [1]. The addition should be performed carefully, potentially in a stepwise manner, to minimize osmotic shock.
Aliquoting and Loading: Aliquot the cell suspension into cryogenic vials. Load the vials into the controlled-rate freezer chamber. For qualification runs, the load should include a range of masses, container configurations, and include temperature probes for mapping.
Program Execution: Initiate the freezing program. A typical program cools at a rate of -1°C/min until the release of the latent heat of fusion is detected. The program may include a rapid cooling segment to counteract this temperature rise, then resume cooling at -1°C/min until reaching a final temperature (e.g., -80°C to -100°C) before transfer to long-term storage [12].
Transfer to Long-Term Storage: Promptly transfer the cryopreserved vials to a liquid nitrogen freezer for storage at ≤ -135°C to ensure long-term stability [1].
Protocol for Passive Freezing
This protocol outlines the passive freezing method as validated for certain cell types like HPCs [12] [1]:
Cell Preparation and Cryoprotectant Addition: Steps 1 and 2 are identical to the CRF protocol, ensuring the cell product is suspended in a freezing medium containing a permeating cryoprotectant like DMSO.
Aliquoting and Container Loading: Aliquot the cell suspension into cryogenic vials. Place the vials into a passive freezing device (e.g., a "Mr. Frosty"-type container saturated with isopropanol at room temperature). Note that vial location (inner vs. outer ring) can impact the cooling profile [38].
Freezing Initiation: Place the closed container immediately into a -80°C mechanical freezer for 18-24 hours. The isopropanol and insulation are intended to approximate a cooling rate of -1°C/min, though actual rates can be variable and non-linear [38].
Transfer to Long-Term Storage: After the freezing period, transfer the vials to a long-term liquid nitrogen storage system (≤ -135°C). The product can remain in the -80°C freezer for a longer period if necessary, but this is not ideal for long-term stability [12].
Thawing Protocol
The ISCT survey emphasizes that the thawing process is equally critical and frequently overlooked [7]. A standardized thawing protocol is vital for product consistency.
Rapid Thawing: Retrieve the vial from long-term storage and immediately thaw it rapidly, typically by gentle agitation in a 37°C water bath or using a validated automated thawing device. The established good practice is a warming rate of approximately 45°C/min [7].
Dilution and Cryoprotectant Removal: Immediately after the ice crystal disappears, wipe the vial with 70% alcohol and carefully transfer the cell suspension to a larger volume of pre-warmed appropriate medium (e.g., wash buffer or culture medium). This step dilutes the cytotoxic DMSO.
Washing and Assessment: Centrifuge the cell suspension to pellet the cells, remove the supernatant containing the cryoprotectant, and resuspend the cell pellet in the final formulation buffer or culture medium for immediate use or further processing.
Viability and Function Assessment: Perform post-thaw cell counts, viability assays (e.g., trypan blue exclusion), and where appropriate, potency or functionality assays.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Reagents and Materials for Cryopreservation Protocols
| Item Category | Specific Examples | Function and Application Notes |
|---|---|---|
| Cryoprotectant Media | CryoStor CS10 (serum-free, defined) | A ready-to-use, GMP-manufactured medium containing 10% DMSO; provides a protective environment during freeze-thaw [1]. |
| Cell-Type Specific Media | mFreSR (for hES/iPS cells), MesenCult-ACF (for MSCs) | Chemically defined, serum-free media optimized for specific stem cell types to maximize recovery and maintain phenotype [1]. |
| Primary Containers | Corning Cryogenic Vials (internal-threaded recommended) | Single-use, sterile vials designed for low-temperature storage; internal threads help prevent contamination [1]. |
| Passive Freezing Devices | Nalgene Mr. Frosty (isopropanol-based), Corning CoolCell (isopropanol-free) | Insulated containers placed in a -80°C freezer to approximate a -1°C/min cooling rate for passive freezing [1]. |
| Controlled-Rate Freezer | Planer Kryo Series and other commercial CRFs | Programmable instrument that dynamically controls chamber temperature to execute precise, documented freezing profiles [7] [38]. |
| Controlled Thawing Devices | ThawSTAR and other automated systems | Provide consistent, controlled-rate thawing, reducing contamination risks associated with water baths and improving reproducibility [7]. |
The 2025 ISCT Cryopreservation Survey reveals an industry at a maturation point, with controlled-rate freezing as the dominant methodology for late-phase and commercial cell therapies due to its superior control and documentation. However, emerging evidence, such as the equivalence of passive freezing for HPC engraftment, demonstrates that the optimal method is cell-type and context dependent. The survey underscores that scaling, standardization, and thawing processes represent significant collective challenges. For researchers and developers, the key is a science-driven approach: investing in CRF for sensitive cells or advanced clinical stages, while considering the cost-effectiveness of PF for robust cells in early development. Ultimately, the success of any cryopreservation strategy hinges on rigorous process understanding, qualification, and a holistic view that treats both freezing and thawing as equally critical to final product quality.
The advancement of cell-based therapies and biomedical research is fundamentally reliant on the precise and reproducible manipulation of living cells. Central to this success is the selection of appropriate protocols tailored to the unique biological and functional characteristics of each cell type. This guide provides a structured framework for protocol selection for Hematopoietic Progenitor Cells (HPCs), induced Pluripotent Stem Cells (iPSCs), and T-Cells, with a particular focus on cryopreservation methodologies. The choice between controlled-rate freezing (CRF) and passive freezing (PF) serves as a critical case study, illustrating how protocol selection directly impacts cell viability, functionality, and therapeutic efficacy. As research progresses, understanding these nuances becomes indispensable for maintaining the integrity of biological samples in applications ranging from basic research to clinical transplantations and immunotherapies.
Cryopreservation Fundamentals: Controlled-Rate vs. Passive Freezing
Cryopreservation is essential for the long-term storage of cells that will be infused more than 48-72 hours after collection. The process requires a cryoprotectant, such as dimethyl sulfoxide (DMSO), to prevent lethal ice crystal formation. The core challenge lies in cooling cells at a rate that minimizes intracellular ice formation and osmotic stress.
Controlled-Rate Freezing (CRF) is widely regarded as the gold standard. This method uses a programmable freezer to decrement the product temperature incrementally according to a preset program. A typical CRF protocol cools the HPC product at a rate of 1°C/min until freezing occurs. The unit then counteracts the release of latent heat of fusion with rapid cooling before resuming a 1°C/min cooling rate until the desired final temperature (typically below -150°C for long-term storage in liquid nitrogen) is reached. CRF provides a precise thermal profile, a racking system, and a fully programmable controller, but it is a complicated, expensive, and time-consuming procedure [12].
Passive Freezing (PF), or non-controlled rate freezing, employs a -80°C mechanical freezer. In this method, HPC products in metal cassettes may be wrapped in disposable absorbent pads or styrofoam insulation to approximate a cooling rate of 1–2°C/min. PF is a simple, convenient, and cost-effective method for cryopreservation. However, nucleation is uncontrolled, cooling rates are not easily or consistently achievable, and temperature monitoring during the freezing process is not performed [12].
Table: Comparison of Controlled-Rate Freezing and Passive Freezing Methods
| Feature | Controlled-Rate Freezing (CRF) | Passive Freezing (PF) |
|---|---|---|
| Core Principle | Programmable, precise cooling rate | Uncontrolled, approximate cooling in a -80°C freezer |
| Cooling Rate | Precisely controlled at ~1°C/min | Variable, aimed at 1–2°C/min via insulation |
| Process Control | High; includes thermal profiling | Low; nucleation and cooling are inconsistent |
| Equipment Cost | High (complicated equipment) | Low (uses standard lab freezer) |
| Operational Complexity | High (requires staff attention) | Low (convenient and simple) |
| Time Consumption | Time-consuming procedure | Less time-consuming for staff |
Cell-Type-Specific Protocol Selection and Experimental Data
Hematopoietic Progenitor Cells (HPCs)
HPCs, collected via apheresis (HPC(A)) or bone marrow (HPC(M)), are critical for transplantation. A recent retrospective study of 50 HPC products directly compared outcomes between CRF (n=25) and PF (n=25) [12] [6]. All products were cryopreserved with a cryoprotectant solution of 15% DMSO and 9% albumin in Plasmalyte-A.
Experimental Findings for HPCs: The study yielded key quantitative outcomes, summarized in the table below. While a statistically significant difference was observed in post-thaw total nucleated cell (TNC) viability, the most critical metrics for transplant success—CD34+ cell viability and engraftment times—showed no significant difference between the two methods [12] [6].
Table: Comparative Post-Thaw Viability and Engraftment of HPCs [12] [6]
| Outcome Measure | Controlled-Rate Freezing (CRF)Mean ± SD | Passive Freezing (PF)Mean ± SD | P-value |
|---|---|---|---|
| TNC Viability (Post-Thaw) | 74.2% ± 9.9% (N=25) | 68.4% ± 9.4% (N=25) | 0.038 |
| CD34+ Cell Viability (Post-Thaw) | 77.1% ± 11.3% (N=13) | 78.5% ± 8.0% (N=25) | 0.664 |
| Days to Neutrophil Engraftment | 12.4 ± 5.0 (N=12) | 15.0 ± 7.7 (N=16) | 0.324 |
| Days to Platelet Engraftment | 21.5 ± 9.1 (N=12) | 22.3 ± 22.8 (N=16) | 0.915 |
Conclusion for HPCs: The study concluded that despite the lower TNC viability with PF, the comparable CD34+ viability and engraftment times demonstrate that PF is an acceptable and comparable alternative to CRF for the initial cryopreservation of HPCs before long-term storage in a liquid nitrogen freezer [6]. This makes PF a viable option, especially in resource-limited settings or during high-throughput scenarios like the COVID-19 pandemic, which strained CRF capacity [12].
Induced Pluripotent Stem Cells (iPSCs)
iPSCs, with their immense potential in disease modeling and regenerative medicine, require stringent protocols to maintain their pluripotent state. While the search results do not provide a direct comparative freezing study for iPSCs like they do for HPCs, they detail comprehensive culture and differentiation workflows.
Key Protocols for iPSCs (feeder-free culture):
- Maintenance: Culture in optimized media such as Essential 8 or StemFlex. Passaging is performed using EDTA or enzyme-free dissociation reagents to maintain cell viability and pluripotency [39].
- Differentiation: Protocols guide the directed differentiation into the three germ layers:
- Ectoderm: Using PSC Neural Induction Medium to generate neural stem cells, which can be further differentiated into dopaminergic neurons or glial cells [39].
- Mesoderm: Involves single-cell dissociation and replating to produce high-purity cardiomyocytes [39].
- Endoderm: Provides a critical starting point for generating liver and pancreatic cell types [39].
- Cryopreservation Consideration: Although not explicitly compared, the sensitivity of iPSCs typically necessitates high-control freezing protocols like CRF to ensure high post-thaw viability and retention of pluripotency.
T-Cells
T-Cells are the workhorses of adaptive immunity and form the basis of powerful immunotherapies like CAR-T and TCR-T cells. Their protocols, therefore, focus on activation, expansion, and antigen-specificity assays.
Activation and Expansion Protocols:
- Traditional Bead-Based Activation: A common method uses synthetic beads coated with anti-CD3 and anti-CD28 antibodies to mimic antigen-presenting cell (APC) stimulation, effectively triggering T-cell activation and proliferation. A limitation of this method is the potential for overstimulation and the challenging physical separation of beads from cells post-expansion [40].
- Cytokine Combinations: The cytokine milieu critically influences the phenotype of expanded T-cells.
- Innovative Microbubble Technology: Akadeum Life Sciences has developed a novel microbubble-based kit that binds target T-cells and gently floats them to the top of the sample for separation. This integrated method for selection, activation, and expansion is gentle on cells, maintains high viability, and avoids overstimulation by allowing daughter cells to naturally detach from the microbubbles [40].
Antigen-Specific T-Cell Expansion Protocol: A high-throughput protocol for expanding and detecting antigen-specific T-cells from human peripheral blood mononuclear cells (PBMCs) involves the following key steps [41]:
- Peptide Preparation: Synthesize and dissolve peptides spanning the target protein sequence in DMSO to create a 2-10 mM stock, aliquoting to avoid freeze-thaw cycles.
- Cell Culture: Isolate PBMCs and culture in complete RPMI media.
- T-Cell Stimulation: Stimulate PBMCs with the prepared peptides. The protocol can be performed using bulk T-cells or co-cultures of sorted T-cell subsets with autologous or MHC-matched Antigen Presenting Cells (APCs).
- Enhance Stimulation: Add co-stimulatory antibodies (e.g., anti-CD28 and anti-CD49d) to the cultures.
- Cytokine Detection: After an appropriate expansion period, re-stimulate cells and detect antigen-specific effector cytokine formation (e.g., IFN-γ, TNF-α, IL-2) by intracellular cytokine staining and flow cytometry.
The Scientist's Toolkit: Essential Research Reagents
Successful execution of cell-based protocols relies on a foundation of high-quality reagents. The table below lists key materials used in the featured experiments and their critical functions.
Table: Research Reagent Solutions for Cell Culture and Assays [40] [39] [41]
| Reagent / Material | Cell Type | Function and Application |
|---|---|---|
| Anti-CD3 / Anti-CD28 Beads | T-Cells | Synthetic APC analogs for robust T-cell activation and expansion. |
| Essential 8 / StemFlex Media | iPSCs | Optimized, defined media for feeder-free maintenance of pluripotent stem cells. |
| Cryoprotectant (DMSO/Albumin) | HPCs, others | Prevents intracellular ice formation during cryopreservation (e.g., 15% DMSO, 9% albumin). |
| Recombinant Cytokines (IL-2, IL-7, IL-15) | T-Cells | Drives specific T-cell proliferation and differentiation into effector or memory phenotypes. |
| PSC Neural Induction Medium | iPSCs | Streamlined medium for directed differentiation of iPSCs into neural stem cells (Ectoderm). |
| Flow Cytometry Antibodies (e.g., anti-CD3, CD8, IFN-γ) | T-Cells | Enables multi-parameter detection of cell surface markers and intracellular cytokines for immunophenotyping. |
| Lipofectamine Stem Transfection Reagent | iPSCs | Efficient delivery of DNA or RNA into PSCs for genome engineering and reprogramming. |
| Peptide Pools | T-Cells | Used to stimulate antigen-specific T-cells for functional immune response assays. |
Protocol selection is not a one-size-fits-all endeavor but a critical strategic decision. The comparative analysis of CRF and PF for HPCs demonstrates that for some robust cell types, simpler and more cost-effective methods can be equivalent to the gold standard for key clinical outcomes like engraftment. However, this may not hold true for more sensitive cells like iPSCs.
When selecting a protocol, researchers must consider:
- Desired Cell Characteristics: The protocol directly influences the final product (e.g., effector vs. memory T-cells).
- Scale and Resources: PF and microbubble technologies offer scalable and resource-conscious alternatives to CRF and traditional bead-based activation.
- Downstream Application: Clinical cell therapy manufacturing demands rigorous, validated protocols (often GMP-grade), whereas basic research may prioritize flexibility and discovery.
This guide underscores that an informed choice, based on empirical evidence and a clear understanding of cell biology, is fundamental to success in the laboratory and the clinic.
For researchers, scientists, and drug development professionals, navigating the landscape of Current Good Manufacturing Practice (cGMP) is fundamental to transforming research into compliant, market-ready therapies. The U.S. Food and Drug Administration (FDA) defines cGMP as containing the minimum requirements for the methods, facilities, and controls used in manufacturing, processing, and packing a drug product [42]. These regulations ensure that a product is safe for use and that it possesses the identity, strength, quality, and purity it claims to have [42]. A robust cGMP framework is built upon two pillars: meticulous documentation and rigorous process validation.
Within this framework, critical manufacturing decisions—such as the choice between controlled-rate freezing and passive freezing in cell and gene therapy products—must be justified through scientifically sound, well-documented validation studies that demonstrate control over the process and its impact on Critical Quality Attributes (CQAs). This guide provides a technical roadmap for integrating these cGMP principles into your research and development workflow.
The Pillars of cGMP: Documentation and Validation
Good Documentation Practices (GDP)
Good Documentation Practices (GDP) form the backbone of any quality system. In 2025, GDP ensures that all data generated during development and manufacturing is reliable, attributable, and accurate [43].
Core Principles of GDP [43]:
- Legibility and Clarity: All records must be easy to read and unambiguous.
- Accuracy: Documentation must precisely reflect the activities performed and results observed.
- Completeness: All necessary information must be recorded, with no blank sections.
- Timeliness: Activities must be documented at the time of performance, not retrospectively.
- Consistency: Uniform terminology and formats should be used across all records.
- Attributability: Every entry must be traceable to the individual who made it.
The regulatory landscape is increasingly digital. For 2025, key trends include the widespread adoption of Electronic Document Management Systems (EDMS), the integration of AI and automation for error checking, and a heightened focus on data integrity to prevent breaches or falsification [43].
Process Validation Framework
The FDA defines process validation as the "collection and evaluation of data, from the process design stage through commercial production, which establishes scientific evidence that a process is capable of consistently delivering a quality product" [44]. This is not a one-time event but a lifecycle approach.
The Three Stages of Process Validation [45] [44]:
- Process Design: The commercial manufacturing process is defined based on knowledge gained through development and scale-up activities.
- Process Qualification: The process design is confirmed to be capable of reproducible commercial manufacturing. This includes both equipment qualification (IQ/OQ) and Process Performance Qualification (PPQ).
- Continued Process Verification: Ongoing assurance is gained during routine production that the process remains in a state of control.
For a freezing process, this means first designing the cycle parameters, then qualifying the equipment and process, and finally continuously monitoring the performance of every batch.
cGMP Application in Freezing Process Validation
Critical Quality Attributes (CQAs) and Critical Process Parameters (CPPs)
The validation of any process, including freezing, begins with identifying CQAs and CPPs. CQAs are physical, chemical, biological, or microbiological properties that must be controlled within appropriate limits to ensure the product meets its quality standards [45]. For hematopoietic progenitor cells (HPCs), a key CQA is cell viability post-thaw, as it directly impacts engraftment efficacy [6] [12].
CPPs are process parameters whose variability has a direct impact on a CQA. For freezing processes, key CPPs include the freezing rate and the final temperature. Controlling these parameters is essential to prevent intracellular ice crystal formation, which can reduce viable cell recovery [12].
Table 1: Typical CQAs and CPPs in Freezing Process Validation
| Category | Parameter | Description & Impact |
|---|---|---|
| CQAs | Total Nucleated Cell (TNC) Viability | A measure of overall cell health post-thaw; impacts product potency [6]. |
| CD34+ Cell Viability | Viability of specific hematopoietic progenitor cells; a direct indicator of therapeutic potential [6] [12]. | |
| Residual Moisture | Critical for lyophilized products; high levels can compromise stability [46]. | |
| Engraftment Potential | The ultimate functional CQA, measured by days to neutrophil and platelet engraftment in patients [12]. | |
| CPPs | Freezing Rate | A slow, controlled rate (~1°C/min) is often critical to prevent cell damage from ice crystals [12]. |
| Final Temperature | Temperature for long-term storage (e.g., < -150°C in liquid nitrogen) must be validated to ensure stability [6]. | |
| Cryoprotectant Concentration | Concentration of agents like DMSO must be optimized and consistent to protect cells during freeze-thaw [12]. |
Equipment Qualification (IQ/OQ/PQ)
All equipment used in cGMP processes, including controlled-rate freezers and mechanical freezers, must undergo a rigorous qualification process.
- Installation Qualification (IQ): Verifies that the equipment is delivered, installed, and configured correctly according to the manufacturer's specifications and user requirements [44]. This includes documentation of utilities and environmental conditions.
- Operational Qualification (OQ): Demonstrates that the equipment operates as intended across all anticipated operating ranges. For a controlled-rate freezer, OQ would include testing shelf temperature uniformity (e.g., ± 0.5°C), cooling and heating rate accuracy, and verification of the vacuum system if applicable [45] [44].
- Performance Qualification (PQ): While sometimes part of equipment qualification, PQ is often integrated with the Process Performance Qualification (PPQ), where the equipment, process, and personnel are tested together to show the entire system can consistently produce a product meeting its pre-determined quality attributes [44].
Table 2: Key IQ/OQ Tests for Lyophilization and Freezing Equipment [45]
| Qualification Test | Description and Target Specification |
|---|---|
| Shelf Temperature Mapping | The shelf surface temperature at any spot is within ± 0.5 °C of the average after equilibration. |
| Vacuum System Test | A target pressure (e.g., 50 mTorr) can be achieved within a specified time and maintained within ± 5 units of the setpoint. |
| Leak Rate Test | The chamber leak rate meets sterility specification (e.g., 2 × 10⁻² mbar·L/s). |
| Condenser Capacity Test | The condenser can trap a weighed amount of water vapor exceeding its stated capacity. |
| Stoppering Test | All vials are stoppered correctly at the target pressure range with no breakage. |
| Cleaning & Sterilization | Cycles are validated to prevent cross-contamination and maintain sterility. |
Process Performance Qualification (PPQ) for Freezing
The PPQ is the culminating validation exercise that combines the qualified equipment, trained personnel, and approved procedures to demonstrate the commercial manufacturing process is reproducible. For a freezing process, a PPQ protocol must include a defined strategy for:
- Batch Size Determination: The batch size must be representative of commercial scale, and its effect on the process (e.g., drying time in lyophilization) must be understood [45].
- Sampling Plans: A statistically sound sampling plan is required to demonstrate homogeneity within the batch and reproducibility between batches. This often involves sampling from multiple locations (e.g., top, middle, bottom shelves) and multiple batches (typically a minimum of three consecutive successful batches) [45].
- Acceptance Criteria: Pre-defined criteria for all relevant CQAs must be established. For example, a PPQ for a cell therapy product may set acceptance criteria for post-thaw viability and cell count.
Case Study: Validation of Controlled-Rate vs. Passive Freezing
A 2025 retrospective study directly compared Controlled-Rate Freezing (CRF) and Passive Freezing (PF) for Hematopoietic Progenitor Cells (HPCs), providing a model for designing a cGMP-compliant validation study [6] [12].
Experimental Protocol and Methodology
Objective: To compare the effects of CRF and PF on Total Nucleated Cell (TNC) viability, CD34+ cell viability, and engraftment outcomes [12].
Materials and Reagents:
- HPC Products: 50 donor products (apheresis-derived and marrow-derived) [12].
- Cryoprotectant Solution: 15% Dimethyl Sulfoxide (DMSO) + 9% albumin in Plasmalyte-A [12]. DMSO prevents intracellular ice crystal formation.
- Equipment: Controlled-rate freezer (CRF) and -80°C mechanical freezer for PF [12].
Procedure:
- Preparation: HPC products were concentrated to an optimal cell concentration of 600–800 × 10⁶ TNC/mL [12].
- Mixing: The product was mixed with the cryoprotectant solution in a 1:1 ratio [12].
- Freezing:
- Storage: All products were transferred to long-term storage in a liquid nitrogen freezer (< -150°C) [6].
- Testing: Post-thaw, products were analyzed for TNC viability and CD34+ cell viability. Engraftment was tracked in patients via days to neutrophil and platelet recovery [6] [12].
Diagram: Experimental Workflow for Freezing Method Comparison. The process begins with cell collection and preparation, branches into the two freezing methodologies, and converges for storage and analysis of Critical Quality Attributes (CQAs).
Data Presentation and Comparative Analysis
The study generated quantitative data that was statistically analyzed to support its conclusions. Presenting such data clearly is crucial for regulatory submissions.
Table 3: Comparative Post-Thaw Viability and Engraftment Outcomes [6] [12]
| Parameter | Controlled-Rate Freezing (CRF) | Passive Freezing (PF) | P-value | Conclusion |
|---|---|---|---|---|
| TNC Viability (Mean ± SD) | 74.2% ± 9.9% (N=25) | 68.4% ± 9.4% (N=25) | 0.038 | Statistically significant difference, but both methods yield acceptable viability. |
| CD34+ Viability (Mean ± SD) | 77.1% ± 11.3% (N=13) | 78.5% ± 8.0% (N=25) | 0.664 | No significant difference. |
| Days to Neutrophil Engraftment (Mean ± SD) | 12.4 ± 5.0 (N=12) | 15.0 ± 7.7 (N=16) | 0.324 | No significant difference in clinical outcome. |
| Days to Platelet Engraftment (Mean ± SD) | 21.5 ± 9.1 (N=12) | 22.3 ± 22.8 (N=16) | 0.915 | No significant difference in clinical outcome. |
The Scientist's Toolkit: Essential Reagents and Materials
Table 4: Key Research Reagent Solutions for Freezing Process Validation
| Item | Function in Experiment |
|---|---|
| Dimethyl Sulfoxide (DMSO) | A cryoprotectant that penetrates cells, preventing the formation of damaging intracellular ice crystals during the freezing process [12]. |
| Human Serum Albumin | Used in the cryoprotectant solution (e.g., at 9%) to provide a stable protein environment, protecting cell membranes from freeze-thaw stress [12]. |
| Plasmalyte-A | A balanced electrolyte solution used as the base for the cryoprotectant solution, maintaining physiological pH and osmolarity during the freezing process [12]. |
| Viability Assay Kits (e.g., 7-AAD) | Essential for measuring post-thaw cell viability, a critical quality attribute (CQA). These assays distinguish between live and dead cells [12]. |
| CD34+ Antibody Panels | Used in flow cytometry to identify and count specific hematopoietic progenitor cells, allowing for the measurement of CD34+ cell viability and recovery [12]. |
Regulatory Submission and Maintaining Compliance
Integrating Validation Data into Submissions
When filing a marketing application (e.g., under 21 CFR Part 314), the FDA reviews the manufacturer's compliance with cGMP, which includes a assessment of their validation data [42]. The submission should clearly demonstrate the link between CPPs and CQAs, and provide evidence from PPQ batches that the process is robust and reproducible. The case study data shown in Table 3 is an example of the evidence required to justify a manufacturing process.
A Quality Systems Approach
The FDA encourages a Quality Systems Approach to cGMP. This means that quality should be built into the product, and not just tested in at the end. A proactive approach, utilizing risk management and quality principles at every stage of development and manufacturing, is viewed favorably by regulators [47]. For freezing processes, this means understanding not just if a process works, but how and why it works, and what variables must be controlled to ensure it consistently produces a quality product.
In the realm of advanced therapies, where processes like controlled-rate and passive freezing are critical, robust validation and impeccable documentation are non-negotiable. The retrospective study on HPC freezing demonstrates that while CRF and PF are both acceptable, the choice must be justified by a validation study that rigorously examines its impact on CQAs from cell viability to clinical engraftment. By adopting a science-based, data-driven approach to cGMP—where every process parameter is understood, controlled, and documented—drug development professionals can successfully navigate the regulatory pathway and ensure that their innovative therapies are both efficacious and compliant.
Solving Cryopreservation Challenges: From Viability Loss to Scaling Hurdles
The cryopreservation of cellular products represents a critical bottleneck in biomedical research, drug development, and cell-based therapies. When post-thaw viability falls below expectations, researchers face a complex diagnostic challenge: determining whether the freezing process or thawing process is primarily responsible for cell death. This distinction is not merely academic; it directly influences how protocols are optimized and resources allocated. Within the broader research context comparing controlled-rate freezing (CRF) to passive freezing (PF), understanding the origin of cryoinjury becomes paramount for developing robust, reproducible preservation protocols. Current industry surveys indicate that cryopreservation processes and post-thaw analytics represent the area where the most challenges are encountered and resources are dedicated [7]. This technical guide provides a structured approach to diagnosing the root causes of post-thaw viability loss, offering detailed methodologies for differentiating between freezing-associated and thawing-associated damage.
The fundamental challenge in diagnosing post-thaw failure lies in the interconnected nature of cryopreservation damage. Freezing-induced stresses may only manifest fully during or after thawing, creating ambiguity in attribution. As illustrated below, the diagnostic pathway requires systematic investigation of multiple parameters and their relationships.
Fundamental Mechanisms of Cryoinjury
Freezing-Associated Damage Pathways
During the freezing phase, cells encounter multiple potentially lethal stressors. Intracellular ice formation (IIF) represents one of the most destructive events, occurring when cooling rates are too rapid to permit sufficient cellular dehydration [48]. IIF physically disrupts intracellular organelles and membrane structures, typically resulting in immediate necrotic cell death. The solution effects damage pathway encompasses injury resulting from prolonged exposure to hypertonic solutions as water freezes out, concentrating extracellular solutes to toxic levels and causing severe osmotic shrinkage [49] [48]. This effect is particularly pronounced at slow cooling rates where cells experience prolonged exposure to concentrated solutes.
Cryoprotectant toxicity constitutes another significant freezing-associated damage mechanism. While dimethyl sulfoxide (DMSO) remains the most widely used penetrating cryoprotectant, it exhibits concentration- and temperature-dependent toxicity [50] [51]. DMSO has been shown to alter cytoskeletal organization, shift cellular metabolism, and modify membrane fluidity even before freezing occurs [36]. At temperatures above freezing, DMSO can induce epigenetic changes, DNA methylation, and histone modifications that compromise cellular function post-thaw [52] [50]. Chilling injury refers to damage that occurs at temperatures above the freezing point but below physiological normothermia, affecting membrane lipid phases and protein function in temperature-sensitive cell types [48].
Thawing-Associated Damage Pathways
The thawing process introduces its own distinct damage mechanisms. Recrystallization occurs during slow warming when small, stable ice crystals merge into larger, more destructive structures [51] [48]. This phenomenon is particularly damaging when the warming rate is insufficient to rapidly traverse the critical temperature zone (-50°C to -20°C) where ice crystal growth is most active. Osmotic shock during thawing manifests when cells are rapidly exposed to hypotonic conditions as extracellular ice melts, causing uncontrolled water influx and potential membrane rupture [36]. This damage pathway is especially significant when cryoprotectant removal is not properly managed.
Devitrification represents a specific thawing injury in which the amorphous glassy state formed during successful vitrification reverts to crystalline ice during overly slow warming [50] [48]. Cryoprotectant dilution stress occurs when the rapid extracellular reduction of cryoprotectant concentration creates osmotic gradients that drive water into cells faster than cryoprotectant can exit, potentially causing lethal cell swelling [36]. The period of post-thaw DMSO exposure before washing or administration also contributes to toxicity, as cells remain in contact with damaging concentrations of cryoprotectant at physiological temperatures where toxicity is magnified [36].
Diagnostic Methodologies
Differential Assessment Protocols
Differentiating between freezing and thawing damage requires strategic assessment at multiple timepoints using complementary assays. The following experimental workflow provides a systematic approach to isolate the primary source of viability loss.
Immediate post-thaw viability assessment provides the first diagnostic data point. Membrane integrity assays using trypan blue, propidium iodide, or 7-AAD staining performed within minutes post-thaw primarily reflect ice crystal damage during freezing and thawing [52] [36]. When immediate viability is low, particularly with evidence of physical membrane rupture, ice formation is the likely culprit.
Morphological analysis of freshly thawed cells can distinguish between freezing and thawing damage patterns. Intracellular ice formation typically causes obvious physical disruption of cellular architecture, with fragmented membranes and disordered internal structures [48]. In contrast, osmotic shock during thawing often produces membrane blebbing and cell swelling without the extreme fragmentation characteristic of intracellular ice [36].
Osmotic stress response testing involves challenging post-thaw cells with controlled osmotic stress. Cells that survived freezing with minimal damage but are compromised by thawing processes will exhibit particular sensitivity to hypotonic conditions [36]. This assay specifically probes membrane and volume regulation integrity affected by thawing processes.
Delayed apoptosis assessment is crucial for detecting freezing-associated damage that manifests hours after thawing. Using Annexin V staining or caspase-3/7 activation assays at 4-24 hours post-thaw reveals apoptotic pathways initiated during freezing [52] [53]. Research demonstrates that measuring viability immediately post-thaw can yield false positives, as apoptosis takes time to manifest [52].
Post-thaw recovery culture over 24-72 hours provides the most clinically relevant assessment of functional survival. Cells that attach, spread, and resume proliferation have survived both freezing and thawing processes with minimal cumulative damage [52] [53]. Persistent metabolic impairment or failure to reattach after 24 hours typically indicates freezing-associated damage to intracellular systems [53].
Quantitative Analysis of Post-Thaw Outcomes
The table below summarizes key parameters for differentiating freezing from thawing damage based on experimental assessments.
Table 1: Diagnostic Parameters for Differentiating Freezing vs. Thawing Damage
| Assessment Method | Freezing Damage Indicators | Thawing Damage Indicators |
|---|---|---|
| Immediate Viability | Low viability with membrane fragmentation | Low viability with cell swelling |
| Morphology | Evidence of intracellular ice crystals | Membrane blebbing, osmotic shock signs |
| 24h Apoptosis | High caspase activation (>40% increase) | Minimal delayed apoptosis |
| Metabolic Activity | Persistent reduction at 24h (>50% decrease) | Partial recovery by 24h |
| Adhesion/Spreading | Failure to reattach (<30% of control) | Initial adhesion with subsequent detachment |
| Colony Formation | Significant reduction (<50% of control) | Moderate reduction |
Freezing Method Comparison: CRF vs. Passive Freezing
Technical Foundations and Performance Data
The choice between controlled-rate freezing (CRF) and passive freezing (PF) significantly influences the nature and extent of cryoinjury. CRF employs specialized equipment to precisely control cooling rates, typically following an optimized profile such as -1°C/min through the critical phase change temperature zone [12] [7]. This controlled cooling allows sufficient time for cellular dehydration while minimizing intracellular ice formation. In contrast, PF relies on placing samples in an insulated container within a -80°C mechanical freezer, resulting in variable, uncontrolled cooling rates that may differ between samples and freezer locations [12] [6].
Recent comparative studies provide quantitative performance data between these approaches. The table below summarizes key findings from direct comparisons of CRF and PF methods.
Table 2: Quantitative Comparison of Controlled-Rate Freezing vs. Passive Freezing Outcomes
| Performance Metric | Controlled-Rate Freezing | Passive Freezing | Study References |
|---|---|---|---|
| TNC Viability Post-Thaw | 74.2% ± 9.9% | 68.4% ± 9.4% | [12] [6] |
| CD34+ Cell Viability | 77.1% ± 11.3% | 78.5% ± 8.0% | [12] [6] |
| Neutrophil Engraftment Days | 12.4 ± 5.0 | 15.0 ± 7.7 | [12] [6] |
| Platelet Engraftment Days | 21.5 ± 9.1 | 22.3 ± 22.8 | [12] [6] |
| Process Control | High precision cooling rate | Variable cooling rates | [7] |
| Equipment Cost | High ($15,000-$50,000) | Low (<$500) | [7] |
| Technical Expertise Required | Specialized training needed | Minimal training | [7] |
Diagnostic Indicators for Method-Specific Failure
When diagnosing post-thaw failure in CRF protocols, evidence of incorrect cooling rate selection typically manifests as either intracellular ice formation (too rapid cooling) or excessive solute effects (too slow cooling) [49] [48]. Uncontrolled nucleation can create heterogeneous ice formation between samples, leading to variable post-thaw outcomes despite identical protocols [51].
For PF systems, cooling rate inconsistency represents the most common failure mode, with samples in different locations within the freezing device experiencing different thermal histories [12]. Insufficient cooling rate control in PF often produces mixed populations of cells experiencing both freezing damage (from overly rapid cooling) and solute effects (from overly slow cooling) simultaneously [49].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Cryopreservation Research
| Reagent/Material | Function/Application | Examples/Notes |
|---|---|---|
| DMSO | Penetrating cryoprotectant | Most common CPA; concentration-dependent toxicity [50] [51] |
| Glycerol | Penetrating cryoprotectant | Lower toxicity than DMSO; used for sensitive cell types [50] |
| CELLBANKER Series | Commercial cryopreservation media | Serum-free, defined formulations available [50] |
| Polyampholytes | Macromolecular cryoprotectant | Emerging class; membrane stabilization [52] |
| Antifreeze Proteins | Ice recrystallization inhibition | Bio-inspired cryoprotectants [51] |
| Annexin V Assays | Apoptosis detection | Identifies delayed apoptosis from freezing damage [52] |
| Caspase 3/7 Detection | Apoptosis pathway activation | Measures execution-phase apoptosis [52] |
| 7-AAD/Propidium Iodide | Membrane integrity assessment | Immediate post-thaw viability [12] |
| CoolCell/Cell Freezing Containers | Passive freezing devices | Provide approximately -1°C/min cooling [53] |
| Controlled-Rate Freezers | Programmable freezing equipment | Precise cooling rate control [7] |
Optimization Strategies and Protocol Recommendations
Evidence-Based Protocol Improvements
Based on diagnostic outcomes, specific optimization strategies can address identified failure modes. For freezing damage dominance, implement cooling rate optimization through systematic testing of rates between 0.5°C/min to 2.0°C/min for sensitive cell types [49]. Consider CPA cocktail development using combinations of penetrating and non-penetrating cryoprotectants at reduced concentrations to minimize toxicity while maintaining protection [52] [51]. Controlled nucleation techniques can be employed at approximately -5°C to ensure consistent ice formation between samples [51].
For thawing damage dominance, focus on warming rate optimization using validated methods achieving >50°C/min to avoid recrystallization [36] [7]. Implement optimized CPA removal protocols through stepwise dilution or use of extracellular non-penetrating agents to minimize osmotic shock [36]. Consider direct infusion without washing for certain therapeutic applications where DMSO concentration is sufficiently low [36].
Protocol Template for Differential Diagnosis
Controlled-Rate Freezing Protocol for Diagnostic Testing:
- Prepare cells in optimized cryoprotectant solution (e.g., 5-10% DMSO with extracellular cryoprotectant)
- Use controlled-rate freezer with programmed profile:
Thawing Protocol Variations for Diagnostic Testing:
- Rapid thaw method: 37°C water bath with gentle agitation until last ice crystal disappears
- Assess immediate viability and membrane integrity
- Perform parallel thawing with stepwise CPA dilution vs. direct culture
- Compare osmotic fragility between immediately diluted and gradually diluted samples [36]
Diagnosing the primary source of post-thaw viability loss requires systematic investigation of both freezing and thawing processes. Through the application of differential assessment protocols, researchers can distinguish between damage mechanisms and implement targeted optimization strategies. The growing evidence comparing controlled-rate and passive freezing methods indicates that both approaches can yield clinically acceptable results when properly optimized and controlled [12] [6]. Future advances in cryopreservation research will likely focus on the development of less toxic cryoprotectant cocktails, improved real-time monitoring of ice formation, and standardized quality assessment metrics that better predict long-term functional recovery [51] [48]. As cell and gene therapies continue to advance, robust diagnostic approaches for cryopreservation failure will remain essential for manufacturing consistency and therapeutic efficacy.
In the context of ongoing research into controlled-rate freezing (CRF) versus passive freezing (PF), the scientific community has rightly focused on optimizing cooling protocols to enhance cell viability and product stability. However, the thawing process represents an equally critical, yet often underestimated, phase where significant cellular and macromolecular damage can occur. While recent investigations, such as those demonstrating the clinical equivalence of PF and CRF for hematopoietic progenitor cell (HPC) engraftment, have refined our understanding of freezing methodologies [ [6] [12]], the thawing imperative demands parallel attention. This technical guide examines the scientific rationale for controlled warming, provides detailed experimental methodologies for its evaluation, and establishes why a holistic approach—encompassing both freezing and thawing—is fundamental to advancing biopreservation sciences in therapeutic development.
The Science of Thawing: Mechanisms of Damage During Warming
The process of thawing subjects biological materials to multiple, simultaneous stressors. Understanding these mechanisms is the first step in developing effective controlled warming protocols.
Ice Recrystallization and Shear Stress
During thawing, especially at slow rates, small ice crystals undergo recrystallization—a process where larger ice crystals grow at the expense of smaller ones. This phenomenon is driven by the tendency of the system to reduce its surface energy. The resulting larger crystals can cause mechanical damage to cellular structures and plasma membranes [ [9]]. Furthermore, the phase change from solid to liquid generates significant shear forces within the solution, which can damage sensitive structures like proteins and cell membranes [ [9]].
Protein Denaturation at Interfaces
As ice melts, new air-liquid interfaces form rapidly. Proteins, particularly therapeutic monoclonal antibodies (mAbs), can adsorb to these interfaces, leading to partial unfolding, aggregation, and loss of biological activity [ [9] [54]]. One control strategy involves using formulation excipients that act as stabilizers (e.g., surfactants and sugar cryoprotectants) to reduce the level of protein denaturation induced upon exposure to these interfaces [ [9]].
Solution Effects and Osmotic Imbalance
During freezing, solutes become concentrated in the unfrozen fraction, creating hypertonic conditions. If thawing occurs too rapidly, cells may experience dramatic osmotic shifts as they transition from this hypertonic environment back to isotonic conditions, potentially causing volumetric stress and membrane rupture [ [9] [4]].
The diagram below illustrates the primary damage mechanisms that occur during the thawing process:
Experimental Evidence: Quantifying the Impact of Thawing Methodologies
Comparative Studies on Thawing Rates
Systematic investigations have demonstrated that thawing rates significantly influence the recovery of viable cells and stability of biopharmaceuticals. Research has categorized thawing rates as slow (1–5°C/min), intermediate (>5°C/min), or rapid (achieved through water baths) [ [9]]. The optimal rate depends on the biological material and its intended application.
A retrospective study of 50 hematopoietic progenitor cell (HPC) products found that while freezing method (CRF vs. PF) affected total nucleated cell (TNC) viability post-thaw, it did not significantly impact CD34+ cell viability or engraftment outcomes, suggesting that post-thaw handling and warming protocols may play a compensatory role [ [6] [12]]. This underscores the importance of standardizing both freezing and thawing processes.
Case Study: Systematic Approach to Freeze-Thaw Characterization
A comprehensive study on monoclonal antibody (mAb-1) stability provides a framework for evaluating thawing parameters [ [54]]. The researchers implemented two distinct protocols to understand the interaction between freezing and thawing rates:
- Protocol A (Slow Freeze-Fast Thaw): Freeze from 5 to -50°C at 0.03°C/min, then thaw at 1°C/min to 5°C.
- Protocol B (Fast Freeze-Slow Thaw): Freeze to -50°C at 1°C/min, then thaw at 0.03°C/min to -25°C, hold for 24 hours, then ramp to 5°C at 0.03°C/min.
The study found that fast thawing was generally preferred for protein stability as it minimized the time for destructive processes like ice recrystallization [ [54]]. This approach also proved beneficial for HPC recovery, where rapid thawing in a 37°C water bath is standard practice to minimize exposure to cytotoxic cryoprotectant agents like DMSO [ [12]].
Table 1: Impact of Thawing Rates on Biological Materials
| Material Type | Slow Thawing (1-5°C/min) | Fast Thawing (>5°C/min) | Key Metrics Affected |
|---|---|---|---|
| Therapeutic Proteins/mAbs | Increased aggregation due to extended exposure to interfaces [ [9]] | Reduced aggregation; maintained activity [ [54]] | SE-HPLC aggregation levels; biological activity |
| Hematopoietic Progenitor Cells | Extended DMSO exposure; potential osmotic shock [ [12]] | Standard practice (37°C water bath); better viability [ [12]] | CD34+ viability; neutrophil engraftment days |
| Ovarian Tissue | Ice recrystallization; structural damage [ [55]] | Optimized protocol with precise temperature steps [ [55]] | Follicular survival; tissue integrity |
Optimization of Thawing Protocols for Complex Tissues
Research on ovarian tissue cryopreservation demonstrates the sophisticated approach required for complex tissues. An optimized thawing protocol consisted of "a 3.5-minute step in a cold chamber to reach slowly Tg' (glass transition temperature), limiting thermal and mechanical shocks, and then a 2-minute incubation at 37°C to quickly reach Tm (melting temperature)" [ [55]]. This two-stage approach—slow warming through the critical glass transition phase followed by rapid melting—preserved tissue architecture and function comparable to fresh tissue [ [55]].
Table 2: Comparative Analysis of Thawing Methods in Research Studies
| Study Reference | Biological Material | Thawing Methods Compared | Key Findings |
|---|---|---|---|
| Bioprocess International (2023) [ [9]] | mAb Drug Substance | Passive (room temp/2-8°C) vs. Controlled (programmable baths) | Controlled thawing reduced aggregation; passive thawing resulted in protein aggregation |
| Scientific Reports (2021) [ [54]] | mAb-1 Fusion Enzyme | Slow thaw (0.03°C/min) vs. Fast thaw (water bath) | Fast thawing significantly reduced freeze-thaw damage and aggregation |
| Cytotherapy (2025) [ [6] [12]] | Hematopoietic Progenitor Cells | 37°C water bath (standardized across freezing methods) | No significant difference in engraftment between CRF and PF when using standardized rapid thaw |
| Fertility Preservation (2025) [ [55]] | Ovarian Tissue | Two-stage protocol (slow to Tg', then fast to Tm) | Tissue quality similar to fresh; resumed folliculogenesis after culture |
Methodologies: Experimental Design for Thawing Optimization
Small-Scale Study Design for Freeze-Thaw Characterization
Early in development, material availability is often limited, creating a need for reliable small-scale models that mimic large-scale processes [ [9] [54]]. A systematic approach to designing such studies includes:
- Selection of Representative Formulation and Container-Closure System: Product stability depends on formulation compatibility with contact materials during thawing [ [9]].
- Appropriate Scale-Down: Choose fill volumes with representative head space or surface-area-to-volume ratios similar to large scale [ [9]].
- Evaluation of Different Thawing Rates: Test both active-control and passive conditions to determine critical process parameters [ [9]].
- Stability Assessment: Evaluate protein attributes or cell viability under different storage conditions after thawing [ [9]].
The experimental workflow below outlines a systematic approach to optimize freeze-thaw protocols:
Analytical Techniques for Assessing Thawing Outcomes
Comprehensive analysis of post-thaw recovery requires multiple orthogonal techniques:
- Cellular Viability Assessments: Flow cytometry with 7-AAD staining, CD34+ cell counts for HPCs [ [12]], and colony-forming unit (CFU) assays to measure functional capacity [ [12] [5]].
- Protein Stability Measurements: Size exclusion HPLC (SE-HPLC) for aggregate quantification [ [54]], analytical ultracentrifugation (AUC) [ [54]], and biological activity assays.
- Low-Temperature Thermal Analysis: Differential scanning calorimetry (DSC) to characterize thermal transitions, electrical resistance measurements, and freeze-drying microscopy to observe sample behavior during warming [ [54]].
The Scientist's Toolkit: Essential Materials for Controlled Thawing Research
Table 3: Key Research Reagent Solutions for Thawing Optimization
| Item/Category | Function in Thawing Research | Application Examples |
|---|---|---|
| Programmable Water Baths | Provide precise temperature control during thawing; enable reproducible warming rates [ [54]] | mAb thawing studies; standardized cell thawing protocols |
| Controlled-Rate Freeze-Thaw Systems | Active control of both freezing and thawing rates with documentation capabilities [ [9]] | Systematic studies of freeze-thaw interaction effects |
| Cryoprotectant Agents (DMSO, Sucrose) | Reduce ice crystal formation and stabilize proteins/cells during phase transitions [ [55] [12]] | HPC cryopreservation; ovarian tissue preservation |
| Surfactants (Polysorbate 80) | Minimize protein aggregation at air-liquid interfaces during thawing [ [9] [54]] | mAb formulations prone to surface-induced aggregation |
| Size Exclusion HPLC | Quantify protein aggregation levels post-thaw [ [54]] | Assessment of mAb stability under different thawing conditions |
| Viability Stains (7-AAD) | Distinguish live/dead cells for recovery calculations [ [12]] | HPC and cell therapy product quality assessment |
The evidence unequivocally demonstrates that controlled warming constitutes a critical component of the cryopreservation workflow, equally as important as controlled freezing for maintaining the viability and function of biological materials. As research continues to refine our understanding of freezing methodologies—including the appropriate applications of passive freezing—parallel attention must be directed toward standardizing and optimizing thawing protocols.
The successful translation of cell and gene therapies, biopharmaceuticals, and regenerative medicine products depends on a holistic approach to biopreservation that acknowledges both the freezing and thawing processes as interconnected determinants of product quality and clinical efficacy. By implementing systematic thawing characterization, employing appropriate analytical techniques, and adopting controlled warming technologies, researchers and drug development professionals can significantly enhance the recovery, consistency, and therapeutic potential of their cryopreserved products.
Controlled-rate freezing (CRF) has established itself as a cornerstone technology in the cryopreservation of biological materials, particularly for cell therapies and biobanking. While passive freezing using devices like "Mr. Frosty" in -80°C freezers has gained acceptance for its simplicity and cost-effectiveness [6], the precise control offered by CRF systems remains crucial for maintaining the viability and functionality of sensitive cell types. The fundamental thesis governing cryopreservation research posits that while passive freezing methods can provide adequate results for some applications, optimized CRF protocols deliver superior consistency, quality, and control—factors that become increasingly critical as therapies advance through clinical development toward commercialization [7].
The emerging challenge in the field is no longer simply choosing between controlled-rate and passive freezing, but rather recognizing that default CRF profiles, often set at a standard -1°C/min, may be suboptimal for many novel cell types and advanced therapies. Current industry surveys indicate that approximately 60% of users rely on these default freezer profiles [7], potentially overlooking significant opportunities to enhance post-thaw recovery and functionality. This technical guide examines the scientific basis for optimizing cooling rates, provides experimental methodologies for protocol development, and explores the implications for the rapidly advancing field of cell and gene therapy.
The Limitations of Default Freezing Profiles
The Thermodynamic Basis of Cryopreservation
Cryopreservation is not merely a process of cooling cells to low temperatures; it is a careful balancing act between multiple thermodynamic and biochemical phenomena. The cooling rate directly influences two primary mechanisms of cell damage: intracellular ice formation at rapid cooling rates and solution effects (dehydration and solute concentration) at slow cooling rates [56]. The optimal cooling rate achieves a balance where sufficient water exits the cell to minimize lethal intracellular ice formation, while avoiding excessive dehydration and prolonged exposure to cryoprotective agent toxicity [7].
Default CRF profiles typically employ a standard cooling rate of -1°C/min, based on historical optimization for robust cell lines and hematopoietic stem cells. However, this universal approach fails to account for fundamental differences in cell size, membrane permeability, and biological function across diverse cell types. Evidence suggests that specialized cell types—including iPSCs, hepatocytes, cardiomyocytes, and engineered cells—often demonstrate superior post-thaw outcomes with customized cooling protocols [7] [56].
Industry Survey Findings on CRF Practices
Recent survey data from the ISCT Cold Chain Management & Logistics Working Group provides insight into current industry practices and challenges. The findings reveal significant gaps in how controlled-rate freezers are qualified and utilized, highlighting opportunities for improved optimization.
Table 1: Key Findings from ISCT Cryopreservation Survey
| Survey Aspect | Finding | Implication |
|---|---|---|
| CRF Adoption | 87% use controlled-rate freezing | High prevalence in cell therapy manufacturing |
| Default Profile Usage | 60% use default CRF profiles | Potential under-optimization for specific cell types |
| Profile Optimization | 33% dedicate resources to freezing process development | Significant portion recognizing need for customization |
| Qualification Approach | Nearly 30% rely solely on vendors for system qualification | Potential gaps in understanding of system limitations |
| Scale Challenges | 22% identify "Ability to process at large scale" as biggest hurdle | Scaling optimized protocols remains challenging |
The survey further revealed that users experiencing challenges with default profiles typically work with more sensitive or specialized cell types, including iPSCs, hepatocytes, cardiomyocytes, photoreceptor cells, macrophages, and certain immune cells [7]. This underscores the biological diversity that necessitates moving beyond one-size-fits-all approaches to cryopreservation.
Comparative Analysis of Freezing Methodologies
Passive vs. Controlled-Rate Freezing: Technical Comparison
The fundamental distinction between passive freezing and controlled-rate freezing lies in the precision of temperature control throughout the critical phase change process. The following comparison examines the operational characteristics and performance metrics of each approach.
Table 2: Technical Comparison of Passive and Controlled-Rate Freezing Methods
| Parameter | Passive Freezing | Controlled-Rate Freezing |
|---|---|---|
| Cooling Control | Uncontrolled rate; approximately -1°C/min in early stages only [38] | Precisely programmable cooling rate (typically -0.3°C to -10°C/min) |
| Temperature Profile | Non-linear, accelerating and decelerating throughout process [38] | Linear or multi-step profiles with precise control through phase change |
| Ice Nucleation | Spontaneous, unpredictable timing and location | Controlled initiation at precise temperature (seeding) |
| Container Effects | Significant variation between vial positions (inner vs. outer rings) [38] | Minimal variation with proper load configuration and mapping |
| Capital Cost | Low ($100-$500 for passive devices) | High ($10,000-$50,000+ for commercial systems) |
| Operational Complexity | Simple, minimal training required | Requires specialized expertise for optimization and operation |
| Documentation | Limited process data recording | Comprehensive data logging for regulatory compliance |
| Batch Consistency | Higher variability between containers [38] | Excellent reproducibility with qualified protocols |
| Best Applications | Early R&D, robust cell lines, budget-limited settings | GMP manufacturing, sensitive cell types, late-stage clinical products |
Impact on Post-Thaw Cell Quality and Function
Recent comparative studies provide quantitative evidence regarding the functional outcomes of different freezing methodologies. In a retrospective study of 50 hematopoietic progenitor cell (HPC) products, researchers found that while total nucleated cell (TNC) viability post-thaw was greater for CRF (74.2% ± 9.9%) compared to passive freezing (68.4% ± 9.4%), there was no significant difference in the more clinically relevant metrics of CD34+ cell viability or engraftment outcomes [6]. This suggests that for some primary cell types, passive freezing may achieve functionally equivalent results despite differences in crude viability measures.
However, studies with more sensitive cell models reveal a different picture. Research using HepG2 cells demonstrated that the freezing profile significantly affected cell recovery and function in downstream toxicity assays [38]. Cells frozen using passive methods showed impaired recovery and greater susceptibility to methotrexate toxicity compared to those frozen with optimized CRF protocols, highlighting how cryopreservation-induced stress can alter cellular responses in functional assays.
Experimental Approaches for Cooling Rate Optimization
Protocol for Cooling Rate Optimization Studies
Establishing an optimized cooling protocol requires systematic investigation of multiple parameters. The following methodology provides a framework for conducting cooling rate optimization studies:
Step 1: Pre-freeze Cell Preparation
- Culture cells to log-phase growth with >80% confluency and >90% viability [16] [1]
- Characterize cells and confirm absence of microbial contamination [1]
- Harvest cells gently using appropriate dissociation reagents [16]
- Centrifuge at 100-400 × g for 5-10 minutes and resuspend in cryopreservation medium [16]
Step 2: Cryopreservation Medium Formulation
- Base medium appropriate for cell type (e.g., Leibovitz L-15 for ovarian tissue) [55]
- Cryoprotectant (typically 5-10% DMSO or alternative CPA) [56]
- Osmoprotectant (e.g., 0.1M sucrose for ovarian tissue) [55]
- Protein source (e.g., human serum albumin or FBS) [55]
Step 3: Experimental Freezing Protocol
- Aliquot cell suspension at consistent density (typically 1×10^3 - 1×10^6 cells/mL) [1]
- Program CRF with test cooling rates (e.g., -0.3°C/min, -1°C/min, -3°C/min) [57] [56]
- Include controlled nucleation (seeding) if applicable [55]
- Transfer to final storage temperature (-135°C to -196°C) after freezing cycle completion [16] [1]
Step 4: Post-Thaw Assessment
- Rapid thaw in 37°C water bath or automated thawing device [1] [56]
- Assess viability via trypan blue exclusion or automated cell counting [16]
- Evaluate specific functionality markers (e.g., motility for sperm [57], plating efficiency for HepG2 [38])
- Conduct longer-term culture to assess recovery and proliferation [38]
Figure 1: Experimental Workflow for Cooling Rate Optimization
Temperature Profiling and Monitoring Techniques
A critical aspect of cooling rate optimization is accurate temperature measurement throughout the freezing process. As demonstrated in detailed profiling studies, the assumption that passive freezing devices maintain a consistent -1°C/min cooling rate is inaccurate [38]. Actual temperature profiles show significant deviation, with rates accelerating before freezing, slowing during phase change, and accelerating again afterward.
To obtain accurate thermal profiles:
- Insert thin thermocouple probes directly into cell suspension through modified cryovial caps [38]
- Record temperature at frequent intervals (e.g., every second) throughout freezing process
- Compare temperature profiles across different vial positions (inner vs. outer rings in passive devices) [38]
- Repeat measurements across multiple independent freezing runs (n≥6 recommended) to assess reproducibility [38]
Case Studies in Cooling Rate Optimization
Stallion Sperm Cryopreservation: A 2025 study demonstrated the impact of precisely controlled pre-freeze cooling rates on post-thaw quality. Researchers compared slow (0.3°C/min), moderate (1°C/min), and fast (approximately 30°C/min) cooling from room temperature to 5°C before freezing. The fast cooling protocol resulted in significantly higher post-thaw total motility (51.8% vs. 45.0% for slow cooling), while maintaining equivalent progressive motility and cellular integrity markers [57]. This challenges conventional wisdom that slower cooling always produces superior results.
Ovarian Tissue Cryopreservation: Research on human ovarian tissue cryopreservation employed differential scanning calorimetry to characterize the thermodynamic properties of freezing medium, determining key parameters including glass transition temperature (-120.49°C), crystallization temperature (-20°C at 2.5°C/min), and melting temperature (-4.11°C) [55]. These parameters informed an optimized multi-step freezing protocol: 1°C/min to -7°C, controlled nucleation, 0.3°C/min to -40°C, and rapid cooling to -140°C. This approach preserved tissue quality equivalent to fresh controls and supported folliculogenesis during culture.
Advanced Applications and Emerging Trends
DMSO-Free Cryopreservation and Cooling Rate Considerations
The movement toward DMSO-free cryopreservation presents new challenges and opportunities for cooling rate optimization. Traditional slow-freeze protocols with DMSO-free media often yield suboptimal post-thaw viability [56]. However, optimizing freezing profiles offers a promising strategy to enhance performance.
A 2025 study on platelet cryopreservation demonstrated successful DMSO-free freezing using controlled-rate freezing with only NaCl solution, achieving post-thaw recovery >85% while maintaining functional integrity [58]. The addition of a deep eutectic solvent (10% choline chloride-glycerol) did not significantly enhance outcomes, suggesting that the controlled freezing process itself was the critical factor. This highlights how cooling rate optimization can enable alternative cryoprotective strategies that eliminate concerns about DMSO cytotoxicity in cell therapies [56].
Scaling Challenges and Technological Solutions
As cell therapies advance toward commercialization, scaling cryopreservation processes presents significant hurdles. Industry surveys identify "ability to process at large scale" as the biggest challenge for 22% of respondents [7]. Most organizations (75%) cryopreserve all units from an entire manufacturing batch together, while 25% divide batches, introducing potential variability between sub-batches.
Advanced technologies that may address scaling challenges include:
- Directional Freezing Systems: Incorporating precisely controlled pre-freeze cooling rates within highly controlled environments [57]
- Automated CRF Systems: Reducing operator dependency and improving batch-to-batch consistency
- Advanced Container Systems: Engineered cryocontainers with improved heat transfer characteristics
- Process Analytical Technology: Implementing in-line monitoring and control for critical quality attributes
Figure 2: Addressing Scaling Challenges Through Process Optimization
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful cooling rate optimization requires access to specialized reagents and equipment. The following toolkit outlines essential materials for conducting rigorous cryopreservation studies.
Table 3: Essential Research Reagents and Equipment for Cooling Rate Optimization
| Category | Specific Products/Models | Function and Application |
|---|---|---|
| Controlled-Rate Freezers | Planer Kryo Series [38] | Precise programmable control of cooling rates for optimization studies |
| Passive Freezing Devices | Nalgene Mr. Frosty [1] [38] | Isopropanol-based containers for uncontrolled-rate freezing comparisons |
| Temperature Profiling | Thin thermocouple probes with data loggers [38] | Direct measurement of sample temperature during freezing process |
| Cryopreservation Media | CryoStor CS10 [1], Synth-a-Freeze [16], mFreSR [1] | Commercially optimized formulations with defined compositions |
| Custom Media Components | DMSO [16], sucrose [55], HSA [55] | Base cryoprotectants and additives for formulation optimization |
| Cell Assessment Tools | Automated cell counters (Countess) [16], flow cytometers [57] [58] | Viability, phenotype, and functional assessment pre- and post-thaw |
| Specialized Assays | CASA (Computer-Assisted Sperm Analyzer) [57], RT-CES (Roche) [38] | Cell-type specific functional assessment post-thaw |
| Cryogenic Storage | Internal-threaded cryogenic vials [1], liquid nitrogen tanks [16] | Secure long-term storage of optimized cell banks |
Moving beyond default CRF profiles represents both a challenge and opportunity for the field of cryopreservation. The evidence clearly demonstrates that optimized cooling protocols can significantly enhance post-thaw cell quality and function, particularly for sensitive primary cells and advanced therapy products. While the standard -1°C/min cooling rate provides a reasonable starting point, researchers must recognize that this universal approach is unlikely to yield optimal results across diverse cell types.
The future of cryopreservation optimization lies in developing cell-type specific protocols based on fundamental biophysical principles and rigorous experimental data. As the field advances toward increasingly complex cell therapies and larger-scale manufacturing, the investment in cooling rate optimization will yield dividends in product consistency, efficacy, and ultimately, patient outcomes. By embracing the approaches outlined in this technical guide—systematic profiling, functional assessment, and strategic protocol development—researchers can overcome the limitations of default freezing profiles and unlock the full potential of their cryopreserved cell products.
The transition from laboratory-scale success to commercial-scale production represents one of the most significant challenges in the development of advanced therapeutic medicinal products (ATMPs). As the cell and gene therapy field advances, manufacturing scalability has emerged as a critical bottleneck, with cryopreservation identified as a major constraint by 22% of industry professionals [7]. The complexity intensifies when dealing with sensitive cell products that require precise thermal management during freezing to maintain viability, potency, and critical quality attributes (CQAs).
This technical guide examines scale-up bottlenecks within the specific context of controlled-rate freezing (CRF) versus passive freezing (PF) methodologies. While 87% of survey participants reported using controlled-rate freezing for cell-based products, particularly in late-stage and commercial applications, scaling these processes introduces formidable obstacles in infrastructure, process control, and economic viability [7]. Successfully navigating these challenges requires integrated approaches that combine advanced engineering solutions with sophisticated process control and monitoring systems to bridge the gap between laboratory innovation and commercial manufacturing success.
Controlled-Rate vs. Passive Freezing: A Technical Comparison
The selection between controlled-rate and passive freezing methods represents a fundamental decision point in process design, with significant implications for scalability, consistency, and product quality.
Scientific Principles and Mechanisms
Controlled-rate freezing employs specialized equipment to precisely regulate cooling velocity throughout the freezing process. This control allows manufacturers to manage critical parameters: the cooling rate before nucleation (affecting chilling injury and cryoprotectant toxicity), the temperature of ice nucleation (impacting osmotic stress and intracellular ice formation), and the cooling rate after nucleation (influencing dehydration and intracellular ice) before reaching the final storage temperature [7]. This precision is particularly crucial for delicate cell types including iPSCs, hepatocytes, cardiomyocytes, and engineered cells [7].
Passive freezing utilizes simple freezing devices like mechanical -80°C freezers without active cooling control. While this approach offers operational simplicity, it provides limited control over critical process parameters, potentially resulting in inconsistent freezing rates and product quality [7]. Recent evidence suggests that for some cell types, including hematopoietic progenitor cells (HPCs), passive freezing can achieve clinically comparable outcomes to controlled-rate freezing, with studies showing no significant difference in CD34+ cell viability post-thaw (77.1% ± 11.3% CRF vs. 78.5% ± 8.0% PF, P = 0.664) or engraftment parameters [6].
Comparative Analysis for Scale-Up Applications
Table 1: Technical and Economic Comparison of Freezing Methodologies for Scale-Up
| Parameter | Controlled-Rate Freezing | Passive Freezing |
|---|---|---|
| Process Control | High control over critical process parameters and CQAs | Limited control over freezing parameters |
| Capital Investment | High-cost infrastructure and consumables | Low-cost, low-consumable infrastructure |
| Operational Complexity | Specialized expertise required for use and optimization | Low technical barrier to adoption |
| Scalability | Bottleneck for batch scale-up; 60% use default profiles | Easier scaling; simple, one-step operation |
| Product Consistency | Excellent batch-to-batch consistency | Potential variability between batches |
| Suitable Clinical Stage | All stages, especially late-stage and commercial | Mainly early-stage (up to Phase II) |
| Typical Cell Types | Sensitive cells (iPSCs, cardiomyocytes, engineered cells) | Robust primary cells (HSCs, MSCs, some T-cells) |
Scale-Up Bottlenecks in Cryopreservation Processes
Equipment and Infrastructure Limitations
Traditional controlled-rate freezers present significant capacity constraints for commercial-scale manufacturing. The majority of respondents (75%) in industry surveys cryopreserve all units from an entire manufacturing batch together, indicating that manufacturing scale remains relatively small in the industry [7]. This approach becomes increasingly problematic as batch sizes grow, creating substantial bottlenecks in production scheduling and equipment utilization.
The economic burden of scaling controlled-rate freezing infrastructure presents another formidable challenge. High capital investment in specialized equipment, substantial liquid nitrogen consumption, and requirement for specialized technical staff create significant barriers to scale-up [7]. These economic factors must be carefully evaluated against product quality requirements when designing commercial-scale cryopreservation processes.
Process Transfer and Validation Challenges
The qualification complexity of controlled-rate freezing systems represents a major scale-up hurdle. Currently, little consensus exists on how to properly qualify controlled-rate freezers, with nearly 30% of organizations relying solely on vendor qualifications [7]. This approach often fails to represent final use cases, as vendor qualifications may not adequately address the full range of mass, container configurations, and temperature profiles encountered in production environments.
Effective qualification strategies should incorporate:
- Comprehensive mapping of temperature across a grid of locations within the freezing chamber
- Freeze curve analysis across different container types and load configurations
- Mixed load validation to establish performance boundaries for various sample configurations [7]
The regulatory landscape further complicates scale-up, particularly when transitioning from passive to controlled-rate freezing methods. Such changes alter multiple critical process parameters, potentially requiring significant comparability studies and process revalidation [7]. Adopting controlled-rate freezing early in clinical development can avoid challenging manufacturing changes later, but requires substantial resource investment at earlier stages.
Strategic Solutions for Scaling Cryopreservation Processes
Advanced Equipment and Modular Approaches
Single-use systems and modular bioreactors have transformed approaches to scaling challenges across bioprocessing. These technologies enable scale-out strategies rather than traditional scale-up methodologies, allowing manufacturers to maintain process conditions that closely mirror laboratory-scale environments while achieving commercial production volumes through parallel operation of multiple smaller units [59]. This approach proves particularly advantageous for autologous cell therapies where individual patient doses require separate processing streams.
Automated rate-controlled freezers with advanced monitoring capabilities represent another critical technological solution. Modern systems incorporate sophisticated process control capabilities that address historical scale-up challenges through automated feedback control systems maintaining optimal freezing parameters throughout the production process [60]. The integration of data logging and remote monitoring features allows manufacturers to track temperature trends and system performance in real-time, providing crucial documentation for regulatory compliance and quality control.
Process Optimization and Quality by Design
Implementing Quality by Design (QbD) principles provides a structured framework for addressing scale-up bottlenecks through systematic risk assessment and mitigation strategies. This approach identifies potential failure modes early in development, enabling proactive design modifications that prevent issues during commercial production [59]. Establishing a well-defined design space creates operational boundaries within which consistent product quality can be assured while maintaining regulatory compliance.
Computational Fluid Dynamics (CFD) modeling offers powerful tools for characterizing and optimizing freezing processes. CFD simulations can map fluid flow patterns and homogeneous distribution of hydrodynamic forces in freezing systems, guiding the development of optimized protocols [61]. These engineering approaches enable researchers to understand and control freezing processes in a way that addresses major technology bottlenecks in bioprocessing knowledge.
Table 2: Scale-Up Strategy Selection Framework
| Scale-Up Challenge | Proposed Solution | Implementation Considerations |
|---|---|---|
| Limited CRF Capacity | Modular scale-out approach with parallel freezing units | Capital investment, facility space, operational complexity |
| Process Consistency | QbD principles with defined design space | Resource-intensive initial characterization, regulatory alignment |
| System Qualification | Comprehensive mapping strategy beyond vendor protocols | Expertise requirements, time investment, validation workload |
| Economic Viability | Technology-specific cost-benefit analysis | Trade-offs between control level and infrastructure cost |
| Post-Thaw Variability | Integrated freezing and thawing process control | Equipment compatibility, staff training requirements |
Experimental Protocols and Methodologies
Controlled-Rate Freezer Qualification Protocol
Objective: To establish comprehensive performance qualification of controlled-rate freezing systems for large-scale production environments.
Methodology:
- Temperature Mapping: Perform full versus empty chamber mapping across a three-dimensional grid of locations to identify thermal gradients and cold spots [7]
- Load Configuration Studies: Evaluate a range of mass values, container types, and fill volumes representing production-scale batches
- Freeze Curve Analysis: Monitor and record thermal profiles at multiple locations within product containers to verify consistent freezing kinetics
- Mixed Load Validation: Assess system performance with heterogeneous load configurations simulating actual production conditions
Acceptance Criteria: All locations within the chamber maintain temperatures within ±2°C of setpoint during critical freezing phases, and freeze curves demonstrate consistent profiles across container locations [7].
Freezing Process Optimization Protocol
Objective: To determine optimal freezing parameters for specific cell types and container systems.
Methodology:
- Cooling Rate Screening: Evaluate cell viability, recovery, and functionality across a range of cooling rates (typically -0.5°C/min to -10°C/min)
- Nucleation Control: Assess impact of manual vs. automated nucleation on product consistency
- Container-Specific Validation: Characterize thermal transfer properties for specific container systems and fill volumes
- Post-Thaw Functionality Assessment: Evaluate not only immediate viability but also longer-term functionality including potency markers and engraftment potential [62]
Analytical Methods: Incorporate freeze curve monitoring as part of the release process, establishing action limits to identify changes in CRF performance [7].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagents and Equipment for Cryopreservation Scale-Up
| Item | Function | Scale-Up Considerations |
|---|---|---|
| Controlled-Rate Freezer | Precisely regulates cooling rate during freezing | Capacity limitations, liquid nitrogen consumption, qualification complexity |
| Cryoprotective Agents (CPAs) | Protect cells from freezing damage (e.g., DMSO) | Biochemical toxicity, osmotic stress during addition/removal, quality specifications |
| Primary Containers | Hold product during freezing (cryobags, vials) | Thermal transfer properties, fill volume optimization, compatibility with freezing systems |
| Temperature Monitoring System | Track thermal profiles throughout freezing process | Number of sensors, data logging capabilities, placement strategy |
| Cryogenic Storage Systems | Long-term preservation at <-150°C | Inventory management, retrieval processes, capacity planning |
| Controlled Thawing Devices | Standardize warming process at clinical site | Warming rate control, contamination prevention, ease of use |
Visualization of Scale-Up Decision Framework
The following workflow diagram outlines a systematic approach for selecting and implementing scale-up strategies for large-batch cryopreservation processes:
Conquering the scale-up bottleneck in large-batch processing requires a multifaceted approach that balances scientific principles with practical manufacturing constraints. The selection between controlled-rate and passive freezing methods represents a critical decision point that must consider cell-specific sensitivities, stage of clinical development, and commercial viability.
Successful scale-up implementations integrate advanced technologies including modular equipment approaches, comprehensive process characterization, and robust quality systems with freeze curve monitoring. As the industry continues to evolve, the organizations that effectively bridge the gap between laboratory innovation and commercial manufacturing will be those that embrace these integrated strategies while maintaining focus on fundamental process understanding.
Future directions point toward increased automation, digital twin technology for process simulation, and advanced process analytical technologies (PAT) for real-time quality assessment. By leveraging these emerging technologies while implementing the foundational strategies outlined in this guide, researchers and manufacturing professionals can overcome the scale-up bottlenecks that have historically constrained the widespread availability of advanced cell and gene therapies.
Cryopreservation is a cornerstone technology in biomedical research and pharmaceutical development, enabling the long-term storage of cells, tissues, and other biologics. Within this field, controlled-rate freezing and passive freezing represent two fundamental approaches for achieving sample stability at ultra-low temperatures. Controlled-rate freezing utilizes programmable equipment to precisely manage the cooling process, while passive freezing involves simpler, uncontrolled cooling in mechanical freezers [63] [6]. This technical guide focuses on two advanced techniques derived from these approaches: interrupted cooling protocols for slow freezing and vitrification, a rapid cooling method. The selection of a cryopreservation strategy is critical, as it must mitigate various freeze-thaw stresses, including intracellular ice formation, osmotic shock, and solute-related damage, to ensure high post-thaw viability and functionality [23]. This whitepaper provides an in-depth analysis of the principles, methodologies, and applications of interrupted cooling and vitrification, serving as a resource for researchers and drug development professionals in optimizing their preservation protocols.
Fundamentals of Interrupted Cooling
Principles and Definition
Interrupted cooling protocols are sophisticated slow freezing strategies where the cooling process is deliberately paused at one or more specific sub-zero temperatures before samples are ultimately plunged into liquid nitrogen for long-term storage [23]. The core objective of these protocols is to exert control over the physical stresses that cells experience during freezing. The plunge temperature, the temperature at which cooling is interrupted and samples are transferred to liquid nitrogen, is a critical parameter that significantly impacts post-thaw cell survival and must be optimized for each cell type [23]. By strategically halting the cooling process, these methods allow for controlled cellular dehydration, reducing the potential for lethal intracellular ice formation during subsequent cooling phases.
Key Protocol Variations
Interrupted cooling encompasses several specific techniques, each designed to manage different aspects of freeze-induced stress.
Two-Step Freezing: This method involves an initial rapid cooling of the sample from room temperature to a predetermined hold temperature. The sample is maintained at this intermediate sub-zero temperature for a set duration, allowing time for intracellular water to exit the cell, thereby reducing the probability of intracellular ice formation during the final cooling phase to liquid nitrogen temperature [23]. Finding the critical sub-zero hold temperatures and optimizing the holding times are the two primary objectives for improving cell survival in this protocol.
Graded Freezing: This is a more complex form of interrupted cooling where the sample is cooled through a series of progressively lower temperatures, with holds at each step. This graded approach allows researchers to elucidate how different types of cryoprotectants protect cells across varying temperature ranges [23]. It provides a powerful tool for understanding fundamental cryobiological concepts and optimizing preservation conditions for novel cell types.
The following diagram illustrates the logical decision-making process and the key steps involved in designing an interrupted cooling protocol.
Figure 1: Logic flow for designing interrupted cooling protocols.
Experimental Protocol: Two-Step Freezing for Cells in Suspension
The following methodology provides a detailed template for implementing a two-step freezing protocol, which can be adapted for specific cell types.
Step 1: Sample Preparation
- Harvest and concentrate cells according to standard protocols.
- Resuspend the cell pellet in an appropriate freezing medium containing a cryoprotectant such as 10% DMSO. The medium should often be supplemented with proteins (e.g., albumin) or other non-penetrating agents to provide extracellular protection [63] [23].
- Aliquot the cell suspension into cryovials.
Step 2: Initial Cooling and Hold
- Place the cryovials in a freezing chamber pre-cooled to 4°C.
- Initiate cooling. For the two-step method, rapidly cool the samples from +4°C to the predetermined hold temperature (e.g., -40°C) at a rate of -1°C to -3°C per minute [23].
- Hold the samples at the target temperature (e.g., -40°C) for a defined period, typically 5-15 minutes. This hold time is critical as it permits cellular dehydration, reducing the risk of intracellular ice formation upon subsequent plunging into liquid nitrogen [23].
Step 3: Final Plunge and Storage
- After the hold period, immediately transfer the cryovials to liquid nitrogen (-196°C) for long-term storage.
Step 4: Thawing
- Rapidly thaw the cryovials in a 37°C water bath with gentle agitation until only a small ice crystal remains.
- Immediately and gradually dilute the thawed cell suspension with pre-warmed culture medium or a specified diluent solution to minimize osmotic shock during cryoprotectant removal [63].
Table 1: Key Parameters for Two-Step Freezing of Different Cell Types
| Cell Type | Cryoprotectant | Cooling Rate to Hold Temp | Hold Temperature | Hold Time | Plunge Temperature |
|---|---|---|---|---|---|
| Hematopoietic Progenitor Cells | 10% DMSO | -1°C to -3°C/min | -40°C to -50°C | 10 min | -40°C [6] |
| Fibroblasts | 10% DMSO | -1°C to -3°C/min | -50°C | 5-15 min | -50°C [23] |
| Testicular Tissue | 10% DMSO | -1°C to -3°C/min | -40°C to -80°C* | 5-10 min | -80°C [63] |
| Lymphocytes | 10% DMSO | -1°C to -3°C/min | -50°C | 10 min | -50°C [23] |
Note: For passive freezing of testicular tissue, the hold is often achieved by placing vials in a -80°C freezer overnight [63].
Fundamentals of Vitrification
Principles and Definition
Vitrification represents a paradigm shift from traditional slow freezing. It is an ultra-rapid cooling process that converts a liquid solution directly into a solid, glass-like state without the formation of ice crystals [63] [64]. This is achieved by combining high cooling rates with high concentrations of cryoprotective agents (CPAs). The extremely fast cooling prevents water molecules from organizing into a crystalline structure, while the CPAs increase the solution's viscosity, effectively "freezing" the molecular arrangement in place [63] [65]. The primary advantage of vitrification is the complete avoidance of both intra- and extracellular ice crystal formation, which is a major source of damage in slow freezing protocols [64]. However, the requirement for high CPA concentrations introduces the risk of chemical toxicity and osmotic shock to the cells, making the timing of the procedure critical [64] [66].
Vitrification Devices: Open vs. Closed Systems
A key practical consideration in vitrification is the choice of device, which directly impacts cooling rate and biosafety.
Open Devices (e.g., Cryotop, Cryoloop): These allow the sample in its tiny volume of vitrification solution to come into direct contact with liquid nitrogen. This direct contact facilitates extremely high cooling and warming rates, which are beneficial for survival [65]. However, this poses a potential risk of contamination from pathogens that may be present in the liquid nitrogen, a risk that has been demonstrated under experimental conditions [65].
Closed Devices (e.g., Rapid-i Kit): These systems encapsulate the sample within a sealed straw or container before immersion in liquid nitrogen. While the cooling rate is marginally lower than that of open systems, closed devices offer 100% protection from cross-contamination, a significant advantage for clinical applications and biobanking [65]. Studies have shown that closed devices like the Rapid-i Kit can achieve high survival rates (94.6% for oocytes) that are comparable to, and sometimes better than, open systems [65].
Experimental Protocol: Cryotop Vitrification for Embryos
The following protocol details the vitrification of in vitro-produced embryos using the Cryotop system, a minimum-volume open device.
Step 1: Equilibration
- Prepare the base medium and vitrification solutions. The base medium is often TCM-199 or another suitable holding medium supplemented with 20% serum [66].
- Transfer one to three embryos into a drop of equilibration solution (e.g., 7.5% ethylene glycol + 7.5% DMSO) for 10-15 minutes at room temperature. During this step, the embryos will undergo initial dehydration and permeation of CPAs, observable as shrinkage followed by gradual return to original volume [66].
Step 2: Vitrification Solution Exposure
- After equilibration, immediately move the embryos to a drop of vitrification solution (e.g., 15% ethylene glycol + 15% DMSO + 0.5 M sucrose) for 60 seconds at room temperature. The exposure time must be strictly timed to minimize CPA toxicity while ensuring sufficient dehydration [66].
Step 3: Loading and Cooling
- Using the Cryotop device, place the embryos in a minimal volume (less than 1 µl) of the vitrification solution onto the film strip.
- Immediately plunge the loaded Cryotop directly into liquid nitrogen. The ultra-thin film and minimal volume ensure a cooling rate exceeding 20,000°C/min, which is necessary for successful vitrification [65].
Step 4: Storage and Warming
- Secure the protective cap over the film strip while submerged in liquid nitrogen and transfer to a storage tank.
- For warming, quickly remove the Cryotop from liquid nitrogen and immediately immerse it directly into a pre-warmed (37°C) thawing solution (e.g., 1.0 M sucrose) for 1 minute. The direct immersion provides the ultra-rapid warming rate required to prevent devitrification (ice crystal formation during warming) [66] [65].
- Sequentially transfer embryos through a series of dilution solutions (e.g., 0.5 M sucrose for 3 minutes) and washing solutions to gradually remove the CPAs [63] [66].
Table 2: Comparison of Vitrification versus Slow Freezing Outcomes in Different Biological Materials
| Biological Material | Technique | Survival / Viability Rate | Key Functional Outcome | Source |
|---|---|---|---|---|
| Neonatal Bovine Testicular Tissue | Vitrification | N/A | Effective preservation of cell membrane integrity, promotion of proliferation, protection against apoptosis. Germ cell colonies formed after in vitro culture. | [63] |
| Neonatal Bovine Testicular Tissue | Controlled Slow Freezing | N/A | 47.9% of seminiferous tubules showed >70% attachment to basement membrane. Germ cell colonies formed after in vitro culture. | [63] |
| Human Oocytes | Vitrification | 89.7% | Comparable pregnancy and implantation rates to modified slow freezing. | [67] |
| Human Oocytes | Slow Freezing (Modified Rehydration) | 89.8% | High pregnancy, implantation, and birth rates, comparable to vitrification. | [67] |
| Hematopoietic Progenitor Cells | Controlled-Rate Freezing | 74.2% (TNC Viability) | Neutrophil engraftment: 12.4 days; Platelet engraftment: 21.5 days. | [6] |
| Hematopoietic Progenitor Cells | Passive Freezing | 68.4% (TNC Viability) | Neutrophil engraftment: 15.0 days; Platelet engraftment: 22.3 days. | [6] |
| Cỏ Goat Embryos (Zygote Stage) | Cryotop Vitrification | Lower survival vs. blastocyst vitrification | Successfully produced blastocysts post-warming, though at lower rates. | [66] |
The following workflow summarizes the key steps and critical decisions in the vitrification process, highlighting the differences between open and closed systems.
Figure 2: Workflow for sample vitrification.
The Scientist's Toolkit: Essential Reagents and Materials
Successful implementation of interrupted cooling and vitrification protocols requires a suite of specialized reagents and equipment. The following table details key solutions and their specific functions in the cryopreservation process.
Table 3: Key Research Reagent Solutions for Cryopreservation Protocols
| Reagent / Material | Composition Example | Function in Protocol |
|---|---|---|
| Permeating Cryoprotectants | Dimethyl sulfoxide (DMSO), Ethylene Glycol (EG), Propylene Glycol (PROH) | Penetrate the cell membrane, lower the freezing point, and reduce the amount of ice formed by increasing the total solute concentration. Essential for both slow freezing and vitrification [23] [64]. |
| Non-Permeating Cryoprotectants | Sucrose, Trehalose, Ficoll, Albumin | Do not enter the cell; create an osmotic gradient that draws water out of the cell, promoting dehydration and reducing intracellular ice formation. Critical in vitrification solutions [63] [66]. |
| Base Medium | Hanks’ Balanced Salt Solution (HBSS), TCM-199, DPBS | Serves as the ionic and nutrient foundation for cryoprotectant solutions. Often supplemented with antibiotics and proteins like albumin [63] [66]. |
| Vitrification Solution | 15% EG + 15% DMSO + 0.5 M Sucrose in base medium | High concentration cocktail of permeating and non-permeating agents used immediately before plunging into LN₂. Induces a glassy state [66]. |
| Equilibration Solution | 7.5% EG + 7.5% DMSO in base medium | Lower concentration CPA solution used for initial exposure, allowing for partial cellular dehydration and CPA permeation with reduced osmotic shock [66]. |
| Thawing / Warming Solution | 1.0 M Sucrose in base medium | High osmotic strength solution used for the first step of warming to prevent osmotic shock during the influx of water as CPAs leave the cell [63] [66]. |
| Dilution & Washing Solutions | 0.5 M Sucrose, then sucrose-free base medium | Series of solutions with progressively lower osmotic strength used to gradually and safely remove CPAs from the sample after thawing/warming [63]. |
Interrupted cooling and vitrification represent two advanced, yet distinct, pathways for achieving successful cryopreservation within the broader context of controlled-rate and passive freezing research. The choice between these techniques is not a matter of superiority but of strategic application. Interrupted cooling protocols offer a methodical approach to managing the physics of ice formation through controlled dehydration, making them particularly suitable for a wide range of cell suspensions where the biophysical parameters are well-characterized [23] [6]. Vitrification, by contrast, avoids ice formation entirely through speed and high solute concentration, proving highly effective for more complex and sensitive structures like oocytes, embryos, and tissues [63] [64] [66].
The ongoing research in this field continues to refine both approaches. Optimization of plunge temperatures and hold times enhances the efficacy of interrupted cooling [23], while developments in closed vitrification systems successfully address the critical issue of biosafety without compromising survival rates [65]. Furthermore, modifications to supporting protocols, such as improved rehydration methods for slow-frozen oocytes, can dramatically elevate the performance of traditional techniques [67]. For researchers and drug development professionals, the optimal cryopreservation strategy must be empirically determined, balancing factors such as cell type, desired throughput, required post-thaw functionality, and regulatory considerations. A deep understanding of the principles and methodologies outlined in this guide provides a solid foundation for making these critical decisions and advancing the science of preservation.
Data-Driven Decisions: Clinical Outcomes and Cost-Benefit Analysis
The cryopreservation of hematopoietic progenitor cells (HPCs) represents a critical enabling technology for modern transplantation medicine, allowing for the long-term storage of cellular products for autologous and allogeneic transplants. Within this field, a fundamental technical dichotomy exists between controlled-rate freezing (CRF) and passive freezing (PF) methodologies. CRF has traditionally been regarded as the gold standard, offering precise control over cooling parameters through programmable equipment. In contrast, PF—an uncontrolled-rate method utilizing mechanical freezers set at -80°C—has gained prominence as a simpler, more accessible alternative, particularly in resource-constrained settings [6] [12]. The core debate centers on whether the technical simplicity of PF compromises cellular integrity, specifically the viability and functional capacity of total nucleated cells (TNCs) and CD34+ hematopoietic stem cells post-thaw. This in-depth technical guide synthesizes current evidence to directly compare these methods, providing researchers and drug development professionals with a data-driven framework for selecting and optimizing cryopreservation protocols.
Experimental Designs and Comparative Outcomes
Core Comparative Study: A Retrospective Analysis
A pivotal 2025 retrospective study provides a direct, head-to-head comparison of CRF and PF, analyzing 50 HPC products (25 per method) [6] [12].
Methodology:
- Cell Sources: Apheresis-derived (HPC(A)) and marrow-derived (HPC(M)) products.
- Cryoprotectant: A uniform solution of 15% Dimethyl Sulfoxide (DMSO) and 9% albumin in Plasmalyte-A was used for both methods.
- Controlled-Rate Freezing (CRF): Products were frozen in a controlled-rate freezer with a programmed cooling profile.
- Passive Freezing (PF): Products were placed in a -80°C mechanical freezer. To achieve a cooling rate of approximately 1–2°C/min, products in metal cassettes were wrapped in absorbent pads or styrofoam insulation [12].
- Storage: All products were transferred to long-term storage in a liquid nitrogen freezer at <-150°C after the initial freezing process.
- Assessment: Post-thaw viability of TNCs and CD34+ cells was analyzed. Engraftment outcomes, including days to neutrophil and platelet recovery, were tracked in patients [6].
Long-Term Stability Assessment
A separate 2025 study evaluated the viability of CD34+ progenitor cell products after long-term storage at -80°C [68]. This single-center, retrospective study analyzed 72 cryopreserved products from 25 patients, stored for a median of 868 days (approximately 2.4 years). Viability was assessed using both acridine orange (AO) staining and 7-AAD flow cytometry, providing a robust comparison of analytical methods alongside stability data [68].
The following tables consolidate key quantitative findings from the cited research, offering a clear comparison of post-thaw viability and functional outcomes.
Table 1: Direct Comparison of Post-Thaw Viability and Engraftment (CRF vs. PF)
| Parameter | Controlled-Rate Freezing (CRF) | Passive Freezing (PF) | P-value |
|---|---|---|---|
| TNC Viability (Mean ± SD) | 74.2% ± 9.9% (N=25) | 68.4% ± 9.4% (N=25) | 0.038 |
| CD34+ Cell Viability (Mean ± SD) | 77.1% ± 11.3% (N=13) | 78.5% ± 8.0% (N=25) | 0.664 |
| Days to Neutrophil Engraftment | 12.4 ± 5.0 (N=12) | 15.0 ± 7.7 (N=16) | 0.324 |
| Days to Platelet Engraftment | 21.5 ± 9.1 (N=12) | 22.3 ± 22.8 (N=16) | 0.915 |
Source: Cytotherapy. 2025 Sep;27(9):1145-1149 [6] [12].
Table 2: Long-Term Stability at -80°C and Viability Assessment Methods
| Parameter | Findings | Notes |
|---|---|---|
| Median Post-Thaw Viability | 94.8% | After median 868 days storage [68] |
| Viability Decline Rate | ~1.02% per 100 days | R² = 0.283, p < 0.001 [68] |
| Mean Viability Loss (Delayed Assessment) | 9.2% (AO); 6.6% (7-AAD) | AO demonstrated greater sensitivity to delayed degradation (p < 0.001) [68] |
| Engraftment Correlation | Preserved kinetics in most patients | Outcomes primarily influenced by disease type, not storage duration or donor age [68] |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Cryopreservation Studies
| Item | Function / Application | Example |
|---|---|---|
| Cryoprotectant | Prevents intracellular and extracellular ice crystal formation; reduces osmotic shock. | Dimethyl Sulfoxide (DMSO) [12] [69] |
| Protein Supplement | Provides a protective matrix; traditionally used in freezing media. | Fetal Bovine Serum (FBS) [69] |
| Serum-Free Media | Xeno-free alternative to FBS; mitigates infection risk and unwanted immune responses. | CryoStor CS10, NutriFreez D10 [69] |
| Viability Stain | Fluorescent dye for microscopy-based viability count; excludes viable cells. | Acridine Orange (AO) / Ethidium Bromide (EB) [68] |
| Viability Dye (Flow Cytometry) | Nucleic acid dye that penetrates compromised membranes; identifies dead cells via flow cytometry. | 7-Aminoactinomycin D (7-AAD) [68] |
| Controlled-Rate Freezer | Equipment for precise, programmable control of cooling rates. | Programmable freezer (e.g., Planer Kryo) [38] |
| Passive Freezing Device | Insulating container placed in a -80°C freezer to moderate cooling rate. | Mr. Frosty (isopropanol-filled) [38] |
Workflow and Critical Process Parameters
The experimental workflow for a typical comparative cryopreservation study, from cell collection to data analysis, can be visualized as follows. This diagram outlines the key stages and critical parameters that directly influence post-thaw viability.
Discussion and Technical Interpretation
Decoupling Viability from Functionality
The data reveals a critical distinction between cell viability and clinical functionality. While the TNC viability was statistically superior in the CRF group (74.2% vs. 68.4%, p=0.038) [6], this difference did not translate into a meaningful divergence in clinical outcomes. The most significant finding is the equivalent post-thaw viability of CD34+ hematopoietic stem cells between the two methods (77.1% vs. 78.5%, p=0.664) [6]. This is a crucial metric, as the CD34+ cell population is primarily responsible for bone marrow reconstitution and engraftment. The comparable neutrophil and platelet engraftment kinetics further confirm that PF preserves the functional integrity of the most critical cell population for transplantation success [6] [68].
The Impact of Temperature Excursions
A critical consideration in cryopreservation is the management of transient warming events (TWEs), which occur when samples are briefly exposed to warmer temperatures during retrieval from deep storage. Ice crystal growth, or recrystallization, can occur at temperatures above the glass transition temperature (Tg, approximately -135°C for pure water), leading to cellular damage [70]. While DMSO offers some protection, its capacity to inhibit ice recrystallization is limited. Research indicates that TWEs, particularly those cycling above -135°C, can reduce colony-forming units (a potency assay) by up to 20% [70]. This underscores the importance of rigorous cold chain management, regardless of the initial freezing method, to maintain product quality during long-term storage.
Protocol Standardization and Industry Trends
Achieving consistent results with PF requires careful attention to protocol standardization. The cooling rate in passive freezers is not inherently uniform and can be influenced by factors such as the type of insulation and the freezer's thermal mass [38]. Wrapping products in absorbent pads or styrofoam is a documented method to achieve the target cooling rate of 1–2°C/min [12]. Despite the demonstrated efficacy of PF, an industry survey by the ISCT Cold Chain Management & Logistics Working Group found that 87% of respondents use controlled-rate freezing for cell-based therapies, with adoption near-universal for late-stage and commercial products [7]. This suggests that while PF is a technically valid alternative, CRF remains the preferred method in advanced therapeutic settings, likely due to its enhanced process control, documentation capabilities, and regulatory familiarity.
The body of evidence demonstrates that passive freezing is an acceptable and effective alternative to controlled-rate freezing for the cryopreservation of hematopoietic progenitor cells. The key differentiator in post-thaw outcomes is not the freezing method itself, but the rigorous application of a standardized protocol that ensures an appropriate cooling rate and consistent handling. For resource-constrained settings, late-phase clinical trials, or commercial applications where process control and documentation are paramount, the investment in CRF is justified. However, for many research and early-phase clinical applications, PF provides a technically sound, cost-effective solution that robustly preserves the critical CD34+ cell population and ensures successful engraftment, making it a viable and practical choice in the fundamental toolkit of cell therapy research and development.
In the field of hematopoietic stem cell transplantation (HSCT), the successful reconstitution of a patient's blood and immune system represents the fundamental gateway to therapeutic success. Neutrophil and platelet engraftment times serve as the ultimate quantitative metrics for evaluating this critical recovery process. Neutrophil engraftment is clinically defined as the first of three consecutive days where the absolute neutrophil count (ANC) recovers to ≥0.5 × 10⁹/L, while platelet engraftment is achieved on the first of three consecutive days with platelet counts ≥20 × 10⁹/L without transfusion support [71]. These precisely measured endpoints provide an objective assessment of graft function and have profound implications for patient outcomes, including risk of infection, bleeding complications, and overall survival.
The cryopreservation method employed for hematopoietic progenitor cells (HPCs)—specifically the comparison between controlled-rate freezing (CRF) and passive freezing (PF)—represents a significant variable that can influence these crucial engraftment metrics. As cell and gene therapies advance, understanding the interplay between cryopreservation methodology and engraftment kinetics becomes increasingly vital for both clinical outcomes and process optimization in pharmaceutical development. This whitepaper examines the current evidence regarding cryopreservation effects on engraftment kinetics, providing researchers and clinicians with evidence-based guidance for protocol development.
Comparative Analysis of Controlled-Rate vs. Passive Freezing
Fundamental Principles and Current Practices
Cryopreservation techniques aim to preserve cellular viability and function through carefully controlled temperature reduction. Controlled-rate freezing (CRF) utilizes programmable equipment to decrement temperature incrementally according to preset protocols, typically cooling at approximately -1°C/minute until freezing occurs, followed by rapid cooling to counteract the latent heat of fusion released during ice formation, then resuming gradual cooling until reaching final storage temperatures [12]. In contrast, passive freezing (PF), also referred to as uncontrolled-rate or "dump" freezing, involves placing HPC products in a -80°C mechanical freezer, often with insulation materials to moderate the cooling rate [12].
Recent industry surveys reveal that 87% of cell therapy facilities utilize CRF for cryopreservation, while only 13% employ passive freezing, with the latter predominantly used in early clinical development phases (up to Phase II) [7]. This distribution reflects the perceived advantages of each method: CRF offers precise parameter control and comprehensive documentation capabilities essential for cGMP manufacturing, while PF provides operational simplicity, lower costs, and easier scaling [7].
Direct Comparative Evidence: Engraftment Outcomes
A recent retrospective study of 50 HPC products provides the most current direct comparison of engraftment outcomes between cryopreservation methods. The findings demonstrate comparable clinical efficacy despite differences in cellular viability metrics:
Table 1: Comparative Analysis of Cryopreservation Methods on HPC Metrics and Engraftment
| Parameter | Controlled-Rate Freezing (CRF) | Passive Freezing (PF) | P-value |
|---|---|---|---|
| TNC Viability Post-Thaw | 74.2% ± 9.9% | 68.4% ± 9.4% | 0.038 |
| CD34+ Cell Viability Post-Thaw | 77.1% ± 11.3% | 78.5% ± 8.0% | 0.664 |
| Days to Neutrophil Engraftment | 12.4 ± 5.0 | 15.0 ± 7.7 | 0.324 |
| Days to Platelet Engraftment | 21.5 ± 9.1 | 22.3 ± 22.8 | 0.915 |
This study concluded that despite statistically significant differences in total nucleated cell (TNC) viability post-thaw, the more clinically relevant metrics—CD34+ cell viability and engraftment times—showed no significant differences between methods [6]. The research affirms that "cryopreservation outcomes using CRF or PF are comparable so PF is an acceptable alternative to CRF for initial cryopreservation before long-term storage in a liquid nitrogen freezer" [6].
Additional clinical evidence supports these findings. A 2023 study of 93 allogeneic transplant patients comparing frozen versus fresh products found comparable neutrophil engraftment (median 14 vs. 16 days, p=0.393) though platelet engraftment was slightly delayed in the frozen product group (median 15 vs. 12 days, p<0.001) [72]. Importantly, this study identified no significant differences in graft failure, graft-versus-host disease, relapse, or survival between groups, supporting the safety of cryopreserved products [72].
Cell-Type Specific Responses to Cryopreservation
Different cellular populations exhibit varying sensitivity to cryopreservation methodologies. Research indicates that platelet recovery and functionality appear superior following CRF compared to PF, with one study demonstrating 91.0% ± 5.5% platelet recovery with CRF versus significantly lower recovery with uncontrolled-rate methods [73]. Lymphocyte recovery and viability also favor CRF conditions, though proliferative responses show minimal intergroup differences [73].
These cell-type specific responses highlight the importance of tailoring cryopreservation protocols to the specific biologic product. For HPCs, the critical CD34+ progenitor population demonstrates equivalent post-thaw viability regardless of freezing method, explaining the comparable engraftment kinetics observed in clinical studies [6] [12].
Experimental Methodologies for Engraftment Analysis
Standardized Cryopreservation Protocols
To ensure reproducible results in cryopreservation research, standardized protocols must be rigorously followed. For HPC cryopreservation, the following methodology represents current best practices:
HPC Cryopreservation Protocol
- Preparation of Cryoprotectant Solution: Prepare a solution containing 15% dimethyl sulfoxide (DMSO) and 9% albumin in Plasmalyte-A [12].
- Cell Concentration Adjustment: Concentrate or dilute HPC products to reach an optimal cell concentration of 600-800 × 10⁶ TNC/mL [12].
- Cryoprotectant Addition: Combine HPC product with cryoprotectant solution in a 1:1 ratio to achieve final concentrations of 7.5% DMSO and 4.5% albumin [12].
- Controlled-Rate Freezing: Using a controlled-rate freezer, cool samples at -1°C/minute to -5°C, followed by a rapid cooling phase to counteract latent heat of fusion, then resume cooling at -1°C/minute to -40°C, followed by a final cooling at -5°C/minute to -100°C [12].
- Passive Freezing: Place samples in a -80°C mechanical freezer using insulating materials (e.g., disposable absorbent pads or styrofoam) to approximate a cooling rate of 1-2°C/minute [12].
- Long-Term Storage: Transfer all samples to liquid nitrogen freezers ≤-150°C for long-term storage [12].
Table 2: Essential Research Reagents for Cryopreservation Studies
| Reagent/Condiment | Function | Application Notes |
|---|---|---|
| DMSO (5-15%) | Primary cryoprotectant preventing intracellular ice crystal formation | Concentration varies by institution (5-15%); 7.5% common in HPC cryopreservation [12] [74] |
| Human Serum Albumin | Membrane stabilizer and additive to reduce cryoprotectant toxicity | Typically used at 4.5-9% concentration in cryoprotectant solutions [12] |
| Plasmalyte-A | Balanced salt solution providing physiological buffer | Base solution for cryoprotectant mixtures [12] |
| Hydroxyethyl Starch | Extracellular cryoprotectant reducing ice crystal formation | Used in some protocols as DMSO adjunct [74] |
| Cryostor CS10 | Commercial, serum-free, GMP-manufactured cryopreservation medium | Pre-formulated alternative to lab-made solutions [1] |
Engraftment Assessment Methodologies
Rigorous assessment of engraftment requires both in vitro and in vivo analyses:
In Vitro Quality Metrics
- Viable CD34+ Analysis: Flow cytometry with 7-aminoactinomycin D (7-AAD) viability staining to quantify viable CD34+ cell populations post-thaw [6] [12].
- Total Nucleated Cell Recovery: Automated cell counting with trypan blue exclusion to assess total cell viability [12].
- Colony-Forming Unit (CFU) Assay: Semisolid methylcellulose cultures to quantify hematopoietic progenitor frequency and functionality [75].
In Vivo Engraftment Monitoring
- Neutrophil Recovery: Daily complete blood counts following infusion until achievement of ANC ≥0.5 × 10⁹/L for three consecutive days [71].
- Platelet Recovery: Daily platelet counts until achievement of ≥20 × 10⁹/L for three consecutive days without transfusion [71].
- Chimerism Analysis: STR-PCR or FISH analysis to confirm donor-derived hematopoiesis [72].
The following workflow diagram illustrates the experimental design for comparative cryopreservation studies:
Figure 1: Experimental Workflow for Cryopreservation Comparison Studies
Factors Influencing Engraftment Success Beyond Freezing Methodology
While cryopreservation method represents one variable in engraftment success, numerous additional factors significantly impact recovery kinetics:
Table 3: Multifactorial Influences on Engraftment Kinetics
| Factor Category | Specific Variables | Impact on Engraftment |
|---|---|---|
| Product Characteristics | CD34+ cell dose, TNC count, granulocyte content, product volume | Higher CD34+ cell dose (>2.5×10⁶/kg) correlates with faster engraftment; higher granulocyte content associates with reduced post-thaw viability [75] |
| Patient Factors | Diagnosis, disease status, prior therapy, body weight | More extensive prior therapy associates with delayed engraftment; higher body weight requires higher cell doses [75] |
| Transplant Protocol | Conditioning regimen intensity, GvHD prophylaxis, HLA matching | Myeloablative conditioning delays engraftment versus reduced-intensity regimens [71] |
| Post-Thaw Handling | Thawing rate, DMSO removal, infusion timing | Rapid thawing (45°C/min) critical for optimal recovery; delayed infusion reduces viability [7] |
Understanding these multifactorial influences is essential when designing cryopreservation studies and interpreting engraftment results. The following decision framework provides guidance for selecting appropriate cryopreservation methods based on research objectives:
Figure 2: Cryopreservation Method Decision Framework
The quantitative metrics of neutrophil and platelet engraftment times provide critical insights into the functional efficacy of cryopreserved hematopoietic progenitor cells. Current evidence demonstrates that while controlled-rate freezing remains the gold standard for cGMP manufacturing with superior process control and documentation capabilities, passive freezing produces equivalent engraftment outcomes despite differences in some cellular viability parameters. This finding has significant implications for resource-constrained environments and scaling challenges in cell therapy manufacturing.
Future research directions should focus on optimizing cryopreservation protocols for novel cell therapies, including CAR-T cells, iPSC-derived products, and engineered hematopoietic stem cells. Additionally, further investigation into the impact of long-term cryostorage beyond 15 years remains warranted, as current evidence suggests stable preservation of hematopoietic potential [75]. As the field advances, standardization of cryopreservation methodologies and engraftment assessment protocols will enhance comparability across studies and strengthen the evidence base supporting clinical applications.
For researchers and therapy developers, method selection should be guided by product stage, regulatory requirements, and resource constraints rather than presumed superiority in clinical outcomes, as both controlled-rate and passive freezing can produce excellent engraftment results when appropriately implemented.
The choice between controlled-rate freezing (CRF) and passive freezing (PF) represents a critical decision point in the research and development of biopharmaceuticals and cellular therapies. This decision carries significant implications for project budgets, facility planning, and operational workflows. While CRF offers precise, programmable freezing profiles, it requires substantial capital investment and complex infrastructure. Conversely, PF provides a lower-cost, simpler alternative but may introduce variability concerns. This technical guide examines the fundamental trade-offs between these approaches within the broader context of cryopreservation research, providing researchers and drug development professionals with a comprehensive framework for evaluating their freezing strategy based on scientific, economic, and operational requirements.
Technical Fundamentals and Comparative Analysis
Core Principles of Freezing Methodologies
Controlled-rate freezing employs specialized equipment to precisely regulate the cooling rate of biological samples according to predefined protocols. These systems typically use liquid nitrogen or mechanical refrigeration to maintain specific temperature profiles, often following a standard rate of -1°C/minute from ambient temperature to the desired endpoint [4]. A critical function of CRF systems is managing the latent heat of fusion—the heat released when water transitions from liquid to solid state at approximately -2°C to -5°C [4]. This exothermic event can cause undesirable temperature fluctuations without proper control mechanisms.
Passive freezing relies on placing samples in insulated containers within standard -80°C mechanical freezers, achieving cooling through thermal inertia. The cooling rate is determined by the insulation properties of the container system rather than active control [5]. While simple and cost-effective, this approach provides less consistent cooling rates and limited documentation capabilities compared to CRF systems.
Comparative Performance Analysis
Recent clinical evidence suggests that for some cell types, PF can achieve outcomes comparable to CRF. A 2025 retrospective study of 50 hematopoietic progenitor cell (HPC) products found no significant differences in total nucleated cell recovery, CD34+ cell viability, or engraftment outcomes between CRF and PF methods [12]. Both methods employed identical cryoprotectant solutions (15% DMSO and 9% albumin in Plasmalyte-A), with products cryopreserved within 48 hours of collection.
However, the suitability of each method depends heavily on cell type and application. Larger, more complex cells like mammalian cells often require greater control of cooling rates to minimize detrimental effects of undercooling and manage the heat liberated during phase change [4]. For these sensitive applications, CRF provides distinct advantages through artificial ice nucleation and precise thermal management.
Table 1: Technical Comparison of Freezing Methodologies
| Parameter | Controlled-Rate Freezing | Passive Freezing |
|---|---|---|
| Cooling Rate Control | Precise, programmable (typically -1°C/min) | Variable, dependent on insulation |
| Latent Heat Management | Active compensation through rapid cooling | Uncontrolled, dependent on thermal mass |
| Documentation Capability | Comprehensive thermal profile recording | Limited or none without additional monitoring |
| Typical Cell Viability Recovery | High and consistent for sensitive cell types | Variable; comparable for some hematopoietic cells [12] |
| Ice Nucleation Control | Artificial seeding possible | Spontaneous only |
| Process Validation | Fully validatable | Challenging to validate consistently |
Economic Analysis
Capital Investment and Operational Costs
The economic implications of freezing technology selection extend beyond initial purchase prices to encompass substantial operational expenditures. CRF systems represent a significant capital investment, with sophisticated units costing approximately $15,000-$20,000 [76]. These systems may also require supporting infrastructure such as liquid nitrogen supply lines, with installation projects involving assessment of flow requirements, inlet pressure specifications, and vacuum-insulated transfer line placement [33].
PF approaches have significantly lower capital requirements, utilizing existing -80°C mechanical freezers with simple insulation devices. The "box-in-box" PF system described in literature provides a low-cost alternative using aluminum enclosures with polyethylene foam insulation to achieve desired cooling rates [5].
Ultra-low temperature (ULT) freezers for storage represent one of the most energy-intensive equipment categories in research facilities, with consumption patterns varying dramatically based on efficiency and operating temperature:
Table 2: Operational Cost Analysis for ULT Freezers
| Freezer Type | Daily Energy Consumption | Annual Energy Cost ($0.12/kWh) | Annual Energy Cost ($0.20/kWh) |
|---|---|---|---|
| Standard Efficiency (-80°C) | 20 kWh/day | $876 | $1,460 |
| High Efficiency (-80°C) | 10 kWh/day | $438 | $730 |
| Standard Efficiency (-70°C) | 14 kWh/day | $613 | $1,022 |
Energy consumption data from [76] indicates that operating ULT freezers at -70°C instead of -80°C can reduce energy consumption by up to 30%, saving approximately 2,190 kWh annually per freezer. Many institutions including AstraZeneca, Genentech, and Harvard have adopted -70°C standards for appropriate applications [76].
Total Cost of Ownership Analysis
When evaluating freezing technologies, researchers should consider the total cost of ownership (TCO) over a typical 10-year equipment lifecycle:
Table 3: 10-Year Total Cost of Ownership Projection
| Cost Component | Controlled-Rate Freezer | Passive Freezing Setup |
|---|---|---|
| Capital Equipment | $15,000-$20,000 | $500-$2,000 (insulation systems) |
| Annual Energy Cost | Varies by ULT freezer efficiency | Varies by ULT freezer efficiency |
| Liquid Nitrogen Consumption | $500-$2,000/year (cryogenic systems) | $0 |
| Maintenance & Service | $1,000-$3,000/year | Minimal |
| Validation & Documentation | Higher (complex protocols) | Lower |
| Estimated 10-Year TCO | $35,000-$70,000+ | $10,000-$25,000 |
Beyond direct costs, operational factors significantly impact productivity. Working in frozen warehouse environments (-18°C or below) can reduce labor productivity by 30-40% compared to chilled environments, with mandatory warming breaks further impacting efficiency [77]. These factors highlight the importance of workflow integration in freezing technology selection.
Infrastructure Requirements
Facility and Utility Considerations
CRF systems demand substantial infrastructure support, particularly for cryogenic units requiring liquid nitrogen (LN2) supply. Installation typically involves a network of vacuum-insulated transfer lines connecting storage dewars to the freezer, with possible need for quality-enhancing components like automatic gas vents, degassers, or subcoolers [33]. Mechanical CRF systems still require significant electrical capacity and generate substantial heat, increasing HVAC loads.
PF approaches primarily rely on standard -80°C mechanical freezers, which consume 200-350 kWh/m³/year [77]. These units generate significant heat that must be removed by facility HVAC systems, creating a multiplier effect on energy costs—a ULT freezer outputting 14,500 BTUs requires continuous heat removal by air conditioning systems [76].
Storage Infrastructure Complexities
Frozen storage facilities demand robust construction with insulation thickness of 150-200mm compared to 80-100mm for chilled storage [77]. Frozen warehouses also require sophisticated frost protection systems including sub-floor heating to prevent structural damage from frost heave. The temperature differentials in frozen storage create powerful moisture drives, making vapor barriers critical to prevent ice accumulation within insulation [77].
Long-term storage stability requires temperatures below -130°C for sensitive cells like mammalian tissue cultures, hybridomas, and stem cells [4]. Liquid nitrogen freezers providing vapor phase storage at -150°C or below offer optimal stability while avoiding the risks associated with liquid phase storage, such as LN2 entrapment in vials [4].
Experimental Design and Protocol Implementation
Protocol for Comparative Freezing Method Evaluation
Objective: To evaluate the impact of controlled-rate freezing versus passive freezing on specific cell types or biological materials.
Materials:
- Cryoprotectant solution (e.g., 15% DMSO, 9% albumin in Plasmalyte-A) [12]
- Test samples (cell suspensions, tissue fragments, or protein solutions)
- Controlled-rate freezer (e.g., CryoMed CRF system)
- Passive freezing device (e.g., "box-in-box" system or Mr. Frosty)
- -80°C mechanical freezer
- Temperature monitoring system (e.g., thermocouples or FBG sensors)
Methodology:
- Prepare identical sample aliquots in cryovials or bags (n≥5 per condition)
- Add cryoprotectant solution following standardized protocols
- For CRF group: Program freeze rate of -1°C/min from room temperature to -40°C, then -5°C/min to -100°C [12] [5]
- For PF group: Place samples in passive freezing device and transfer to -80°C freezer
- Monitor and document temperature profiles using calibrated sensors
- Transfer all samples to long-term storage in vapor phase liquid nitrogen
- After 1-7 days, thaw samples rapidly in 37°C water bath with gentle agitation
- Assess outcomes via viability assays, recovery counts, and functional tests
Outcome Measures:
- Post-thaw viability (e.g., trypan blue exclusion, 7-AAD)
- Functional recovery (e.g., colony-forming assays for HPCs [12])
- Phenotypic markers (e.g., CD34+ cell counts for hematopoietic cells [12])
- Process documentation (cooling rate verification, thermal profile analysis)
Research Reagent Solutions and Materials
Table 4: Essential Materials for Freezing Protocol Implementation
| Item | Function | Application Notes |
|---|---|---|
| Dimethyl Sulfoxide (DMSO) | Cryoprotectant | Typically used at 5-15% concentration; prevents ice crystal formation [12] |
| Hydroxyethyl Starch | Cryoprotectant | Alternative or supplement to DMSO; reduces toxicity concerns |
| Programmable CRF | Controlled freezing | Enables precise cooling rates (e.g., -1°C/min); manages latent heat [4] |
| Box-in-Box Device | Passive freezing | Aluminum/plastic enclosure with insulation; provides ~-1°C to -3.5°C/min [5] |
| Mr. Frosty | Passive freezing | Commercial isopropanol-based system; provides ~-1°C/min cooling |
| Fibre-Bragg-Grating Sensors | Temperature monitoring | Enables precise freezing time detection; resists freezing-point depression issues [78] |
| Liquid Nitrogen Storage Tank | Long-term preservation | Maintains temperatures below -130°C; vapor phase reduces contamination risk [4] |
Decision Framework and Implementation Strategy
Technology Selection Algorithm
The following workflow diagram outlines a systematic approach for selecting the appropriate freezing methodology based on research requirements, regulatory constraints, and resource considerations:
Process Optimization and Validation
Implementation of either freezing methodology requires careful process characterization and validation. For CRF systems, this involves qualifying equipment performance across the intended operating range, establishing temperature mapping, and validating alarm systems. For PF approaches, validation should focus on consistency of cooling rates across multiple runs and positions within the freezing device.
Researchers should employ advanced temperature monitoring techniques such as derivative-based temperature analysis to accurately detect freezing endpoints independent of freezing-point depression effects in concentrated solutions [78]. The Plank equation can provide theoretical freezing time predictions, though it often requires empirical correction factors for pharmaceutical applications [78].
Freeze-thaw cycle stress studies should evaluate both low (~1/3×) and high (~3×) protein concentration levels to identify optimal cryoprotectant formulations [79]. For cellular therapeutics, functional assessments post-thaw are essential, as viability markers alone may not reflect therapeutic potential [62].
The decision between controlled-rate and passive freezing technologies involves balancing scientific requirements with practical constraints. CRF offers precision, documentation, and process control essential for regulated environments and sensitive biological materials. PF provides a cost-effective, operationally simpler alternative suitable for robust cell types and research applications with limited resources. Emerging evidence that PF achieves comparable results to CRF for some cell types [12] suggests that blanket recommendations are inappropriate—the optimal approach depends on specific research contexts, cell types, and quality requirements. By applying the structured evaluation framework presented in this guide, researchers can make informed decisions that align freezing technology selection with their scientific objectives and operational realities.
Within the rapidly advancing field of cell and gene therapy (CGT), cryopreservation has evolved from a simple storage step to a critical process parameter with significant implications for product quality, regulatory strategy, and commercial viability. The fundamental choice between controlled-rate freezing (CRF) and passive freezing (PF) represents a strategic decision that increasingly follows a distinct pattern across the therapy development lifecycle. Current industry data reveals a clear trend: while 87% of survey respondents use controlled-rate freezing for their cell-based products, the remaining 13% utilizing passive freezing have products exclusively in the earlier stages of clinical development (up to phase II) [7]. This distribution underscores a growing consensus on aligning freezing methodologies with development phase requirements, resource constraints, and regulatory considerations.
This technical guide examines the scientific, operational, and regulatory foundations driving this industry trend, providing researchers, scientists, and drug development professionals with evidence-based frameworks for cryopreservation strategy. By synthesizing recent comparative studies, industry survey data, and technological advancements, we aim to establish a comprehensive foundation for decision-making in cryopreservation protocol selection.
Fundamental Principles: CRF vs. Passive Freezing
Controlled-Rate Freezing (CRF)
Controlled-rate freezing employs specialized programmable equipment to decrease product temperature according to predefined, optimized profiles. The process typically follows a precise sequence: cooling at approximately -1°C/min until freezing initiation, countering the exothermic latent heat of fusion release during phase change, then resuming controlled cooling until reaching the target temperature (e.g., -80°C to -150°C) [12]. This method enables exact control over critical process parameters including:
- Cooling rate before and after nucleation
- Nucleation temperature
- Final temperature before transfer to long-term storage
Passive Freezing (PF)
Passive freezing, also called uncontrolled-rate or non-controlled rate freezing, utilizes insulated containers placed in standard -80°C mechanical freezers to achieve relatively controlled cooling through thermal mass principles rather than active programming. Advanced passive freezing devices incorporate proprietary materials like closed-cell polyethylene foam and solid alloy thermal cores to achieve consistent cooling rates approximating the -1°C/minute profile optimal for many cell types [80]. Unlike CRF, however, nucleation in PF systems remains uncontrolled and cooling rates are not easily modified for different product requirements.
Quantitative Comparison: Performance and Outcomes
Cell Viability and Recovery Metrics
Recent comparative studies provide compelling quantitative data on the performance of both freezing methodologies across critical quality attributes. A 2025 retrospective analysis of 50 hematopoietic progenitor cell (HPC) products offers particularly relevant insights:
Table 1: Comparative Post-Thaw Cell Viability and Engraftment Outcomes
| Parameter | Controlled-Rate Freezing (CRF) | Passive Freezing (PF) | P-value |
|---|---|---|---|
| TNC Viability Post-Thaw | 74.2% ± 9.9% | 68.4% ± 9.4% | 0.038 |
| CD34+ Viability Post-Thaw | 77.1% ± 11.3% | 78.5% ± 8.0% | 0.664 |
| Days to Neutrophil Engraftment | 12.4 ± 5.0 | 15.0 ± 7.7 | 0.324 |
| Days to Platelet Engraftment | 21.5 ± 9.1 | 22.3 ± 22.8 | 0.915 |
Data source: Cytotherapy, 2025;27(9):1145-1149 [12] [6]
While the study found a statistically significant advantage for CRF in total nucleated cell (TNC) viability, it demonstrated no significant difference in the more clinically relevant metrics of CD34+ cell viability and engraftment times for both neutrophils and platelets [6]. This finding is particularly significant for early-stage development where demonstrating product viability and basic efficacy is paramount.
Operational and Economic Considerations
Beyond biological outcomes, practical considerations significantly influence methodology selection across development phases:
Table 2: Operational and Economic Comparison
| Consideration | Controlled-Rate Freezing (CRF) | Passive Freezing (PF) |
|---|---|---|
| Infrastructure Cost | High ($\$ \$) | Low ($) |
| Consumables Requirement | High | Low |
| Specialized Expertise Needed | Yes | Minimal |
| Process Development Resources | Significant | Minimal |
| Batch Scale-up Capability | Bottleneck potential | Easily scalable |
| Footprint | Large | Small |
| Maintenance Requirements | Complex, regular | Minimal to none |
| Documentation Capability | Extensive automated | Manual |
Data source: ISCT Survey, 2025 [7]
The operational profile of each method reveals why PF dominates early-stage development where resources are constrained and process flexibility is valued, while CRF's documentation and control advantages become essential later in development.
Industry Adoption Trends: Survey Data Analysis
The International Society for Cell & Gene Therapy (ISCT) 2025 survey on cryopreservation practices provides robust insights into current industry standards. The survey of professionals across the CGT sector revealed that:
- 87% of respondents use controlled-rate freezing in their current practice [7]
- 13% use passive freezing, with 86% of these having products exclusively in Phase I/II clinical development [7]
- 60% of CRF users employ default freezing profiles without customization [7]
- 22% identified "Ability to process at a large scale" as the biggest hurdle for cryopreservation [7]
This distribution pattern demonstrates a strategic approach where the simpler, more cost-effective PF method serves adequately during early development and proof-of-concept stages, while the greater control and documentation capabilities of CRF become necessary as products approach regulatory submission and commercialization.
Figure 1: Decision Framework for Cryopreservation Method Selection Across Development Stages
Experimental Evidence: Key Studies and Protocols
Hematopoietic Progenitor Cell Study
Experimental Protocol: A 2025 retrospective study directly compared CRF and PF methods for hematopoietic progenitor cell cryopreservation [12] [6].
- Cell Types: Apheresis-derived and marrow-derived HPCs
- Cryoprotectant: Standardized solution containing 15% DMSO and 9% albumin in Plasmalyte-A
- Cell Concentration: Optimized to 600-800 × 10⁶ TNC/mL
- CRF Protocol: Controlled-rate freezer with precise -1°C/minute cooling rate
- PF Protocol: Passive freezing in -80°C mechanical freezer using insulated containers
- Assessment Metrics: Post-thaw TNC viability, CD34+ viability, neutrophil engraftment, platelet engraftment
- Storage: All products ultimately stored in liquid nitrogen vapor phase below -150°C
Key Findings: The research concluded that while TNC viability showed a statistically significant advantage for CRF (74.2% ± 9.9% vs. 68.4% ± 9.4%, p=0.038), the more clinically relevant metrics of CD34+ cell viability and engraftment times showed no significant differences between methods [6]. This demonstrates that PF can deliver equivalent biological performance for critical therapeutic outcomes.
T-cell Therapy Clinical Trial Protocol
Experimental Protocol: A Phase IIb clinical trial for Ovasave (an autologous T-cell therapy for Crohn's disease) implemented a rigorous comparison between freezing methods [80].
- Cell Types: Peripheral blood mononuclear cells (PBMCs) and antigen-specific regulatory T-cells (Ova-Tregs)
- Freezing Methods Comparison: Programmable freezer vs. CoolCell PF container
- Cooling Rate: Both methods targeted -1°C/minute
- Viability Assessment: Pre-freezing and post-thaw viability via propidium iodide staining and flow cytometry
- Cleanroom Compliance: EU GMP cleanroom guidelines with particle emission and microbial contamination testing
Key Findings: The study demonstrated equivalent post-thaw viability between CRF (91.7% ± 4.0%) and PF (91.7% ± 3.7%) for the therapeutic T-cell product [80]. The passive freezing container also met stringent GMP cleanroom requirements after appropriate decontamination procedures, validating its suitability for clinical manufacturing environments.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Cryopreservation Research
| Material/Equipment | Function | Application Notes |
|---|---|---|
| Controlled-Rate Freezer | Precisely controls cooling rate according to programmed parameters | Essential for late-stage development; enables process parameter control |
| Passive Freezing Container | Provides consistent cooling rate via thermal mass principles in -80°C freezer | Cost-effective for early development; validated for GMP use |
| DMSO Cryoprotectant | Prevents intracellular ice crystal formation; typically used at 10-15% concentration | Requires toxicity management; critical for cell viability |
| Cryogenic Vials | Contain product during freezing and storage; must withstand extreme temperatures | Closed-system vials recommended for GMP applications |
| Liquid Nitrogen Storage | Long-term storage below -150°C | Final storage for both CRF and PF products |
| Viability Assays | Assess post-thaw cell recovery and function (e.g., flow cytometry with propidium iodide) | Critical quality attribute measurement |
| Temperature Monitoring | Documents temperature profile during freezing process | Enhanced documentation for CRF; possible with advanced PF systems |
Strategic Implementation Framework
Decision Matrix for Method Selection
The choice between CRF and PF depends on multiple factors across technical, regulatory, and operational domains:
Figure 2: Key Decision Factors Influencing Cryopreservation Method Selection
Technology Transfer Considerations
Transitioning from PF to CRF represents a significant manufacturing process change that requires comprehensive comparability studies. The ISCT survey notes that adopting controlled-rate freezing early in clinical development can avoid the challenging effort of making a significant manufacturing change and establishing comparability subsequently [7]. When such transition is necessary, a phased approach is recommended:
- Analytical Comparability: Extensive side-by-side testing of critical quality attributes
- Process Validation: Demonstrating consistent performance with the new method
- Regulatory Documentation: Comprehensive documentation of the change and its justification
- Quality System Updates: Revision of standard operating procedures and specifications
The prevailing industry trend of utilizing passive freezing for early-stage therapies and controlled-rate freezing for late-stage and commercial products represents a rational alignment of methodology with development priorities and constraints. Robust scientific evidence demonstrates that passive freezing can deliver equivalent engraftment outcomes for critical cell therapies including hematopoietic progenitor cells and T-cell products [12] [80], making it adequate for proof-of-concept establishment in early development.
As therapies advance toward commercialization, the enhanced process control, documentation capabilities, and regulatory alignment of controlled-rate freezing justify its increased cost and operational complexity [7]. This strategic approach enables efficient resource allocation while maintaining focus on product quality throughout the development lifecycle, ultimately supporting the advancement of safe and effective cell and gene therapies for patients in need.
In the development of advanced cell and gene therapies, the cryopreservation process is not merely a storage convenience but a critical determinant of product quality, efficacy, and ultimately, patient outcomes. Within the context of ongoing research into controlled-rate freezing (CRF) versus passive freezing (PF), data logging and freeze curve analysis emerge as fundamental technologies for process validation, optimization, and regulatory compliance. Controlled-rate freezing enables precise, programmable temperature reduction, while passive freezing involves placing samples in a pre-cooled environment (e.g., -80°C) without active control over the cooling rate [6] [12]. Although recent studies in hematopoietic progenitor cell (HPC) cryopreservation demonstrate comparable engraftment outcomes between these methods, this equivalence does not diminish the value of data-rich processes [6] [12]. Instead, it underscores the necessity of rigorous process understanding and control, whether through the inherent programmability of CRF or the validated characterization of a PF protocol. This whitepaper details how the strategic implementation of data logging and freeze curve analysis provides the evidence-based foundation required to future-proof bioprocesses against increasing regulatory scrutiny and process scale-up challenges.
The Science of Freezing: From Fundamentals to Ice Crystal Morphology
The Physics of Phase Transition
The freezing of aqueous solutions, particularly those containing biologics, is a complex thermodynamic process. As a liquid is cooled below its equilibrium freezing point, it enters a metastable supercooled state before ice nucleation begins stochastically [81]. Once nucleation occurs, the system releases the latent heat of fusion, causing a transient temperature rise until the equilibrium freezing point is reached and ice crystallization proceeds [4] [81]. The rate at which heat is removed during this phase transition directly determines ice crystal size and distribution. Rapid cooling promotes the formation of numerous small intracellular ice crystals, while slower cooling rates allow water to exit the cell before freezing, leading to extracellular ice formation and potential solution effects including osmotic stress, pH shifts, and cryoconcentration of solutes and proteins [4] [81] [10]. For large, sensitive mammalian cells, a uniform cooling rate of approximately 1°C per minute is often effective for a wide variety of cells, as it balances these competing damaging mechanisms [4].
Critical Parameters Impacting Cell Viability and Product Quality
- Ice Crystal Formation: The size, location, and morphology of ice crystals directly cause mechanical damage to cell membranes and subcellular structures [81].
- Cryoconcentration: As ice forms, solutes (salts, cryoprotectants, proteins) are excluded from the crystal lattice, becoming concentrated in the remaining liquid phase. This can lead to protein aggregation, denaturation, and extreme pH shifts in the unfrozen fraction [10].
- Cryoprotectant Toxicity: While cryoprotective agents (CPAs) like dimethyl sulfoxide (DMSO) are essential to mitigate ice damage, they can exhibit toxicity that is dependent on concentration, temperature, and exposure time [81].
Freeze Curve Analysis: The Decoded Process Signature
Deconstructing the Freezing Curve
A freezing curve is a time-temperature trace that serves as a process signature, providing a complete narrative of the thermal events during cryopreservation. Advanced controlled-rate freezers monitor and log multiple curves simultaneously to form a comprehensive dataset [82].
Table 1: Key Components of a Freezing Curve
| Curve Type | Monitored Variable | Analytical Value | Data Source |
|---|---|---|---|
| Theoretical Curve | Pre-programmed temperature setpoints | The target path representing the ideal cooling profile | Programmable Freezer [82] |
| Chamber Probe Curve | Air temperature within the freezer chamber | Represents the environmental conditions applied to the entire sample inventory | Chamber Probe [82] |
| Sample Probe Curve | Temperature within a representative sample | Reveals the sample's actual response to the chamber conditions, including phase changes | Sample Probe [82] |
Identifying Critical Transition Points
The sample probe curve is paramount for process understanding, as it reveals four critical stages of the freezing process [82]:
- Liquid Cooling: The sample temperature decreases sensibly from the starting point to its nucleation point.
- Phase Transition (Latent Heat Release): The temperature plateaus or rises exothermically as ice nucleation occurs and latent heat is released. The duration and shape of this plateau are critical indicators of the freezing dynamics.
- Crystalline Cooling: Once the phase change is complete, the frozen sample's temperature begins to decrease sensibly again.
- Stable State: The sample reaches its target final temperature (e.g., below -40°C) and is ready for transfer to long-term storage.
Figure 1: Stages of a Sample Freezing Curve. The diagram maps the sequential thermal stages a sample undergoes during a controlled freeze, highlighting key transition points like nucleation and the end of the phase change.
A Protocol for Freeze Curve Analysis and Process Validation
Experimental Setup and Data Acquisition
Objective: To characterize and validate a cryopreservation protocol for a specific cell type (e.g., Induced Pluripotent Stem Cells - iPSCs) using data logging and freeze curve analysis.
Materials and Reagents: Table 2: Essential Research Reagent Solutions for Cryopreservation Studies
| Item | Function / Rationale | Example |
|---|---|---|
| Programmable CRF | Provides active cooling, data logging of chamber and sample temperatures, and ensures process repeatability. | Digitcool, CryoMed [82] [4] |
| Passive Freezing Device | A -80°C mechanical freezer used for uncontrolled-rate freezing protocols. | Standard -80°C Freezer [6] [12] |
| Cryoprotectant | Permeating agent that reduces intracellular ice formation; requires optimization for concentration and exposure time. | DMSO, Glycerol [81] [12] |
| Cell Viability Assay | Quantifies post-thaw cell survival and functionality. | Flow cytometry (e.g., 7-AAD), CD34+ viability, colony-forming assays [6] [12] |
| Formulation Buffer | Provides the base solution matrix; its composition can affect ice formation and solute behavior. | Plasmalyte-A with albumin [12] |
Methodology:
- Instrument Calibration: Calibrate all temperature probes (chamber and sample) of the controlled-rate freezer against a NIST-traceable standard prior to experimentation.
- Sample Preparation: Prepare identical aliquots of the cell product (i.e., iPSCs). Add cryoprotectant solution (e.g., containing 10% DMSO) in a controlled manner. Load samples into cryocontainers, ensuring one representative sample is equipped with an external temperature probe for the CRF.
- Process Execution:
- CRF Arm: Place samples in the CRF. Initiate a programmed freeze profile (e.g., -1°C/min to -40°C, then -5°C/min to -100°C, followed by transfer to liquid nitrogen vapor phase storage) [4].
- PF Arm: Place samples within an insulated container (e.g., wrapped in absorbent pads or placed in a polystyrene box) inside a -80°C mechanical freezer to approximate a cooling rate of 1-2°C/min, before transfer to long-term storage [12].
- Data Logging: For the CRF run, configure the software to record the theoretical, chamber, and sample probe curves at a high frequency (e.g., every 5-10 seconds) [82].
Data Analysis and Correlation with Quality Attributes
- Curve Overlay and Deviation Analysis: Export data and overlay the theoretical, chamber, and sample curves. Calculate the area between the theoretical and sample curves as a quantitative measure of process deviation.
- Parameter Extraction: From the sample curve, extract key parameters:
- Supercooling Degree: The difference between the equilibrium freezing point and the actual nucleation temperature.
- Phase Transition Time: The duration of the latent heat release plateau.
- Effective Cooling Rate: The calculated cooling rate during the liquid and crystalline cooling stages.
- Post-Thaw Quality Assessment: After a standardized storage period, thaw samples using a controlled method. Assess critical quality attributes (CQAs) including:
- Cell Viability: Via flow cytometry (e.g., 7-AAD) and specific functional markers (e.g., CD34+ viability) [6] [12].
- Functionality: Through colony-forming unit assays or other potency assays relevant to the cell type.
- Recovery: Calculate the percentage of total nucleated cells (TNC) and viable CD34+ cells recovered post-thaw [6].
- Statistical Correlation: Use multivariate analysis to correlate extracted freezing parameters (supercooling degree, phase transition time) with the post-thaw CQAs to establish a design space for the process.
Figure 2: Freeze Curve Validation Workflow. This workflow outlines the systematic approach for using experimental data to correlate freezing process parameters with final product quality.
The true power of freeze curve analysis is realized when quantitative data is linked to clinical or product outcomes. A retrospective study of 50 hematopoietic progenitor cell (HPC) products provides a powerful example of this data-driven approach.
Table 3: Comparative Analysis of Controlled-Rate vs. Passive Freezing for HPCs [6] [12]
| Parameter | Controlled-Rate Freezing (CRF) | Passive Freezing (PF) | Statistical Significance (P-value) |
|---|---|---|---|
| Total Nucleated Cell (TNC) Viability Post-Thaw | 74.2% ± 9.9% | 68.4% ± 9.4% | P = 0.038 |
| CD34+ Cell Viability Post-Thaw | 77.1% ± 11.3% | 78.5% ± 8.0% | P = 0.664 (Not Significant) |
| Days to Neutrophil Engraftment | 12.4 ± 5.0 | 15.0 ± 7.7 | P = 0.324 (Not Significant) |
| Days to Platelet Engraftment | 21.5 ± 9.1 | 22.3 ± 22.8 | P = 0.915 (Not Significant) |
Analysis of Outcomes:
- While a statistically significant difference was observed in TNC viability, the more critical potency measures—CD34+ cell viability and, most importantly, time to engraftment—showed no significant difference between the two methods [6] [12].
- This demonstrates that for HPCs, PF is an acceptable alternative to CRF, provided the process is well-characterized and controlled. The data provides the evidence to support this protocol decision.
- For other, more sensitive cell types like iPSCs, the analysis might reveal a stronger correlation between specific curve features (e.g., minimal supercooling) and high viability, thereby justifying the use of CRF.
Future-Proofing Through Data: Compliance and Scalability
Meeting Regulatory Expectations
Regulatory agencies increasingly advocate for a Quality by Design (QbD) framework. A database of fully annotated freeze curves, linked to product CQAs, builds a robust Process Performance Qualification (PPQ) package. This data provides objective evidence that the process is operating within a validated state of control, facilitating regulatory submissions and approvals [4] [10]. In the event of a process deviation or a product failure investigation, historical freeze curve data can be invaluable for root cause analysis, helping to distinguish between process-related and product-related failures.
Enabling Process Scalability and Technology Transfer
The transition from research-scale to clinical and commercial-scale manufacturing presents significant cryopreservation challenges. Heat and mass transfer dynamics change with volume, impacting the effective cooling rate and the degree of cryoconcentration in large volumes like bulk drug substance [10]. Using freeze curve analysis at small scale (e.g., in a cryowedge that mimics the geometry of a large vessel) allows for the predictive modeling of freezing behavior at full scale. This data-driven approach de-risks scale-up, ensures consistency across manufacturing sites, and provides a scientific rationale for any necessary protocol adjustments during technology transfer.
The debate between controlled-rate and passive freezing is not about declaring a universal winner, but about making a scientifically defensible and economically sound choice for a specific product and process. Freeze curve analysis is the critical tool that transforms cryopreservation from an art into a science. By meticulously logging and interpreting this data, researchers and drug developers can build an unassailable knowledge base that validates their current process, guides future optimization, and provides the agility to adapt to new regulatory and scaling challenges. In an era of increasingly personalized and advanced medicines, a future-proof process is, by definition, a data-rich process.
Conclusion
The choice between controlled-rate and passive freezing is not a simple declaration of a superior method, but a strategic decision based on clinical stage, cell type, and operational resources. Recent 2025 evidence confirms that passive freezing can achieve equivalent engraftment outcomes for certain cell types like HPCs, offering a simple, cost-effective solution for early-stage research and autologous transplants. However, controlled-rate freezing provides unparalleled process control, critical for sensitive cells (iPSCs, CAR-T) and late-stage commercial therapies where consistency and rigorous documentation are paramount. The future of cryopreservation lies in smarter, more integrated technologies—leveraging freeze curve data for real-time process control, developing advanced rewarming techniques to complement freezing, and creating scalable solutions to support the booming cell and gene therapy market. Researchers are advised to adopt controlled-rate freezing early for pivotal clinical programs to avoid costly process changes later, while considering passive freezing as a validated, efficient alternative for appropriate applications.
References
- 1. Protocols and Best Practices for Freezing Cells [https://www.stemcell.com/cryopreservation-basics-protocols-and-best-practices-for-freezing-cells]
- 2. Cell Culture Fundamentals: Cryopreservation and Storage ... [https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/cell-culture-and-cell-culture-analysis/mammalian-cell-culture/cryopreservation-storage-cells?srsltid=AfmBOoqNyHG8WBNSOmSzxt-KnrI6hxQju22k-rnfhfRSqcrIChBIQhFj]
- 3. The role of preservation in the variability of regenerative ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7677879/]
- 4. The Importance of Controlling the Rate of Freezing [https://www.thermofisher.com/blog/life-in-the-lab/the-importance-of-controlling-the-rate-of-freezing/]
- 5. Development of a Reliable, Low-cost, Controlled Cooling ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3022343/]
- 6. Passive freezing is equivalent to controlled-rate ... [https://pubmed.ncbi.nlm.nih.gov/40323255/]
- 7. Cryopreservation - Key Survey Findings from the ISCT ... [https://www.isctglobal.org/telegrafthub/blogs/ken-ip1/2025/05/28/cryopreservation-key-survey-findings-from-the-isct]
- 8. Comparison of liquid nitrogen-free slow freezing protocols ... [https://www.sciencedirect.com/science/article/abs/pii/S0011224023003358]
- 9. Evaluating Freeze–Thaw Processes in Biopharmaceutical ... [https://www.bioprocessintl.com/fill-finish/evaluating-freeze-thaw-processes-in-biopharmaceutical-development-small-scale-study-designs]
- 10. Large-Scale Freezing of Biologics: Understanding Protein ... [https://www.biopharminternational.com/view/large-scale-freezing-biologics-understanding-protein-and-solute-concentration-changes-cryovessel-par]
- 11. Cryopreservation Freezing Methods and... | Technology Networks [https://www.technologynetworks.com/cell-science/articles/cryopreservation-freezing-methods-and-equipment-292855]
- 12. Passive freezing is equivalent to controlled-rate ... [https://www.sciencedirect.com/science/article/abs/pii/S1465324925006796]
- 13. Relevance of controlled cooling and freezing phases in T- ... [https://www.sciencedirect.com/science/article/abs/pii/S0011224024000622]
- 14. Innovations in Platelet Cryopreservation: Evaluation of DMSO ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12563202/]
- 15. Cryopreservation: An Overview of Principles and Cell-Specific Considerations - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7995302/]
- 16. Cell Freezing Protocols | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/cell-culture-protocols/freezing-cells.html]
- 17. Reassessing DMSO–lipid interactions: Improved AMBER ... [https://www.sciencedirect.com/science/article/abs/pii/S0005273625000756]
- 18. Mechanisms of freezing damage [https://pubmed.ncbi.nlm.nih.gov/3332492/]
- 19. Natural Cryoprotective and Cytoprotective Agents in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9145333/]
- 20. Cryoprotective Agent - an overview [https://www.sciencedirect.com/topics/nursing-and-health-professions/cryoprotective-agent]
- 21. Strategies in developing dimethyl sulfoxide (DMSO)-free cryopreservation protocols for biotherapeutics - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9579275/]
- 22. Post-thaw dimethyl sulfoxide reduction in autologous ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12398813/]
- 23. Interrupted cooling protocols in cryopreservation: A review ... [https://www.sciencedirect.com/science/article/abs/pii/S0011224025001452]
- 24. A comparison of cryopreservation methods: Slow-cooling vs ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3196719/]
- 25. Localization of freezing injury in articular cartilage [https://pubmed.ncbi.nlm.nih.gov/8156798/]
- 26. Interrupted cooling protocols in cryopreservation: A review ... [https://pubmed.ncbi.nlm.nih.gov/41192344/]
- 27. Thermostability of Biological Systems - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC3137158/]
- 28. Enthalpy of fusion [https://en.wikipedia.org/wiki/Enthalpy_of_fusion]
- 29. 6.1: Freezing of Food [https://eng.libretexts.org/Bookshelves/Biological_Engineering/Introduction_to_Biosystems_Engineering_(Holden_et_al.)/06%3A_Processing_Systems/6.01%3A_Freezing_of_Food]
- 30. Effect of different freezing rates during cryopreservation of rat ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3465236/]
- 31. Innovations Driving Controlled-Rate Freezer Market 2025- ... [https://www.datainsightsmarket.com/reports/controlled-rate-freezer-228891]
- 32. Controlled Rate Freezers [https://www.cryo-products.com/product/crf/]
- 33. Controlled rate freezer: the cryogenic infrastructure [https://demaco-cryogenics.com/blog/controlled-rate-freezer-the-cryogenic-infrastructure/]
- 34. What is a Controlled-Rate Freezer [https://strexcell.com/blog/overview-of-the-controlled-rate-freezer/]
- 35. Controlled Rate Freezer FM-CRF-A100 [https://www.fison.com/controlled-rate-freezer/fm-crf-a100?srsltid=AfmBOoo4zXXETc1uwqbwebNilj9rCSQcprM9fF0aGZsUaQTK7vz2SZG8]
- 36. Impact of Freeze–Thaw Processes on the Quality of Cells [https://www.insights.bio/cell-and-gene-therapy-insights/journal/article/365/Impact-of-FreezeThaw-Processes-on-the-Quality-of-Cells]
- 37. Alcohol-Free Cell Freezing Container 6.2 CAGR Growth Outlook ... [https://www.archivemarketresearch.com/reports/alcohol-free-cell-freezing-container-285863]
- 38. Standardizing Cell-Based Assays By Reducing ... [https://www.bioprocessintl.com/cell-therapies/variables-in-passive-cryopreservative-methods-standardizing-cell-based-assays-by-reducing-cryopreservation-induced-variability]
- 39. Pluripotent Stem Cell (PSC) Protocols [https://www.thermofisher.com/us/en/home/references/protocols/cell-culture/stem-cell-protocols/ipsc-protocols.html]
- 40. Choosing the Best T Cell Activation and Expansion Protocol [https://www.akadeum.com/technology/t-cell-activation-expansion-techniques/?srsltid=AfmBOoq1WClxhMvX8DiHYzjGawv2fHCiRQbduLxFewpbDkcd-L4ZacBd]
- 41. A T-cell-based immunogenicity protocol for evaluating human ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8377590/]
- 42. Current Good Manufacturing Practice (CGMP) Regulations [https://www.fda.gov/drugs/pharmaceutical-quality-resources/current-good-manufacturing-practice-cgmp-regulations]
- 43. Documenting Compliance: Essential GDP Tips for 2025 [https://jafconsulting.com/documenting-compliance-essential-gdp-tips-for-2025/]
- 44. Pharmaceutical Validation Guide: Proper Implementation [https://dicksondata.com/en-gb/guide-to-pharmaceutical-validation-for-effective-implementation?srsltid=AfmBOor9e5Y3CuWhG1n7rhEr2IWRF4wRaZxsqrXFMIF4Ai3gnmCbdXjM]
- 45. Recommended Best Practices for Lyophilization Validation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8575750/]
- 46. Applications of Freeze - Drying in Biopharmaceuticals • Asita Freeze ... [https://asitafreezedryer.com/applications-of-freeze-drying-in-biopharmaceuticals/]
- 47. Quality Systems Approach to Pharmaceutical Current Good ... [https://www.fda.gov/regulatory-information/search-fda-guidance-documents/quality-systems-approach-pharmaceutical-current-good-manufacturing-practice-regulations]
- 48. Winter is coming: the future of cryopreservation - BMC Biology [https://bmcbiol.biomedcentral.com/articles/10.1186/s12915-021-00976-8]
- 49. Comparison of different freezing rates on post-thaw viability ... [https://www.sciencedirect.com/science/article/abs/pii/S0011224025000094]
- 50. Cryopreservation: A Review Article - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9756256/]
- 51. Chemical approaches to cryopreservation [https://www.nature.com/articles/s41570-022-00407-4]
- 52. Post-Thaw Culture and Measurement of Total Cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7362331/]
- 53. Quantitative assessment of the impact of cryopreservation on ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-020-02054-2]
- 54. Freeze–thaw characterization process to minimize ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8166975/]
- 55. Optimization of freezing and thawing protocols for human ... [https://www.sciencedirect.com/science/article/pii/S0011224025000513]
- 56. Optimizing cryopreservation strategies for scalable cell therapies [https://pmc.ncbi.nlm.nih.gov/articles/PMC11659802/]
- 57. The Impact of Precisely Controlled Pre-Freeze Cooling Rates ... [https://sciety.org/articles/activity/10.20944/preprints202511.0836.v1?utm_source=sciety_labs_article_page]
- 58. Innovations in Platelet Cryopreservation [https://pubmed.ncbi.nlm.nih.gov/41155307]
- 59. Overcoming Scale Up Bottlenecks in Cell Culture ... [https://www.worldpharmatoday.com/biopharma/overcoming-scale-up-bottlenecks-in-cell-culture-bioprocesses/]
- 60. The Benefits of an Automated Rate Controlled Freezer for ... [https://cryometrix.com/2025/01/21/the-benefits-of-an-automated-rate-controlled-freezer-for-labs/]
- 61. Overcoming bioprocess bottlenecks in the large-scale ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-020-02109-4]
- 62. Impact of Cryopreservation and Freeze-Thawing on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9045030/]
- 63. A comparative analysis of vitrification and two slow freezing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11950551/]
- 64. Advantages of vitrification preservation in assisted ... [https://clinicalepigeneticsjournal.biomedcentral.com/articles/10.1186/s13148-022-01355-y]
- 65. Comparison of closed vs open vitrification devices [https://blog.vitrolife.com/togetheralltheway/comparison-of-closed-vs-open-vitrification-devices]
- 66. Effects of the cryotop vitrification method for ... [https://www.sciencedirect.com/science/article/pii/S0011224025001294]
- 67. Equivalent outcomes of human oocytes after vitrification or ... [https://pubmed.ncbi.nlm.nih.gov/40234976/]
- 68. Viable and Functional: Long-Term −80 °C Cryopreservation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12525932/]
- 69. Evaluation of the viability and functionality of human ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2025.1627973/full]
- 70. Very cold but still too warm; functional characterization of ... [https://www.sciencedirect.com/science/article/abs/pii/S146532492500903X]
- 71. Improving Engraftment and Immune Reconstitution in ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2014.00068/full]
- 72. The effect of cryopreservation on engraftment kinetics in ... [https://pubmed.ncbi.nlm.nih.gov/37775358/]
- 73. (PDF) Microprocessor- controlled vs. "dump- freezing " platelet and... [https://www.academia.edu/10965951/Microprocessor_controlled_vs_dump_freezing_platelet_and_lymphocyte_cryopreservation_a_quantitative_and_qualitative_comparative_study]
- 74. Current practices in peripheral blood stem cell processing ... [https://www.bloodresearch.or.kr/journal/view.html?doi=10.1007/s44313-025-00090-6]
- 75. Long-term effects of cryopreservation on clinically prepared ... [https://www.sciencedirect.com/science/article/abs/pii/S1465324914004514]
- 76. How Much Does It REALLY Cost to Run Ultra-Low Temp ... [https://www.farrarscientific.com/blog/ultra-low-temperature-freezer-operating-costs]
- 77. Chilled Warehouse vs. Frozen Warehouse: Cost & Risk ... [https://paragonlogistics.com/chilled-warehouse-vs-frozen-warehouse-cost-risk-comparison/]
- 78. Temperature Based Process Characterization of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8062970/]
- 79. Large-Scale Freezing of Biologics [https://www.bioprocessintl.com/fill-finish/large-scale-freezing-of-biologics]
- 80. Innovative Temperature Standardization Technology ... [https://drug-dev.com/standardization-technology-innovative-temperature-standardization-technology-supports-cell-therapy-clinical-trials/]
- 81. Walking on thin ice: controlled freezing & thawing of ... [https://www.insights.bio/cell-and-gene-therapy-insights/journal/article/2677/Walking-on-thin-ice-controlled-freezing-thawing-of-pharmaceutical-active-molec]
- 82. Data in Motion: Freezing Curves in the Digitcool [https://www.imv-technologies.us/academy/data-in-motion-freezing-curves-in-the-digitcool]