Optimizing Leukapheresis for Autologous Cell Therapy: A Guide for Researchers and Developers
This article provides a comprehensive overview of leukapheresis processing, a critical initial step in autologous cell therapy manufacturing.
Optimizing Leukapheresis for Autologous Cell Therapy: A Guide for Researchers and Developers
Abstract
This article provides a comprehensive overview of leukapheresis processing, a critical initial step in autologous cell therapy manufacturing. Tailored for researchers, scientists, and drug development professionals, it covers foundational principles, from market drivers to device technology. It delves into methodological applications for CAR-T cell therapy, including yield optimization and material selection (fresh vs. cryopreserved). The content further addresses key challenges such as high costs and process standardization, offering troubleshooting and optimization strategies. Finally, it presents a comparative analysis of different platforms and materials, validating their impact on final cell product quality and functionality, thereby serving as a holistic resource for advancing therapeutic development.
The Critical Role of Leukapheresis in the Evolving Cell Therapy Landscape
The Chimeric Antigen Receptor T-cell (CAR-T) therapy market is experiencing unprecedented growth, transforming from a novel immunotherapy into a multi-billion dollar sector driven by technological innovation and increasing clinical adoption. This expansion is fundamentally reshaping treatment paradigms for hematological malignancies and creating new opportunities for autologous cell therapy research.
Quantitative Market Projections
Table 1: Global CAR-T Therapy Market Size Projections (2024-2034)
| Year | Market Size (USD Billion) | CAGR Period | Compound Annual Growth Rate (CAGR) | Primary Reference Source |
|---|---|---|---|---|
| 2024 | $5.42 - $5.51 | 2025-2032 | 26.0% | [1] |
| 2032 | $34.42 | 2025-2032 | 26.0% | [1] |
| 2034 | $146.55 | 2025-2034 | 38.83% | [2] |
Table 2: CAR-T Market Analysis by Segment (2024 Dominance)
| Market Segment | Leading Sub-category | Market Share (2024) | Fastest-Growing Sub-category | Projected CAGR |
|---|---|---|---|---|
| Target Antigen | CD19 | 63% | BCMA | 46.15% |
| Indication | Hematologic Malignancies | 94% | Solid Tumors | 45.68% |
| Therapy Type | Autologous | 80% | Allogeneic | 44.35% |
| Manufacturing Method | Centralized | 54% | In Vivo CAR-T | 47.28% |
| End User | Hospitals | 44% | Cancer Treatment Centers | Rapid Growth |
| Region | North America | 49-64% | Asia-Pacific | 40.22% |
Primary Growth Catalysts
The remarkable expansion of the CAR-T therapy market is fueled by several interconnected factors:
Rising Cancer Prevalence and Unmet Need: Increasing incidence of hematologic malignancies, particularly relapsed/refractory cases, creates sustained demand for novel therapies. The high efficacy of CAR-T treatments in patients who have exhausted conventional options drives clinical adoption [3] [4].
Technological Innovation Platforms: Next-generation CAR-T developments include allogeneic (off-the-shelf) products, dual/multitargeted CARs, armored CARs with enhanced persistence, and safety switch-equipped cells [5]. These innovations address limitations of early-generation therapies.
Expanding Clinical Applications: While currently dominant in hematologic malignancies (94% market share), research into solid tumor applications represents the fastest-growing segment with a projected CAGR of 45.68% from 2025-2034 [2]. Additionally, research is expanding into autoimmune diseases including systemic lupus erythematosus, myasthenia gravis, and multiple sclerosis [3].
Manufacturing and Supply Chain Advancements: The adoption of cryopreserved leukapheresis as starting material has revolutionized CAR-T manufacturing by decoupling collection from processing, providing scheduling flexibility and logistical resilience [6] [7].
Experimental Protocols: Leukapheresis Processing for CAR-T Manufacturing
Protocol: Closed-System Automated Leukapheresis Cryopreservation
Objective: To establish a standardized protocol for cryopreserving leukapheresis products that maintains cellular viability, recovery, and CAR-T manufacturing potential [7].
Materials and Reagents:
- Leukapheresis collection kit (closed-system)
- Clinical-grade cryoprotectant (CS10 with 10% DMSO)
- Automated cryopreservation system (e.g., Thermo Profile 4)
- Cryogenic storage bags
- Centrifuge with temperature control
- Cell counting and viability assay (e.g., flow cytometry with 7-AAD)
Procedure:
- Leukapheresis Collection: Collect mononuclear cells via standard leukapheresis procedure. Maintain samples at room temperature and process within 2-8 hours of collection.
Initial Quality Assessment:
- Determine total nucleated cell count and viability
- Measure hematocrit (target: 5-10%)
- Assess CD3+ T-cell percentage via flow cytometry
Centrifugation and Impurity Removal:
- Centrifuge at 400-500 × g for 15 minutes at room temperature
- Carefully remove supernatant containing platelets and plasma
- Reduce residual red blood cells and non-cellular impurities
Cryoprotectant Addition:
- Resuspend cell pellet in CS10 cryoprotectant
- Maintain DMSO concentration ≥7.5% in final cryomedium
- Achieve target cell concentration of 5×10⁷ to 8×10⁷ cells/mL
- Dispense 20 mL aliquots per cryobag (target: ≥1×10⁹ cells/bag)
Controlled-Rate Freezing:
- Initiate freezing within 120 minutes of cryoprotectant addition
- Use programmed freeze cycle: 1°C/minute to -45°C, then 10°C/minute to -100°C
- Transfer to vapor phase liquid nitrogen for long-term storage (-135°C to -150°C)
Quality Control Metrics:
- Post-thaw viability: ≥90%
- CD3+ T-cell recovery: ≥85%
- Sterility testing: Negative for microbial contamination
Protocol: Comparative Analysis of Fresh vs. Cryopreserved Leukapheresis in CAR-T Manufacturing
Objective: To systematically evaluate the impact of cryopreserved versus fresh leukapheresis on CAR-T cell quality attributes and functionality across multiple manufacturing platforms [7].
Experimental Design:
- Sample Groups: Parallel processing of fresh and cryopreserved leukapheresis from the same donor (n≥5)
- CAR-T Platforms: Compare non-viral, lentiviral, and rapid manufacturing platforms
- Timepoints: Assess at initiation, during expansion, and final product
Methodology:
- Sample Preparation:
- Fresh leukapheresis: Process within 24 hours of collection
- Cryopreserved: Thaw rapidly at 37°C, dilute with warm medium, and wash to remove DMSO
CAR-T Manufacturing:
- T-cell activation: Anti-CD3/CD28 beads or alternatives
- Genetic modification: Lentiviral transduction or mRNA electroporation
- Expansion culture: IL-2 supplemented media, 10-14 days
- Final formulation: Cryopreserve or use fresh
Quality Attribute Assessment:
- Viability and Expansion: Daily cell counts, viability staining
- Phenotype Characterization: Flow cytometry for T-cell subsets (CD4/CD8, memory phenotypes)
- CAR Expression: Detection of transgene expression
- Functional Potency:
- Cytotoxicity assays against target cells
- Cytokine secretion (IFN-γ, IL-2) upon antigen exposure
- Exhaustion marker profiling (PD-1, LAG-3, TIM-3)
Statistical Analysis:
- Paired t-tests for fresh vs. cryopreserved comparisons
- ANOVA for multiple platform comparisons
- Significance threshold: p<0.05
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Leukapheresis Processing and CAR-T Manufacturing
| Reagent/Material | Function | Application Notes | Quality Standards |
|---|---|---|---|
| CS10 Cryoprotectant | Prevents ice crystal formation during freezing; contains 10% DMSO | Clinical-grade; optimized for leukapheresis; requires gradual addition to cells | GMP-grade; endotoxin tested |
| Anti-CD3/CD28 Activator | T-cell activation and expansion | Magnetic beads or soluble antibodies; critical for initiation of CAR-T manufacturing | Clinical-grade; purity >95% |
| Lentiviral Vectors | CAR gene delivery | Third-generation safety-modified; various transgene options; titer >1×10⁸ IU/mL | GMP-grade; replication incompetent |
| Cell Culture Media | Supports T-cell growth and expansion | Serum-free formulations; cytokine supplemented (IL-2, IL-7, IL-15) | Xeno-free; endotoxin <1EU/mL |
| Flow Cytometry Antibodies | Cell phenotype and CAR expression analysis | Multi-color panels for T-cell subsets, memory markers, exhaustion markers | Validated for cell therapy applications |
| Cytotoxicity Assay Kits | Functional potency assessment | Measures target cell killing (e.g., luciferase-based, flow cytometry) | High sensitivity; reproducible |
| Cytokine Detection Assays | Functional characterization | ELISA or multiplex platforms for IFN-γ, IL-2, IL-6 measurement | Validated for cell culture supernatants |
Emerging Trends and Future Directions
Next-Generation CAR-T Platforms
The CAR-T therapy landscape is rapidly evolving with several innovative platforms entering clinical development:
Allogeneic (Off-the-Shelf) CAR-Ts: Derived from healthy donors, these products aim to overcome manufacturing delays and variability of autologous approaches. Companies including Allogene Therapeutics and Caribou Biosciences are advancing these platforms, with some showing promising Phase I results [3] [5].
In Vivo CAR-T Delivery: Novel platforms such as Interius BioTherapeutics' INT2104 and Umoja Biopharma's UB-VV111 aim to generate CAR-T cells directly within the patient's body, eliminating complex ex vivo manufacturing [3] [5]. These approaches use viral vectors or nanoparticle delivery systems for in vivo T-cell engineering.
Armored CAR-T Cells: Fourth-generation CARs engineered to express additional supportive molecules (cytokines, co-stimulatory ligands) to enhance persistence and overcome immunosuppressive tumor microenvironments [5].
Dual/Tandem CAR-Ts: Targeting multiple antigens simultaneously to prevent antigen escape, a common resistance mechanism. Miltenyi Biomedicine's zamtocabtagene autoleucel (targeting CD19 and CD20) has received PRIME designation in the EU [3].
Manufacturing Innovation: The Cryopreservation Advantage
The adoption of cryopreserved leukapheresis represents a paradigm shift in CAR-T manufacturing, addressing critical bottlenecks:
Supply Chain Resilience: Decouples leukapheresis from manufacturing scheduling, providing flexibility for patients and protection against shipping delays [6] [7].
Quality Preservation: Recent studies demonstrate that optimized cryopreservation maintains lymphocyte profiles (66.59% in cryopreserved vs. 68.68% in fresh) and T-cell fitness comparable to fresh leukapheresis [7].
Distributed Manufacturing Enablement: Supports the development of regional manufacturing networks by enabling longer storage and transportation of starting materials.
Clinical Outcome Consistency: The Novartis manufacturing process for tisagenlecleucel utilizing cryopreserved leukapheresis has demonstrated high success rates in global clinical trials across 50+ centers in 12 countries [6].
The continued optimization of leukapheresis processing and cryopreservation protocols remains fundamental to scaling CAR-T therapy availability while maintaining product quality and therapeutic efficacy. As the market expands toward solid tumors and autoimmune applications, these manufacturing innovations will be crucial for broader patient access and commercial sustainability.
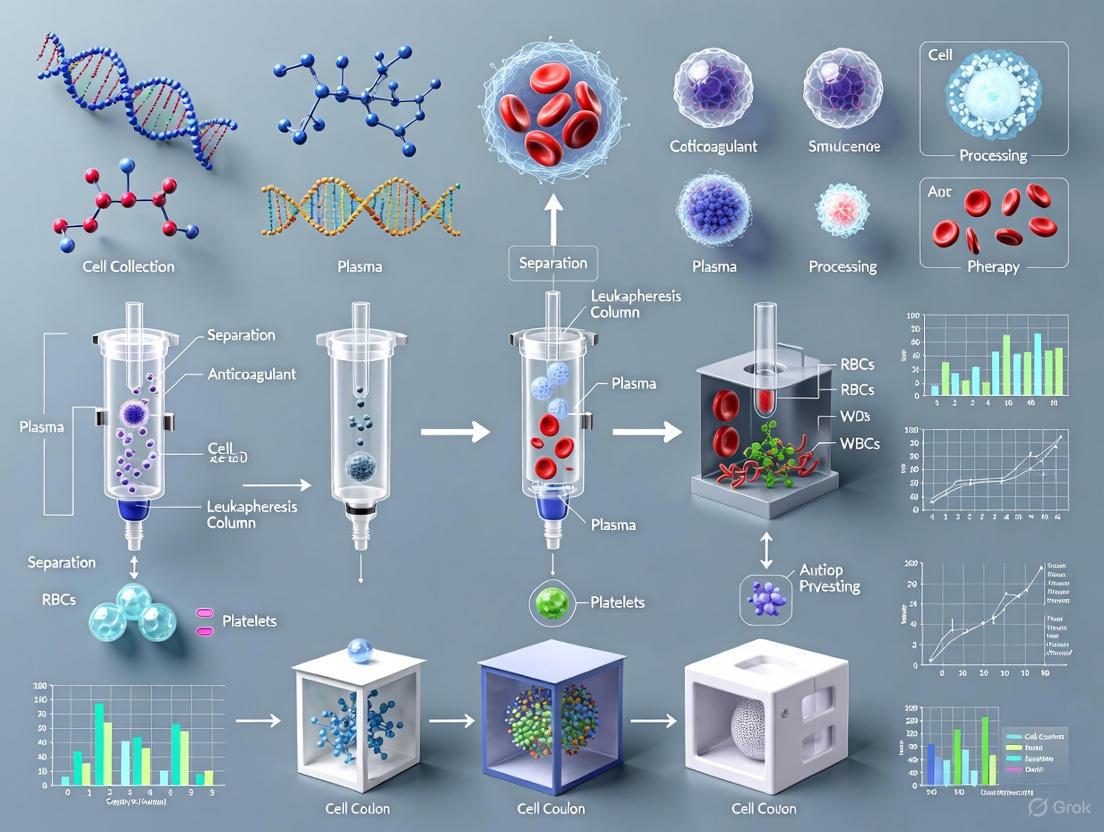
Leukapheresis is a specialized apheresis technique used to separate and collect white blood cells (leukocytes) from a patient's blood, serving as the critical first step in manufacturing autologous cell therapies such as Chimeric Antigen Receptor (CAR) T-cell treatments [8] [9]. In autologous therapy, a patient's own cells are collected, processed, and subsequently reinfused back into the same patient [10]. This approach fundamentally differs from allogeneic therapies that use donor-derived cells. The leukapheresis procedure yields a leukapheresis product (often called a leukopak) rich in mononuclear leukocytes, particularly lymphocytes and monocytes, which can be further processed for therapeutic applications [8]. For researchers and drug development professionals, understanding and optimizing leukapheresis is paramount, as the quality of this starting material directly impacts downstream manufacturing success and ultimately, therapeutic efficacy [11].
Technical Principles and Procedural Framework
The Leukapheresis Procedure
Leukapheresis operates on the principle of centrifugal separation. During the procedure, blood is drawn from the patient and passed through an automated cell separator apparatus [8]. Within this device, blood components are separated into layers based on density [9]. The leukocyte-rich layer is selectively removed, while the remaining blood constituents—including red blood cells, platelets, and plasma—are returned to the patient [8] [9]. This process is typically performed over 2–5 hours, during which 2–4 times the patient's total blood volume may be processed to obtain a sufficient cell yield [12]. The procedure is commonly performed using devices such as the COBE Spectra or Spectra Optia systems (Terumo BCT), which require specialized equipment and trained personnel [8] [12].
Key Output: The Leukopak
The product of this procedure, the leukopak, is a bag containing a highly concentrated sample of leukocytes [9]. A single leukopak can contain up to 20 billion leukocytes with an average volume of approximately 167 mL [8] [9]. Compared to alternative starting materials like buffy coat (a byproduct of standard blood centrifugation), leukopaks offer a substantially higher concentration of leukocytes from a single donor, which helps minimize donor-to-donor variability in research and manufacturing contexts [9]. The following table summarizes typical leukopak characteristics and composition.
Table 1: Typical Composition and Characteristics of a Leukopak
| Parameter | Typical Value/Range | Notes |
|---|---|---|
| Total Volume | 81–345 mL [8] | Varies by apheresis system and protocol |
| Total Nucleated Cells (TNC) | 98 × 108 (range: 9–341 × 108) [12] | Varies with patient/donor health and disease status |
| CD3+ T Cells | 38 × 108 (range: 4–232 × 108) [12] | Primary starting material for CAR-T manufacturing |
| Key Constituents | Lymphocytes, monocytes, granulocytes, platelets, some RBCs [9] | Requires further processing to isolate specific cell types |
Quantitative Framework for Leukapheresis in Research and Therapy
Successful cell therapy manufacturing depends on collecting a sufficient number of viable cells from the leukapheresis starting material. Research indicates that lymphocyte collection is feasible even from heavily pre-treated patients with low peripheral blood counts [12]. The processing volume and target cell counts are critical decision points in protocol design. The following table outlines key performance data and operational parameters for leukapheresis in an autologous therapy context.
Table 2: Leukapheresis Performance Data and Operational Parameters
| Parameter | Findings/Values | Clinical/Research Context |
|---|---|---|
| Minimum Peripheral Lymphocyte Count | As low as 0.18/nL [12] | Sufficient lymphocytes can be harvested even at very low pre-apheresis counts |
| Processed Blood Volume for Low ALC | 12–15 L [12] | Recommended for patients with ALC ≤ 1.0/nL |
| Standard Processed Blood Volume | 2–4 times total blood volume [12] | Standard processing range for most patients |
| Average Procedure Duration | 2–5 hours [12] | Varies based on processing volume and flow rate |
| Anticoagulant | ACD-A [8] [12] | Standard anticoagulant used (blood:anticoagulant ratio of 12–15:1) |
| Cell Viability Post-Cryopreservation | Viability maintained with optimized protocols [13] | Cryopreservation allows for flexibility in manufacturing scheduling |
Detailed Experimental Protocol for Lymphocyte Collection for CAR-T Manufacturing
Pre-Apheresis Considerations
- Patient Eligibility Assessment: Verify that the patient meets inclusion criteria for leukapheresis and subsequent CAR-T therapy. Key considerations include absolute lymphocyte count (ALC), disease status, and performance status [12].
- Timing and Medication Hold: Schedule leukapheresis at a point in the patient's treatment cycle that allows for adequate lymphocyte recovery. Discontinue T-cell impairing medications with appropriate washout periods prior to apheresis [12].
- Venous Access Assessment: Evaluate peripheral venous access. The procedure is typically performed via peripheral veins. If access is difficult, consider ultrasound-guided puncture of peripheral veins [12].
Leukapheresis Procedure
- Equipment Setup: Utilize a Spectra Optia apheresis system or equivalent. Ensure all disposable sets and collection bags are sterile and properly installed [12].
- Anticoagulation Protocol: Use Acid Citrate Dextrose Solution A (ACD-A) as anticoagulant at a blood-to-anticoagulant ratio of 12:1 to 15:1 [12].
- Processing Parameters:
- For patients with ALC > 1.0/nL: Process 2–3 times the patient's total blood volume.
- For patients with ALC ≤ 1.0/nL: Process 12–15 L of blood volume to ensure adequate cell yield [12].
- Monitor the patient for citrate-induced hypocalcemia symptoms (paresthesia, lightheadedness) and manage with calcium supplements as needed [14] [15].
- Product Collection: Collect the leukapheresis product into the approved leukopak collection bag. Gently mix the bag periodically during collection to prevent cell clumping [12].
Post-Apheresis Product Handling and Analysis
- Sample Testing: Aseptically remove samples from the leukopak for the following analyses [12]:
- Total Nucleated Cell (TNC) Count: Using an automated hematology analyzer.
- Cell Composition Analysis: by flow cytometry to determine CD3+ T-cell count and percentage.
- Viability Assessment: Using propidium iodide staining and automated cell counter.
- Sterility Testing: Inoculate BACTEC culture bottles for bacterial and fungal testing.
- Product Shipment or Cryopreservation:
- For fresh shipment: Transport the leukopak at controlled temperature (ambient or 4°C based on manufacturer's specifications) to the manufacturing facility.
- For cryopreservation: Cryopreserve the leukapheresis product according to the CAR-T cell manufacturer's specifications using controlled-rate freezing and transfer to vapor-phase liquid nitrogen for storage [12].
Workflow Integration: From Leukapheresis to CAR-T Cell Product
The journey from leukapheresis to a finished CAR-T cell therapy product is a multi-stage process. The leukapheresis step provides the foundational starting material upon which all subsequent manufacturing operations depend. The following diagram illustrates this integrated workflow, highlighting the role of leukapheresis within the broader context of autologous CAR-T cell therapy.
Diagram 1: Autologous CAR-T Cell Therapy Workflow.
Optimization Strategies for Research and Manufacturing
Addressing Starting Material Variability
The composition of leukapheresis products can vary significantly between individuals, particularly in heavily pre-treated patients [11]. This variability poses a substantial challenge for standardized manufacturing. To address this, research indicates that strategic enrichment of the leukapheresis product can significantly improve downstream manufacturing outcomes.
- Monocyte (CD14+) Depletion: Monocytes can adhere to manufacturing surfaces and activation beads, negatively impacting T cell activation and transduction [11]. Studies show that CD14+ depletion can lead to a 2-fold improvement in T cell activation levels and an increase in CAR expression by up to 64% compared to using unsorted leukapheresis [11].
- B Cell (CD19+) Depletion: In patients with B-cell malignancies, depleting CD19+ B cells from the leukapheresis product minimizes the risk of transducing malignant cells during the CAR vector introduction process [11].
Technological Advancements in Cell Processing
Following leukapheresis, the isolation of specific cell types (e.g., T cells) from the leukopak is a critical step. While traditional methods like Fluorescence-Activated Cell Sorting (FACS) and Magnetic-Activated Cell Sorting (MACS) are widely used, newer technologies such as Buoyancy-Activated Cell Sorting (BACS) employing microbubbles are emerging [9]. These platforms can offer gentler cell handling, potentially improving cell viability and function for subsequent manufacturing steps [9].
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for Leukapheresis and Downstream Processing
| Item | Function/Application | Examples/Notes |
|---|---|---|
| Apheresis System | Automated separation of blood components to collect leukocytes. | Spectra Optia (Terumo BCT), COBE Spectra (Terumo BCT) [8] [12] |
| Anticoagulant | Prevents blood clotting during the apheresis procedure. | Acid Citrate Dextrose Solution A (ACD-A) [8] [12] |
| Cell Isolation Kits | Isolation of specific cell populations (e.g., T cells) from the leukopak. | Magnetic kits (e.g., for CD14+ or CD19+ depletion); Microbubble-based kits (e.g., Akadeum's BACS) [9] [11] |
| Activation Reagents | Stimulate T cell proliferation prior to genetic modification. | CD3/CD28 Dynabeads, TransAct [11] |
| Cytokines | Promote T cell growth and survival during ex vivo expansion. | Interleukin-2 (IL-2) at 300 IU/mL [11] |
| Viral Vectors | Deliver genetic material encoding the Chimeric Antigen Receptor (CAR). | Retroviral or lentiviral vectors [12] |
Leukapheresis represents a foundational pillar in the development and manufacture of autologous cell therapies. A deep understanding of its technical principles, optimized protocols for lymphocyte collection—even from challenging patient populations—and strategies to manage starting material variability are crucial for research scientists and drug development professionals. As the field of cell therapy continues to evolve, further standardization and innovation in leukapheresis and initial cell processing will be key to enhancing manufacturing consistency, scalability, and ultimately, the success of transformative autologous treatments.
Leukapheresis, the procedure for separating and collecting white blood cells from blood, serves as the critical first step in autologous cell therapy manufacturing, particularly for Chimeric Antigen Receptor T-cell (CAR-T) therapies [16] [17]. The quality of the leukapheresis product directly impacts downstream manufacturing success, making optimized collection protocols essential for research and clinical applications [16] [7]. Global adoption of leukapheresis technologies varies significantly across regions, influenced by healthcare infrastructure, regulatory landscapes, research investment, and healthcare priorities [18] [19]. This application note provides a comparative analysis of leukapheresis adoption across three major regions—North America, Europe, and Asia-Pacific—and presents standardized protocols to support autologous cell therapy research across diverse operational environments. The analysis is framed within the context of advancing leukapheresis processing for autologous cell therapy research, addressing the distinct requirements of researchers, scientists, and drug development professionals working in this field.
The global leukapheresis market demonstrates robust growth driven by increasing prevalence of blood cancers, expanding cell and gene therapy pipelines, and growing adoption of personalized medicine approaches [20] [21]. Market projections indicate consistent expansion, though estimates vary based on segment definitions and methodology.
Table 1: Global Leukapheresis Market Size Projections
| Source | Base Year Market Size | Projected Market Size | Forecast Period | CAGR |
|---|---|---|---|---|
| Polaris Market Research [21] | USD 84.58 million (2024) | USD 196.14 million (2034) | 2025-2034 | 8.8% |
| Research Nester [20] | USD 3.22 billion (2025) | USD 7.76 billion (2035) | 2026-2035 | 9.2% |
| Precedence Research [22] | USD 219.47 million (2024) | USD 628.80 million (2034) | 2025-2034 | 11.10% |
Regional adoption patterns reflect disparities in healthcare infrastructure, regulatory maturity, research investment, and market penetration of advanced therapies. The following section provides a detailed comparative analysis of major geographic markets.
Table 2: Regional Adoption Trends in Leukapheresis Markets
| Region | Market Share (2024) | Projected CAGR | Key Growth Drivers | Primary Challenges |
|---|---|---|---|---|
| North America | 36-45.84% [20] [18] | ~8-9% [18] | High CAR-T therapy adoption, Strong R&D infrastructure, Favorable reimbursement policies [18] [22] | High procedural costs, Skilled personnel shortages [20] [18] |
| Europe | 28% [20] | ~5.5% [23] | Established regulatory pathways, Cross-border research collaborations, Government healthcare investments [23] [18] | Economic disparities between member states, Regulatory complexity [18] [19] |
| Asia-Pacific | Growing [18] | 11.57-12.4% [18] [22] | Healthcare infrastructure expansion, Rising cancer incidence, Government initiatives, Medical tourism [18] [19] | Economic disparities, Skilled personnel shortage, Import dependencies [19] |
North American Market Characteristics
North America represents the mature segment of the global leukapheresis market, characterized by advanced healthcare infrastructure, established regulatory pathways, and high adoption of innovative therapies [18] [22]. The United States contributes the majority share of the regional market, supported by numerous FDA-approved CAR-T therapies and favorable reimbursement frameworks [18] [22]. The region's leadership is further reinforced by the concentration of key industry players, advanced research facilities, and substantial investments in cell therapy manufacturing infrastructure [18]. Recent Medicare reimbursement expansions for therapeutic apheresis have further enhanced market growth by improving financial viability for healthcare providers [18].
European Market Dynamics
Europe maintains a mature yet dynamically evolving leukapheresis market, characterized by standardized regulatory oversight through the European Medicines Agency and robust national healthcare systems [20] [18]. The region demonstrates strong collaboration through cross-border clinical research networks and government-funded initiatives for advanced therapy development [23] [18]. Germany, France, and the United Kingdom lead regional adoption, investing in integrated apheresis suites as part of comprehensive cancer treatment programs [18]. The European Blood Alliance's campaign to expand donor pools has incentivized centers to adopt continuous-flow platforms that maximize cell yield and donor comfort [18]. However, economic disparities between Western and Eastern European countries create uneven market penetration, with advanced technologies primarily concentrated in major metropolitan areas [19].
Asia-Pacific Emerging Market Profile
The Asia-Pacific region represents the fastest-growing leukapheresis market globally, driven by massive healthcare infrastructure investments, rising cancer incidence, and expanding biotechnology sectors [18] [22] [19]. Japan leads the regional market with advanced healthcare infrastructure and established cellular therapy programs, while China demonstrates the most rapid growth fueled by substantial government investments in healthcare modernization [19]. The region benefits from growing medical tourism, particularly in Singapore, Thailand, and India, which drives investments in cutting-edge medical technologies including advanced leukapheresis systems [19]. Emerging economies across the region are developing regulatory frameworks to accelerate approval processes for advanced therapies, further stimulating market growth [18].
Technical Protocols for Leukapheresis in Autologous Cell Therapy Research
Pre-Apheresis Patient Assessment and Preparation
Objective: To ensure patient eligibility and optimize collection efficiency for autologous cell therapy research.
Materials:
- Hematology analyzer
- Vascular access supplies (16-18 gauge needles or apheresis catheters)
- Anticoagulant (ACD-A)
- Calcium supplements
Procedure:
- Patient Evaluation:
Timing Considerations:
Pre-procedure Preparation:
- Ensure adequate hydration
- Confirm availability of calcium supplements for citrate-induced hypocalcemia prophylaxis [16]
Leukapheresis Collection Parameters
Objective: To obtain sufficient MNC yield while maintaining cell viability and function for autologous cell therapy research.
Materials:
- Apheresis system (continuous-flow centrifugation preferred) [18]
- Sterile leukapheresis kit/disposables
- Anticoagulant (ACD-A)
- Collection bags
Procedure:
- Device Setup:
- Prime system according to manufacturer instructions
- Configure anticoagulant ratio (typically 8:1 to 12:1 blood:ACD-A) [16]
Parameter Optimization:
- Process 2-3 total blood volumes for patients with lymphocyte counts >1,000/μL [16]
- Extend processing to 3-4 total blood volumes for lymphopenic patients (<500/μL) [16]
- Adjust inlet flow rate based on vascular access and patient tolerance (30-70 mL/min) [16]
- Maintain collection rate to optimize MNC yield while minimizing granulocyte contamination [16]
Patient Monitoring:
- Monitor for citrate toxicity symptoms (paresthesia, nausea)
- Provide calcium supplementation as needed
- Assess patient tolerance throughout procedure
Post-Collection Processing and Cryopreservation
Objective: To preserve cell viability and function for downstream autologous cell therapy applications.
Materials:
- Cryopreservation solution (CS10 with 10% DMSO)
- Controlled-rate freezer
- Cryogenic storage containers
- Cell counting equipment
Procedure:
- Product Assessment:
- Determine total nucleated cell count and viability
- Perform immunophenotyping to assess lymphocyte subsets [7]
Cryopreservation Optimization:
Quality Control:
Figure 1: Comprehensive leukapheresis workflow for autologous cell therapy research, illustrating the sequential stages from patient screening to product release.
Regional Operational Considerations
Technology Access and Implementation
North America: Characterized by widespread adoption of continuous-flow apheresis systems with automated interface management [18]. These systems offer real-time adjustment of plasma-to-anticoagulant ratios, reducing citrate toxicity events by approximately 40% [18]. Research facilities typically employ latest-generation apheresis equipment with closed-system capabilities to maintain sterility and process consistency.
Europe: Utilizes similar technological platforms as North America, with strong emphasis on compliance with Good Manufacturing Practice (GMP) standards and regulatory requirements [23] [18]. European research centers often participate in cross-border technology assessment programs and standardized training initiatives.
Asia-Pacific: Demonstrates variable technology access, with advanced centers in Japan, Singapore, and Australia utilizing state-of-the-art systems comparable to Western standards, while emerging markets may rely on older equipment or imported devices [19]. Import dependencies create vulnerabilities to trade tensions and supply chain disruptions [19].
Regulatory and Quality Considerations
North America: FDA oversight provides comprehensive regulatory framework for cell collection and processing [16] [18]. Compliance with current Good Tissue Practices (cGTP) and Good Manufacturing Practices (cGMP) is required [16]. The FACT-JACIE accreditation system provides additional quality standards for centers engaged in cellular therapy [16].
Europe: The Advanced Therapy Medicinal Products (ATMP) framework governs cell therapy products, with the Hospital Exemption provision allowing non-routine treatment in hospital settings [16]. The European Medicines Agency provides centralized authorization procedures for advanced therapies [18].
Asia-Pacific: Regulatory standards vary significantly across countries, with Japan and South Korea establishing advanced regulatory frameworks for cell therapies, while other markets are developing appropriate oversight mechanisms [18] [19]. This regulatory complexity complicates market entry strategies and increases compliance costs for international manufacturers [19].
Figure 2: Regional innovation pathways in leukapheresis technology and implementation, highlighting distinct focus areas across different geographic markets.
Research Reagent Solutions and Essential Materials
Table 3: Essential Research Reagents and Materials for Leukapheresis Processing
| Category | Specific Products | Research Application | Regional Availability Considerations |
|---|---|---|---|
| Apheresis Systems | Spectra Optia (Terumo BCT), Amicus (Fresenius) | Continuous-flow MNC collection | Widely available in North America and Europe; expanding in Asia-Pacific [18] [19] |
| Leukapheresis Disposables | Sterile collection kits, tubing sets, apheresis catheters | Single-use sterile collection | Global availability with regional preferences for specific brands [21] [22] |
| Cell Separation Media | Ficoll-Paque, Lymphoprep | Density gradient separation | Standard global availability with potential supply chain variations [7] |
| Cryopreservation Media | CS10, CryoStor, CellBanker | Cell freezing and preservation | Global availability with regional GMP compliance variations [7] |
| Quality Assessment Reagents | Flow cytometry antibodies, viability stains, cell counting kits | Product characterization | Comprehensive panels readily available in North America and Europe; limited in emerging markets [7] [19] |
Leukapheresis represents the foundational step in autologous cell therapy research, with global adoption patterns reflecting regional healthcare infrastructure, regulatory maturity, and research priorities. North America maintains leadership in technology adoption and therapy integration, while Europe demonstrates strengths in collaborative research and regulatory standardization. The Asia-Pacific region exhibits the most dynamic growth trajectory, fueled by healthcare infrastructure expansion and increasing government support for advanced therapies. The standardized protocols presented in this application note provide researchers with methodologies adaptable to diverse operational environments, supporting the ongoing globalization of autologous cell therapy research. As the field evolves, continued harmonization of standards and practices across regions will be essential for advancing the development and accessibility of these promising therapies.
Quantitative Analysis of Key Industry Challenges
The leukapheresis industry faces several interconnected constraints that impact both therapeutic applications and research workflows. The table below summarizes the quantitative data and regional impact of these primary challenges.
Table 1: Impact Analysis of Major Leukapheresis Industry Challenges
| Challenge | Quantitative Impact | Geographic Relevance & Timeline | Key Supporting Data |
|---|---|---|---|
| High Procedure Costs [23] [18] | -1.4% impact on CAGR forecast; patient invoices for CAR-T therapies can exceed USD 500,000, with leukapheresis comprising a meaningful early cost share [18]. | Global, most pronounced in emerging markets; Short term (≤ 2 years) [18]. | Stand-alone private collection centers operate 32% cheaper than hospital-based settings, yet most low- and middle-income regions lack such facilities [18]. |
| Donor/Patient Cell Variability [24] | Mononuclear cell product content varies significantly by clinical indication (e.g., CLL vs. lymphoma), directly impacting manufacturing success rates [24]. | A global and fundamental biological constraint, relevant to all autologous therapy production [24]. | Different cellular contaminants (e.g., monocytes, granulocytes) can inhibit T cell proliferation or selectively induce apoptosis of activated T cells [24]. |
| Skilled Professional Shortages [18] [13] | -1.1% impact on CAGR forecast; the National Marrow Donor Program warns that physician retirements will outpace new specialists by 2027 [18]. | Global, acute in North America and Europe; Long term (≥ 4 years) [18]. | Quality standards (e.g., FACT-JACIE) require robust quality management plans and trained staff, increasing the talent burden on centers [13]. |
Experimental Protocols for Challenge Mitigation
Protocol: Mitigating Cellular Variability through Monocyte Depletion
This protocol is adapted from research demonstrating that the removal of specific cell populations from the leukapheresis starting material can enhance the consistency and performance of subsequent CAR T-cell products [25].
1. Principle The success of CAR T-cell manufacturing and final product function is significantly influenced by the composition of the donor starting material. Myeloid cells, particularly monocytes (CD14+), can introduce variability by negatively affecting T-cell activation and transduction efficiency. This protocol outlines a method for enriching leukapheresis material to improve processing outcomes [25].
2. Reagents and Equipment
- Leukapheresis product from a healthy donor or patient
- Immunomagnetic cell separation system (e.g., CliniMACS Prodigy)
- CD14-specific immunomagnetic beads (GMP-grade)
- Cell culture bags and appropriate media
- Flow cytometer for analysis (CD3, CD14, CD19 antibodies)
3. Step-by-Step Procedure 1. Leukapheresis Receipt: Aseptically transfer the leukapheresis product into a sterile processing environment. 2. Cell Preparation: Adjust the cell concentration and volume according to the immunomagnetic separation system's specifications. 3. Antibody Incubation: Incubate the cell suspension with GMP-grade CD14 microbeads. The beads selectively bind to CD14-expressing monocytes. 4. Magnetic Separation: Load the cell-bead mixture onto the separation system. The CD14+ monocytes are retained in the magnetic field. 5. Collection: Collect the flow-through, which is the monocyte-depleted leukapheresis product. This product is enriched for lymphocytes, including T-cells. 6. Quality Control: Analyze the pre- and post-depletion samples by flow cytometry to determine the percentage of CD14+ monocytes and CD3+ T-cells.
4. Expected Outcomes and Validation
- Activation: A 2-fold improvement in the level of T-cell activation compared to unsorted leukapheresis, achieving a level similar to initiating with purified T-cells [25].
- Transduction: In culture bag systems, monocyte depletion significantly improves transduction consistency and can increase CAR expression levels by up to 64% [25].
- Functionality: CAR T-cell products from monocyte-depleted starting material demonstrate superior and more consistent cytotoxicity in target cell killing assays and display a more rested and naive memory phenotype [25].
Protocol: Standardizing Cryopreserved Leukapheresis for Supply Chain Resilience
This protocol establishes a standardized, closed automated process for cryopreserving leukapheresis material, decoupling cell collection from manufacturing and mitigating risks associated with the short viability of fresh samples and skilled operator dependency [26] [6].
1. Principle Fresh leukapheresis exhibits time-sensitive viability decay, typically requiring processing within a 24-72 hour window [26]. Cryopreservation provides flexibility for patients, protects against shipping delays, and removes manufacturing time constraints, thereby building resilience into the supply chain [6]. This protocol uses a closed-system automated platform to ensure standardization and minimize operator-induced variability.
2. Reagents and Equipment
- Closed-system automated cell processing platform
- Cryoprotectant: Clinical-grade CS10 (10% DMSO)
- Controlled-rate freezer (e.g., Thermo Profile 4 system)
- Cryogenic storage bags
- Liquid nitrogen storage system
3. Step-by-Step Procedure 1. Initial Processing: Subject the leukapheresis product to a centrifugation-based step to reduce non-cellular impurities (e.g., residual red blood cells, platelets) [26]. 2. Mixing with Cryoprotectant: Combine the processed cell product with CS10 cryoprotectant. The final target DMSO concentration should be 7.5% - 10% (v/v) [26]. 3. Formulation: Adjust the cell concentration to a target of 5–8 × 10^7 cells/ml. The recommended formulation volume is 20 ml per cryobag [26]. 4. Time-Sensitive Freezing: Critical Step: Initiate controlled-rate freezing within 120 minutes of cryoprotectant addition to prevent ice crystal formation. Use a validated freezing curve [26]. 5. Storage: Transfer the frozen cryobags to a liquid nitrogen vapor-phase storage system.
4. Expected Outcomes and Quality Attributes
- Viability: Post-thaw viability of ≥ 90% [26].
- Cell Recovery: Maintenance of key cell populations; post-thaw CD3+ T lymphocyte proportions should show minimal variation from pre-cryopreservation levels (e.g., ~45% post-thaw vs. ~48% pre-cryo) [26].
- Compatibility: The cryopreserved leukapheresis product should be functionally comparable to fresh material across various CAR-T manufacturing platforms (non-viral, lentiviral, Fast CAR-T) in terms of cell expansion, phenotype, and cytotoxicity [26].
Workflow Visualization
Cryopreservation Standardization Workflow
Monocyte Depletion for Enhanced Manufacturing
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents and Materials for Leukapheresis Research & Processing
| Item | Function & Application |
|---|---|
| CS10 Cryoprotectant | A clinical-grade, serum-free cryopreservation medium containing 10% DMSO. It is essential for the long-term storage of leukapheresis products and PBMCs, ensuring high post-thaw viability and functional recovery [26]. |
| Immunomagnetic Beads (CD14+) | GMP-grade, antibody-coupled magnetic beads for the specific depletion of monocytes from leukapheresis products. This enrichment step is critical for reducing process variability and improving T-cell activation and transduction [25]. |
| Closed-System Automated Formulator | An automated, closed-system platform for processing and formulating leukapheresis product with cryoprotectant. It standardizes the pre-cryopreservation workflow, reduces operator error, and minimizes contamination risk [26]. |
| Controlled-Rate Freezer | A instrument that precisely controls the cooling rate during the cryopreservation process. This is vital for preventing lethal intracellular ice crystal formation and achieving the target post-thaw viability of ≥ 90% [26]. |
| Leukapheresis Disposables (Kits, Tubing) | Single-use, sterile kits and tubing sets designed for specific apheresis systems. They ensure procedural hygiene, prevent cross-contamination, and are indispensable for both therapeutic and research-grade cell collection [23] [18]. |
Application Note: Integrated Automation and AI in Leukapheresis Processing
The landscape of leukapheresis processing for autologous cell therapy is being reshaped by three convergent technological forces: advanced automation, artificial intelligence (AI) for real-time monitoring, and decentralized manufacturing models. These innovations collectively address critical bottlenecks in cell therapy production, including vein-to-vein time, product quality variability, and patient access limitations [27] [28].
Decentralized Manufacturing Models are transitioning from fixed-site facilities to mobile and point-of-care solutions. The world's first mobile leukapheresis center, launched by BBG Advanced Therapies, exemplifies this shift. This mobile unit is a fully functional leukapheresis suite on a bus, designed to mirror the controls and quality standards of a fixed site, complete with redundant power supplies, real-time environmental monitoring, and on-board cell analysis equipment [28]. This model brings the collection capability directly to patients, reducing travel burdens and expanding access for rural and underserved communities [29].
Automation and Closed Systems are crucial for standardizing processes in decentralized settings. Platforms like the Cocoon system provide closed, automated, single-use cassette-based bioreactor manufacturing, which reduces contamination risk and minimizes the required facility footprint [27]. This level of automation is essential for transferring complex processes from centralized cleanrooms to local hospitals or mobile settings.
AI-Assisted Monitoring leverages intelligent algorithms to optimize processes and ensure quality. AI-driven systems are being deployed for real-time data analysis during leukapheresis, enhancing yield prediction, and monitoring cell quality [23] [30]. Integrated digital platforms, such as the xCellit system, enable real-time process monitoring and electronic batch records, providing the necessary oversight for multi-site manufacturing networks [27].
Table 1: Quantitative Impact of Emerging Leukapheresis and Manufacturing Models
| Technology Trend | Reported Metric | Impact/Performance | Source/Context |
|---|---|---|---|
| Decentralized Manufacturing | Vein-to-vein time | Median of 7 days; novel 20-hour process reported [27] | Galapagos platform; Ohio State University |
| Mobile Leukapheresis | Service area coverage | >63,000 square miles [28] | BBG Advanced Therapies mobile unit |
| AI & Automation | Market Growth (CAGR) | 5.5% (2025-2035) for leukapheresis products [23] | Global leukapheresis products market |
| Therapeutic Apheresis Equipment | Market Value | $2.01M (2023) to $2.20M (2032) [31] | Global apheresis equipment market |
Experimental Validation and Workflow
Recent clinical validations underscore the efficacy of these integrated platforms. A decentralized model employing an automated, closed system demonstrated the ability to produce fresh, stem-like CAR-T cells with high response rates and manageable safety profiles in lymphoma patients [27]. The key to success in these decentralized workflows is the seamless integration of each technological component.
The following diagram illustrates the integrated workflow of a decentralized manufacturing model, from mobile leukapheresis to final product release, highlighting the role of automation and AI at each stage.
Protocol: Implementation of a Decentralized, Automated Leukapheresis Workflow
Mobile Leukapheresis and Initial Processing
Principle This protocol describes the procedure for performing leukapheresis in a mobile setting, ensuring the collection of high-quality starting material for subsequent automated manufacturing of autologous cell therapies. The mobile unit is designed to maintain the same standards as a fixed site, with controlled environment, redundant power, and integrated analytical capabilities [28].
Materials
- Mobile leukapheresis unit with onboard power generation and environmental monitoring.
- Apheresis device (e.g., Terumo BCT, Haemonetics, or Fresenius systems) [31].
- Single-use, sterile leukapheresis disposable kit.
- Onboard Sysmex cell counter or equivalent for immediate cell count and viability analysis.
- Validated temperature-controlled shippers for leukopak transport.
Procedure
- Pre-Procedural Screening & Setup:
- Upon arrival at the community site, connect the mobile unit to shore power.
- Confirm environmental parameters (temperature, pressure) are within specified ranges using the real-time monitoring system.
- Perform patient/donor eligibility confirmation, informed consent, and venous access assessment (peripheral vs. central line) [28].
Leukapheresis Collection:
- Initiate the leukapheresis procedure using an automated apheresis system. The process typically takes 3–6 hours but may extend to a two-day procedure if target cell yields are not met [28].
- The highly trained nursing team closely monitors the patient and the device parameters throughout the collection.
On-board Sample Analysis & Packaging:
- Upon collection completion, perform an immediate cell count and viability analysis using the onboard Sysmex cell counter [28].
- The leukopak is packaged into a validated temperature-controlled shipping container with appropriate data loggers. All labelling and documentation requirements are completed.
Quality Assurance & Release:
- A quality assurance professional reviews all documentation—from screening to collection and packaging—to ensure specifications are met before the leukopak is released for transport [28].
- Coordinate with a courier for immediate pickup or transport the leukopak to the nearest automated manufacturing node.
Automated Manufacturing with AI Monitoring
Principle This protocol details the automated manufacturing and AI-driven monitoring process that follows mobile leukapheresis. The goal is to rapidly manufacture a fresh CAR-T cell product while preserving T-cell stemness, leveraging closed automation and real-time data analytics for quality control [27].
Materials
- Closed, automated cell processing system (e.g., Cocoon platform).
- Single-use, pre-sterilized processing cassettes/consumables.
- Cell culture media and activation/transduction reagents.
- Integrated digital platform for process monitoring (e.g., xCellit).
- QC sampling kit for in-process controls.
Procedure
- System Priming & Leukopak Loading:
- Load a single-use processing cassette into the automated system within a decentralized manufacturing facility (e.g., a local hospital lab or a blood center).
- The system primes the fluidic path with media. The leukopak is aseptically connected to the system's input port.
Initiation of Automated Process:
- The automated system performs cell isolation, activation, viral transduction (if applicable), and culture. In ultra-rapid protocols (e.g., 20-hour process), traditional isolation and expansion steps are bypassed to preserve T-cell stemness [27].
- The system operates as a closed, integrated unit, minimizing manual intervention and contamination risk.
AI-Assisted In-Process Monitoring:
- The integrated digital platform (e.g., xCellit) collects real-time data on critical process parameters (CPPs) like gas exchange, pH, and cell concentration [27].
- AI algorithms analyze this data stream to predict final cell yield and product quality, and to identify any process deviations for immediate corrective action [23] [30]. Electronic batch records are automatically generated.
Harvest and Final Product Formulation:
- Upon process completion, the system automatically harvests the cell product into a final bag for infusion.
- The final product is transported to the clinical site for fresh infusion, bypassing the need for cryopreservation and its associated logistics and cell viability challenges [27].
Table 2: The Scientist's Toolkit - Key Reagents and Materials for Automated Leukapheresis Research
| Item Category | Specific Examples | Function in Workflow |
|---|---|---|
| Leukapheresis Disposables | Single-use leukapheresis kits, tubing sets, collection bags [23] | Ensure sterility and prevent cross-contamination during the initial cell collection from patients/donors. |
| Automated Cell Processing System | Cocoon platform [27] | Provides a closed, automated environment for cell culture, activation, and transduction, standardizing manufacturing. |
| Cell Culture Media | Serum-free media, activation cytokines, transduction reagents | Supports T-cell growth and enables genetic modification for CAR-T cell production. |
| Integrated Digital Platform | xCellit digital platform [27] | Enables real-time process monitoring, data management, and electronic batch record generation for quality control. |
| Validated Shipping System | Temperature-controlled shippers with data loggers [28] | Maintains the required temperature of the leukopak during transport from the collection to the manufacturing site. |
The following diagram summarizes the core feedback loop enabled by AI-assisted monitoring in the automated manufacturing process, ensuring continuous quality control.
From Vein to Vector: Methodological Strategies for Effective Leukapheresis in CAR-T Manufacturing
The global market for apheresis and leukapheresis products is experiencing significant growth, driven by the expansion of cell therapies, particularly in oncology. The table below summarizes key market metrics and regional growth trends.
Table 1: Global Market Overview for Leukapheresis Products and Automated Processing Systems
| Market Segment | Key Metrics | Projected CAGR & Value |
|---|---|---|
| Global Leukapheresis Products Market [23] | Market Size in 2025: USD 1,340.6 Million | CAGR (2025-2035): 5.5%Market Size in 2035: USD 2,290.1 Million |
| Automated & Closed Cell Therapy Processing Systems Market [32] | Market Size in 2025: USD 2.22 Billion | CAGR (2025-2034): 19.9%Market Size in 2034: USD 11.36 Billion |
| Global Apheresis Equipment Market [33] | Market Size in 2024: USD 3.2 Billion | CAGR (2025-2034): 9.2%Market Size in 2034: USD 7.7 Billion |
Table 2: Regional Market Analysis and Growth Projections
| Region | Market Position & Trends | Country-Specific CAGR (2025-2035) |
|---|---|---|
| North America | Dominates the market due to high prevalence of blood disorders, well-established healthcare infrastructure, and strong presence of key market players [23]. Held a 51.0% share in the automated systems market in 2024 [32]. | USA: 5.6% [23] |
| Europe | Advances at a gradual pace with Germany, France, and the UK at the forefront, integrating leukapheresis procedures to improve patient outcomes [23]. | European Union: 5.5% [23]UK: 5.4% [23] |
| Asia-Pacific | Expected to be the fastest-growing region, fueled by rising urbanization, growing disposable incomes, and increasing demand for sophisticated therapeutic options [23] [32]. | Japan: 5.5%South Korea: 5.6% [23] |
Key Automated Closed-System Platforms
The transition from manual, open processes to automated, closed-system manufacturing is critical for scaling autologous cell therapy production. These systems enhance consistency, minimize contamination risks, and reduce manual labor [34] [35]. The following table profiles leading platforms.
Table 3: Profiles of Key Automated Closed-System Platforms for Cell Therapy Manufacturing
| Platform (Company) | Key Features & Throughput | Primary Applications & Market Context |
|---|---|---|
| Cocoon Platform (Lonza) | Fully closed, automated system. Each unit processes one patient batch at a time. ~10 days vein-to-vein time (V2VT) [34]. | Supports decentralized manufacturing. Used for commercial-scale gene therapies (e.g., Vertex's CASGEVY). Holds 18%-22% market share in automated systems [34]. |
| Cell Shuttle (Cellares) | FDA AMT designation (2025). Processes 16 batches in parallel. Up to 1,000+ annual batches per shuttle [34]. | Designed for high-throughput "smart factories." Estimated market share of 10%-14% [34]. |
| CliniMACS Prodigy (Miltenyi Biotec) | Fully closed, end-to-end automated system from cell selection to formulation. Enables processing in Grade C cleanrooms [34]. | CAR-T production; also used for NK cell differentiation from CD34+ cells [34] [36]. Market share of 4%-8% [34]. |
| CTS Rotea System (Thermo Fisher Scientific) | Closed-loop system for leukopak processing. Processes at 5.3 L/hour with >90% PBMC recovery in <30 minutes [34]. | Automates leukopak processing for autologous and allogeneic therapies. A modular system that requires connection to other platforms for end-to-end automation [34]. |
| Sefia Platform (Cytiva) | Modular platform with Select and Expansion systems. Increases manufactured doses by up to 50% per year. Reduces manual operators by 40% [34]. | Scalable from clinical (10 doses/year) to commercial production (1,000 doses/year) [34]. Market share of 7%-11% [34]. |
Experimental Protocol: NK Cell Manufacturing from Cord Blood on CliniMACS Prodigy
This protocol details the use of the CliniMACS Prodigy system for the initial enrichment of CD34+ hematopoietic stem cells (HSCs) from umbilical cord blood (UCB), a critical first step in manufacturing allogeneic Natural Killer (NK) cells [36].
Materials and Reagents
Table 4: Research Reagent Solutions for CD34+ HSC Enrichment
| Item | Function | Example / Specification |
|---|---|---|
| Umbilical Cord Blood (UCB) Unit | Source of CD34+ HSCs. | Fresh UCB, supplied by an accredited cord blood bank. Must contain ≥2.0E06 CD34+ cells (R&D) or ≥3.5E06 CD34+ cells (GMP) [36]. |
| CliniMACS PBS/EDTA Buffer | Washing buffer; prevents cell clumping. | Supplemented with 0.5% Human Serum Albumin (HSA) [36]. |
| CliniMACS CD34 Reagent | Magnetic antibody for target cell selection. | Biotinylated anti-CD34 antibody and Anti-Biotin MACiBeads [36]. |
| FcR Blocking Reagent | Blocks non-specific antibody binding. | 5% IgG solution [36]. |
| Elution Buffer | Medium for collecting enriched cells. | Proprietary Glycostem Basal Growth Medium (GBGM) or equivalent [36]. |
| TS310 Tubing Set | Single-use, disposable fluid path. | Pre-assembled set for the LP-34 Enrichment Protocol on the Prodigy [36]. |
Step-by-Step Procedure
Pre-process UCB Validation:
- Verify UCB unit data, including volume, total nucleated cell (TNC) count, and CD34+ cell content upon receipt.
- Ensure the unit was transported at 15–25°C and was not X-ray screened [36].
System Setup (Performed in a GMP-grade cleanroom):
- Load the CliniMACS Prodigy with the sterile, single-use TS310 tubing set.
- Prime the system with CliniMACS PBS/EDTA buffer with 0.5% HSA.
- Select the "LP-34 Enrichment Protocol" (version 2.2) on the Prodigy touchscreen interface [36].
Cell Processing:
- Connect the UCB unit to the system.
- The process is fully automated and includes the following key steps: a. Cell Sampling: A small sample is automatically taken for initial cell count. b. FcR Blocking and Labeling: The instrument mixes the UCB with FcR blocking reagent and the CliniMACS CD34 Reagent. c. Incubation: A defined period for magnetic antibody binding. d. Washing and Magnetic Separation: Cells are washed and passed through a magnetic column. Labeled CD34+ cells are retained, while unlabeled cells are washed to waste. e. Elution: The target CD34+ cells are flushed from the column and collected into an output bag containing ~80 mL of elution buffer [36].
Post-process Quality Control:
- Aseptically collect a 1 mL sample from the output bag.
- Perform flow cytometry analysis to determine CD34+ cell recovery, viability, and purity [36].
Expected Outcomes and Performance
Across 36 manufacturing runs, the CliniMACS Prodigy demonstrated robust performance for CD34+ cell enrichment from UCB [36]:
- Cell Recovery: Average CD34+ cell recoveries of 68.18% to 71.94%, depending on the starting CD34+ cell content of the UCB unit.
- Purity: Achieved purities of 57.48% to 69.73%, with higher purity correlated with higher starting CD34+ cell content.
- Consistency: Factors like UCB age, TNC count, and RBC/platelet content had no significant impact on performance, highlighting the process robustness [36].
Experimental Protocol: PBMC Isolation and Processing from Leukopaks
For autologous cell therapy, the leukopak obtained via leukapheresis is the foundational starting material. This protocol covers the receipt and processing of leukopaks to isolate high-viability Peripheral Blood Mononuclear Cells (PBMCs) for downstream CAR-T manufacturing.
Materials and Reagents
Table 5: Research Reagent Solutions for PBMC Processing
| Item | Function | Example / Specification |
|---|---|---|
| Leukopak | Apheresis product rich in leukocytes. | Contains up to 20 billion leukocytes from a single donor. Must be transported in a validated temperature-controlled shipper [37] [38]. |
| Density Gradient Medium | Separates PBMCs based on density. | Ficoll-Paque PREMIUM or equivalent [38]. |
| Wash Buffer | Dilutes sample and washes cells. | PBS (without Ca2+/Mg2+), potentially supplemented with a low percentage of serum or albumin [38]. |
| Cryopreservation Medium | Protects cells during freeze-thaw. | Cell culture medium containing 10% DMSO [38]. |
| Closed-System Cell Processor | Automates and closes the processing step. | Thermo Fisher Scientific's CTS Rotea Counterflow Centrifugation System [34]. |
Step-by-Step Procedure
Leukopak Receipt and Inspection:
- Upon arrival, immediately inspect the leukopak shipping container and data loggers to ensure it was maintained at the correct temperature (ambient or 2-8°C).
- Record the time of receipt and inspect for any signs of damage or leakage [38].
PBMC Isolation via Automated Closed System (Recommended):
- System: Use the CTS Rotea system or equivalent.
- Process: Follow the manufacturer's instructions. The system automates washing and concentration steps using counterflow centrifugation.
- Performance: This method can process a leukopak at 5.3 L/hour in <30 minutes, achieving >90% PBMC recovery and >95% cell viability, significantly outperforming manual methods [34].
PBMC Isolation via Manual Density Gradient (Alternative):
- Temperature Equilibration: Ensure the leukopak and all reagents are at room temperature (15-25°C) to ensure proper density separation [38].
- Dilution: Dilute the leukopak sample 1:1 with wash buffer.
- Centrifugation: Carefully layer the diluted blood over the density gradient medium. Centrifuge at 400-800 × g for 20-30 minutes with the brake off.
- PBMC Collection: After centrifugation, collect the opaque interface layer (the "buffy coat") containing the PBMCs, using a pipette.
- Washing: Wash the collected PBMCs with buffer 2-3 times to remove residual platelets, Ficoll, and plasma proteins [38].
Cryopreservation (If not used immediately):
- DMSO Handling: Resuspend the final PBMC pellet in pre-chilled cryopreservation medium. Work quickly to minimize DMSO exposure before freezing.
- Controlled-Rate Freezing: Use an isopropanol freezing chamber (e.g., "Mr. Frosty") or a controlled-rate freezer to freeze cells at approximately -1°C/minute.
- Storage: Transfer frozen vials to the vapor phase of liquid nitrogen for long-term storage [38].
Critical Factors for Success
- Donor Variability: Acknowledge that donor health and collection parameters are major contributors to variability in PBMC recovery and performance. Using verified donor programs can improve consistency [38].
- Minimize Clotting: Ensure proper mixing with anticoagulant during the initial blood draw. Avoid continuous rocking of the leukopak during storage, as this can induce micro-clotting [38].
- Avoid Granulocyte Contamination: Transporting or storing whole blood at 2-8°C for >24 hours can increase granulocyte contamination in the PBMC fraction, which can suppress T-cell function in downstream assays [38].
Workflow Visualization
The following diagram illustrates the integrated workflow for autologous cell therapy, from leukapheresis to final formulation, highlighting the role of automated closed-system platforms.
Challenges and Future Outlook
Despite the clear benefits, the adoption of automated closed-system platforms faces several hurdles:
- Financial Barriers: High upfront capital expenditure, with a payback period often exceeding three to five years, can be prohibitive, especially for small to midsize biotechs [34].
- Technical and Operational Barriers: Process rigidity in non-modular platforms can limit adaptability for novel therapies. Interoperability gaps between different vendors' systems can force manufacturers into proprietary ecosystems, increasing switching costs [34].
- Regulatory Uncertainty: Changing or introducing a new automated platform after IND approval typically triggers full comparability studies, adding 12-18 months to development timelines [34].
Future trends point toward greater integration of artificial intelligence (AI) for yield optimization, the rise of point-of-care leukapheresis technologies, and the development of platform technologies for allogeneic "off-the-shelf" therapies, all of which will continue to shape the apheresis device technology landscape [23] [32].
The initial leukapheresis procedure is a critical determinant of success in autologous chimeric antigen receptor (CAR) T-cell therapy. The quality and quantity of collected T-cells directly impact manufacturing outcomes and, ultimately, clinical efficacy [39] [40]. Traditional apheresis protocols utilizing fixed processing volumes often result in either inadequate cell yields requiring repeat procedures or excessive collection times that increase patient burden [41]. This application note details a yield-driven collection strategy that dynamically adjusts processed blood volume and procedure duration based on pre-apheresis CD3+ cell counts, optimizing resource utilization while ensuring sufficient material for manufacturing.
Background and Rationale
The Impact of Starting Material on CAR-T Cell Therapy
The foundational principle of yield-driven collection recognizes that pre-apheresis T-cell counts are highly variable among patients, influenced by factors including prior therapy lines, disease burden, and recent immunosuppressive treatments [39] [42]. Despite this variability, a direct correlation exists between pre-apheresis CD3+ counts and successful manufacturing outcomes [39] [40]. Collections yielding higher numbers of non-senescent CD3+CD27+CD28+ T-cells demonstrate significantly greater manufacturing success rates, with one analysis identifying a cutoff of ≥34.58 × 10^8 CD3+CD27+CD28+ cells for an 89.5% probability of successful CAR-T cell production [39].
Furthermore, pre-apheresis CD3+ cell counts possess prognostic value for clinical outcomes. Patients with CD3+ counts >553/μL prior to leukapheresis demonstrated significantly superior 1-year progression-free survival (68.3% vs. 17.3%) and overall survival compared to those with lower counts [40].
Limitations of Standardized Processing Volumes
Current apheresis procedures for different CAR-T products often employ standardized processing volumes, leading to substantial variability in collected cell yields. A recent analysis found that axi-cel protocols required significantly larger processing volumes (12L vs. 10L for tisa-cel) and longer processing times (240min vs. 203min), resulting in higher but more variable CD3+ yields and frequently exceeding estimated blood volumes needed to achieve target cell counts [41] [43]. This "one-size-fits-all" approach underscores the need for personalized collection strategies.
Quantitative Foundation for Yield-Driven Protocols
Key Parameter Correlations
Analysis of apheresis data reveals consistent relationships between pre-apheresis counts, processing parameters, and final yields:
Table 1: Correlation Between Pre-Apheresis CD3+ Counts and Collection Outcomes
| Pre-Apheresis CD3+ (cells/μL) | Recommended Processing Volume | Expected Collection Efficiency | Probability of Achieving Manufacturing Target |
|---|---|---|---|
| < 300 | 12-15 L | 60-65% | 40-50% |
| 300-600 | 10-12 L | 60-65% | 70-80% |
| > 600 | 8-10 L | 60-65% | >90% |
Data synthesized from [41] [39] [42]
Product-Specific Considerations
Different CAR-T products have varying cell dose requirements and manufacturing processes, necessitating protocol adaptations:
Table 2: Processing Parameters and Outcomes by CAR-T Product
| CAR-T Product | Typical Processing Volume | Average Processing Time (min) | Mean CD3+ Yield (×10^9 cells) | Manufacturing Success Rate |
|---|---|---|---|---|
| Axi-cel | 12 L | 240 | 3.04 | Not specified |
| Liso-cel | 12 L | 204 | 1.54 | Not specified |
| Tisa-cel | 10 L | 203 | 1.59 | 61% |
Experimental Protocol: Yield-Driven Leukapheresis
Pre-Apheresis Assessment and Patient Preparation
Materials Required:
- Complete blood count (CBC) with differential
- Flow cytometry reagents for CD3, CD4, CD8, CD27, CD28 quantification
- Calcium supplements for prophylactic administration
- Spectra Optia apheresis system (Terumo BCT) or equivalent
Procedure:
- Patient Evaluation (7-10 days pre-apheresis):
- Obtain CBC with manual differential if blasts are suspected
- Perform comprehensive lymphocyte phenotyping via flow cytometry to quantify:
- Absolute CD3+, CD4+, CD8+ counts
- CD3+CD27+CD28+ non-senescent T-cell population
- Exclusion of circulating blasts (if >10%, consider cytoreduction)
- Assess venous access; plan for central venous catheter if peripheral access inadequate
- Timing Considerations:
- Schedule apheresis at least 2 weeks after last bendamustine therapy or other T-cell depleting regimens
- Ensure platelet count >50×10^3/μL and hemoglobin >8.0 g/dL before procedure
- For patients with absolute lymphocyte count (ALC) ≤1.0/μL, plan for 12-15L processing volume [42]
Processing Volume Calculation Algorithm
The processed blood volume (V) can be calculated using the following formula based on pre-apheresis CD3+ count:
V (L) = Target CD3+ Yield / (Pre-apheresis CD3+ × Collection Efficiency)
Where:
- Target CD3+ Yield = Minimum 0.6×10^9 cells (manufacturing minimum)
- Pre-apheresis CD3+ = Absolute CD3+ count (cells/μL)
- Collection Efficiency = 0.62 (median efficiency, range 0.55-0.70) [41]
Practical Implementation:
- For minimum yield (0.6×10^9 CD3+ cells): Vmin = 0.6×10^9 / (CD3pre × 0.62)
- For target yield (2.0×10^9 CD3+ cells): Vtarget = 2.0×10^9 / (CD3pre × 0.62)
- Set processing volume to V_target, not to exceed 15L total blood volume
- If V_min exceeds 15L, plan for two-day collection or consider manufacturing with lower cell dose
Leukapheresis Procedure
Setup and Monitoring:
- Instrument Configuration:
- Use Spectra Optia with continuous mononuclear cell (CMNC) collection chamber
- Set anticoagulant (ACD-A) to blood ratio at 12:1 to 15:1
- Program collection flow rate at 1.0-1.5 mL/min
- Configure interface management for optimal lymphocyte collection
Procedure Execution:
- Process calculated blood volume based on CD3+ algorithm
- For patients with pre-apheresis CD3+ <300/μL: Process 12-15L regardless of calculation
- For patients with pre-apheresis CD3+ >800/μL: Limit processing to 8-10L unless product-specific requirements dictate higher yields
- Monitor patient for symptoms of hypocalcemia; administer calcium prophylaxis as needed
Product Handling:
- Determine total nucleated cell (TNC) count and CD3+ content immediately post-collection
- For cryopreservation, use DMSO-containing cryomedium at controlled rate freezing
- Maintain chain of identity and chain of custody documentation
Quality Assessment and Release Criteria
Product Specifications:
- Minimum acceptable CD3+ yield: ≥0.6×10^9 cells [44]
- Target CD3+ yield: ≥2.0×10^9 cells for manufacturing flexibility
- Viability: ≥90% by propidium iodide exclusion
- Sterility: Negative bacterial/fungal culture after 14-day incubation
Workflow Visualization
Figure 1. Yield-Driven Leukapheresis Protocol Workflow. This algorithm guides processing volume determination based on pre-apheresis CD3+ counts to optimize cell yield while minimizing patient burden.
Validation Data and Performance Metrics
Clinical Validation
Implementation of yield-driven protocols has demonstrated significant improvements in apheresis outcomes:
- Reduced Processing Time: Patients with pre-apheresis CD3+ >600/μL underwent apheresis for mean 173 minutes versus 247 minutes for those with counts <300/μL [41]
- Improved Manufacturing Success: Collections containing ≥34.58×10^8 CD3+CD27+CD28+ cells achieved 89.5% manufacturing success rate versus 35% for lower yields [39]
- Predictable Yields: Protocol-based processing volumes yielded CD3+ collections within target range in 92% of cases versus 68% with fixed-volume approaches [41] [42]
Troubleshooting Guide
Table 3: Common Challenges and Resolution Strategies
| Challenge | Potential Causes | Resolution Strategies |
|---|---|---|
| Low CD3+ yield despite adequate processing volume | Low collection efficiency, high non-T cell contamination | Verify instrument setup, adjust interface management, consider two-day collection |
| Excessive granulocyte contamination | Poor interface control, high pre-apheresis granulocyte count | Optimize centrifuge speed, review collection rate settings |
| Product volume exceeding specifications | Over-collection, high anticoagulant ratio | Implement product concentration steps, adjust ACD ratio |
| Manufacturing failure despite adequate CD3+ numbers | High senescent T-cell population, cellular dysfunction | Assess CD27/CD28 expression pre-apheresis, consider alternative manufacturing approach |
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Reagents for Apheresis Quality Control and Cellular Analysis
| Reagent/Category | Specific Examples | Research Application |
|---|---|---|
| Flow Cytometry Antibodies | Anti-CD3, CD4, CD8, CD45, CD27, CD28 | Immunophenotyping of pre-apheresis blood and final product |
| Cell Viability Assays | Propidium iodide, 7-AAD | Determination of cell viability and apoptosis assessment |
| Cell Separation Media | Ficoll-Paque PLUS, Lymphoprep | Density gradient separation for product analysis |
| Cryopreservation Media | CryoStor CS10, Bambanker | Cryopreservation of apheresis product for shipping |
| Microbiological Culture | BACTEC, BacT/ALERT | Sterility testing for bacterial and fungal contamination |
| Cell Counting Reagents | Trypan blue, AO/PI stains | Automated cell counting and viability assessment |
Yield-driven leukapheresis protocols represent a significant advancement in autologous cell therapy manufacturing, moving from standardized approaches to personalized collection strategies. By tailoring processed blood volumes to individual patient characteristics, particularly pre-apheresis CD3+ counts, institutions can optimize resource utilization, reduce patient burden, and improve manufacturing success rates. The protocol detailed herein provides a validated framework for implementation, with specific guidance on volume calculation, quality parameters, and troubleshooting strategies. As CAR-T therapy expands to new indications and patient populations, such refined collection methodologies will be essential for maximizing therapeutic potential and improving patient access.
For researchers and drug development professionals working in autologous cell therapy, the logistics of sourcing viable cellular starting material is a critical determinant of manufacturing success. The choice between using fresh leukapheresis products, traditionally considered the gold standard, and cryopreserved leukapheresis has profound implications for supply chain resilience, process flexibility, and ultimately, therapeutic output. In autologous therapies, where a patient's own cells are the starting material, cell quality can be significantly impacted by their disease state and prior treatments [7] [45]. This application note provides a structured, data-driven comparison to guide the selection and implementation of leukapheresis material logistics, framing the discussion within scalable manufacturing paradigms.
Comparative Analysis: Fresh vs. Cryopreserved Leukapheresis
A comprehensive evaluation of fresh and cryopreserved leukapheresis products reveals distinct trade-offs across logistical and quality attributes. The data, synthesized from recent comparative studies, is summarized in the table below.
Table 1: Comparative Analysis of Fresh and Cryopreserved Leukapheresis for Scalable Manufacturing
| Attribute | Fresh Leukapheresis | Cryopreserved Leukapheresis | Key Findings & Supporting Data |
|---|---|---|---|
| Logistical Flexibility | Limited; requires immediate processing and transport within a strict 24-72 hour window [7]. | High; enables long-term storage (≥30 months [46]), decouples collection from manufacturing, and facilitates global shipment [6]. | Cryopreservation allows for "just-in-case" inventory, mitigating risks from transport delays and manufacturing scheduling conflicts [6] [46]. |
| Supply Chain Resilience | Vulnerable to transport delays, temperature excursions, and external disruptions [7]. | Enhanced; stable material allows for a more robust and distributed manufacturing model [7]. | Centralized manufacturing for global clinical trials (e.g., in 12 countries) has been successfully achieved using cryopreserved starting material [6]. |
| Initial Cell Viability | High; typically >99% at collection [7]. | Slightly reduced but acceptable; post-thaw viability consistently ≥90%, with reports of ≥80% being sufficient for manufacturing [7] [47]. | While a statistically significant drop from fresh, the post-thaw viability meets manufacturing acceptance criteria and does not compromise functional outcomes [7] [46]. |
| Cell Recovery & Phenotype | Preserves native immune cell profile. | Excellent recovery (85-97%) with comparable phenotypic profiles to fresh material [46]. May exhibit a higher lymphocyte proportion (66.59%) vs. cryopreserved PBMCs (52.20%) [7]. | Key T-cell subsets (CD3+, CD4+, CD8+), crucial for CAR-T manufacturing, show minimal variation post-thaw [7] [46]. |
| Manufacturing Compatibility & Clinical Outcome | Compatible with standard processes. | Demonstrated compatibility across multiple platforms, including lentiviral CAR-T, non-viral CAR-T, and Fast CAR-T platforms [7]. | Final products derived from cryopreserved leukapheresis show comparable expansion potential, cytotoxicity, and transduction efficiency to those from fresh material [7] [6]. Clinical outcomes with products like Kymriah validate its efficacy [6] [48]. |
The logistical workflow for each option underscores the operational differences, with cryopreservation introducing a "pause" point that significantly de-risks the process.
Figure 1: Logistical workflows for fresh versus cryopreserved leukapheresis material, highlighting the time-critical nature of the fresh pathway versus the stability and flexibility of the cryopreserved pathway.
Protocols for Processing and Utilizing Cryopreserved Leukapheresis
Protocol: Standardized Cryopreservation of Leukapheresis Material
This protocol outlines key steps for preparing cryopreserved leukapheresis products to ensure high post-thaw viability and recovery, based on optimized processes from recent studies [7] [47].
- Objective: To preserve leukapheresis material with ≥90% post-thaw viability and maintained cell phenotype for subsequent cell therapy manufacturing.
Materials:
Method:
- Initial Processing: Perform a centrifugation step to reduce non-cellular impurities like red blood cells and platelets. This is critical for minimizing DMSO concentration variability and improving cryoprotection efficacy [7].
- Formulation: Resuspend the cell pellet in cryoprotectant. Studies have validated both 5% and 10% DMSO concentrations; 5% DMSO is often selected for its favorable toxicity profile while maintaining high recovery [47].
- Target Concentration: Adjust the final cell concentration to a target of 5 × 10⁷ to 8 × 10⁷ cells/mL [7].
- Time-sensitive Freezing: The interval from cryoprotectant addition to the initiation of controlled-rate freezing should be strictly limited to ≤120 minutes to maintain cell viability [7].
- Controlled-Rate Freezing: Use a validated freezing profile. For example, the Thermo Profile 4 system is cited for preventing damaging ice crystal formation [7].
- Storage: Transfer frozen bags to the vapor phase of liquid nitrogen for long-term storage. Stability is validated for at least 24 months, with ongoing studies supporting up to 48 months [47].
Protocol: Thawing and Quality Assessment of Cryopreserved Leukapheresis
A standardized thawing process is crucial for maximizing cell recovery and function.
- Objective: To rapidly thaw cryopreserved leukapheresis material with high recovery and validate its quality for manufacturing.
Materials:
- Pre-warmed water bath (37°C)
- Thawing medium (e.g., complete media with DNase)
- Centrifuge
- Trypan Blue or automated cell counter
- Flow cytometer with antibody panels (CD3, CD4, CD8, CD19, CD56, etc.)
Method:
- Rapid Thaw: Remove the cryobag from LN₂ and immediately thaw in a 37°C water bath with gentle agitation until only a small ice crystal remains.
- Dilution & Washing: Immediately transfer the cell suspension into a pre-warmed, large volume of thawing medium. DNase can be added to prevent clumping from released DNA.
- Centrifugation: Centrifuge the cell suspension to remove the cryoprotectant and cell debris. Resuspend the pellet in appropriate culture medium.
- Quality Assessment:
- Viability & Recovery: Assess using Trypan Blue exclusion. Acceptable post-thaw viability is typically ≥80% [47], with optimized processes achieving ≥90% [7]. Calculate percentage recovery relative to pre-freeze cell count.
- Phenotyping: Perform flow cytometry to confirm the proportions of key immune cells (T, B, NK cells) and critical T-cell subsets (e.g., naïve and central memory T cells). The profile should be comparable to pre-freeze analysis [7] [46].
The Scientist's Toolkit: Essential Reagents and Materials
Successful implementation of cryopreserved leukapheresis in manufacturing relies on specific, high-quality reagents and systems.
Table 2: Key Research Reagent Solutions for Cryopreserved Leukapheresis Workflows
| Item | Function/Application | Examples / Key Specifications |
|---|---|---|
| Cryoprotectant | Protects cells from ice crystal damage during freeze-thaw cycles. | CryoStor (protein-free, serum-free) or CS10 (10% DMSO). 5% DMSO is a validated concentration for leukopaks [47]. |
| Closed-System Cell Processor | Automated, sterile processing and formulation of leukapheresis product. | Reduces operator error and contamination risk, standardizes the centrifugation and formulation steps [7]. |
| Controlled-Rate Freezer | Enables reproducible freezing with optimized cooling profiles. | Thermo Profile 4 system. Critical for achieving high post-thaw viability [7]. |
| Apheresis System | Initial collection of leukocytes from the patient/donor. | Spectra Optia. Provides automated, continuous-flow centrifugation for high-yield, high-purity leukapheresis collections [49] [18]. |
| Cell Counting & Viability Assay | Quantifying cell number and assessing post-thaw health. | Trypan Blue exclusion or automated cell counters. |
| Flow Cytometry Panels | Comprehensive immunophenotyping of pre- and post-thaw products. | Antibodies against CD3, CD4, CD8, CD45RO, CCR7 to assess T-cell subsets and differentiation status [7] [45]. |
The collective evidence strongly supports cryopreserved leukapheresis as a robust and superior logistical solution for scalable autologous cell therapy manufacturing. While fresh products retain a marginal advantage in initial viability, cryopreservation delivers critical operational benefits: unmatched flexibility in scheduling, enhanced supply chain resilience against delays, and the ability to implement distributed manufacturing models [7] [6]. Furthermore, comprehensive comparability studies confirm that CAR-T cells manufactured from cryopreserved starting material exhibit equivalent expansion, phenotype, and most importantly, cytotoxicity and clinical outcomes [7] [6] [48].
For successful implementation, research and development teams should:
- Invest in Protocol Standardization: Establish and rigorously validate in-house protocols for cryopreservation and thawing, focusing on controlling critical process parameters like freeze-rate and DMSO concentration.
- Prioritize Supply Chain Partnerships: Collaborate with suppliers and CDMOs that have expertise in cryopreservation logistics, including validated cold-chain storage and shipping capabilities [47].
- Adopt a Proactive Cell Banking Strategy: For autologous therapies, consider the strategic advantage of collecting and cryopreserving a patient's leukapheresis material during a healthier disease stage, preserving T-cell fitness for future manufacturing [45].
The transition to cryopreserved leukapheresis is a pivotal step toward more reliable, scalable, and globally accessible cell therapies.
The success of autologous cell therapies, particularly chimeric antigen receptor (CAR) T-cell treatments, is fundamentally dependent on the initial collection of a sufficient number of lymphocytes via leukapheresis. For heavily pre-treated patients with advanced B-cell malignancies, this first step presents a significant clinical challenge. These patients often exhibit profound lymphopenia and compromised immune function as a consequence of their disease and prior therapies, which can critically impair lymphocyte collection efficiency (CE) and jeopardize subsequent manufacturing success. This Application Note details evidence-based strategies and standardized protocols to optimize lymphocyte collection from this vulnerable patient population, ensuring reliable starting material for cell therapy manufacturing.
Quantitative Analysis of Key Challenge Factors
Understanding the patient-specific and procedural factors that influence collection success is the first step in developing a mitigation strategy. The following table summarizes the impact of key variables on lymphocyte collection efficiency (CE), based on retrospective clinical analyses.
Table 1: Factors Affecting Lymphocyte Collection Efficiency (CE) in Heavily Pre-Treated Patients
| Factor | Impact on CE | Supporting Data |
|---|---|---|
| Disease Type | Appreciably associated with low CE (<40%) [50]. | Diagnosis of Acute Lymphocytic Leukemia (ALL) was associated with lower CE compared to Non-Hodgkin Lymphoma (NHL) or Chronic Lymphocytic Leukemia (CLL) (OR for NHL vs. ALL: 0.20, p=0.009) [50]. |
| Patient Age | Advancing age is a negative predictor [50]. | Every 10-year increase in age was associated with higher odds of low CE (OR=1.51, p=0.034) [50]. |
| Pre-Apheresis Platelet Count | Higher counts are associated with lower CE [50]. | Every 10 × 103/𝜇L increase in pre-collection platelets was associated with higher odds of low CE (OR=1.07, p=0.005) [50]. |
| Pre-Apheresis Absolute Lymphocyte Count (ALC) | A strong indicator of final CD3+ yield [51]. | Pre-collection ALC and CD3+ count significantly correlate with apheresis product CD3+ count (p<0.0001). Successful collection is feasible even with ALC < 100/μL [51]. |
| Prior Therapies | Heavy pretreatment contributes to low blood counts, but does not preclude successful collection [50] [51]. | Studies included patients with advanced, relapsed/refractory diseases who had received multiple lines of therapy, including prior transplants [50]. |
The workflow below illustrates the strategic approach to addressing these challenges, from patient assessment to the final collection product.
Figure 1: Strategic Workflow for Optimizing Lymphocyte Collection from Challenging Patients
Pre-Apheresis Optimization Strategies
Patient Assessment and Timing
A comprehensive pre-collection evaluation is crucial. While a low pre-apheresis ALC is common, it should not be considered an absolute contraindication for collection, as studies confirm that CD3+ T cells can still be successfully harvested even with ALC < 100/μL [51]. The key is to schedule leukapheresis at a point of relative medical stability, avoiding periods of active infection or rapid disease progression. Furthermore, integrating a cryopreservation strategy from the outset provides critical flexibility, allowing collection to occur when the patient's health is optimal, decoupling the apheresis procedure from the manufacturing schedule [52].
Strategic Protocol Selection
The choice of apheresis platform and protocol can influence product quality. Both the COBE Spectra and Spectra Optia platforms have been successfully used for lymphocyte collections [50]. On the Spectra Optia, the Continuous Mononuclear Cell (CMNC) protocol is generally preferred. Critical parameters to monitor and optimize include:
- Anticoagulant: Acid Citrate Dextrose A (ACD-A) is standard. The inlet-to-AC ratio should be maintained, typically at 15:1 [50] [51].
- Flow Rates: Inlet flow rates should be kept below 100 mL/min to ensure efficiency and patient safety [50].
- Processed Volume: The goal should be to process 2-4 times the patient's total blood volume (TBV). Under-processing (low total volume) has been associated with out-of-specification final products due to low cell viability [51].
Detailed Experimental Protocol for Leukapheresis
Title: Standardized Protocol for Mononuclear Cell Collection from Heavily Pre-Treated Patients
Objective: To reliably collect a sufficient number of MNCs from patients with low peripheral blood counts for subsequent CAR-T cell manufacturing.
Materials & Equipment:
- Apheresis system (COBE Spectra or Spectra Optia with CMNC protocol)
- ACD-A anticoagulant
- Access kit (peripheral 16-18G needles or central venous catheter)
- Blood collection bags
- CBC analyzer with differential (e.g., Sysmex)
Pre-Procedure Steps:
- Patient Consent and Assessment: Obtain informed consent. Assess venous access; place a central catheter if peripheral access is inadequate.
- Pre-Apheresis Labs: Within 24 hours, obtain a complete blood count (CBC) with differential, including ALC and platelet count.
Procedure Steps:
- System Priming: Prime the apheresis system and circuit according to manufacturer instructions using ACD-A.
- Patient Connection: Establish venous access and connect the patient to the system.
- Parameter Setup:
- Set ACD-A to blood ratio to 15:1 to 20:1.
- Set inlet flow rate to 50-100 mL/min, adjusting for patient tolerance.
- Set collect pump flow rate to 1-2 mL/min [50].
- Processing: Process a total volume of 2-4 times the patient's TBV. The typical total processed volume is ~12 Liters [50].
- Monitoring: Monitor the patient throughout the procedure for signs of hypocalcemia or other adverse events.
- Product Collection: Upon completion, clamp and seal the product bag. Disconnect the patient.
Post-Procedure Steps:
- Product Analysis: Perform a CBC with differential on the apheresis product to determine total nucleated cell count and lymphocyte count.
- Calculate Collection Efficiency (CE): Use the formula: Lymphocyte CE (%) = [ (product lymphocyte count × product volume) / (process blood volume × average peripheral blood lymphocyte count) ] × 100 where the average peripheral blood lymphocyte count = (pre-count + post-count) / 2 [50].
- Product Handling: If not processing immediately, cryopreserve the leukapheresis product using a standardized, controlled-rate freezing protocol.
Post-Collection and Manufacturing Strategies
Cryopreservation as a Strategic Tool
The use of cryopreserved leukapheresis material is a validated strategy to overcome logistical and clinical hurdles. It provides flexibility in scheduling, allows for manufacturing buffer times, and enables collection when the patient's T-cell fitness is optimal [52]. A standardized, closed, and automated process for cryopreservation is critical for success.
Table 2: Key Parameters for Cryopreservation of Leukapheresis Material
| Parameter | Target Specification | Function/Rationale |
|---|---|---|
| Cryoprotectant | CS10 (10% DMSO) | Clinical-grade; ensures consistent cryoprotection while minimizing osmotic shock [7]. |
| Cell Concentration | 5 × 107 – 8 × 107 cells/mL | Optimizes recovery and post-thaw viability; accommodates high-density requirements [7]. |
| Freezing Timeframe | ≤ 120 minutes from cryoprotectant addition to freezing | Prevents ice crystal formation and maintains cell viability [7]. |
| Post-Thaw Viability | ≥ 90% | Critical Quality Attribute (CQA) indicating successful preservation and fitness for manufacturing [7]. |
Gentle Cell Handling
Lymphocytes are fragile, and rough handling can induce activation, cell death, or genetic changes, compromising their therapeutic potential [53]. To maintain viability and functionality:
- Use slow and controlled pipetting.
- Avoid unnecessary vortexing, shaking, or high-speed centrifugation.
- Minimize the time cells spend outside their optimal environment (e.g., room temperature).
- Consider gentle cell separation technologies, such as buoyancy-activated cell sorting (BACS) with microbubbles, which minimizes mechanical stress compared to column-based methods [53].
The Scientist's Toolkit: Essential Reagent Solutions
Table 3: Key Research Reagents and Materials for Lymphocyte Collection and Processing
| Reagent/Material | Function/Application | Example |
|---|---|---|
| ACD-A Anticoagulant | Prevents coagulation during apheresis; maintains cell viability. | Terumo BCT systems [50] [51]. |
| Lymphocyte Cryopreservation Medium | Protects cells from freezing-induced damage. | CS10 (10% DMSO formulation) [7]. |
| Closed-System Automated Processing Platform | Standardizes cryopreservation, reduces variability, and ensures aseptic processing. | IntegriCellTM Platform [54]. |
| Gentle Cell Isolation Kits | Isolates target cell populations (e.g., T cells, NK cells) with minimal activation and loss. | Microbubble-based negative selection kits [53]. |
| GMP-Grade Cytokines | Expands and maintains T-cell or NK-cell cultures post-thaw. | IL-2, IL-15 [55]. |
| Cell Culture Surfaces | Influences cell proliferation and cluster formation; charged surfaces can enhance NK cell expansion. | Poled β-PVDF films [55]. |
Quantitative Comparison of Leukapheresis Performance Across Engineering Platforms
Table 1: Performance of Cryopreserved Leukapheresis in Viral vs. Non-Viral CAR-T Manufacturing Platforms [26] [7]
| Performance Metric | Non-Viral CAR-T Platform | Lentiviral CAR-T Platform | Fast CAR-T Platform |
|---|---|---|---|
| Post-Thaw Viability | ≥ 90% | ≥ 90% | ≥ 90% |
| Cell Expansion | Comparable to fresh leukapheresis | Comparable to fresh leukapheresis | Comparable to fresh leukapheresis |
| CAR+ Cell Proportion | Comparable to fresh leukapheresis | Comparable to fresh leukapheresis | Comparable to fresh leukapheresis |
| Cytotoxicity | Comparable to fresh leukapheresis | Comparable to fresh leukapheresis | Comparable to fresh leukapheresis |
| Cell Phenotype | Comparable phenotypic profile | Comparable phenotypic profile | Comparable phenotypic profile |
Table 2: Key Cryopreservation Parameters for Protocol Standardization [26]
| Process Parameter | Specification |
|---|---|
| Target Cell Concentration | 5–8 × 107 cells/mL |
| Final DMSO Concentration | 7.5% – 10% (v/v) |
| Formulation Volume per Bag | 20 mL |
| Formulation Duration | ≤ 120 minutes |
| Freezing Protocol | Controlled-rate freezing (e.g., Thermo Profile 4) |
Experimental Protocols for Platform Compatibility
Protocol 1: Standardized Cryopreservation of Leukapheresis Products
Objective: To prepare a cryopreserved leukapheresis product with high post-thaw viability and compatibility with downstream genetic engineering platforms [26] [7].
Materials:
- Leukapheresis product (e.g., ~1 mL per 1x109 cells, hematocrit 5-10%)
- Clinical-grade cryoprotectant (e.g., CS10)
- Centrifuge with temperature control
- Controlled-rate freezer
- Cryogenic bags
Method:
- Initial Processing: Perform a centrifugation-based procedure on the leukapheresis product to reduce non-target cellular impurities, such as residual red blood cells and platelets [26] [7].
- Formulation:
- Resuspend the cell pellet in the cryoprotectant to achieve the target cell concentration of 5–8 × 107 cells/mL.
- Ensure the final DMSO concentration is between 7.5% and 10%.
- Dispense the cell suspension into cryogenic bags, with a recommended volume of 20 mL per bag [26].
- Time-Sensitive Freezing:
- Initiate controlled-rate freezing within 120 minutes of cryoprotectant addition.
- Use a validated freezing profile (e.g., Thermo Profile 4) to prevent ice crystal formation [26].
- Storage: Transfer cryopreserved bags to long-term storage in the vapor phase of liquid nitrogen.
Protocol 2: CAR-T Generation Using the PiggyBac Transposon System from Cryopreserved PBMCs
Objective: To generate functional CAR-T cells from cryopreserved peripheral blood mononuclear cells (PBMCs) using a non-viral PiggyBac transposon electroporation system [45].
Materials:
- Cryopreserved PBMCs
- PiggyBac transposon plasmid carrying the CAR construct
- PiggyBac transposase plasmid
- Electroporation system
- T-cell activation beads (e.g., CD3/CD28)
- Cell culture media and cytokines (e.g., IL-2)
Method:
- Thaw and Enrich:
- Rapidly thaw cryopreserved PBMCs in a 37°C water bath.
- Isulate T cells using CD4/CD8 magnetic bead enrichment. Assess CD3+ purity, which should be unaffected by long-term cryopreservation [45].
- Activate:
- Activate enriched T cells with activation beads for 48 hours [45].
- Electroporation:
- Electroporate the activated T cells with the PiggyBac CAR transposon and transposase plasmids [45].
- Culture and Expand:
- Culture the electroporated cells for approximately 11 days to generate CAR-T cells.
- Monitor cell viability, expansion, and phenotype throughout the culture process. The resulting CAR-T cells exhibit comparable expansion potential, phenotype, and cytotoxicity to those derived from fresh PBMCs [45].
Workflow Visualization
Cross-Platform Manufacturing Workflow
Post-Electroporation Recovery Optimization
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Leukapheresis-Based CAR-T Manufacturing [26] [45] [7]
| Reagent/Material | Function | Application Notes |
|---|---|---|
| Clinical-Grade Cryoprotectant (e.g., CS10) | Protects cells from ice crystal damage during freezing and thawing. | Contains 10% DMSO. Final concentration should be 7.5-10% in the formulated product [26] [7]. |
| PiggyBac Transposon System | Non-viral gene delivery; integrates large CAR gene constructs into T-cell genome. | Comprises transposon plasmid (carrying CAR) and transposase plasmid. Enables cost-effective manufacturing [45]. |
| Lentiviral Vectors | Viral gene delivery; stably integrates CAR gene into T-cell genome. | Commonly used second-generation vectors with safety modifications. Suitable for a wide range of T-cell subtypes [26] [7]. |
| T-Cell Activation Beads | Mimics antigen presentation to activate T cells prior to genetic modification. | Critical for successful transduction/transfection. Typically used for 48 hours pre-electroporation/transduction [45]. |
| Controlled-Rate Freezer | Ensures consistent, reproducible cooling rates during cryopreservation. | Prevents destructive ice crystal formation. Use of a standardized profile (e.g., Thermo Profile 4) is recommended [26]. |
Overcoming Hurdles: Practical Solutions for Leukapheresis Process Optimization
Cryopreservation of leukapheresis-derived starting material is a critical enabling technology for the scalable and distributed manufacturing of autologous cell therapies, such as Chimeric Antigen Receptor T-cell (CAR-T) treatments. This application note provides a detailed protocol and supporting data for standardizing the cryopreservation process to maximize post-thaw viability and functionality. By optimizing key parameters—including cryoprotectant composition, controlled-rate freezing, and thawing methods—researchers can achieve post-thaw viabilities consistently ≥90% and maintain critical T-cell functions essential for successful therapeutic outcomes. The procedures outlined herein are validated against current industrial practices and are designed to integrate seamlessly into research and clinical-scale cell therapy workflows.
The transition from fresh to cryopreserved leukapheresis material decouples cell collection from manufacturing, overcoming significant logistical hurdles in autologous cell therapy production. Cryopreservation provides flexibility in clinical scheduling, allows for quality control testing on the starting material, and enables the banking of cells when the donor's T-cells are healthiest [46]. While a historical concern has been that the freeze-thaw process diminishes cell quality, recent industrial studies have demonstrated that with optimized and standardized protocols, cryopreserved leukapheresis performs comparably to fresh material in CAR-T manufacturing, supporting equivalent expansion, phenotype, and cytotoxicity [7] [52]. The success of commercial products like tisagenlecleucel (Kymriah), manufactured from cryopreserved leukapheresis, validates this approach in a global, multicenter context [52] [46]. This document details the core principles and standardized methods to achieve such successful outcomes.
Scientific Principles and Rationale
Mechanisms of Cryoprotection
Cryopreservation inflicts two primary types of stress on cells: the formation of intracellular ice crystals that mechanically disrupt membranes, and lethal increases in solute concentration in the remaining liquid phase as water freezes [56]. Cryoprotective Agents (CPAs) are used to mitigate these damages.
- Permeating Cryoprotectants: Small molecules like Dimethyl Sulfoxide (DMSO), glycerol, and ethylene glycol readily cross cell membranes. They depress the freezing point of water and reduce the amount of water available to form ice by forming hydrogen bonds, thereby promoting an amorphous, glass-like state (vitrification) instead of crystalline ice [56]. DMSO, typically used at a 10% concentration, also increases membrane porosity, facilitating water efflux during freezing [56].
- Non-Permeating Cryoprotectants: Larger molecules like sucrose, trehalose, and hydroxyethyl starch act extracellularly. They induce osmotic dehydration of the cell prior to freezing, reducing the chance of intracellular ice formation, and also contribute to extracellular vitrification [56]. Trehalose, a natural disaccharide produced by some freeze-tolerant organisms, offers exceptional stability due to its non-reducing glycosidic bond [56].
The Critical Role of Cooling and Thawing Rates
The rate of temperature change is a primary determinant of cell survival.
- Cooling Rate: A slow, controlled cooling rate (approximately -1°C/min) is optimal for many nucleated cells, including lymphocytes and mesenchymal stem cells [56] [57] [58]. This rate allows sufficient time for water to exit the cell before intracellular freezing occurs, minimizing mechanical damage from ice crystals.
- Thawing Rate: Rapid thawing is generally recommended to minimize the time cells are exposed to the cytotoxic effects of concentrated solutes and to prevent ice recrystallization during the warming phase [57] [58]. However, recent evidence indicates that for T-cells cooled at -1°C/min, the warming rate has a minimal impact on viability. The critical interaction occurs with rapid cooling; cells cooled rapidly (-10°C/min) show significant viability loss when thawed slowly, an effect correlated with observed ice recrystallization [59].
Standardized Protocol for Leukapheresis Cryopreservation
The following protocol is optimized for the cryopreservation of leukapheresis products for subsequent T-cell therapy manufacturing.
Pre-processing and Cryoprotectant Formulation
- Objective: To concentrate leukocytes and formulate them with a protective medium for freezing.
- Materials:
- Leukapheresis product
- Cryoprotectant medium: Clinical-grade CryoStor CS10 (10% DMSO) is recommended [7] [57]. Alternatively, 10% DMSO in human platelet lysate (HPL) or fetal bovine serum (FBS) can be used, though defined, serum-free media are preferred for clinical applications [60].
- Sterile centrifuge tubes and pipettes
- Biologic safety cabinet
- Method:
- Initial Processing: Perform a soft centrifugation step on the leukapheresis product to reduce non-cellular impurities like red blood cells and platelets, which can interfere with consistent cryoprotection [7].
- Cell Counting: Determine total nucleated cell count and viability (e.g., via Trypan Blue exclusion). Ensure pre-freeze viability is high (>95%) [46].
- Formulation: Resuspend the cell pellet in pre-chilled (4°C) cryoprotectant medium to a final concentration of 5-8 x 10^7 cells/mL [7]. Gently mix to ensure homogeneity.
- Aliquoting: Dispense the cell suspension into appropriate cryogenic vials or bags. For bags, a typical formulation volume is 20 mL, ensuring a total of ≥1 x 10^9 cells per bag as a Critical Quality Attribute (CQA) [7].
Table 1: Key Pre-Cryopreservation Parameters and Specifications
| Parameter | Target Specification | Rationale |
|---|---|---|
| Pre-freeze Viability | >95% [46] | High starting viability is critical for post-thaw recovery. |
| Final Cell Concentration | 5-8 x 10^7 cells/mL [7] | Prevents osmotic stress from high density and avoids excessive storage space. |
| Cryoprotectant | 10% DMSO (e.g., CryoStor CS10) [7] | Standard, effective concentration for immune cells; ensures vitrification. |
| Processing Time | ≤120 minutes from CPA addition to freezing [7] | Limits exposure to potentially toxic CPA at room temperature. |
Controlled-Rate Freezing and Storage
- Objective: To freeze cells at an optimal, reproducible rate and transfer them to long-term storage.
- Materials:
- Controlled-rate freezer or a passive freezing container (e.g., CoolCell or Mr. Frosty)
- -80°C freezer
- Liquid nitrogen storage tank (vapor or liquid phase)
- Method:
- Cooling: Place filled cryocontainers into a controlled-rate freezer programmed to cool at -1°C/min from +4°C to at least -40°C, before transferring to -80°C [57] [58]. If using a passive freezing container, place it directly in a -80°C freezer for a minimum of 4 hours (or overnight), which approximates the -1°C/min cooling rate [58] [60].
- Long-term Storage: After 24 hours, promptly transfer cryocontainers to the vapor phase of liquid nitrogen (below -135°C) for stable long-term storage [57] [61]. Storage at -80°C is acceptable only for short periods (up to one week), as viability declines over time at this temperature [58].
Thawing and Post-Thaw Assessment
- Objective: To rapidly recover cells with maximal viability and functionality.
- Materials:
- 37°C water bath or bead bath (cGMP-compliant closed systems are available)
- Pre-warmed complete culture medium (e.g., XVIVO-15 + 5% AB serum)
- Centrifuge
- Method:
- Thawing: Rapidly thaw cryovials by gently swirling in a 37°C water bath until only a small ice crystal remains (approximately 1-2 minutes) [58]. For cryobags, use a validated bag-thawing system.
- Dilution & Washing: Immediately upon thawing, dilute the cell suspension drop-wise with a large volume (e.g., 10-fold) of pre-warmed medium. This step rapidly reduces the concentration of DMSO, mitigating its toxicity [57] [58].
- Centrifugation: Centrifuge the diluted cell suspension at 300-500 x g for 5 minutes to remove the cryoprotectant-containing supernatant [58]. This "indirect" revival method is standard, though some protocols for primary cells suggest "direct" seeding after dilution may be effective for certain cell types [60].
- Assessment: Resuspend the cell pellet in fresh medium. Perform a cell count and viability assay (e.g., Trypan Blue, flow cytometry with viability dyes). Assess recovery and phenotype via flow cytometry (e.g., CD3+, CD4+, CD8+ percentages).
Table 2: Post-Thaw Quality Control Acceptance Criteria
| Quality Attribute | Target Post-Thaw Performance | Measurement Method |
|---|---|---|
| Viability | ≥90% [7] [46] | Trypan Blue exclusion or automated cell counter |
| Cell Recovery | ≥85% of pre-freeze nucleated cell count [46] | Cell counting pre-freeze and post-thaw |
| T-cell Purity (CD3+) | Proportion comparable to pre-freeze profile [7] | Flow cytometry |
| Functionality | Successful expansion and CAR-T manufacture [7] [52] | In vitro proliferation and cytotoxicity assays |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Leukapheresis Cryopreservation
| Item | Function/Application | Example Products/Notes |
|---|---|---|
| Cryoprotectant Medium | Protects cells from ice crystal damage and osmotic shock during freeze-thaw. | CryoStor CS10 (cGMP, defined); FBS + 10% DMSO (research-grade) [57] [60] |
| Controlled-Rate Freezer | Provides a reproducible, optimal cooling rate (-1°C/min). | Programmable freezer; CoolCell (passive, isopropanol-free) [57] [58] |
| Cryogenic Storage Vials/Bags | Secure, sterile container for long-term storage in LN₂. | Internal-threaded cryovials; cGMP cryobags [57] |
| Liquid Nitrogen Storage | Long-term storage at <-135°C to halt all metabolic activity. | Vapor phase storage is recommended to prevent contamination [57] [61] |
| Thawing Device | Enables rapid, consistent, and sterile thawing. | cGMP-compliant water bath or dry-thawing instrument (e.g., ThawSTAR) [57] |
Experimental Workflow and Cryopreservation Impact
The following diagram illustrates the complete workflow for processing cryopreserved leukapheresis into a final cell therapy product, highlighting the critical parameters and quality checkpoints.
The standardization of cryopreservation protocols for leukapheresis material is no longer a technical barrier but a fundamental enabler for robust and scalable cell therapy manufacturing. By adhering to optimized parameters for cryoprotectant proportion (10% DMSO), cooling rate (-1°C/min), and post-thaw handling (rapid thaw with immediate dilution), researchers and developers can ensure high viability and functional recovery of T-cells. The implementation of these detailed protocols supports the creation of a resilient supply chain, decouples manufacturing logistics, and ultimately contributes to the reliable delivery of effective autologous cell therapies to patients.
In autologous CAR-T cell therapy, the leukapheresis material serves as the foundational starting material whose quality directly impacts the success of subsequent manufacturing steps and the final therapeutic efficacy. Substantial variability in CD3+ cell yield and composition presents a critical challenge for researchers and drug development professionals working to establish robust, scalable processes [62]. This variability stems from multiple factors, including patient-specific characteristics, differences in leukapheresis procedures, and the specific requirements of different CAR-T products [41]. A European survey conducted by the T2Evolve Consortium highlighted the urgent need to standardize analytical methods for characterizing apheresis materials, drug products, and post-infusion patient samples to enable comparability across clinical trials [62]. This application note provides detailed protocols and analytical frameworks to address these variability challenges within leukapheresis processing for autologous cell therapy research.
Quantitative Analysis of CD3+ Cell Yield Variability
Recent clinical data reveals significant differences in CD3+ cell collection efficiency based on the targeted CAR-T product. A retrospective analysis of 80 lymphoma patients undergoing leukapheresis for three different CD19-targeted CAR-T products demonstrated substantial procedural variations impacting CD3+ yield [41].
Table 1: CD3+ Cell Yield Variability Across CAR-T Products
| CAR-T Product | Processing Blood Volume | Processing Time (minutes) | CD3+ Cell Yield (×10^9 cells) | Yield Standard Deviation | Cases Exceeding Estimated Volume for 5×10^9 CD3+ Cells |
|---|---|---|---|---|---|
| Axi-cel | 12 L | 240 | 3.04 | ± 3.04 | 45.0% |
| Liso-cel | 12 L | 204 | 1.54 | ± 1.54 | 17.2% |
| Tisa-cel | 10 L | 203 | 1.59 | ± 1.59 | 16.1% |
Data adapted from Cytotherapy, 2025 [41]
The axi-cel group required significantly larger processing blood volumes and longer processing times, resulting in higher but more variable CD3+ cell yields compared to liso-cel and tisa-cel [41]. Notably, 45% of axi-cel procedures exceeded estimated blood volumes needed to achieve the target CD3+ cell count of 5×10^9 cells, highlighting the challenge of standardizing collection protocols across products [41].
Standardized Leukapheresis Processing Protocol
Pre-apheresis Patient Assessment
- Complete Blood Count (CBC) with Differential: Perform within 24 hours prior to apheresis to assess absolute lymphocyte count (ALC) and CD3+ cell count
- CD3+ Cell Enumeration: Use flow cytometry to determine baseline CD3+ levels; optimal ALC >500/μL correlates with improved yield [41]
- Patient Clinical History Review: Document prior therapies, especially lymphotoxic treatments, and time since last therapy
Leukapheresis Procedure Specifications
- Collection Volume: Process 10-12 L of blood volume over 200-240 minutes, adjusted based on pre-apheresis CD3+ count [41]
- Anticoagulation: Use ACD-A anticoagulant at a ratio of 1:12 to 1:14 (ACD:whole blood)
- Inlet Flow Rate: Maintain 40-60 mL/min based on patient tolerance and access
- Collection Rate: Set interface control to collect 1-2 mL of concentrated leukocytes per minute
- Product Volume: Target 100-200 mL final collection volume
Quality Control Testing of Leukapheresis Product
Table 2: Acceptance Criteria for Leukapheresis Material
| Parameter | Target Value | Acceptance Range | Testing Method |
|---|---|---|---|
| Total Nucleated Cell Count | - | ≥2.0×10^9 cells | Hematology analyzer |
| Viability | ≥90% | ≥80% | Trypan blue exclusion/flow cytometry |
| CD3+ Cell Content | - | ≥1.0×10^9 cells | Flow cytometry |
| CD3+ % of Lymphocytes | ≥70% | ≥60% | Flow cytometry |
| Sterility | No growth | No growth | BacT/ALERT or equivalent |
| Endotoxin | <5.0 EU/kg | <5.0 EU/kg | LAL test |
Cryopreservation Protocol for Leukapheresis Products
Cryopreservation decouples leukapheresis from manufacturing, enabling centralized processing and quality verification. Standardized cryopreservation maintains cellular diversity and function critical for CAR-T manufacturing [7].
Cryopreservation Procedure
- Pre-processing: Centrifuge leukapheresis product at 400×g for 15 minutes to reduce red blood cells and platelets
- Cryoprotectant: Use CS10 (10% DMSO) cryoprotectant medium
- Cell Concentration: Adjust to 5×10^7 cells/mL in cryoprotectant medium
- Freezing Container: Use controlled-rate freezing container or programmed freezer
- Cooling Rate: -1°C per minute to -40°C, then -10°C per minute to -100°C
- Storage: Transfer to vapor phase liquid nitrogen (-135°C to -150°C) for long-term storage
Post-thaw Quality Assessment
Table 3: Cryopreserved Leukapheresis Quality Metrics
| Parameter | Pre-cryopreservation | Post-thaw | Performance vs. Fresh |
|---|---|---|---|
| Viability | 94.0–96.2% | 90.9–97.0% | Slightly lower (91.0% vs. 99.0% fresh) |
| CD3+ % | 41.2–56.5% | 42.0–51.2% | Comparable |
| Lymphocyte % | 66.6% | 66.6% | Higher than PBMCs (52.2%) |
| Formulation Time | 43–108 minutes | - | - |
Data adapted from Scientific Reports, 2025 [7]
Cryopreserved leukapheresis products maintain critical quality attributes, with post-thaw viability ≥90% and lymphocyte proportions significantly higher than cryopreserved PBMCs (66.6% vs. 52.2%), correlating with enhanced CAR-T manufacturing potential [7].
Yield-Driven Optimization Strategy
A yield-driven approach optimizes apheresis based on pre-procedure CD3+ counts and product-specific requirements [41]. This strategy reduces patient burden while ensuring adequate cell yield for manufacturing.
Processing Volume Calculation
- Step 1: Determine pre-apheresis peripheral blood CD3+ count (cells/μL)
- Step 2: Calculate estimated blood volume (EBV) needed to achieve target CD3+ yield:
- EBV (L) = Target CD3+ yield / (Pre-apheresis CD3+ count × Collection Efficiency)
- Collection efficiency typically ranges from 50-70% [41]
- Step 3: Adjust processing volume based on product-specific requirements:
- Axi-cel: Maximum 12 L processing volume
- Liso-cel/tisa-cel: 10-12 L processing volume
Experimental Workflow for Process Optimization
The following diagram illustrates the comprehensive workflow for managing leukapheresis variability from collection through manufacturing:
Leukapheresis Variability Management Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Reagents for Leukapheresis Processing and Analysis
| Reagent/Category | Specific Examples | Function/Application | Technical Notes |
|---|---|---|---|
| Cell Separation Media | Ficoll-Paque PLUS, Lymphoprep | Density gradient separation of PBMCs from leukapheresis product | Maintain at room temperature; avoid refrigeration |
| Cryopreservation Media | CS10, CryoStor CS10 | Cryoprotectant with 10% DMSO | Clinical grade; contain extracellular cryoprotectants |
| Viability Assays | Trypan blue, 7-AAD, Annexin V/PI | Assessment of cell viability and apoptosis | Use multiple methods for comprehensive assessment |
| Flow Cytometry Antibodies | Anti-CD3, CD4, CD8, CD45, CD19, CD56 | Immunophenotyping of lymphocyte subsets | Include viability dye to exclude dead cells |
| Cell Activation Reagents | Anti-CD3/CD28 beads, IL-2, IL-7, IL-15 | T-cell activation and expansion | Concentration optimization required for each donor |
| Cell Counting Platforms | Automated cell counters, Hemocytometers | Quantification of cell concentration and viability | Calibrate regularly; use duplicate measurements |
| Sterility Testing Kits | BacT/ALERT culture bottles, Mycoplasma detection | Microbial contamination screening | Incubate for 14 days for final assessment |
Analytical Methods for Composition Assessment
Standardized immunophenotyping is essential for characterizing leukapheresis products. The T2Evolve survey identified significant variability in analytical methods across European centers, underscoring the need for harmonization [62].
Comprehensive Immunophenotyping Panel
- T-cell Subsets: CD3, CD4, CD8, CD45RA, CD62L, CD95 (for naive, memory subsets)
- Activation Markers: CD25, CD69, HLA-DR
- Exhaustion Markers: PD-1, LAG-3, TIM-3
- B-cells and NK-cells: CD19, CD20, CD56, CD16
- Viability Stain: Fixable viability dye (e.g., Zombie dye)
Sample Processing Protocol
- Staining Volume: 100μL of cell suspension (1×10^6 cells)
- Antibody Incubation: 20 minutes at room temperature, protected from light
- Red Blood Cell Lysis: Use ammonium chloride or commercial lysing solution
- Fixation: 1-2% paraformaldehyde for intracellular staining
- Acquisition: Acquire within 24 hours on flow cytometer; minimum 10,000 CD3+ events
Advanced Approaches for Variability Mitigation
CRISPR-Based Screening for CAR-T Enhancement
The CELLFIE platform enables systematic discovery of genetic modifications that enhance CAR-T cell function despite variable starting material [63]. Genome-wide CRISPR screens in human primary CAR-T cells identified several gene knockouts that boost efficacy, including RHOG and FAS knockouts, which enhanced anti-tumor activity across multiple models and donors [63].
Universal CAR-T Platforms
Allogeneic UCAR-T cells derived from healthy donors offer an alternative approach to overcome limitations of autologous starting material [64]. Key engineering strategies include:
- TRAC Locus Disruption: Ablates endogenous TCR to prevent GvHD
- B2M Knockout: Reduces HLA class I expression to mitigate host rejection
- HLA-E Expression: Protects against NK cell-mediated lysis via "missing-self" response [64]
Managing product variability in leukapheresis requires a multifaceted approach combining yield-driven collection strategies, standardized cryopreservation protocols, and comprehensive analytics. Implementation of the frameworks and protocols detailed in this application note will enable researchers and drug development professionals to better control CD3+ cell yield and composition, ultimately enhancing the consistency and success of autologous CAR-T cell manufacturing. As the field advances, integrated approaches combining optimized leukapheresis with genetic engineering platforms like CELLFIE [63] and universal CAR-T strategies [64] will further address the challenges of product variability.
In autologous cell therapy research, the vein-to-vein time—the total duration from leukapheresis to the infusion of the final therapeutic product back into the patient—is a critical metric. It directly impacts patient outcomes, as prolonged ex vivo culture can lead to T-cell exhaustion and diminished therapeutic potency [65] [66]. Furthermore, extended and complex manufacturing processes are a primary driver of high Cost of Goods Sold (CoGS), creating significant barriers to the widespread adoption of life-saving therapies like Chimeric Antigen Receptor (CAR) T-cell treatments [67] [66].
This application note details the implementation of an integrated and automated platform that streamlines the entire CAR-T manufacturing workflow. By consolidating key unit operations into a single, closed system, this approach significantly shortens production timelines, reduces manual handling, and enhances process consistency, offering a viable path to more affordable and accessible autologous cell therapies.
Integrated Automated Platform for CAR-T Manufacturing
The cornerstone of this streamlined workflow is the integration of three critical CAR-T manufacturing steps—T-cell activation, lentiviral transduction, and cell expansion—onto a single, functionally closed, automated platform [65]. This 3-in-1 integration eliminates the need for multiple, open-process steps that are typical of traditional manual manufacturing.
Key Performance Data
The quantitative benefits of implementing this integrated platform are substantial, as shown in the table below.
Table 1: Performance Metrics of the Integrated 3-in-1 CAR-T Manufacturing Platform [65]
| Performance Metric | Traditional Manual Process | Integrated Automated Platform |
|---|---|---|
| Total Manufacturing Time | 2-3 weeks [66] | 7-8 days [65] |
| Transduction Efficiency | Baseline (Variable) | 2-fold increase [65] |
| CAR-T Cell Yield | Variable | >12 billion cells [65] |
| Key Process Features | Open, multi-step transfers; serum-containing media | Functionally closed, single system; serum-free culture conditions |
Impact on Vein-to-Vein Time and CoGS
Shortening the manufacturing timeline directly addresses two major challenges. Firstly, a reduced vein-to-vein time is clinically beneficial as it mitigates the risk of T-cell differentiation into less potent phenotypes and allows critically ill patients to receive treatment faster [66]. Secondly, process intensification through automation and integration substantially lowers CoGS by:
- Reducing Labor Costs: Minimizing the need for highly skilled manual labor and extensive cleanroom occupancy [68] [66].
- Lowering Facility Overhead: Utilizing closed systems can reduce the requirement for stringent and expensive cleanroom environments [68].
- Improving Process Consistency: Automation reduces human error and batch-to-batch variability, leading to higher product success rates and less material waste [68] [65].
- Eliminating Cryopreservation: In some decentralized models, rapid manufacturing allows for the use of fresh products, removing the costs associated with cryopreservation and complex frozen logistics [66].
Detailed Protocol: 3-in-1 CAR-T Cell Manufacturing on an Automated Hollow-Fiber Platform
The following protocol is adapted from a published methodology demonstrating the consolidation of activation, transduction, and expansion on the Quantum Cell Expansion System [65].
Starting Material and Preparation
- Leukapheresis Product: Obtain a leukopak via leukapheresis using a system such as the Spectra Optia Apheresis System. Process the leukopak to isolate Peripheral Blood Mononuclear Cells (PBMCs) using a cell wash and concentration system (e.g., Rotea Counterflow Centrifugation System or a partner technology) [68] [65].
- Reagents: Use serum-free, GMP-grade cell culture medium and supplements. Prepare a lentiviral vector stock at the required titer.
- Equipment Setup: Assemble the single-use set for the hollow-fiber bioreactor according to the manufacturer's instructions. Prime the system with culture medium.
Experimental Workflow
The following diagram illustrates the consolidated, automated workflow that replaces multiple traditional manual steps.
Step-by-Step Methodology
System Loading:
- Resuspend the PBMC pellet in pre-warmed, serum-free medium.
- Load the cell suspension into the prepared hollow-fiber bioreactor system.
- Initiate circulation according to the system's standard protocol.
Integrated Activation, Transduction, and Expansion:
- Activation: Introduce GMP-grade T-cell activation beads or reagents directly into the bioreactor. Allow the activation to proceed for 24 hours within the system.
- Transduction: Without opening the system, administer the calculated volume of lentiviral vector directly into the circulating medium. The hollow-fiber design enhances cell-vector contact, improving transduction efficiency.
- Expansion: Allow the culture to continue in perfusion mode for a total of 7-8 days. The system automatically manages gas exchange, nutrient delivery, and waste removal. Monitor cell density and viability daily using integrated or off-line sampling ports.
Harvest:
- Once the target cell count is achieved (e.g., >12 billion CAR-T cells), initiate the harvest procedure as per the system's protocol.
- Collect the final cell product into a sterile, closed bag.
- Perform quality control assays, including cell count, viability, transduction efficiency, and sterility testing.
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful implementation of this automated protocol relies on key reagents and instruments designed for robustness and GMP compliance.
Table 2: Key Research Reagent Solutions for Automated CAR-T Manufacturing
| Item | Function | Application Note |
|---|---|---|
| Spectra Optia Apheresis System | Automated collection of mononuclear cells from patient blood via leukapheresis. | Provides a consistent and high-quality leukapheresis starting material [65]. |
| Quantum Cell Expansion System | A closed, hollow-fiber bioreactor for automated cell culture, expansion, and now integrated activation/transduction. | Enables the consolidated 3-in-1 workflow and explosive T-cell expansion in a GMP-friendly system [65]. |
| Gibco CTS Rotea Counterflow Centrifugation System | A closed system for cell processing, including PBMC separation, washing, and concentration. | Used for preparing leukopaks before loading onto the Quantum system; offers high cell recovery and viability [68]. |
| GMP-Grade Leukopaks (e.g., LeukoCer) | Commercially available, high-quality, leukapheresis-derived starting materials. | Useful for process development and standardization, ensuring a reliable and consistent cell source [69]. |
| Serum-Free, GMP-Grade Cell Culture Medium | A chemically defined medium for the ex vivo culture of T-cells. | Eliminates lot-to-lot variability and contamination risks associated with fetal bovine serum, crucial for a standardized process [68] [65]. |
The integration of activation, transduction, and expansion into a single, automated hollow-fiber platform presents a transformative strategy for autologous CAR-T manufacturing. This protocol demonstrates that it is feasible to significantly reduce vein-to-vein time to under 8 days while simultaneously improving critical process metrics like transduction efficiency and cell yield. By transitioning from manual, open processes to standardized, closed automation, researchers and developers can achieve the dual objectives of enhancing product quality and substantially lowering CoGS. This approach is a critical step toward making personalized cell therapies more scalable, reliable, and accessible to a broader patient population.
In autologous cell therapy research, the leukapheresis collection is the foundational raw material that determines the success of downstream manufacturing. An optimal leukapheresis product is essential for cell therapies like Chimeric Antigen Receptor (CAR) T-cells, where the quality and T-cell content of the starting material directly impact manufacturing success and patient outcomes [70]. Implementing rigorous, standardized quality control (QC) checks post-collection is therefore not merely a regulatory formality but a critical step to ensure product consistency, safety, and efficacy.
This document provides detailed application notes and protocols for core QC checks—sterility, cell count, and viability—specifically within the context of leukapheresis processing for autologous therapies. The procedures outlined are designed to be integrated into a research Quality Management System, providing a foundation for robust process control and reliable data generation.
Critical Quality Attributes (CQAs) and Testing Framework
Post-collection, the leukapheresis product must be evaluated against several Critical Quality Attributes (CQAs). These metrics provide an immediate assessment of the sample's suitability for further processing and cryopreservation.
Table 1: Key Quality Control Metrics for Leukapheresis Products
| Quality Attribute | Target Specification | Testing Method | Significance |
|---|---|---|---|
| Total Nucleated Cell Count | Varies by protocol & patient | Automated cell counter | Ensures sufficient cellular yield for manufacturing [7] |
| Cell Viability | ≥ 90% (post-thaw) [7] | Trypan blue exclusion/flow-based assays | Induces cellular health and fitness for culture |
| CD3+ T-cell Proportion | ~40-60% (varies) [7] | Flow cytometry | Confirms presence of key effector cells for CAR-T manufacturing |
| Sterility | No microbial growth | Automated culture systems (e.g., BACTEC) [71] | Ensures product safety and prevents culture contamination |
| Cell Composition (Phenotyping) | Lymphocyte-rich | Flow cytometry | Higher lymphocyte proportion in leukapheresis vs. PBMCs enhances CAR-T potential [7] |
A recent survey of hematopoietic stem cell transplantation centers highlights significant heterogeneity in processing practices, underscoring the need for standardization. For instance, 28.6% of patients did not undergo post-thaw quality assessment, creating a major gap in quality assurance [72]. Adhering to a standardized QC framework mitigates such risks.
Detailed Experimental Protocols
Protocol for Cell Count and Viability Assessment
Principle: Cell count and viability are determined using a dual-fluorescence staining kit (e.g., acridine orange/propidium iodide) on an automated cell counter. Viable cells with intact membranes exclude propidium iodide, while non-viable cells with compromised membranes incorporate it.
Materials:
- Automated cell counter (e.g., NC-200)
- Fluorescence cell counting slides
- Dual-fluorescence viability dye (AO/PI)
- Phosphate-buffered saline (PBS)
- Microcentrifuge tubes
Procedure:
- Sample Preparation: Thaw the leukapheresis sample rapidly in a 37°C water bath if frozen. For fresh samples, proceed directly. Mix the sample gently to ensure homogeneity.
- Dilution and Staining: Dilute an aliquot of the cell suspension in PBS to fall within the instrument's linear range (e.g., 2x10^5 - 2x10^6 cells/mL). Mix 10 µL of the diluted cell suspension with 10 µL of AO/PI stain in a microcentrifuge tube.
- Incubation and Loading: Incubate the mixture for 5-10 minutes at room temperature, protected from light. Load 10 µL of the stained mixture into a counting slide.
- Analysis: Insert the slide into the automated cell counter and initiate the analysis. Record the following parameters:
- Total Nucleated Cell Count (cells/mL)
- Total Viable Cell Count (cells/mL)
- Percentage Viability
Acceptance Criteria: A post-thaw viability of ≥ 90% is a benchmark for cryopreserved leukapheresis products intended for CAR-T manufacturing [7]. Samples with viability below this threshold may not perform optimally in downstream manufacturing steps.
Protocol for Sterility Testing Using Automated Systems
Principle: This method uses automated culture systems to detect microbial contamination (bacteria and fungi) through monitoring of CO₂ production, which is a byproduct of microbial metabolism.
Materials:
- Automated sterility testing system (e.g., BACTEC)
- BACTEC Peds Plus T/F aerobic and anaerobic culture bottles
- Class II Biological Safety Cabinet
- Sterile syringes and needles
Procedure:
- Sample Preparation: Work under aseptic conditions within a Biological Safety Cabinet. Thaw the leukapheresis sample if frozen.
- Inoculation:
- Aseptically inoculate 1-2 mL of the leukapheresis product into a BACTEC Peds Plus T/F aerobic culture bottle.
- Aseptically inoculate 1-2 mL of the product into a BACTEC Peds Plus T/F anaerobic culture bottle.
- Incubation and Monitoring: Load the inoculated bottles into the BACTEC automated instrument. The system will incubate the bottles at 35±1.5°C and monitor them continuously for 14 days for signs of microbial growth.
- Result Interpretation: A bottle is flagged as positive by the instrument when microbial growth is detected. If a bottle is positive, subculture it onto solid media for further identification. The sample passes sterility testing only if both aerobic and anaerobic bottles show no growth after the full incubation period.
Validation: Studies have confirmed that this automated method can reliably detect microorganisms close to the limit of detection (10-50 colony forming units) within an acceptable incubation time, making it a suitable alternative to the manual compendial method [71].
Protocol for Cellular Composition Analysis by Flow Cytometry
Principle: Flow cytometry uses fluorescently-labeled antibodies to identify and quantify specific cell populations within a heterogeneous leukapheresis sample, such as T-cells (CD3+), B-cells (CD19+), and NK-cells (CD56+).
Materials:
- Flow cytometer
- Fluorescently-conjugated antibodies (e.g., anti-CD3, CD19, CD56)
- Flow cytometry staining buffer (PBS with 1-2% FBS)
- Fixation buffer (e.g., 1-4% paraformaldehyde)
- FACS tubes
Procedure:
- Sample Staining:
- Aliquot 100 µL of well-mixed leukapheresis sample into a FACS tube.
- Add the pre-titrated antibody cocktail (e.g., CD3-FITC, CD19-PE, CD56-APC).
- Vortex gently and incubate for 20-30 minutes at room temperature, protected from light.
- Washing and Fixation: Add 2 mL of staining buffer to the tube, centrifuge at 300-500 x g for 5 minutes, and carefully decant the supernatant. Repeat the wash step once. Resuspend the cell pellet in 200-500 µL of fixation buffer.
- Data Acquisition and Analysis: Acquire the sample on a flow cytometer, collecting a minimum of 10,000 events in the lymphocyte gate. Analyze the data using flow cytometry software to determine the percentage of each cell population of interest.
Expected Outcomes: Cryopreserved leukapheresis products have been shown to maintain a higher lymphocyte proportion (66.59%) compared to cryopreserved PBMCs (52.20%), which is advantageous for T-cell therapies [7].
Diagram 1: Post-collection quality control workflow for leukapheresis products.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for Post-Collection QC
| Reagent/Material | Function | Example Products/Notes |
|---|---|---|
| Automated Cell Counter | Precisely counts total and viable cells | Instruments using AO/PI fluorescence staining (e.g., NC-200) |
| Controlled-Rate Freezer | Ensures consistent, reproducible freezing | Critical for cryopreservation; used by 100% of centers surveyed [72] |
| Cryoprotectant | Protects cells from ice crystal damage during freeze-thaw | Clinical-grade DMSO (e.g., CryoStor CS10); concentrations vary from 5-15% [72] |
| Automated Sterility System | Detects bacterial/fungal contamination | BACTEC culture bottles; validated for cell therapy products [71] |
| Flow Cytometry Antibodies | Identifies and quantifies specific immune cell subsets | Anti-CD3, CD19, CD56 for lymphocyte phenotyping |
| Cryopreservation Media | Formulation for maintaining cell viability during storage | Combinations of DMSO with albumin, cell culture media (RPMI, IMDM), or buffered solutions [72] |
Diagram 2: Integrated QC in leukapheresis cryopreservation workflow.
Troubleshooting and Best Practices
- Low Post-Thaw Viability: Ensure the time from cryoprotectant addition to initiation of controlled-rate freezing is strictly limited to ≤ 120 minutes [7]. Optimize the final cell concentration for cryopreservation to ~5 x 10⁷ cells/mL.
- Sterility Test Failures: Reinforce aseptic techniques during all open processing steps. Implement strict environmental monitoring of laminar flow hoods and cleanrooms. Consider using closed-system automated processing platforms where possible.
- Inconsistent Cell Yields/Recovery: Standardize sample mixing procedures before aliquoting for any testing or processing. Validate all centrifugation steps to minimize unintended cell loss.
- Combating Contamination: Implement a rigorous quarantine system for all new or incoming cell lines, performing preliminary mycoplasma testing before they are introduced into the main laboratory workflow [73]. Regular testing for mycoplasma is essential, as it can alter cell growth and gene expression without visible signs.
For autologous cell therapies, the vein-to-vein workflow presents a monumental logistics challenge. The starting material—a patient's own cells collected via leukapheresis—is both precious and perishable. Traditional models relying on fresh leukapheresis shipment are fraught with risk, as the cells have a limited viability window of approximately 24-72 hours post-collection [74] [75]. Any delay due to transportation, manufacturing slot unavailability, or unforeseen disruptions can compromise cell quality and viability, potentially jeopardizing the entire therapeutic product [76] [77].
This application note explores how the integrated application of cryopreservation and distributed manufacturing models directly addresses these critical bottlenecks. By transforming the leukapheresis material into a stable, storable commodity, these strategies de-risk the supply chain, enhance scheduling flexibility, and ultimately build the resilience required for scalable and globally accessible autologous cell therapies.
The Bottleneck: Logistical Vulnerabilities in Fresh Leukapheresis Logistics
The conventional supply chain for autologous cell therapies is linear and time-sensitive. Fresh leukapheresis material must be transported from the clinical collection site to a centralized manufacturing facility, a process that must be completed within a narrow window to maintain cell viability and functionality [75]. Beyond this window, cells undergo significant metabolic decline [74]. This model is vulnerable to a multitude of logistical bottlenecks:
- Scheduling Complexity: Apheresis appointments, manufacturing slots, and patient conditioning must be perfectly synchronized across different time zones and institutions [77] [75].
- Transportation Risks: Unforeseen events like weather, customs delays, or courier failures can lead to costly shipments and complete product loss [76] [74].
- Geographical Constraints: Patients in remote or rural areas may have limited access to apheresis centers, and long transit times to centralized manufacturing facilities can be prohibitive [77].
- Manufacturing Inflexibility: Centralized facilities have limited capacity and cannot easily adapt to fluctuations in patient enrollment or recover from upstream delays without causing a cascade of scheduling conflicts [7].
The Solution: An Integrated Approach of Cryopreservation and Distributed Manufacturing
Cryopreservation: Creating a Stable Cellular Starting Material
Cryopreservation halts biological activity by storing leukapheresis material at ultra-low temperatures (typically ≤ -150°C), effectively creating a long-term, stable starting material [74] [75]. This decouples the leukapheresis collection from the manufacturing process, introducing critical flexibility into the supply chain.
Quantitative Evidence: Validating Cryopreserved Leukapheresis
Recent comparative studies robustly demonstrate that cryopreserved leukapheresis is a non-inferior starting material for CAR-T manufacturing. The data below summarizes key quality attributes from recent investigations.
Table 1: Comparative Analysis of Fresh vs. Cryopreserved Leukapheresis in CAR-T Manufacturing
| Quality Attribute | Fresh Leukapheresis | Cryopreserved Leukapheresis | Significance & Source |
|---|---|---|---|
| Post-Thaw Viability | >90% (within 24-36h) [75] | ≥90% [7] | Maintains critical viability threshold [7] [75] |
| T Cell Profile (CD3+ %) | Baseline | 42.01–51.21% (post-thaw) [7] | No significant loss of key T-cell population [7] |
| CAR-T Cell Expansion | Baseline | Comparable to fresh [54] [6] | Final product fold expansion is not compromised [54] [6] |
| Transduction Efficiency | Baseline | Comparable to fresh [6] | Genetic modification efficiency is maintained [6] |
| Cytotoxic Function | Baseline | Comparable anti-tumor potency [54] [6] | Functional efficacy of the final CAR-T product is preserved [54] [6] |
| Clinical Response | Baseline | Non-inferior outcomes [6] | Patient outcomes are not adversely affected [6] |
Detailed Protocol: High Cell Concentration Cryopreservation of Leukapheresis
The following protocol, adapted from a validated method, outlines the steps for processing and cryopreserving leukapheresis product to reduce volume and remove non-target cells, thereby optimizing the starting material [78].
Aim: To standardize the cryopreservation of leukapheresis product for CAR-T manufacturing by reducing volume, minimizing non-target cells (e.g., erythrocytes, platelets), and maintaining T lymphocyte viability and function.
Materials:
- Leukapheresis product
- Centrifuge (e.g., Allegra X-15R, Beckman Coulter)
- Centrifuge tubes
- Blood transfer bags (e.g., 600 mL, Grifols)
- Cryopreservation bags (e.g., 15-85 mL EVA, Macopharma)
- Dimethyl sulfoxide (DMSO)
- Autologous plasma
- Programmable controlled-rate freezer (e.g., CM-2010, Carburos Metálicos)
- Liquid nitrogen storage system
Method:
- Pre-processing Assessment: Perform a whole blood count and flow cytometry analysis on the apheresis final product to determine total nucleated cells (TNC) and T-cell percentage and number.
- Overnight Storage (Optional): If processing cannot occur immediately, store the leukapheresis product overnight at 4°C [78].
- Centrifugation:
- Transfer the product to appropriate centrifuge tubes.
- Centrifuge at 300 G for 15 minutes at room temperature (e.g., acceleration=9, no brake) [78].
- Volume Reduction:
- After centrifugation, visually inspect the product for hemolysis, particulates, or platelet clumps.
- Carefully remove the supernatant plasma to adjust the cell pellet to a final volume of approximately 40 mL [78]. This typically reduces the original volume by almost fivefold (e.g., from a median of 185 mL to 40 mL) [78].
- Cryopreservation Formulation:
- Transfer the concentrated cell product to a cryopreservation bag.
- Prepare a cryopreservation solution consisting of at least 20% autologous plasma and 10% DMSO [78].
- Gradually add the cryoprotectant solution to the cell bag within 5 minutes to achieve a final cell concentration of 50–200 x 10^6 cells/mL [78].
- Controlled-Rate Freezing and Storage:
Key Advantages of this Protocol:
- Logistical Simplicity: Volume reduction simplifies handling, saves storage space, and reduces shipping costs [78].
- Purer Starting Material: The process reduces monocytes, platelets, and erythrocytes, providing a more defined starting population for manufacturing [78].
- Scheduling Flexibility: The optional overnight hold and the creation of a stable frozen product decouple apheresis from manufacturing scheduling [78].
Distributed Manufacturing: Localizing Production for Resilience
Distributed manufacturing complements cryopreservation by restructuring the production network. Instead of a single, centralized factory, it employs a network of smaller, geographically dispersed manufacturing facilities [79]. This model leverages cryopreserved starting materials that can be easily shipped to and from these regional centers.
Table 2: Benefits of a Distributed Manufacturing Model for Cell Therapy
| Benefit Category | Specific Advantage | Impact on Supply Chain |
|---|---|---|
| Supply Chain Acceleration | Faster deployment of raw materials and finished goods; reduced inbound/outbound lag time [80]. | Shortens vein-to-vein time by producing therapies closer to the patient. |
| Enhanced Resilience | Reduced dependency on single sources; ability to pivot during regional disruptions [79]. | Mitigates risk of total shutdown from events like natural disasters or political instability. |
| Cost Containment | Lower transportation spending; reduced risk of spoilage [80]. | Optimizes logistics costs and minimizes costly product losses. |
| Regulatory & Market Agility | Reduced tariffs and trade barriers; easier compliance with regional regulations [79] [77]. | Simplifies global market entry and adherence to diverse regional requirements. |
The synergy between these two approaches is powerful. Cryopreservation enables distributed manufacturing by providing a stable starting material that can withstand the logistics of shipment to regional facilities. In turn, distributed manufacturing maximizes the value of cryopreservation by locating production hubs near patients, drastically reducing the final, time-critical leg of the journey for the finished drug product.
Essential Reagents and Materials for Implementation
The successful deployment of these strategies requires specific reagents and infrastructure. The table below lists key materials for establishing a robust cryopreservation workflow for leukapheresis.
Table 3: Research Reagent Solutions for Leukapheresis Cryopreservation
| Item | Function / Application | Example Specification / Notes |
|---|---|---|
| Cryoprotectant | Protects cells from ice crystal formation during freeze-thaw. | Clinical-grade DMSO at 10% final concentration, often in proprietary solutions like CS10 [7]. |
| Cryopreservation Bags | Secure, sterile container for freezing and storing cell products. | EVA bags with a 15-85 mL capacity; validated for leachables and stability at ultra-low temperatures [78]. |
| Controlled-Rate Freezer | Ensures consistent, optimal cooling rate to maximize cell viability. | Programmable freezer (e.g., cooling at 1°C/min to -80°C) [78]. |
| Liquid Nitrogen Storage | Provides long-term storage at ≤ -150°C to halt metabolic activity. | Storage in the gas phase of liquid nitrogen [78] [74]. |
| Closed System Processing | Maintains sterility during centrifugation and formulation. | Automated systems (e.g., IntegriCellTM) or sterile tubing welders for connecting bags [54] [77]. |
| Cell Separation Media | For density gradient centrifugation if PBMC isolation is required. | e.g., Ficoll, though direct leukapheresis cryopreservation is increasingly common [7]. |
Regulatory and Operational Considerations
Implementing cryopreservation and distributed models requires careful navigation of regulatory landscapes. Key agencies like the FDA, EMA, and authorities in APAC regions (e.g., Japan's MHLW, South Korea's ARMAB) provide guidance [77]. Critical considerations include:
- Minimal vs. Substantial Manipulation: In many regions, cryopreservation of starting material is considered a "minimal manipulation," simplifying regulatory pathways. This is contingent on the process not altering biological characteristics [77].
- Closed System Advantage: Using validated closed systems for cryopreservation allows for processing in controlled, non-classified environments, significantly reducing infrastructure costs and contamination risk [77].
- Chain of Identity and Custody: Robust tracking systems are mandatory to maintain the chain of identity from patient to product across a potentially distributed network [77].
The integration of cryopreservation and distributed manufacturing models presents a paradigm shift for the autologous cell therapy supply chain. By transforming a time-sensitive, fragile logistics operation into a stable, flexible, and resilient network, these strategies directly mitigate the critical bottlenecks that have long constrained the field. The validated protocols for leukapheresis cryopreservation ensure that cell quality and functionality are maintained, while the distributed model enhances global access and supply chain robustness. For researchers and drug developers, adopting this integrated approach is no longer just an option but a necessity for achieving scalable, reliable, and globally accessible cell therapies.
Data-Driven Decisions: Validating Leukapheresis Products for Clinical Success
Application Note
The starting material for Chimeric Antigen Receptor T-cell (CAR-T) therapy is a critical determinant of manufacturing success and subsequent clinical efficacy. Autologous cell therapy research has historically relied on fresh leukapheresis, a material constrained by a narrow 24-72-hour viability window that complicates logistics and limits manufacturing flexibility [7] [17]. This creates significant bottlenecks, particularly for distributed manufacturing models and for patients with rapidly progressive disease.
Cryopreserved leukapheresis presents a promising alternative, potentially decoupling cell collection from manufacturing. Despite its advantages, its adoption has been hindered by perceptions of inferior quality; a bibliometric analysis revealed that from 2010 to 2024, only 18.3% of 349 related studies focused on cryopreserved leukapheresis, while the majority investigated cryopreserved Peripheral Blood Mononuclear Cells (PBMCs) [7]. This application note synthesizes recent, comprehensive evidence to demonstrate that with standardized protocols, cryopreserved leukapheresis is a functionally equivalent and logistically superior starting material for autologous CAR-T manufacturing.
Comparative Performance Analysis
A direct, multi-platform comparability study between cryopreserved and fresh leukapheresis was conducted, evaluating key quality attributes from initial cell composition through to the functionality of the final CAR-T product.
Table 1: Post-Thaw Quality Attributes of Cryopreserved Leukapheresis vs. PBMCs
| Quality Attribute | Cryopreserved Leukapheresis | Cryopreserved PBMCs | Significance |
|---|---|---|---|
| Post-Thaw Viability | ≥ 90% [7] | ~90% (Typical supplier spec) [81] | Meets manufacturing thresholds |
| Lymphocyte Proportion | 66.59 ± 2.64% [7] | 52.20 ± 9.29% [7] | p < 0.05; Higher T-cell potential |
| T-cell Proportion (CD3+) | 42.01 – 51.21% [7] | Decreased post-thaw [82] | Better preservation in leukapheresis |
| Viability vs. Fresh | 91.0% (vs. 99.0% fresh) [7] | N/A | Lower initially, but functionally recovers |
The data indicates that cryopreserved leukapheresis not only meets viability benchmarks but also demonstrates a significantly higher lymphocyte proportion compared to cryopreserved PBMCs. This correlates with enhanced potential for T-cell therapies, as the starting material is richer in the target cell population [7]. While initial post-thaw viability is lower than fresh material, the cells undergo functional recovery.
Table 2: CAR-T Manufacturing Outcomes and Clinical Efficacy
| Parameter | Cryopreserved Leukapheresis | Fresh Leukapheresis | Conclusion |
|---|---|---|---|
| Cell Viability & Expansion | Comparable [7] [83] | Comparable [7] [83] | No significant difference |
| CAR+ Cell Proportion | Comparable [7] | Comparable [7] | No significant difference |
| In Vitro Cytotoxicity | Comparable [7] | Comparable [7] | Maintained anti-tumor function |
| Phenotype (T-cell subsets) | Comparable [7] [84] | Comparable [7] [84] | Profile is preserved |
| Clinical Response (CD22 CAR-T) | Comparable [83] | Comparable [83] | No difference in efficacy |
| Incidence of Toxicities | Comparable [83] | Comparable [83] | No difference in safety profile |
Critically, when used as the starting material for CAR-T manufacturing across non-viral, lentiviral, and Fast CAR-T platforms, cryopreserved leukapheresis performed comparably to fresh in all key metrics: cell expansion, transduction efficiency, phenotype, and in vitro cytotoxicity [7]. Most importantly, clinical studies have confirmed that CAR-T products manufactured from cryopreserved starting material yield comparable clinical outcomes. A retrospective analysis of patients receiving anti-CD22 CAR-T cells found no significant differences in in vivo expansion, persistence, incidence of toxicities, or disease response between those who received products made from cryopreserved (n=21) versus fresh (n=19) leukapheresis [83].
Protocol
Standardized Protocol for Leukapheresis Cryopreservation
The following optimized protocol ensures high post-thaw viability and functionality, validated for CAR-T manufacturing.
Materials & Equipment
- Leukapheresis Product: Collected in ACD-A anticoagulant [12].
- Cryoprotectant: Clinical-grade CS10 (10% DMSO) [7].
- Centrifuge: Equipped for controlled centrifugation.
- Cryopreservation Bags
- Controlled-Rate Freezer: Validated system (e.g., Thermo Profile 4) [7].
- Storage: Vapor phase liquid nitrogen (< -40°C) [46].
Step-by-Step Procedure
Initial Processing:
Formulation with Cryoprotectant:
- Mix the cell suspension with an equal volume of pre-chilled cryoprotectant (CS10, 10% DMSO) to achieve a final DMSO concentration of ≥ 7.5% [7].
- Critical Step: The interval from cryoprotectant addition to the initiation of controlled-rate freezing must be ≤ 120 minutes to maintain cell viability [7].
Cryopreservation and Storage:
- Aliquot the formulated product into cryopreservation bags (e.g., 20 mL/bag, target of ≥ 1 × 10^9 cells per bag) [7].
- Transfer bags to a controlled-rate freezer. Use a validated freezing profile.
- Immediately transfer the frozen bags to vapor phase liquid nitrogen for long-term storage (< -40°C). Temperature excursions above -40°C reduce post-thaw viability [46].
Quality Control Checks
- Pre-cryopreservation Viability: Should be ≥ 95% [7].
- Post-thaw Viability: Acceptable at ≥ 90% [7] [46].
- Cell Recovery & Phenotype: Assess CD3+ T-cell proportion post-thaw to ensure no significant loss [7].
Protocol for Functional Validation via CAR-T Manufacturing
This protocol outlines the critical steps for validating a cryopreserved leukapheresis product by using it as starting material in a CAR-T manufacturing process.
Thawing and Recovery
- Rapidly thaw the cryopreserved leukapheresis bag in a 37°C water bath.
- Gently transfer the contents to a container and slowly add pre-warmed medium with 5% FBS or human serum albumin over spaced intervals to dilute the DMSO.
- Wash cells 2-3 times by centrifugation (e.g., 300-350 × g for 10 min) to remove residual cryoprotectant [82] [84].
- Resuspend the cell pellet in complete medium and allow for a brief recovery incubation (several hours) before proceeding to manufacturing.
CAR-T Manufacturing and In Vitro Assays
Manufacturing processes should follow standard operating procedures for the chosen platform (e.g., non-viral, lentiviral). Key performance indicators must be compared against benchmarks from fresh leukapheresis [7].
- Cell Expansion: Track fold expansion over the culture period.
- Transduction Efficiency: Measure the percentage of CAR+ cells by flow cytometry.
- Phenotypic Profiling: Analyze T-cell subsets (CD4+/CD8+, naïve, memory, exhausted) in the final product.
- Potency Assay: Perform an in vitro cytotoxicity assay against CD19+ target cells (e.g., NALM-6) at various Effector:Target ratios to confirm specific killing [84].
Visualizations
Experimental Workflow for Comparability Study
Decision Logic for Selecting Starting Material
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions
| Reagent / Material | Function / Application | Key Considerations |
|---|---|---|
| CS10 Cryoprotectant | Clinical-grade freezing medium containing 10% DMSO. Protects cells from ice crystal damage during freeze-thaw. | Preferred over homemade DMSO solutions for lot-to-lot consistency and reduced risk of contamination [7]. |
| ACD-A Anticoagulant | Acid Citrate Dextrose Solution A. Used during leukapheresis collection to prevent clotting. | Standard anticoagulant for apheresis; gentler on cells compared to heparin for long-term processing [12]. |
| Lymphoprep / Ficoll | Density gradient medium for isolation of PBMCs from leukapheresis product. | Enriches for mononuclear cells; temperature sensitivity and processing ratios can affect cell loss [7] [82]. |
| ACK Lysing Buffer | Ammonium-Chloride-Potassium buffer for osmotic lysis of residual red blood cells. | Improves sample purity and accuracy of cell counting. May enhance antigen sensitivity of memory T cells [82]. |
| RetroNectin | A recombinant fragment of human fibronectin. Used to coat culture surfaces during viral transduction. | Enhances transduction efficiency of retroviral and lentiviral vectors by co-localizing target cells and viral particles [84]. |
| Anti-CD3/CD28 Antibodies | Functional grade antibodies for T-cell activation and stimulation prior to transduction. | Critical for initiating T-cell proliferation. Often used with IL-2 (e.g., 300 IU/mL) to support expansion [84]. |
Leukapheresis serves as the foundational step in autologous chimeric antigen receptor T-cell (CAR-T) therapy manufacturing, with the quality of the collected starting material directly impacting final product characteristics, efficacy, and consistency [7]. For researchers and drug development professionals, understanding the nuanced interplay between leukapheresis material, manufacturing platforms, and resulting CAR-T product phenotypes is crucial for process optimization. Recent investigations reveal that differences in manufacturing processes—including the handling of leukapheresis material—contribute significantly to the compositional and functional heterogeneity observed among commercially available CAR-T products [85] [7]. This application note provides a structured, data-driven framework for the comparative analysis of leukapheresis requirements and yields across leading CD19-directed CAR-T constructs, leveraging recent empirical findings to inform protocol development and raw material standardization.
Comparative Landscape of Commercial CAR-T Products
The three approved CD19-directed CAR-T products for large B-cell lymphoma (LBCL)—axicabtagene ciloleucel (axi-cel), tisagenlecleucel (tisa-cel), and lisocabtagene maraleucel (liso-cel)—differ in their costimulatory domains, manufacturing processes, and final product composition [85] [86]. Axi-cel incorporates a CD28 costimulatory domain, while tisa-cel and liso-cel both use 4-1BB costimulatory domains but differ in other structural components [86]. Critically, these products exhibit distinct manufacturing timelines and starting material requirements, factors that subsequently influence the T-cell composition of the infusion product and its clinical performance profile [85] [87].
Table 1: Key Characteristics of Commercial CD19-Directed CAR-T Products
| Product | Costimulatory Domain | Manufacturing Time | Starting Material | Activation Method |
|---|---|---|---|---|
| Axi-cel | CD28 | Shorter (~41 days) [87] | Peripheral Blood Mononuclear Cells (PBMCs) [85] | Anti-CD3 antibody [85] |
| Tisa-cel | 4-1BB | Prolonged (~52 days) [87] | Isolated CD3+ T cells [85] | Anti-CD3/anti-CD28 beads [85] |
| Liso-cel | 4-1BB | Not Specified | CD4+ & CD8+ T cells (separately) [85] | Anti-CD3/anti-CD28 beads [85] |
Quantitative Analysis of Leukapheresis Processing and Yield
Standardized processing of leukapheresis material is essential for ensuring consistent CAR-T manufacturing outcomes. Recent optimizations have established robust protocols for cryopreserved leukapheresis, enabling detailed comparability assessments between fresh and cryopreserved starting materials.
Table 2: Quality Attributes of Cryopreserved vs. Fresh Leukapheresis Material
| Parameter | Fresh Leukapheresis | Cryopreserved Leukapheresis | Cryopreserved PBMCs |
|---|---|---|---|
| Post-Thaw Viability | 99.2-99.5% [7] | 90.9-97.0% [7] | Not Specified |
| Lymphocyte Proportion | 68.68 ± 1.78% [7] | 66.59 ± 2.64% [7] | 52.20 ± 9.29% [7] |
| CD3+ T-cell Proportion | 43.82-56.31% [7] | 42.01-51.21% [7] | Not Specified |
| Cell Concentration Pre-Cryopreservation | 5.09-9.71 × 10⁷ cells/mL [7] | 4.06-5.12 × 10⁷ cells/mL [7] | Not Specified |
Impact of Manufacturing Timeline on Final Product Composition
The duration of the manufacturing process directly impacts the phenotypic characteristics of the final CAR-T product. Single-cell RNA sequencing analysis of pre-infusion CAR-T products has revealed that axi-cel and tisa-cel are markedly different products at a transcriptional and cellular level [85]. Axi-cel products demonstrate a higher proportion of CD4 and CD8 central memory T cells, which are associated with favorable outcomes and durable responses [85]. In contrast, tisa-cel products, subjected to a longer manufacturing process, contain more proliferative cells but a decreased proportion of naïve and central memory T-cell subsets [85]. This divergence is attributed to the prolonged expansion culture used in tisa-cel manufacturing, which appears to adversely affect the T-cell differentiation state independently of the CAR construct itself [85].
Application Note: Protocol for Standardized Cryopreserved Leukapheresis
Experimental Protocol: Closed-System Leukapheresis Cryopreservation
Objective: To establish a standardized, automated protocol for cryopreserving leukapheresis products that maintains T-cell fitness and compatibility with downstream CAR-T manufacturing platforms.
Materials:
- Leukapheresis Collection: Obtain via automated blood cell separator or mobile leukapheresis unit [28].
- Cryoprotectant: Clinical-grade CS10 (10% DMSO solution) [7].
- Equipment: Closed-system automated processing platform, controlled-rate freezer (validated using Thermo Profile 4 system), temperature-monitored storage vapor-phase liquid nitrogen freezer [7].
- Quality Control: Sysmex cell counter for automated cell counting and viability analysis, flow cytometer for immunophenotyping [28].
Methodology:
- Initial Processing:
- Centrifuge leukapheresis product to reduce non-cellular impurities (residual red blood cells, platelets).
- Adjust cell concentration to a target range of 5 × 10⁷ to 8 × 10⁷ cells/mL in a final formulation volume of 20 mL per cryobag [7].
Cryoprotectant Addition:
- Add CS10 cryoprotectant to achieve a final DMSO concentration of ≥7.5%.
- Maintain the interval from cryoprotectant addition to initiation of controlled-rate freezing to ≤120 minutes to preserve cell viability [7].
Controlled-Rate Freezing:
- Utilize a validated freezing curve with the controlled-rate freezer.
- Transfer cryobags to long-term vapor-phase liquid nitrogen storage post-freezing.
Quality Assessment:
- Post-Thaw Viability: Assess via trypan blue exclusion or automated cell counter. Acceptable viability is ≥90% [7].
- Cell Recovery: Determine total nucleated cell and CD3+ T-cell recovery post-thaw.
- Immunophenotyping: Perform flow cytometry to confirm retention of T-cell subsets (CD4+, CD8+, naïve, memory T cells).
Validation: The protocol's success is validated by demonstrating comparable performance in downstream CAR-T manufacturing platforms (non-viral, lentiviral, Fast CAR-T) relative to fresh leukapheresis, with equivalent metrics in cell viability, expansion, CAR+ cell proportion, and in vitro cytotoxicity [7].
Protocol Schematic: Cryopreserved Leukapheresis Workflow
Figure 1: Cryopreserved leukapheresis workflow for CAR-T manufacturing.
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for Leukapheresis Processing
| Reagent/Material | Function/Application | Example Use Case |
|---|---|---|
| Clinical-Grade CS10 | Cryoprotectant with 10% DMSO; minimizes ice crystal formation and preserves cell viability during freezing [7]. | Standardized formulation for cryopreserving leukapheresis products [7]. |
| Anti-CD3/anti-CD28 Beads | T-cell activation and stimulation; mimics endogenous TCR co-stimulation signaling [85]. | T-cell activation during manufacturing of tisa-cel and liso-cel [85]. |
| Anti-CD3 Antibody | T-cell receptor stimulation; provides primary activation signal for T-cell expansion [85]. | T-cell activation during manufacturing of axi-cel [85]. |
| Closed-System Automated Platform | Ensures aseptic processing, reduces operator error, and enhances process consistency and scalability [7]. | Automated centrifugation, concentration, and formulation of leukapheresis product pre-cryopreservation [7]. |
Analytical Methodologies for Product Characterization
Single-Cell RNA Sequencing for CAR-T Product Analysis
Experimental Protocol:
- Sample Preparation: Obtain pre-infusion CAR-T products (e.g., from infusion bags). Cryopreserve cells for batch analysis [85].
- Library Construction: Perform single-cell RNA sequencing using 10x Genomics Chromium platform to partition single cells and barcode RNA [85].
- CAR+ Cell Identification: Distinguish CAR transgene-positive (CAR+) from negative (CAR−) cells based on the presence of a short, construct-specific sequence (CAR-CD19FvL) within the scRNA-seq data [85].
- Bioinformatic Analysis: Perform differential gene expression analysis, T-cell subset annotation, and Gene Set Enrichment Analysis (GSEA) to compare products (e.g., axi-cel vs. tisa-cel) and identify pathways associated with clinical outcomes like durable response [85].
In Vitro CAR-T Manufacturing Mimicry
Experimental Protocol:
- T-cell Source: Use cryopreserved peripheral blood mononuclear cells (PBMCs) from healthy donors [85].
- T-cell Isolation & Activation:
- Condition A (mimicking tisa-cel/liso-cel): Enrich T cells using a magnetic bead T Cell Isolation Kit. Activate with CD3/CD28 Dynabeads in complete culture media supplemented with 100 IU/mL rhIL-2 [85].
- Condition B (mimicking axi-cel): Use unselected PBMCs. Activate by resuspending in complete culture media supplemented with 50 ng/mL OKT3 antibody and 300 IU/mL rhIL-2 [85].
- Viral Transduction: After 24 hours of activation, transduce cells with either CD19/CD28z or CD19/4-1BBz viral supernatant on retronectin-coated plates [85].
- Analysis: Compare CAR+ and CAR− cells at various time points (early vs. prolonged expansion) using flow cytometry and functional assays to assess differentiation and activation [85].
Logistical Framework and Emerging Models
The traditional centralized leukapheresis model presents significant access barriers. A transformative innovation is the development of mobile leukapheresis centers, which are designed to mirror the quality controls of fixed sites while operating within a bus-sized vehicle [28]. These mobile units incorporate redundant power supplies, real-time environmental monitoring, and the same collection equipment as fixed sites, enabling decentralized leukapheresis collection and expanding access for underserved patient populations [28]. This model directly addresses the critical bottleneck of initial cell collection, which is essential for scaling autologous CAR-T therapy production.
Schematic: Mobile Leukapheresis Operational Workflow
Figure 2: Mobile leukapheresis collection and processing workflow.
The comparative analysis of leukapheresis requirements for commercial CAR-T products underscores that the starting material and its subsequent manufacturing journey are critical determinants of final product phenotype. The standardization of cryopreserved leukapheresis protocols and the advent of decentralized collection models represent significant advancements for the field. These developments promise to enhance supply chain resilience, improve patient access, and provide a more consistent raw material for manufacturing. Future research must focus on large-scale clinical validation of these standardized protocols and further elucidate the precise molecular mechanisms linking leukapheresis material attributes to long-term therapeutic efficacy, ultimately enabling more predictable and potent CAR-T products.
In autologous cell therapy research, the functional equivalence of final cellular products must be established irrespective of variations in the leukapheresis starting material. This is critical for ensuring consistent product quality, safety, and efficacy, as mandated by regulatory bodies [88]. Functional equivalence validation demonstrates that despite inherent biological variability in donor apheresis collections, the resulting therapies consistently exhibit the key biological activities required for clinical success [89]. This Application Note provides a standardized framework for assessing three critical pillars of cellular function: cytotoxicity, expansion potential, and persistence. By implementing these protocols, researchers can robustly qualify manufacturing processes and bridge material changes during development.
Quantitative Functional Attributes
The table below summarizes the core functional attributes and their corresponding quantitative measures that must be evaluated to establish functional equivalence.
Table 1: Key Metrics for Functional Equivalence Validation
| Functional Attribute | Key Quantitative Measures | Assay Platforms |
|---|---|---|
| Cytotoxicity | Specific lysis (%), degranulation (CD107a expression), cytokine release (IFN-γ, IL-2 pg/mL) [88] | Flow cytometry, ELISA/MSD, co-culture assays (e.g., xCelligence, Incucyte) |
| Expansion Potential | Fold expansion, population doublings, viability (%), immunophenotype (e.g., naïve, memory subsets) [88] | Automated cell counters, flow cytometry, metabolic assays (e.g., ATP) |
| Persistence | In vivo expansion & duration, transcriptional profiling for exhaustion/exhaustion, vector copy number (VCN) [88] [90] | qPCR/ddPCR, single-cell RNA-seq, in vivo imaging, TCR sequencing [88] |
Experimental Protocols
Cytotoxicity Assessment
Principle: Measure the ability of CAR-T cells to specifically recognize and kill antigen-expressing target cells [88].
Materials:
- Effector cells (CAR-T from different leukapheresis starting materials)
- Target cells (antigen-positive and antigen-negative cell lines)
- Culture medium
- Flow cytometry antibodies (for CD107a, etc.)
- Cytokine detection kit (e.g., IFN-γ ELISA/MSD)
Procedure:
- Co-culture Setup: Seed target cells and add effector cells at varying Effector:Target (E:T) ratios (e.g., 1:1, 5:1, 10:1). Include target-only and effector-only controls.
- Degranulation Assay: Add anti-CD107a antibody to co-culture. After 1-2 hours, add GolgiStop. Incubate for 4-6 hours total.
- Flow Cytometry Analysis: Harvest cells, stain for surface markers, and analyze by flow cytometry to calculate specific lysis and CD107a expression on effector cells.
- Cytokine Measurement: Collect supernatant from co-culture after 18-24 hours. Quantify IFN-γ and other relevant cytokines (e.g., IL-2, TNF-α) via ELISA or multiplex immunoassays [88].
Expansion Potential
Principle: Evaluate the proliferative capacity and maintenance of viability of cell products during ex vivo culture [88].
Materials:
- Test cells
- Culture medium with appropriate cytokines (e.g., IL-2)
- Cell counter or automated system
- Flow cytometer
- Viability stain
Procedure:
- Initial Seeding: Seed cells at a defined density in culture vessels.
- Long-term Culture: Maintain cultures for 10-14 days, feeding with fresh medium and cytokines as needed.
- Periodic Sampling: Every 2-3 days, remove an aliquot of cells.
- Cell Counting & Viability: Count total cells and assess viability using trypan blue or an automated cell counter.
- Fold Expansion Calculation: Calculate fold expansion: (Total viable cell count at day X) / (Total viable cell count at day 0).
- Phenotyping: At key timepoints, stain cells with antibodies for T-cell subsets (e.g., CD45RA, CD62L for naïve/memory status) and analyze by flow cytometry.
Persistence Assessment
Principle: Assess the long-term survival and functional maintenance of cells post-infusion/administration.
Methods:
- In Vivo Tracking:
- Utilize murine models. Luciferase-expressing cells can be tracked via IVIS imaging.
- Monitor cell numbers in peripheral blood and tissues over time using flow cytometry or PCR-based methods.
- Vector Copy Number (VCN):
- Extract genomic DNA from cell samples.
- Quantify the average number of integrated CAR vectors per cell using droplet digital PCR (ddPCR), a routine quality control assay [88].
- Transcriptomic Profiling:
- Perform single-cell RNA sequencing (scRNA-seq) on infusion products or post-infusion samples.
- Analyze for signatures of T-cell exhaustion, memory, and effector phenotypes, which are epigenetically programmed and critical for long-term efficacy [88].
Signaling Pathways in Engineered Cell Products
The functionality of advanced cell therapies is governed by intracellular signaling pathways activated upon antigen engagement. The diagram below illustrates the key signaling modules in a second-generation CAR, which is the foundation for most currently approved therapies [90] [91].
Research Reagent Solutions
The table below lists essential reagents and tools required for the execution of these functional equivalence protocols.
Table 2: Key Research Reagents for Functional Validation
| Reagent / Tool | Function / Application | Example Use Case |
|---|---|---|
| Anti-CD107a Antibody | Marks lysosomal degranulation, indicating cytotoxic activity [88] | Flow cytometry-based measurement of killing in real-time. |
| Cytokine ELISA/MSD Kits | Quantifies secreted cytokines (IFN-γ, IL-2) as a measure of T-cell activation [88] | Analysis of supernatant from effector-target co-cultures. |
| ddPCR Assays | Precisely quantifies Vector Copy Number (VCN) for persistence and safety [88] | Genomic DNA analysis from final product or post-infusion samples. |
| T-cell Phenotyping Panels | Identifies differentiation states (naïve, memory, effector) via surface markers (e.g., CD45RO, CD62L) [88] | Flow cytometry to profile starting material and final product composition. |
| scRNA-seq Reagents | Enables single-cell resolution analysis of transcriptional states and TCR clonality [88] | Deep profiling of product heterogeneity and exhaustion signatures. |
Application Note & Protocol
Within the development of autologous cell therapies, leukapheresis serves as the critical first step, providing the foundational T-cell population for engineering. A central logistical and biological question is whether using cryopreserved leukapheresis or peripheral blood mononuclear cell (PBMC) products negatively impacts T-cell fitness compared to fresh material. Maintaining T-cell fitness—encompassing differentiation, memory potential, and avoidance of exhaustion—is paramount for manufacturing potent cell therapies like chimeric antigen receptor (CAR)-T cells. This application note synthesizes recent research to compare T-cell fitness attributes between fresh and cryopreserved sources and provides detailed protocols for their evaluation, directly supporting leukapheresis processing strategies in autologous cell therapy research.
The decision to use fresh or cryopreserved starting material involves balancing logistical flexibility against potential impacts on cellular quality. The data below summarize key comparative findings.
Table 1: Post-Thaw Viability and Recovery of Cryopreserved Material
| Material Type | Post-Thaw Viability (%) | Key Cell Recovery Metrics | Source/Study |
|---|---|---|---|
| Cryopreserved Leukapheresis | 90.9% - 97.0% [7] | Lymphocyte proportion: 66.59% (comparable to fresh) [7] | Standardized, automated closed system [7] |
| Cryopreserved PBMCs | 73.7% - 98.4% [48] | Sufficient yield for manufacturing despite viability drop [48] | Panch et al. [48] |
| Cryopreserved CAR-T Final Product | Robust (Specific range not given) [48] | No significant difference in % T cells, transduction efficiency, or CD4:CD8 ratio [48] | Industry (Novartis, Kite) & Academic [48] |
Table 2: Impact on T-cell Differentiation, Memory, and Exhaustion
| T-cell Attribute | Impact of Cryopreservation | Clinical/Functional Correlation |
|---|---|---|
| Memory & Naive Subsets | No significant difference in fold-expansion or CD4:CD8 ratio at harvest [48]. | |
| Stem-like Progenitors | Two distinct CCR7+ progenitor subsets identified: PD-1-TIGIT- (functional lineage) and PD-1+TIGIT+ (exhausted-like lineage) [92]. Cryopreservation's specific effect requires further study. | Presence of PD-1+TIGIT+ progenitors indicates a pre-commitment to dysfunction [92]. |
| Exhaustion Markers | Elevated expression of genes related to apoptosis and cell-cycle damage pathways post-thaw [48]. | Similar in vivo persistence and clinical outcomes in patients treated with fresh vs. cryopreserved CAR-Ts [48]. |
| In Vitro & In Vivo Function | Slower expansion during manufacture, but no difference in activation, cytokine production, or anti-tumor cytotoxicity [93]. | No relationship between post-thaw viability (range 73.7%-98.4%) and clinical response [48]. |
Experimental Protocols for Assessing T-cell Fitness
The following protocols are essential for quantifying the critical quality attributes of T-cells derived from fresh or cryopreserved leukapheresis material.
Protocol: Multicolor Flow Cytometry for T-cell Memory and Exhaustion Phenotyping
This protocol details the procedure for characterizing T-cell differentiation and exhaustion states, crucial for evaluating starting material and final products [48] [92].
1. Research Reagent Solutions
Table 3: Key Reagents for T-cell Fitness Analysis
| Reagent / Material | Function / Application |
|---|---|
| Anti-human Antibody Panels [92] | Surface/CD3, CD4, CD8, CD45RO, CCR7, CD62L (SELL), CD95, CD27, CD28, CD127 (IL7R). Inhibitory Receptors/PD-1, TIGIT, TIM-3, LAG-3 [92] [94]. Transcription Factors/TCF1 (LEF1), T-bet. Cytotoxicity/Granzyme B (GZMB), Granzyme K (GZMK). |
| Flow Cytometry Staining Buffer | PBS with 0.5% BSA for antibody dilution and cell washing [93]. |
| Viability Stain | Fixable live/dead dye to exclude non-viable cells from analysis [93]. |
| Fluorophore-conjugated Anti-FMC63 Antibody [93] | Detection of CD19-specific CAR expression on transduced T-cells. |
| Intracellular Staining Kit | Permeabilization buffers for staining transcription factors (e.g., T-bet) and cytokines. |
2. Procedure Steps
- Cell Preparation: Thaw cryopreserved PBMCs or use fresh leukapheresis-derived cells. Rest cells overnight in complete media (e.g., RPMI-1640 + 10% human serum + IL-2 (100UI/ml)) [93].
- Surface Staining: Resuspend ~1x10^6 cells in 100µL of buffer containing a viability stain and Fc block. Incubate for 30 minutes at room temperature [93]. Add surface antibody cocktail and incubate for 30 minutes at 4°C. Wash cells twice with buffer.
- Intracellular Staining (if required): Fix and permeabilize cells using a commercial kit according to the manufacturer's instructions. Incubate with antibodies against intracellular targets (e.g., transcription factors, cytokines). Wash and resuspend in buffer for acquisition.
- Data Acquisition & Analysis: Acquire data on a flow cytometer capable of detecting 10+ colors. Use UMAP analysis or traditional gating strategies to identify T-cell subsets (e.g., Naive, TSCM, TCM, TEM, TTE) and assess exhaustion marker expression (PD-1, TIGIT) [92].
The following workflow diagram outlines the key steps for processing and analyzing T-cells from leukapheresis.
Protocol: Functional Cytotoxicity and Cytokine Assay
This protocol assesses the effector function of T-cells, a critical determinant of therapeutic potential [93].
1. Materials
- Target Cells: CD19+ tumor cell lines (e.g., K562-hCD19, OCI-LY3) and antigen-negative controls (e.g., K562-null) [93].
- Effector Cells: CAR-T cells or activated T-cells from fresh or cryopreserved PBMCs.
- Culture Media: Appropriate complete media for target and effector cells.
- Cytokine Detection Kit: ELISA or multiplex bead-based array for IFN-γ, TNF-α, IL-2.
2. Procedure Steps
- Co-culture Setup: Collect, wash, and count effector T-cells. Co-culture effector and target cells at a defined Effector-to-Target (E:T) ratio (e.g., 5:1) for 24-72 hours in a 96-well plate [93].
- Cytotoxicity Measurement: After co-culture, measure target cell killing using a real-time cell analyzer (e.g., xCelligence) or flow cytometry-based cytotoxicity assay (e.g., staining for caspase activity or membrane integrity).
- Cytokine Secretion Analysis: Collect cell-free supernatant from co-culture wells. Quantify cytokine concentrations using an ELISA or multiplex immunoassay according to the manufacturer's instructions.
Protocol: Single-Cell RNA Sequencing (scRNA-seq) for Deep Phenotyping
This protocol provides an unbiased, high-resolution map of T-cell states and exhaustion-associated gene signatures [92] [94].
1. Materials
- Single-Cell Platform: 10X Genomics Chromium Controller.
- Reagent Kits: Chromium Single Cell 3' or 5' Reagent Kits.
- Bioinformatic Tools: Cell Ranger, Seurat, or Scanpy for data analysis.
2. Procedure Steps
- Cell Preparation and Sorting: Isolate live CD8+ memory T-cells (e.g., CD3+ CD8+ CD95+) or total PBMCs via fluorescence-activated cell sorting (FACS). Ensure high viability (>90%) [92].
- Library Preparation and Sequencing: Load cells onto the Chromium Chip to generate single-cell gel beads-in-emulsion (GEMs). Proceed with cDNA amplification, library construction, and sequencing following the manufacturer's protocol.
- Bioinformatic Analysis: Align sequencing reads to the reference genome, quantify gene expression, and perform quality control. Use dimensionality reduction (UMAP) and graph-based clustering to identify cell populations. Analyze differentially expressed genes (DEGs) and pathway enrichment between conditions (e.g., fresh vs. cryopreserved) [92] [94].
Signaling Pathways and Molecular Regulation
The molecular landscape governing T-cell differentiation and exhaustion is a key consideration when evaluating starting materials. Research has identified distinct progenitor lineages within the human CD8+ memory T-cell pool. Specifically, two stem-like progenitor subsets are distinguished by the expression of inhibitory receptors PD-1 and TIGIT [92]. Progenitors lacking these markers (PD-1-TIGIT-) are committed to a functional lineage, while those expressing them (PD-1+TIGIT+) are pre-committed to a dysfunctional, exhausted-like lineage, expressing genes like CXCL13 [92] [94]. The diagram below illustrates the differentiation paths and functional fates of these progenitor cells.
Furthermore, scRNA-seq studies on breast cancer tumors reveal that the presence of this PD-1high/CXCL13+ exhausted-like T-cell phenotype is associated with a distinct tumor immune microenvironment (TIME). This "exhausted" environment is characterized by an inflammatory signature, altered cytotoxic profiles (high GZMB, low IFNG), elevated MHC-I on tumor cells, and immature tertiary lymphoid structures [94]. These findings provide a framework for analyzing the impact of cryopreservation on T-cell fate.
The collective evidence indicates that while cryopreservation induces measurable transcriptomic stress and an initial drop in T-cell viability, these alterations do not necessarily translate to impaired functional capacity or clinical efficacy of the final cellular product, such as CAR-T cells [48] [93]. The identification of pre-committed exhausted-like T-cell progenitors in starting material underscores the importance of deep phenotyping beyond simple viability counts [92].
For researchers processing leukapheresis for autologous therapy, the use of cryopreserved starting material presents a viable and logistically advantageous path forward. Success hinges on:
- Standardized Protocols: Implementing closed, automated systems for cryopreservation to ensure post-thaw viability ≥90% and consistent cell recovery [7].
- Comprehensive Fitness Assessment: Employing a combination of flow cytometry, functional assays, and advanced tools like scRNA-seq to fully characterize the T-cell product [48] [92] [94].
- Focus on Critical Subsets: Monitoring the frequency of stem-like memory and pre-exhausted T-cell subsets, as these may be more predictive of long-term persistence and potency than bulk population metrics.
In conclusion, cryopreserved leukapheresis is a robust and scalable starting material for autologous cell therapy research. By adopting the detailed application notes and protocols herein, researchers and drug development professionals can confidently leverage cryopreservation to build more flexible and resilient manufacturing pipelines without compromising on T-cell fitness.
The successful manufacturing of autologous cell therapies, particularly Chimeric Antigen Receptor T-cell (CAR-T) therapies, is fundamentally dependent on the quality of the starting material—the leukapheresis product. Current autologous CAR-T manufacturing faces a paradoxical dilemma: while a significant proportion of patients (up to 33% in lymphoma) are unable to receive treatment due to leukapheresis failure or rapid disease progression, the field remains critically dependent on cells sourced from these same immunocompromised patients [7]. These failure rates directly correlate with therapeutic outcomes, with B-cell acute lymphoblastic leukemia (B-ALL) seeing 15–40% treatment failures, rising to over 50% in B-cell lymphoma [7]. Furthermore, manufacturing failure rates due to product-related issues range between 4 and 7.6% [95], highlighting the urgent need for standardized, high-quality leukapheresis products.
The establishment of universal Critical Quality Attributes (CQAs) for leukapheresis products represents a pivotal step toward ensuring consistent manufacturing success. CQAs are biological properties or characteristics that must be controlled within appropriate limits to ensure the final product achieves the desired safety, efficacy, and stability. For leukapheresis, these encompass measurable metrics such as cell viability, composition, recovery, and functional potency. Standardizing these attributes is essential for creating reproducible, scalable, and distributed manufacturing models for autologous cell therapies [7].
Quantitative Specifications for Leukapheresis Products
Comprehensive protocol standardization begins with defining quantitative benchmarks for leukapheresis products. The following tables summarize key parameter ranges established through recent comparative studies and clinical practice guidelines.
Table 1: Key Cellular Parameters for Leukapheresis Products
| Parameter | Target Range | Clinical Significance |
|---|---|---|
| Post-thaw Viability | ≥ 90% [7] | Indicates effective cryopreservation and predicts T-cell expansion potential. |
| Lymphocyte Proportion | ~66.6% (vs. ~52.2% in PBMCs) [7] | Higher lymphocyte count correlates with enhanced CAR-T potential. |
| CD3+ T-cell Proportion | 42-56% [7] | Ensures adequate starting population for T-cell therapy manufacturing. |
| Cell Concentration (Pre-cryo) | 4.06–5.12 × 10^7 cells/mL [7] | Maintains cell health during processing and cryopreservation. |
| Hematocrit Level | 5-10% [7] | Reduces non-cellular impurities that can impact cryopreservation efficiency. |
Table 2: Critical Process Parameters for Leukapheresis Cryopreservation
| Process Step | Key Parameter | Standardized Specification |
|---|---|---|
| Formulation | Cryoprotectant | Clinical-grade CS10 (10% DMSO) [7] |
| Formulation | Target Cell Concentration | ~5 × 10^7 cells/mL [7] |
| Formulation | Processing Time | 43–108 minutes (via closed automated system) [7] |
| Freezing | Time from CPA addition to freezing | ≤ 120 minutes [7] |
| Freezing | System | Controlled-rate freezer (e.g., Thermo Profile 4) [7] |
| Storage | Format | 20 mL/bag, containing ≥ 1 × 10^9 cells [7] |
Established Experimental Protocols for CQA Validation
Protocol: Comprehensive Functional Profiling of Cryopreserved Leukapheresis Products
This protocol enables a systematic comparison of cryopreserved leukapheresis against traditional peripheral blood mononuclear cell (PBMC) starting materials [7].
- Objective: To evaluate the impact of the freezing process on the performance and phenotypic profile of cryopreserved leukapheresis products versus PBMCs.
- Materials:
- Leukapheresis Product: Collected via automated closed-system apheresis.
- Cryopreservation Medium: CS10 (10% DMSO).
- Equipment: Controlled-rate freezer (e.g., Thermo Profile 4), flow cytometer.
- Methodology:
- Sample Preparation: Starting with an equivalent amount of leukapheresis material, split the product for parallel processing as (a) fresh leukapheresis, (b) cryopreserved leukapheresis, and (c) PBMCs for cryopreservation.
- Cryopreservation: Cryopreserve the leukapheresis product at a target concentration of ~5 × 10^7 cells/mL, strictly limiting the interval from cryoprotectant addition to controlled-rate freezing initiation to ≤ 120 minutes.
- Thawing and Analysis: Thaw samples and assess:
- Viability: Using trypan blue exclusion or automated cell counters.
- Immune Phenotyping: By flow cytometry to quantify major lymphocyte subsets (T cells: CD3+, B cells: CD19+, NK cells: CD56+).
- Lymphocyte Proportion: Compare the percentage of lymphocytes between fresh leukapheresis, cryopreserved leukapheresis, and cryopreserved PBMCs.
- Expected Outcomes: Cryopreserved leukapheresis products are expected to achieve ≥ 90% post-thaw viability and exhibit a statistically significant higher lymphocyte proportion compared to cryopreserved PBMCs, with no significant differences in the proportions of T, B, and NK cell subsets between fresh and cryopreserved leukapheresis [7].
Protocol: Pre-Apheresis Patient Evaluation and Suitability Assessment
A standardized pre-apheresis evaluation is crucial for ensuring the collection of high-quality starting material, especially in heavily pre-treated patients [95].
- Objective: To systematically assess patient eligibility and optimize timing for leukapheresis to maximize lymphocyte yield and fitness.
- Materials:
- Patient Medical Records.
- Blood Analyzer for complete blood count (CBC) with differential.
- Flow Cytometer for immunophenotyping.
- Methodology:
- Eligibility Determination:
- Review medical history, performance status, and underlying hematological disease per established guidelines (e.g., EBMT-JACIE) [95].
- Suitability Assessment:
- Blood Tests: Obtain pre-apheresis values for absolute lymphocyte count (ALC), mononuclear cell (MNC) count, and preferably CD3+ cell count [95].
- Timing: Establish protocols defining the timeline for sharing information between clinicians and apheresis specialists (≤30 days). For lymphoma patients, consider leukapheresis before commencing salvage therapy [95].
- Venous Access: Assess venous access for the procedure [95].
- Eligibility Determination:
- Key Parameters to Monitor:
- Absolute Lymphocyte Count (ALC): A critical determinant of collection yield.
- Previous Therapies: Identify treatments causing lymphocytopenia or interfering with lymphocyte fitness; consider wash-out from chemotherapy and corticosteroids [95].
- Infectious Status: Screen and reassess before the procedure.
This structured evaluation helps identify potential collection challenges early and allows for procedural optimization, contributing to more consistent leukapheresis CQAs.
Regulatory Framework and CQA Definition
The regulatory landscape for CAR-T starting materials is complex but provides essential guidance for defining CQAs. A consensus position among experts is that the process for preparing CAR-T starting materials, including leukapheresis and cryopreservation, should follow current Good Tissue Practice (cGTP) requirements, while subsequent manufacturing steps follow both cGMP and cGTP regulations [96].
Applying cGTP rather than cGMP to the initial collection and cryopreservation steps focuses on preventing the introduction and transmission of communicable diseases, which is the primary safety concern for starting materials. This approach avoids placing an undue cGMP burden on collection facilities (e.g., hospitals, apheresis centers) which would increase complexity and negatively impact patient access to these therapies [96]. This regulatory distinction helps clarify that the CQAs for leukapheresis products are centered on safety, identity, and viability, rather than the more extensive pharmaceutical quality attributes required for the final drug product.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents and Materials for Standardized Leukapheresis Processing
| Reagent/Material | Function | Application Note |
|---|---|---|
| Cryoprotectant CS10 | Prevents ice crystal formation during freezing; contains 10% DMSO. | Ensures consistent cryoprotection; minimizes erythrocyte volume interference with DMSO concentration [7]. |
| Magnetic Cell Sorting Beads | Isolation of specific cell populations (e.g., CD4+/CD8+ T cells) via antibody-conjugated magnetic beads. | Enables defined CD4:CD8 ratios in final product; critical for Treg manufacturing [97] [98]. |
| Rapamycin | mTOR inhibitor used during T-cell expansion. | Selectively inhibits effector T-cell proliferation while allowing robust Treg expansion, maintaining functional phenotype [98]. |
| Closed Automated Processing System | Integrated unit operations for leukapheresis formulation and cryopreservation. | Reduces processing time and variability; essential for scalable and distributed manufacturing [7]. |
Workflow and CQA Relationship Visualization
The following diagram illustrates the integrated workflow for standardized leukapheresis processing, highlighting the critical control points where CQAs are monitored and ensured.
The relationship between the controlled process parameters and the resulting Critical Quality Attributes is fundamental to protocol standardization. The following diagram maps these key interactions, providing a logical framework for quality by design.
The establishment of universal Critical Quality Attributes for leukapheresis products, supported by standardized protocols and a clear regulatory framework, is a foundational pillar for the future of autologous cell therapy. The quantitative benchmarks, experimental methodologies, and structured workflows detailed in this application note provide a roadmap for researchers and drug development professionals to enhance the consistency, scalability, and ultimately, the clinical success of these revolutionary treatments. Continued collaboration between apheresis specialists, hematologists, manufacturers, and regulators remains critical to refine these standards and ensure that advanced therapies can reach all eligible patients.
Conclusion
Leukapheresis is not merely a collection step but a foundational determinant of success in autologous cell therapy. Mastering this process requires a holistic approach that integrates foundational knowledge, robust methodology, proactive troubleshooting, and rigorous validation. The strategic adoption of cryopreserved leukapheresis, coupled with yield-driven collection protocols and automated integrated platforms, presents a clear path toward more scalable, resilient, and cost-effective manufacturing. Future progress hinges on the continued standardization of leukapheresis processes, large-scale clinical validation of optimized materials, and the wider implementation of decentralized manufacturing networks. These advances are critical for breaking current production bottlenecks and ultimately fulfilling the promise of accessible, life-saving cell therapies for a broader patient population.
References
- 1. CAR T-Cell Therapies Market Size to Reach 34.42 billion ... [https://finance.yahoo.com/news/car-t-cell-therapies-market-141000688.html]
- 2. CAR T-Cell Therapy Market Size to Surpass USD 146.55 [https://www.globenewswire.com/news-release/2025/10/30/3177604/0/en/CAR-T-Cell-Therapy-Market-Size-to-Surpass-USD-146-55-Billion-by-2034-Fueled-by-Innovations-in-Cancer-Treatment-and-Growing-Demand-for-Personalized-Therapies.html]
- 3. CAR T-cell Therapy Market Forecasts Indicate Strong ... [https://www.prnewswire.com/news-releases/car-t-cell-therapy-market-forecasts-indicate-strong-growth-trajectory-in-the-7mm-during-the-forecast-period-20252034--delveinsight-302596178.html]
- 4. CAR T-Cell Therapy Market Size to Hit USD ... [https://www.precedenceresearch.com/car-t-cell-therapy-market]
- 5. Next-Gen CAR-T Therapy Market Trends 2025 FDA ... [https://www.towardshealthcare.com/insights/next-gen-car-t-therapy-market-sizing]
- 6. Autologous cryopreserved leukapheresis cellular material ... [https://www.sciencedirect.com/science/article/pii/S1465324919308667]
- 7. a multi-platform comparative study | Scientific Reports [https://www.nature.com/articles/s41598-025-14865-5]
- 8. Leukapheresis - an overview [https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/leukapheresis]
- 9. Leukapheresis Procedure and Removing White Blood ... [https://www.akadeum.com/types-of-cell-separation/leukaphresis/?srsltid=AfmBOorZ1rLV5DrWSVHPW4s_iyldl_WSEu2kZLPWVNFjW831Q0D-Zzb0]
- 10. Autologous Cell Therapy [https://www.akadeum.com/technology/cell-expansion/autologous/?srsltid=AfmBOorAQGwhr2XsTVCCt6_Cr__vSeGhiTx9H0JYikBM6d5JA1wyK44d]
- 11. Enriching leukapheresis improves T cell activation and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7917474/]
- 12. Current Challenges in Providing Good Leukapheresis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7290830/]
- 13. Leukapheresis Market Size, Trends & Forecast 2024–2030 [https://www.strategicmarketresearch.com/market-report/leukapheresis-market]
- 14. Risks of leukapheresis and how to manage them-A non- ... [https://pubmed.ncbi.nlm.nih.gov/30287071/]
- 15. Risks of leukapheresis and how to manage them—A non- ... [https://www.sciencedirect.com/science/article/abs/pii/S1473050218303598]
- 16. How to Plan an Efficient Leukocytapheresis to Collect T Cells ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10098271/]
- 17. Leukapheresis guidance and best practices for optimal ... [https://www.sciencedirect.com/science/article/pii/S1465324922006417]
- 18. Leukapheresis Market Size, Forecast & Share Analysis ... [https://www.mordorintelligence.com/industry-reports/leukapheresis-market]
- 19. Asia Pacific Leukapheresis Market Size, Trends, Scope & Fore [https://www.verifiedmarketresearch.com/product/leukapheresis-market/asia-pacific/]
- 20. Leukapheresis Market Size, Share & Growth Trends 2035 [https://www.researchnester.com/reports/leukapheresis-market/5887]
- 21. Leukapheresis Market Report Size, Share & Global Trends ... [https://www.polarismarketresearch.com/industry-analysis/leukapheresis-market]
- 22. Leukapheresis Market Size to Hit USD 628.80 Mn by 2034 [https://www.precedenceresearch.com/leukapheresis-market]
- 23. Leukapheresis Products Market Outlook 2025 to 2035 [https://www.futuremarketinsights.com/reports/leukapheresis-products-market]
- 24. Addressing Variability in Donor Tissues and Cells - NCBI - NIH [https://www.ncbi.nlm.nih.gov/books/NBK544028/]
- 25. Enriching leukapheresis improves T cell activation and ... [https://discovery.ucl.ac.uk/id/eprint/10123750/]
- 26. a multi-platform comparative study - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC12354897/]
- 27. Advanced Therapies Week 2025: Decentralized ... [https://www.phacilitate.com/insights-resources/advanced-therapies-week-2025-decentralized-manufacturing-point-care?page=9&azLetterField=]
- 28. The road to accessible CAR-T therapies: Inside a world- ... [https://www.clinicaltrialsarena.com/sponsored/the-road-to-accessible-car-t-therapies-inside-a-world-first-innovation-for-decentralised-leukapheresis/]
- 29. Scaling Access: How Mobile Leukapheresis and ... [https://biobridgeglobal.org/uncategorized/scaling-access-mobile-leukapheresis-access-and-innovation-in-cell-and-gene-therapy/]
- 30. Global Leukaphereis Product Market Trends: AI-Driven ... [https://www.linkedin.com/pulse/global-leukaphereis-product-market-trends-ai-driven-marketing-u3jcc/]
- 31. Apheresis Equipment Market Analysis 2025-2035 : Trends ... [https://www.pharmiweb.com/press-release/2025-02-20/apheresis-equipment-market-analysis-2025-2035-trends-innovations-and-growth-prospects]
- 32. Automated and Closed Cell Therapy Processing Systems ... [https://www.biospace.com/press-releases/automated-and-closed-cell-therapy-processing-systems-market-size-to-hit-usd-11-36-billion-by-203]
- 33. Apheresis Equipment Market Size, Share Analysis | CAGR ... [https://media.market.us/global-apheresis-equipment-market-news/]
- 34. Exploring The Market For Closed-Loop Cell Therapy ... [https://www.cellandgene.com/doc/exploring-the-market-for-closed-loop-cell-therapy-production-0001]
- 35. How Single-Use and Closed Systems Work Better Together [https://www.enablebiotech.com/news/maximizing-impact-in-cell-therapy-manufacturing-how-single-use-and-closed-systems-work-better-together]
- 36. Closed-system manufacturing of therapeutic NK cells using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12238007/]
- 37. Leukapheresis Procedure and Removing White Blood ... [https://www.akadeum.com/types-of-cell-separation/leukaphresis/?srsltid=AfmBOooyFG65lIw1peWK5K28RGdTUGJ5MHkH73nbF9oHvIqZQ16OFpBE]
- 38. Optimizing PBMC Viability and Recovery: Updated Best ... [https://cgt.global/pbmc-viability-and-recovery/]
- 39. Impact of cellular composition and T‐cell senescence ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12432817/]
- 40. T-cell counts in peripheral blood at leukapheresis predict ... [https://www.nature.com/articles/s41598-022-23589-9]
- 41. Yield-driven approach optimizes apheresis of CD19 ... [https://pubmed.ncbi.nlm.nih.gov/40590814/]
- 42. Current Challenges in Providing Good Leukapheresis ... [https://www.mdpi.com/2073-4409/9/5/1225]
- 43. Yield-driven approach optimizes apheresis of CD19 ... [https://www.sciencedirect.com/science/article/pii/S146532492500739X?dgcid=rss_sd_all]
- 44. Autologous Lymphapheresis for the Production of Chimeric ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5398918/]
- 45. Comparative analysis and process optimization for ... [https://www.nature.com/articles/s41598-025-89686-7]
- 46. Cryopreserved Leukopaks: Addressing the Viability Myth [https://dls.com/resources/cryopreserved-leukopaks-addressing-the-viability-myth/]
- 47. Advancing Cell Therapy with Cryopreserved Leukopaks [https://www.isctglobal.org/telegrafthub/blogs/isct-head-office1/2025/09/10/advancing-cell-therapy-with-cryopreserved-leuk]
- 48. Fresh versus Frozen: Effects of Cryopreservation on CAR T Cells [https://pmc.ncbi.nlm.nih.gov/articles/PMC6612752/]
- 49. Trends in cell collection and apheresis practices [https://www.sciencedirect.com/science/article/pii/S1473050225001892]
- 50. Factors affecting lymphocyte collection efficiency for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6499685/]
- 51. Factors Affecting Lymphocyte Collection Efficiency and ... [https://www.sciencedirect.com/science/article/pii/S0006497118706678]
- 52. Autologous Cryopreserved Leukapheresis Cellular ... [https://pubmed.ncbi.nlm.nih.gov/31837735/]
- 53. Fragile Immune Cells Require Gentle Handling [https://www.akadeum.com/blog/fragile-immune-cells/?srsltid=AfmBOopRwVLYC4m5AFu1ADlKLXh4F6hOiSfaUia7-8fwgyyBYeSY1oEI]
- 54. Cryo-processed Leukapheresis Using Automated Closed ... [https://www.sciencedirect.com/science/article/abs/pii/S1465324925003779]
- 55. Analysis of the impact of handling and culture ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10451065/]
- 56. Cryopreservation: An Overview of Principles and Cell- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7995302/]
- 57. Protocols and Best Practices for Freezing Cells [https://www.stemcell.com/cryopreservation-basics-protocols-and-best-practices-for-freezing-cells]
- 58. Cryopreservation of mammalian cell lines video protocol [https://www.abcam.com/en-us/technical-resources/protocols/cryopreservation-of-mammalian-cell-lines]
- 59. The Impact of Varying Cooling and Thawing Rates on ... [https://www.nature.com/articles/s41598-019-39957-x]
- 60. Optimisation of cryopreservation conditions, including storage ... [https://bmcmolcellbiol.biomedcentral.com/articles/10.1186/s12860-024-00516-6]
- 61. Cryopreservation as a Key Element in the Successful Delivery ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7727450/]
- 62. European survey on CAR T-Cell analytical methods from ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12058815/]
- 63. Systematic discovery of CRISPR-boosted CAR T cell ... [https://www.nature.com/articles/s41586-025-09507-9]
- 64. Recent advances in universal chimeric antigen receptor T cell ... [https://jhoonline.biomedcentral.com/articles/10.1186/s13045-025-01737-8]
- 65. Terumo Blood and Cell Technologies Demonstrates ... [https://www.terumobct.com/en/gl/our-company/news-and-events/news-releases/2025/terumo-blood-and-cell-technologies-demonstrates-integrated-3-in-.html]
- 66. strategies for more affordable CAR-T cell manufacturing [https://appliedcells.com/affordable-car-t-manufacturing/]
- 67. Forecasting Budgets for Advanced Therapy R&D [https://www.labmanager.com/forecasting-budgets-for-advanced-therapy-r-d-34491]
- 68. Guide to Cell Therapy GMP Manufacturing for Autologous ... [https://www.thermofisher.com/blog/behindthebench/guide-to-cell-therapy-gmp-manufacturing-for-autologous-car-t-and-beyond/]
- 69. Leukapheresis Market Report 2025 [https://www.researchandmarkets.com/reports/6103612/leukapheresis-market-report?srsltid=AfmBOoreEfhELsbpoOrjdZD7h-tm8ZGhnbZWvXd1KsTmOhVJ1KjaMkoW]
- 70. Leukapheresis guidance and best practices for optimal ... [https://pubmed.ncbi.nlm.nih.gov/35718701/]
- 71. Validation of an automated quality control method to test ... [https://www.sciencedirect.com/science/article/pii/S2531137924028414]
- 72. Current practices in peripheral blood stem cell processing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12283502/]
- 73. Essential Quality Control Considerations for Cell Culture [https://absbio.com/white-papers/essential-quality-control-considerations-for-cell-culture/]
- 74. Cryopreservation for Critical Starting Material Products [https://www.organabio.com/cryopreservation-for-critical-starting-material-products-benefits-of-effective-cold-chain-management/?srsltid=AfmBOoq1NxIyblspNPybWAeLLchsxdSP0GZQLxq9h2_mU0wjgzpPfOhc]
- 75. Cryopreservation: Key to Scalable & Global Cell Therapy ... [https://www.atlantisbioscience.com/blog/cryopreservation-in-cell-therapy-enabling-scalability-quality-and-global-distribution/?srsltid=AfmBOor-00jyw6Y8JrQj_593mVpyeawAZJvoohuXM8_v0tta__sjP3a6]
- 76. Solving Logistics Bottlenecks to improve your fulfillment [https://eplogistics.com/blog/logistics-bottlenecks/]
- 77. Practice of Cryopreservation of Cellular Starting Materials from ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12446400/]
- 78. Advantages of high cell concentration prior to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11073629/]
- 79. Distributed manufacturing benefits: Revolutionizing ... [https://ultimaker.com/learn/distributed-manufacturing-benefits-revolutionizing-production/]
- 80. How Localized Logistics Can Eliminate Delays and ... [https://www.lslog.com/how-localized-logistics-can-eliminate-delays-and-accelerate-supply-chain-output/]
- 81. Top PBMC Suppliers for 2026: Fresh, Cryopreserved, and ... [https://cgt.global/top-pbmc-suppliers-for-2026-fresh-cryopreserved-and-gmp-grade-options-compared/]
- 82. Phenotypic and functional comparisons between ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12576105/]
- 83. Cryopreserved anti-CD22 and bispecific anti-CD19/22 CAR T ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9798176/]
- 84. Impact of cryopreservation on CAR T production and ... [https://www.frontiersin.org/journals/oncology/articles/10.3389/fonc.2022.1024362/full]
- 85. Comparison of axicabtagene ciloleucel and tisagenlecleucel ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12306477/]
- 86. lisocabtagene maraleucel versus tisagenlecleucel - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8953336/]
- 87. Axicabtagene ciloleucel compared to tisagenlecleucel for ... [https://haematologica.org/article/view/10697]
- 88. New insights on potency assays from recent advances and ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2025.1597888/full]
- 89. If You're Gonna Play in Cell Therapy, You've Got to.... [https://biobridgeglobal.org/uncategorized/back-to-the-basics-of-access-autologous-cell-therapy-and-mobile-leukapheresis/]
- 90. Emerging CAR immunotherapies: broadening therapeutic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12321946/]
- 91. CAR-T cell therapy for cancer: current challenges and ... [https://www.nature.com/articles/s41392-025-02269-w]
- 92. Two subsets of stem-like CD8+ memory T cell progenitors with ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7610790/]
- 93. CAR-T manufactured from frozen PBMC yield efficient ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2022.1007042/full]
- 94. A comprehensive single-cell map of T cell exhaustion ... [https://www.nature.com/articles/s41467-022-35238-w]
- 95. Managing leukapheresis in adult and pediatric patients ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10645345/]
- 96. Regulatory Requirements For CAR-T Starting Materials [https://www.cellandgene.com/doc/regulatory-requirements-for-car-t-starting-materials-0001]
- 97. CAR-T cell manufacturing: Major process parameters and next ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10791545/]
- 98. Treg cell therapy manufacturability: current state of the art ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12141221/]