Optimizing MSC Proliferation Capacity Under GMP Conditions: A Comprehensive Guide to Scalable and Clinically Compliant Manufacturing
This article provides a comprehensive analysis of strategies for optimizing mesenchymal stem cell (MSC) proliferation capacity while maintaining strict Good Manufacturing Practice (GMP) compliance for clinical applications.
Optimizing MSC Proliferation Capacity Under GMP Conditions: A Comprehensive Guide to Scalable and Clinically Compliant Manufacturing
Abstract
This article provides a comprehensive analysis of strategies for optimizing mesenchymal stem cell (MSC) proliferation capacity while maintaining strict Good Manufacturing Practice (GMP) compliance for clinical applications. Covering foundational biology, advanced expansion methodologies, troubleshooting of common challenges, and validation frameworks, it serves as an essential resource for researchers and drug development professionals working to translate MSC-based therapies from bench to bedside. The content integrates recent advancements in GMP-compliant culture media, automated bioreactor systems, and quality control protocols to enable robust, scalable MSC manufacturing that meets regulatory standards for clinical use.
Understanding MSC Biology and GMP Requirements: The Foundation for Clinical Translation
Core MSC Definitions & The ISCT Criteria
Q1: What are the minimum criteria to define human Mesenchymal Stromal Cells? According to the International Society for Cellular Therapy (ISCT), human MSCs must simultaneously satisfy three minimum criteria:
- Plastic Adherence: The cells must adhere to a plastic surface when maintained under standard culture conditions [1].
- Specific Surface Marker Expression: ≥95% of the cell population must express the positive markers CD73, CD90, and CD105. Conversely, ≤2% of the population must lack expression of the negative markers CD34, CD45, CD11b or CD14, CD79α or CD19, and HLA-DR [1] [2].
- Trilineage Differentiation Potential: Under in vitro stimulating conditions, the cells must possess the capacity to differentiate into osteoblasts (bone), adipocytes (fat), and chondrocytes (cartilage) [1] [3] [2].
Q2: Why is donor source a critical variable in GMP-compliant MSC manufacturing? Donor characteristics significantly impact MSC properties, which is a major source of heterogeneity in clinical-grade products [1] [2]. This variability must be characterized and controlled for in extended passage studies.
- Donor Age: Fetal and young donor MSCs often demonstrate a higher proliferation capacity and can surpass 30 population doublings more reliably than cells from adult donors [2].
- Tissue Source: MSCs can be isolated from bone marrow (BM), adipose tissue (AD), umbilical cord (UC), placenta (PL), and other tissues [1] [3]. The "HOX code," a stable pattern of gene expression, is tissue-specific and can influence differentiation potential [3].
- Breed/Species: Genetic background affects differentiation potential; for instance, osteogenic potential can vary significantly between breeds [2].
Table 1: Impact of Donor Characteristics on MSC Properties
| Donor Characteristic | Impact on Proliferation | Impact on Differentiation | Relevance to GMP |
|---|---|---|---|
| Age (Fetal/Young) | Enhanced: Higher population doublings, lower senescence [2] | Variable effect on adipogenic, osteogenic, and chondrogenic potential [2] | Selecting young donors may improve batch consistency and expansion yield. |
| Tissue Source | Varies; UC and PL-MSCs can proliferate faster than BM-MSCs in specific media [4] | Defines the inherent "HOX code," influencing lineage preference [3] | Critical for defining the Master Cell Bank and its intended therapeutic application. |
| Genetic Background | Can influence proliferation duration and rate [2] | Significant: Breed can strongly affect osteogenic and adipogenic outcomes [2] | Highlights the need for rigorous donor screening and selection criteria. |
Troubleshooting MSC Culture & Characterization
Q3: How can we maintain MSC stemness and potency during extended ex vivo expansion? The loss of stemness during passage is characterized by reduced proliferation, increased senescence, and diminished differentiation capacity [3]. This is regulated by key genetic factors.
- Key Regulators of Stemness:
- Transcription Factors: OCT4, SOX2, TWIST1, and HOX genes maintain the undifferentiated state, promote proliferation, and inhibit senescence. For example, OCT4 overexpression enhances proliferation and chondrogenesis by suppressing senescence markers like p16 and p21 [3].
- Epigenetic Regulators: EZH2 (increased by TWIST1) silences senescence genes via H3K27me3 modification [3].
- Optimized Culture Conditions:
- Serum Choice: Using Human Serum (HS) or animal component-free, GMP-compliant media (e.g., MSC-Brew GMP Medium) can enhance proliferation rates and maintain immunomodulatory properties compared to standard Fetal Bovine Serum (FBS) [5] [4].
- Recombinant Proteins: Growth factors like FGF-basic (FGF-2) are essential for maintaining proliferation and preventing spontaneous differentiation [6].
The following diagram illustrates the molecular network that regulates MSC stemness and how it is influenced by external culture conditions.
Q4: Our MSCs are failing adipogenic or osteogenic differentiation. What could be wrong? Failed differentiation is a common issue often linked to suboptimal culture conditions, donor variability, or cell senescence.
Table 2: Troubleshooting Trilineage Differentiation Assays
| Problem | Possible Causes | Solutions & GMP Considerations |
|---|---|---|
| Weak Adipogenic Differentiation | • High passage number/senescence [3]• Donor age & breed effects [2]• Suboptimal induction cocktail | • Use low-passage cells (P3-P5) [4].• Test multiple donors and select a high-potency cell bank.• Validate reagent concentrations (e.g., IBMX, indomethacin, insulin [4]). |
| Weak Osteogenic Differentiation | • Donor breed/biological variability [2]• Inconsistent β-glycerophosphate addition [4]• Over-confluent cells at induction start | • Select donors with proven high osteogenic potential [2].• Strictly follow protocol timing for β-glycerophosphate [4].• Start induction at 60-80% confluence. |
| General Differentiation Failure | • Mycoplasma contamination• Serum lot variability• Spontaneous differentiation during expansion | • Implement rigorous sterility testing (e.g., Bact/Alert, Mycoplasma assays) [5].• Use large, pre-qualified serum lots or xeno-free media [5] [4].• Check key stemness markers (e.g., OCT4, TWIST1) before induction [3]. |
Essential Experimental Protocols & Reagents
Q5: What are the detailed protocols for confirming trilineage differentiation? The following are standard in vitro protocols for inducing MSC differentiation. Always include unstained controls and undifferentiated MSCs cultured in standard growth medium as negative controls.
A. Adipogenic Differentiation Protocol:
- Seeding: Seed MSCs at a density of 7.5 × 10⁴ cells in a 35-mm² dish in standard growth medium and allow to adhere overnight [4].
- Induction: Replace the medium with adipogenic induction medium. A standard formulation includes:
- DMEM base
- 10% FBS (or validated HS/serum-free alternative)
- 500 μM IBMX (isobutylmethylxanthine)
- 200 μM Indomethacin
- 1 μM Dexamethasone
- 10 μM Insulin [4]
- Maintenance: Culture for 3-4 weeks, replacing the induction medium twice per week.
- Staining & Analysis: Wash, fix with 4% formaldehyde, and stain with 0.3% Oil Red O solution to visualize lipid droplets [4].
B. Osteogenic Differentiation Protocol:
- Seeding: Seed MSCs at a density of 4.5 × 10⁴ cells in a 35-mm² dish [4].
- Induction: Replace with osteogenic induction medium:
- DMEM base
- 10% FBS/HS
- 0.1 μM Dexamethasone
- 300 μM Ascorbic Acid [4]
- Enhancement: On day 7, add 10 mM β-glycerophosphate to the medium [4].
- Maintenance: Culture for 3-4 weeks, replacing the medium every 3 days.
- Staining & Analysis: Wash, fix, and stain with Alizarin Red S to detect calcium deposits [4].
C. Chondrogenic Differentiation Protocol:
- Method: Typically performed in a pellet culture system or micromass to promote cell-cell interactions.
- Induction: Requires a medium supplemented with key factors like Transforming Growth Factor-beta (TGF-β), often TGF-β3, and Ascorbic Acid [3].
- Staining: The resulting cartilage pellet is typically sectioned and stained with Toluidine Blue or Safranin O to detect sulfated proteoglycans in the extracellular matrix.
Q6: What are the key reagents for successful GMP-compliant MSC research? Using qualified, consistent reagents is fundamental for reproducible GMP research.
Table 3: Research Reagent Solutions for MSC Culture & Differentiation
| Reagent Category | Specific Examples & Functions | GMP-Compliant Application |
|---|---|---|
| Basal Media | Dulbecco’s Modified Eagle Medium (DMEM), MEM α [5] | Foundation for all media formulations; requires GMP-grade sourcing. |
| Media Supplements | MSC-Brew GMP Medium: Animal component-free, enhances proliferation & maintains potency [5].Human Serum (HS): Alternative to FBS; reduces immunogenicity risk, enhances proliferation [4]. | Critical for moving from research-grade (FBS) to clinical-grade manufacturing. |
| Growth Factors / Recombinant Proteins | FGF-basic (FGF-2): Maintains proliferation and stemness [6].TGF-β3: Essential for chondrogenic differentiation [3] [6].BMP-4: Can block unwanted differentiation in stem cultures [6]. | Defined, recombinant proteins ensure batch-to-batch consistency and eliminate animal-derived components. |
| Dissociation Enzymes | Trypsin-EDTA, Liberase [2] | GMP-grade enzymes are required for cell passaging and harvesting. |
| Characterization Kits | BD Stemflow Human MSC Analysis Kit (Flow Cytometry) [5] | Standardized, validated kits ensure accurate and consistent ISCT marker profiling. |
The workflow below summarizes the key stages from MSC isolation to full characterization, integrating the critical quality control checkpoints essential for GMP.
The Critical Importance of GMP Standards in MSC-Based Advanced Therapy Medicinal Products
Technical Support Center
Troubleshooting Guides
Guide 1: Addressing Slow MSC Proliferation in Extended Culture
Problem: Decreased doubling times and increased senescence observed during extended passaging under GMP conditions.
Investigation & Resolution:
- Verify Culture Media Composition: Test and compare multiple GMP-compliant, animal component-free media formulations. Research shows MSC-Brew GMP Medium demonstrated superior performance with lower doubling times across passages compared to standard media and other commercial options [7].
- Monitor Critical Quality Attributes (CQAs): Track population doubling level, cell yield, viability, and confluence at each passage as In-Process Controls (IPCs) [8].
- Assess Colony Forming Unit (CFU) Capacity: Seed cells at low density (20-500 cells per dish) and culture for 10 days to evaluate potency preservation. Crystal violet staining quantifies colony formation [7].
- Control Process Parameters: Maintain consistent seeding density (e.g., 5 × 10³ cells/cm²) and passage at 80-90% confluency to ensure standardized growth conditions [7].
Guide 2: Ensuring Consistent Immunomodulatory Potency
Problem: Batch-to-batch variability in MSC immunomodulatory function despite consistent expansion protocols.
Investigation & Resolution:
- Source Selection: Consider tissue source impact on potency. Studies indicate Wharton's Jelly MSCs show strongest inhibition of PBMC proliferation and enhance regulatory T cell populations, while decidua-derived MSCs excel in anti-inflammatory cytokine secretion [9].
- Functional Potency Assays: Implement BrdU proliferation assays to measure PBMC inhibition and flow cytometry to quantify T cell population changes [9].
- Secretome Analysis: Quantify prostaglandin E2 (PGE-2), IL-10, IL-12, and IL-17 levels via ELISA. Transcriptome analysis of IL-6, HGF, and TGF-β provides additional potency markers [9].
- Donor Screening: Document donor age, gender, and health status, as these significantly impact MSC therapeutic efficacy and expansion potential [10].
Table 1: Performance of GMP-Compliant, Animal Component-Free Media Formulations in MSC Culture
| Media Formulation | Average Doubling Time | Colony Forming Capacity | MSC Marker Expression (%) | Post-Thaw Viability (%) |
|---|---|---|---|---|
| MSC-Brew GMP Medium | Lowest reported [7] | Highest colony formation [7] | >95% [7] | >95% [7] |
| MesenCult-ACF Plus Medium | Higher than MSC-Brew [7] | Lower than MSC-Brew [7] | >95% [7] | >95% [7] |
| Standard FBS Media | Highest reported [7] | Lowest colony formation [7] | >95% [7] | Not specified |
Table 2: Immunomodulatory Performance of MSCs from Different Tissue Sources
| MSC Source | PBMC Proliferation Inhibition | Treg Cell Enhancement | PGE-2 Secretion | IL-10 Secretion |
|---|---|---|---|---|
| Wharton's Jelly | Strongest [9] | Strongest [9] | Highest [9] | Moderate [9] |
| Decidua Tissue | Moderate [9] | Moderate [9] | Moderate [9] | Highest [9] |
| Adipose Tissue | Not specified | Not specified | Not specified | Not specified |
| Bone Marrow | Not specified | Not specified | Not specified | Not specified |
Detailed Experimental Protocols
Protocol 1: Media Comparison Study for GMP-Compliant MSC Expansion
Objective: Evaluate efficacy of animal component-free media formulations on MSC proliferation and potency.
Methodology:
- Isolate MSCs from infrapatellar fat pad tissue digested with 0.1% collagenase for 2 hours at 37°C [7]
- Centrifuge at 300 ×g for 10 minutes, filter through 100μm filter, and resuspend in test media [7]
- Culture cells in parallel using:
- Standard MSC media (MEM α + 10% FBS + gentamicin)
- MesenCult-ACF Plus Medium
- MSC-Brew GMP Medium [7]
- Passage at 80-90% confluency with consistent seeding density (5 × 10³ cells/cm²) [7]
- Doubling Time Calculation: Calculate at each passage using formula:
Doubling Time = (duration × ln2) / ln(final concentration/initial concentration)[7] - CFU Assay: Seed at 20, 50, 100, and 500 cells per dish, culture for 10 days, fix with formalin, and stain with Crystal Violet [7]
- Surface Marker Analysis: At passage 3, analyze CD45-, CD73+, CD90+, CD105+ expression using flow cytometry [7]
Protocol 2: GMP-Validation for Clinical-Grade MSC Banking
Objective: Establish reproducible isolation, expansion, and storage protocols meeting GMP standards.
Methodology:
- Donor Screening: Apply strict inclusion/exclusion criteria with ethics committee approval and informed consent [7]
- GMP-Compliant Isolation: Process tissue in accredited cellular therapy facility following established SOPs [7]
- In-Process Controls: Monitor doubling time, population doubling level, morphology, surface marker profile, cell yield, viability, and confluence [8]
- Quality Control Testing:
- Stability Studies: Assess post-thaw viability and marker expression over 180 days at prescribed storage conditions [7]
- Documentation: Maintain batch records for all process parameters and quality control results [10]
Process Workflow Diagrams
GMP-Compliant MSC Manufacturing Workflow
Media Optimization Study Design
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for GMP-Compliant MSC Research
| Reagent/Equipment | Function | GMP Considerations |
|---|---|---|
| MSC-Brew GMP Medium | Animal component-free expansion | Chemically defined, eliminates xeno-contamination risks [7] |
| Human Platelet Lysate (hPL) | FBS substitute for culture | Reduced immunogenicity, but requires disease transmission controls [10] |
| Collagenase | Tissue digestion and cell isolation | GMP-grade enzymes with documented traceability [7] |
| DMSO-free Cryoprotectants | Cell preservation | Xenogeneic-free, chemically defined formulations [10] |
| BD Stemflow MSC Analysis Kit | Phenotypic characterization | Standardized panels for CD45, CD73, CD90, CD105 [7] |
| Bact/Alert System | Microbiological sterility testing | Validated for cell therapy products [7] |
Frequently Asked Questions
Q1: What are the key differences between research-grade and GMP-compliant MSC manufacturing? GMP-compliant manufacturing requires strict adherence to standardized protocols, comprehensive documentation, quality control testing at multiple stages, validated processes, and traceability of all materials from donor to final product [10] [11]. This ensures product consistency, safety, and efficacy for clinical use.
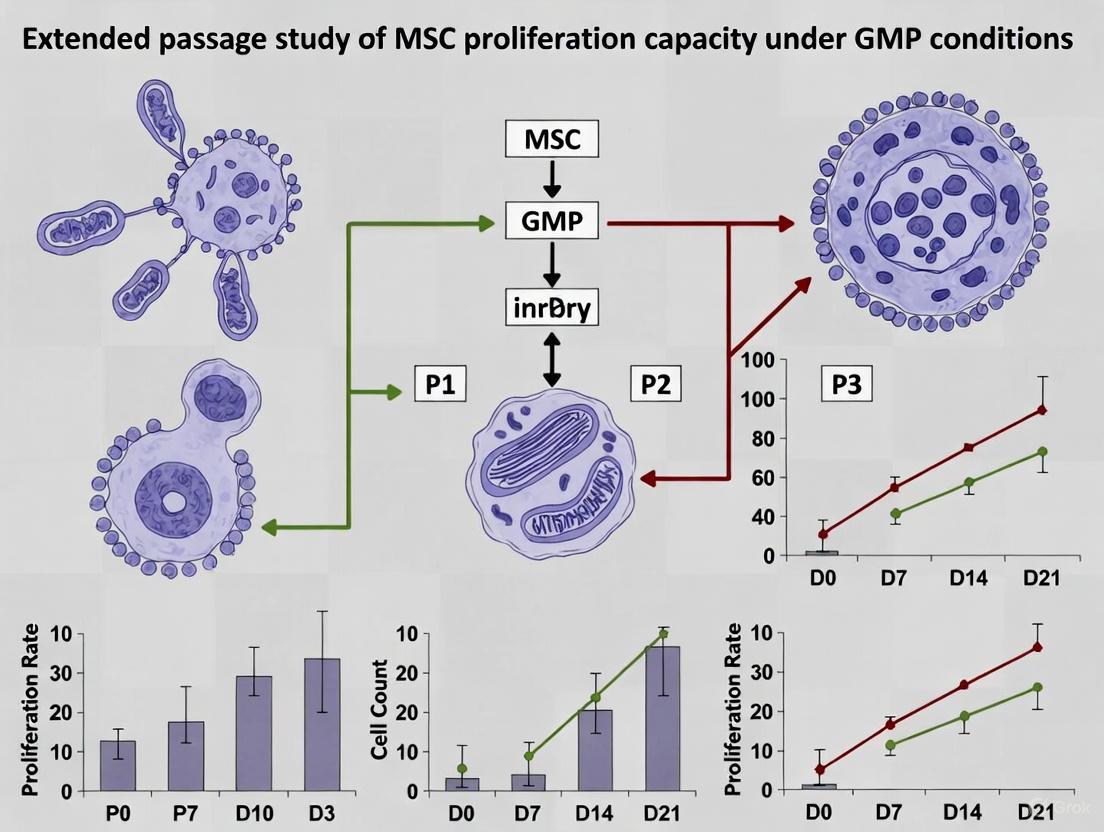
Q2: How does extended passage affect MSC characteristics under GMP conditions? Extended passaging can alter MSC proliferation capacity, differentiation potential, and secretome profile. Regular monitoring of doubling times, karyotypic stability, and surface marker expression is essential. GMP protocols should define maximum population doubling levels or passages to maintain product quality and safety [10].
Q3: What are the EMA's specific GMP requirements for ATMPs? The European Commission has published specific GMP guidelines for ATMPs that adapt EU GMP requirements to ATMP characteristics. These include requirements for quality management, risk management, production, quality control, and specific annexes addressing the novel manufacturing scenarios used for these products [12] [11].
Q4: How should we select between autologous and allogeneic MSC approaches? Autologous therapies eliminate donor-specific immune reactions but pose logistical challenges for acute conditions. Allogeneic 'off-the-shelf' products offer immediate availability but may trigger immune responses. The choice depends on target indication, patient population, and manufacturing capabilities [10].
Q5: What quality control assays are essential for MSC batch release? Essential QC assays include viability testing (>70-95% requirement), sterility (bacteria, fungi), mycoplasma testing, endotoxin levels, identity/purity by flow cytometry, potency assays specific to mechanism of action, and karyotypic analysis to exclude abnormalities [10] [7].
MSC Source Comparison at a Glance
The table below summarizes key quantitative data from comparative studies on Mesenchymal Stem Cells (MSCs) derived from bone marrow (BM), adipose tissue (AT), and umbilical cord blood (UCB) to inform source selection for clinical manufacturing.
| Parameter | Bone Marrow (BM) | Adipose Tissue (AT) | Umbilical Cord Blood (UCB) |
|---|---|---|---|
| Isolation Success Rate | 100% [13] | 100% [13] | 63% [13] |
| Colony Frequency | Intermediate [13] | Highest [13] | Lowest [13] |
| Proliferation/Expansion Potential | Lowest culture period and proliferation capacity [13] | Intermediate [13] | Highest culture period and proliferation capacity [13] |
| Adipogenic Differentiation | Positive [13] | Positive [13] | No adipogenic capacity [13] |
| Osteogenic Differentiation | Positive [14] | Positive [14] | Positive [14] |
| Chondrogenic Differentiation | Positive [14] | Positive [14] | Positive [14] |
| Relative Ease of Harvest | Invasive, can lead to patient morbidity [5] | Minimally invasive, often available as surgical waste [5] [14] | Non-invasive, readily available [14] |
| Initial Cell Yield | Low frequency [14] | High yield (500-fold more MSCs than BM per gram of tissue) [14] | Variable, requires expansion [15] |
Frequently Asked Questions (FAQs) for Clinical-Grade MSC Work
FAQ 1: Why is my isolation of UCB-MSCs failing, and how can I improve the success rate?
The isolation of MSCs from umbilical cord blood has a lower success rate (~63%) compared to bone marrow or adipose tissue [13]. To improve outcomes:
- Use High-Quality Starting Material: Select UCB units with a high mononuclear cell count, for example, ≥1 x 10^6 hematopoietic stem cells/mL [15].
- Optimize Initial Culture Conditions: Supplement the primary isolation medium with 2.5%-10% activated human platelet-rich plasma (aPRP) or AB-human serum instead of FBS to enhance initial cell attachment and growth [15] [16].
- Ensure GMP-Compliant Processing: Implement closed-system processing for mononuclear cell isolation to ensure sterility and compliance [15].
FAQ 2: My MSCs are senescing too quickly during ex vivo expansion. What factors can I adjust?
Premature senescence during expansion is a major hurdle in producing clinically sufficient doses [3]. Key considerations include:
- Culture Media: Switch from FBS to GMP-compliant, xenogeneic-free media formulations like MSC-Brew GMP Medium or StemPro MSC SFM XenoFree. These have been shown to enhance proliferation rates and maintain stemness [5] [16].
- Oxygen Tension: Culture MSCs under physiological hypoxia (e.g., 5% O₂) instead of atmospheric oxygen (20% O₂). Hypoxic conditions have been demonstrated to promote faster expansion and improve functionality [17].
- Passaging Strategy: Avoid over-confluency and high passage numbers. Use consistent, controlled seeding densities (e.g., 5 x 10³ cells/cm²) and characterize the maximum safe passage number for your cell line where stemness is maintained [10].
FAQ 3: How can I reduce contamination from hematopoietic cells in my mouse BM-MSC cultures?
This is a common issue in mouse MSC isolation [17]. Two effective strategies are:
- Use Specialized Supplements: Add supplements like MesenPure to the culture medium, which can significantly reduce hematopoietic (CD45+) cell contamination as early as passage 0 [17].
- Magnetic-Activated Cell Sorting (MACS): Deplete hematopoietic cells using magnetic beads conjugated to antibodies against pan-hematopoietic markers like CD45 [18].
FAQ 4: My MSCs are not differentiating efficiently down the adipogenic lineage. What could be wrong?
Differentiation failure can be source- and protocol-dependent.
- Check Your Cell Source: Be aware that UCB-MSCs have been reported to show no adipogenic differentiation capacity, in contrast to BM- and AT-MSCs [13]. If adipogenesis is a critical endpoint, choose an alternative source.
- Standardize Your Cocktail: For murine MSCs from BM or AT, ensure the use of a robust, standardized 4-component adipogenic cocktail containing Insulin, Dexamethasone, IBMX, and Indomethacin [18].
- Confirm Differentiation Potential: Qualify each new MSC batch by testing its trilineage differentiation potential according to ISCT standards to ensure it meets minimum criteria before starting large-scale experiments [19].
Detailed Experimental Protocols
Protocol 1: GMP-Compliant Isolation and Culture of UCB-MSCs Using aPRP
This protocol outlines a xenogenic-free method for isolating and expanding UCB-MSCs, suitable for clinical applications [15].
Workflow Overview
Materials:
- Iscove modified Dulbecco medium (IMDM)
- Antibiotic-mycotic
- Epidermal Growth Factor (EGF) & basic Fibroblast Growth Factor (bFGF)
- Ficoll Hypaque (density 1.077 g/mL)
- Calcium Chloride (CaCl₂)
Step-by-Step Method:
- UCB Collection: Collect umbilical cord blood from the umbilical vein with informed consent. Use only samples with a high mononuclear cell count (e.g., ≥1 x 10^6 HSCs/mL) for best results [15].
- MNC and aPRP Preparation:
- Centrifuge UCB at 2,000 rpm for 15 min. Separate the cell pellet and plasma [15].
- For aPRP: Centrifuge the plasma at 3,500 rpm for 10 min. Resuspend the platelet pellet in a third of the original plasma volume. Add 100 μL of CaCl₂ per 1 mL of PRP, incubate at 37°C for 30 min to form a clot, and then centrifuge to collect the supernatant (aPRP) [15].
- For MNCs: Dilute the cell pellet 1:1 with PBS and isolate MNCs by density gradient centrifugation using Ficoll Hypaque [15].
- Primary Culture: Plate MNCs at 5 x 10⁴ cells/mL in T-75 flasks in complete IMDM supplemented with 1% antibiotic-mycotic, 10 ng/mL EGF, 10 ng/mL bFGF, and 10% aPRP. Incubate at 37°C with 5% CO₂. Add 6 mL of fresh media after 3 days. Replace the media completely after 7 days, and then every 4 days thereafter until cells reach 70-80% confluence [15].
- Secondary Culture (Expansion): For subculture, switch to complete medium containing 5% aPRP. This concentration has been shown to be suitable for expansion while maintaining cell characteristics [15].
- Characterization: Validate the resulting UCB-MSCs through flow cytometry for standard MSC surface markers (CD73+, CD90+, CD105+, CD14-, CD34-, CD45-, HLA-DR-) and in vitro trilineage differentiation potential (osteogenic and chondrogenic, as adipogenic potential may be limited) [15].
Protocol 2: Switching to Serum-Free and Xeno-Free MSC Expansion
This protocol is critical for transitioning research-grade MSCs to clinically compliant cultures, eliminating the risks associated with fetal bovine serum (FBS) [10] [16].
Materials:
- StemPro MSC SFM XenoFree (Basal Medium & Supplement) or other GMP-compliant media.
- CELLstart Substrate or other GMP-compliant attachment substrate.
- Gentamicin (optional).
- Cryopreserved human MSCs (e.g., from bone marrow or adipose tissue).
Step-by-Step Method:
- Prepare Complete Medium: Aseptically mix 490 mL of MSC SFM Basal Medium, 5 mL of MSC SFM XenoFree Supplement, 5 mL of GlutaMAX Supplement (2 mM final), and 50 μL of Gentamicin (5 μg/mL final, optional). Store at 2-8°C for up to two weeks [16].
- Coat Culture Vessels: Dilute CELLstart substrate 1:100 in DPBS. Add enough solution to cover the culture surface (e.g., 10 mL for a T-75 flask). Incubate at 37°C for 60-120 minutes. Immediately before use, remove the coating solution and replace it with complete medium. Do not rinse [16].
- Recover Cryopreserved MSCs: Thaw cells rapidly in a 37°C water bath. Transfer to a conical tube and slowly dilute with pre-warmed complete medium. Centrifuge at 100-200 x g for 5 minutes. Resuspend the pellet in complete medium and seed at a density of ≥5 x 10³ cells/cm² onto the coated vessels [16].
- Maintain Cultures: Incubate at 37°C in a humidified atmosphere of 4-6% CO₂. Replace the medium every 2-3 days. Passage cells at 80-90% confluency using a gentle enzyme like TrypLE Express [16].
The Scientist's Toolkit: Essential Research Reagents
The table below lists key reagents for the GMP-compliant derivation and expansion of MSCs, as featured in the protocols and studies cited.
| Reagent / Kit Name | Function / Application | Key Feature for GMP Compliance |
|---|---|---|
| MSC-Brew GMP Medium [5] | Expansion of MSCs (e.g., from infrapatellar fat pad). | Animal component-free, GMP-compliant formulation. |
| MesenCult-ACF Plus Medium [5] | Animal component-free culture of MSCs. | Defined, xeno-free supplement supports standardized expansion. |
| StemPro MSC SFM XenoFree [16] | Serum-free, xeno-free expansion of human MSCs and ADSCs. | Completely defined system for clinical applications. |
| CELLstart Substrate [16] | Culture vessel coating for cell attachment in xeno-free systems. | Chemically defined, replaces animal-derived attachment matrices. |
| MesenCult Expansion Kit (Mouse) [17] | Derivation and expansion of mouse MSCs. | Includes MesenPure supplement to reduce hematopoietic cell contamination. |
| Human Platelet Lysate (hPL) / aPRP [15] | Serum replacement for FBS in culture media. | Human-derived, reduces risk of xenogenic immunogenicity and contamination. |
| TrypLE Express [16] | Enzymatic cell dissociation and passaging. | Animal origin-free, recombinant enzyme alternative to trypsin. |
Next Steps in GMP MSC Research
For researchers embarking on GMP-focused MSC studies, the following steps are critical:
- Establish Quality Control (QC) Assays: Implement rigorous QC testing for your final MSC product, including sterility (bacteria, fungi, mycoplasma), viability (>95% is a common release criterion), endotoxin levels, and identity (flow cytometry for MSC markers) [5] [10].
- Conduct Karyotyping and Genetic Stability Tests: Regularly assess genomic stability, especially after multiple passages, to ensure safety for clinical use [10].
- Define Critical Process Parameters: Systematically document and control variables such as seeding density, passage number, confluency at harvest, and the specific media lot used to ensure process consistency and product reproducibility [10].
The Infrapatellar Fat Pad (IPFP) is emerging as a superior source of Mesenchymal Stem Cells (MSCs) for regenerative medicine, particularly for musculoskeletal applications. Sourced from the knee joint, IPFP-derived MSCs (IPFP-MSCs) offer distinct practical and biological advantages over traditional sources like bone marrow, making them highly suitable for Good Manufacturing Practice (GMP) research and clinical-scale production.
Key Advantages for GMP Translation:
- Less Invasive Harvesting: IPFP tissue can be obtained with low morbidity during knee arthroscopy, often as surgical waste, avoiding the painful bone marrow aspiration required for Bone Marrow-derived MSCs (BM-MSCs) [5] [20].
- Abundant Cell Yield: The IPFP provides a rich source of MSCs. One study reported a yield of approximately 2.66 million viable cells from just 2.7 grams of tissue [21].
- High Proliferative Capacity: IPFP-MSCs demonstrate robust growth kinetics. Their proliferation capacity is reported to be less affected by donor age compared to BM-MSCs, a critical factor for consistent cell product quality [22] [20].
- Superior Chondrogenic Potential: IPFP-MSCs exhibit a strong inherent capacity for chondrogenic differentiation, making them an excellent candidate for cartilage regeneration studies and therapies [22] [20].
The following tables summarize key experimental data comparing IPFP-MSCs with other common MSC sources, highlighting their performance in proliferation and differentiation.
Table 1: Comparison of Proliferation and Yield Across MSC Sources
| MSC Source | Reported Doubling Time | Harvest Invasiveness | Key Advantages |
|---|---|---|---|
| Infrapatellar Fat Pad (IPFP) | ~48-60 hours (Passage 2-3) [20] | Low (often from surgical waste) [5] | Age-independent proliferation, high chondrogenic potential [20] |
| Bone Marrow (BM) | Varies with donor age [22] | High (invasive aspiration) [5] | Considered the "gold standard," well-characterized [5] |
| Subcutaneous Fat (SC) | Generally higher than IPFP-MSCs [20] | Low (via liposuction) [22] | Abundantly available, easy access [22] |
| Synovium | Lower than IPFP-MSCs (in some studies) [20] | Moderate (requires specific arthroscopic procedure) | High proliferative and chondrogenic capacity [20] |
Table 2: Chondrogenic Differentiation Potential: Gene Expression Markers
| Experimental Group | COL2 Expression (Collagen Type II) | ACAN Expression (Aggrecan) | Reference |
|---|---|---|---|
| IPFP-MSCs + Hyperacute Serum (HAS) | Significantly higher (p<0.01) | Significantly higher (p<0.001) | [21] |
| IPFP-MSCs + Platelet-Rich Plasma (PRP) | Lower than HAS group | Lower than HAS group | [21] |
| IPFP-MSCs (Standard Culture) | Baseline expression | Baseline expression | [21] |
Essential Experimental Protocols
Protocol 1: GMP-Compliant Isolation & Expansion of IPFP-MSCs
This protocol is adapted from studies focusing on translating research-grade methods to GMP-compliant conditions [5].
Key Steps:
- Tissue Harvest: Arthroscopically collect IPFP tissue using a shaver system (e.g., oscillating arthroscopic shaver). Collect the tissue fragments in a sterile, in-line collection chamber [5].
- Enzymatic Digestion: Mince the tissue and digest with 0.1% collagenase in serum-free media for approximately 2 hours at 37°C with constant agitation [5] [21].
- Cell Separation: Centrifuge the digested suspension. Wash the cell pellet with PBS and filter it through a 100μm strainer to remove debris [5].
- GMP-Compliant Culture: Seed the isolated cells in animal component-free, GMP-compliant media. MSC-Brew GMP Medium has been shown to result in lower doubling times and higher colony formation compared to standard media [5].
- Cell Characterization: Confirm MSC identity via flow cytometry for positive markers (CD73, CD90, CD105 ≥95%) and negative markers (CD34, CD45, CD11b, CD19, HLA-DR ≤2%) according to ISCT standards [23]. Verify trilineage differentiation potential (chondrogenesis, adipogenesis, osteogenesis) in vitro.
Protocol 2: In Vitro Chondrogenic Differentiation Assay
This protocol is used to evaluate the chondrogenic potential of IPFP-MSCs, a key quality attribute [21].
Key Steps:
- Pellet Culture: Harvest expanded MSCs (passage 3-5) and centrifuge 200,000 – 500,000 cells in a conical tube to form a micromass pellet.
- Chondrogenic Induction: Culture the pellets in a defined chondrogenic induction medium. This typically consists of high-glucose DMEM supplemented with ITS (Insulin-Transferrin-Selenium), dexamethasone, ascorbate-2-phosphate, sodium pyruvate, proline, and transforming growth factor-beta (TGF-β3).
- Medium Changes: Replace the induction medium every 2-3 days for 21-28 days.
- Analysis of Differentiation:
- Histology: Fix pellets, embed in paraffin, section, and stain with Alcian Blue to detect sulfated glycosaminoglycans (GAGs), a key component of the cartilage matrix [21].
- Gene Expression: Analyze the expression of cartilage-specific genes (e.g., SOX9, COL2A1, ACAN) via quantitative PCR (qPCR). Compare expression levels to undifferentiated MSCs or control groups [21].
Troubleshooting Guides & FAQs
FAQ 1: The viability of my IPFP-MSCs is low after arthroscopic harvest. Is the tissue still usable?
Answer: Yes. Arthroscopic harvest using a shaver does subject cells to mechanical and thermal stress, but studies confirm it still yields cells with high viability and maintained regenerative potential. Supplementing culture media with blood products like Platelet-Rich Plasma (PRP) or Hyperacute Serum (HAS) post-isolation can significantly enhance metabolic activity and cell recovery [21].
FAQ 2: My IPFP-MSCs show slow proliferation in later passages, affecting my expansion study. How can I improve growth kinetics?
Answer: This is a common challenge in extended passage studies. Consider these solutions:
- Optimize Culture Medium: Use a GMP-compliant, animal component-free medium specifically formulated for MSCs, such as MSC-Brew GMP Medium or MesenCult-ACF Plus Medium, which have been shown to support lower doubling times compared to standard media [5].
- Use Decellularized Extracellular Matrix (dECM): Expanding IPFP-MSCs on a dECM deposited by earlier-passage cells can reduce reactive oxygen species (ROS) and enhance proliferation capacity [20].
- Add Growth Factors: Supplementing media with basic Fibroblast Growth Factor (bFGF) can promote IPFP-MSC proliferation [20].
FAQ 3: How can I ensure my IPFP-MSC culture is of high quality and stable for a GMP-focused thesis?
Answer: Implement rigorous, routine quality control checks:
- Purity and Identity: Regularly perform flow cytometry to confirm surface marker expression meets ISCT standards [23].
- Potency: Conduct periodic trilineage differentiation assays to confirm multipotency is retained across passages.
- Stability: Perform cell doubling time and colony-forming unit (CFU) assays at defined passages to track proliferative stability.
- Sterility: Adhere to strict sterility testing protocols for mycoplasma, bacteria, and fungi, as required for GMP [5].
Key Signaling Pathways in Chondrogenesis
The chondrogenic differentiation of IPFP-MSCs is a tightly regulated process. The SOX trio of transcription factors (SOX9, SOX5, SOX6) is essential for driving the expression of cartilage-specific matrix proteins like collagen type II and aggrecan [22]. The following diagram illustrates the core regulatory network.
Diagram 1: Transcriptional Regulation of Chondrogenesis.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for IPFP-MSC Research & GMP Translation
| Reagent / Material | Function / Purpose | Example Product / Note |
|---|---|---|
| Collagenase, Type I | Enzymatic digestion of IPFP tissue to release stromal cells. | GMP-grade is required for clinical-scale manufacturing [5]. |
| GMP-Compliant Basal Medium | Serum-free, xeno-free base medium for cell expansion. | MEM α, DMEM [5]. |
| Animal Component-Free Supplement | Defines culture environment; eliminates batch variability & pathogen risk. | MSC-Brew GMP Medium (showed enhanced proliferation) [5]. |
| bFGF (Basic Fibroblast Growth Factor) | Growth factor supplement to enhance MSC proliferation rate. | Human recombinant, GMP-grade [21]. |
| Chondrogenic Induction Kit | Defined medium for in vitro chondrogenic differentiation assays. | Often contains TGF-β3, ascorbate, and ITS supplement [21]. |
| Flow Cytometry Antibody Panel | Cell surface marker characterization (CD73, CD90, CD105, etc.). | Essential for identity and purity testing per ISCT standards [23]. |
| Hyperacute Serum (HAS) | Blood product supplement shown to enhance chondrogenic matrix production. | Can be prepared in-house or sourced commercially [21]. |
Troubleshooting Guide: Frequent Issues in GMP-Compliant MSC Expansion
This guide addresses common challenges and their solutions during the isolation and extended culture of Mesenchymal Stem Cells (MSCs) under Good Manufacturing Practice (GMP) conditions.
Problem: Declining Cell Viability or Proliferation Rate Over Extended Passages
- Potential Cause #1: Suboptimal culture medium. The use of media containing animal-derived components (like Fetal Bovine Serum) can lead to batch-to-batch variability and risks of contamination.
- Solution: Transition to a defined, xeno-free (animal-component-free), GMP-compliant culture medium. Validation data shows that media such as MSC-Brew GMP Medium can significantly improve cell doubling times and colony-forming unit (CFU) capacity compared to standard media [5].
- Solution: Re-evaluate and optimize the seeding density. A common practice is to seed cells at a density of 5 × 10³ cells/cm² and passage them at 80-90% confluency to maintain healthy, proliferating cultures [5].
- Potential Cause #2: In-process contamination or failure in aseptic technique.
- Solution: Implement a Closed-System Processing workflow wherever possible. For tissue dissection and digestion, use sterile, closed collection chambers to minimize environmental exposure [5].
- Solution: Integrate advanced sterility testing methods, such as isolator technology, which provides a physically sealed workspace to drastically reduce contamination risk during both production and quality control testing [24].
Problem: Failure to Meet Product Release Specifications
- Potential Cause #1: Incomplete characterization or loss of MSC identity.
- Solution: Adhere strictly to ISCT (International Society for Cell & Gene Therapy) criteria for defining MSCs. This includes testing for the positive expression of surface markers (CD73, CD90, CD105) and negative expression of hematopoietic markers (CD34, CD45, CD11b, CD19, HLA-DR) via flow cytometry. This testing should be performed at various passages, including after cryopreservation, to ensure phenotype stability [5] [25].
- Solution: Implement a robust Identity and Purity Testing panel. This goes beyond the basic ISCT markers and should include endotoxin and mycoplasma testing using validated methods like the Bact/Alert system to ensure product purity [5].
- Potential Cause #2: Inadequate post-thaw recovery.
- Solution: Optimize the cryopreservation and thawing protocol. A study validating a GMP-compliant protocol demonstrated post-thaw viability consistently >95%, which is well above the typical minimum requirement of >70%. Stability assessments confirmed this high viability could be maintained for up to 180 days of storage, validating the robustness of the method [5].
Frequently Asked Questions (FAQs)
Q1: What are the minimum viability requirements for a GMP-compliant MSC product batch release? While specifications are product-specific, GMP validation studies for clinical-grade MSCs often set high standards. Recent protocols have established a minimum viability requirement of >70%, with successful validations consistently achieving post-thaw viability >95% [5]. Your product's specific release criteria must be defined and validated in your Marketing Authorisation application.
Q2: Beyond the basic ISCT criteria, what other quality controls are needed for GMP? The basic ISCT criteria (plastic adherence, marker expression, differentiation potential) are the foundation. GMP requires a comprehensive quality system that adds [5] [25]:
- Sterility: Testing for bacteria, fungi, and mycoplasma.
- Purity: Endotoxin testing below a specified limit.
- Potency: A quantitative assay relevant to the proposed mechanism of action (e.g., immunomodulation), which may include CFU assays to demonstrate clonogenic capacity [5].
- Viability: As per Q1.
- Safety: Karyotyping to check for genetic stability, especially after extended passaging.
Q3: How can I distinguish a proven, regulated cell therapy from an unproven one? The ISCT states that proven and approved CGT products undergo rigorous quality, safety, and efficacy evaluation by regulatory agencies [25]. Be cautious of clinics or products that exhibit these characteristics of being unproven [25]:
- Lack of approval from recognized national regulatory bodies (e.g., FDA, EMA, MHRA).
- Marketing that relies on "tokens of scientific legitimacy" (e.g., citing preliminary research or ethics approvals) without robust clinical trial data.
- Requests for high, upfront payments for the treatment itself ("pay-to-participate").
- Insufficient information disclosed to enable proper informed consent.
Q4: What are the key differences between GMP and cGMP? The terms are often used interchangeably, but "c" in cGMP stands for "current," emphasizing the requirement to use up-to-date technologies and systems [26] [27]. This means manufacturers are expected to employ modern, validated methods and pursue continuous improvement, rather than relying on outdated but once-acceptable techniques [26].
Experimental Protocol: GMP-Compliant Isolation and Extended Culture of MSCs
The following workflow details a methodology adapted from a peer-reviewed study for the GMP-compliant derivation of MSCs from the infrapatellar fat pad (FPMSCs), which can be adapted for other tissue sources [5].
Key Materials and Reagents:
- Tissue: Human infrapatellar fat pad (or other MSC source) obtained with informed consent and ethical approval.
- Digestion Enzyme: 0.1% Collagenase in serum-free media.
- Basal Medium: MEM α or equivalent.
- GMP-Compliant Media: Defined, xeno-free media such as MSC-Brew GMP Medium (Miltenyi Biotec) or MesenCult-ACF Plus Medium (StemCell Technologies) [5].
- Cryopreservation Solution: Typically 10% DMSO in a suitable carrier.
Detailed Procedure:
- Tissue Digestion: Mince the acquired tissue into ~1mm³ pieces. Digest with 0.1% collagenase in serum-free media for 2 hours at 37°C with gentle agitation [5].
- Cell Isolation: Centrifuge the digested mixture at 300 ×g for 10 minutes. Carefully remove the supernatant and surfactant. Wash the cell pellet with PBS and filter it through a 100 μm cell strainer to remove debris. After a final centrifugation, resuspend the cell pellet in your chosen GMP-compliant culture medium [5].
- Primary Culture (P0): Seed the isolated cells in a culture vessel and incubate. This is considered passage 0 (P0).
- Subculture and Extended Expansion: Once cells reach 80-90% confluency, detach them using a GMP-compliant enzyme (e.g., trypsin substitute). For expansion studies, re-seed cells at a density of 5 × 10³ cells/cm² in fresh GMP-compliant medium. Repeat this process for multiple passages to study long-term proliferation capacity [5].
- In-process Monitoring: Monitor cell morphology, confluence, and check for contamination regularly.
- Harvest and Cryopreservation: Once the desired cell numbers are achieved, detach the cells, perform a cell count and viability assessment (e.g., using Trypan Blue). Cryopreserve the cell batch in a controlled-rate freezer using a cryopreservation solution, then transfer to liquid nitrogen for long-term storage [5].
The choice of culture medium is critical for maintaining proliferation capacity during extended passages. The table below summarizes key performance data from a study comparing different media [5].
Table 1: Impact of GMP-Compliant Media on MSC Proliferation and Potency
| Media Formulation | Type | Doubling Time (Representative) | Colony Forming Unit (CFU) Capacity | Key Advantage |
|---|---|---|---|---|
| Standard MSC Media(e.g., MEM-α + 10% FBS) | Research Grade, with animal components | Higher | Standard | Baseline for comparison |
| MSC-Brew GMP Medium | GMP-compliant, Xeno-free | Lower (Enhanced Proliferation) | Higher (Enhanced Potency) | Optimized for clinical-grade manufacturing |
| MesenCult-ACF Plus Medium | GMP-compliant, Xeno-free | Lower than standard | Higher than standard | Defined, animal-component-free formulation |
The Scientist's Toolkit: Essential Reagents for GMP MSC Research
Table 2: Key Research Reagent Solutions for GMP-Compliant MSC Studies
| Reagent / Material | Function in the Protocol | GMP-Compliance Consideration |
|---|---|---|
| Xeno-Free Culture Medium(e.g., MSC-Brew GMP Medium) | Supports cell growth, proliferation, and maintenance of stemness. | Defined formulation eliminates risk from animal-derived components; sourced from qualified suppliers with full traceability. |
| GMP-Grade Enzymes(e.g., Collagenase, Trypsin substitutes) | Tissue dissociation and cell detachment during passaging. | Must be certified for absence of animal and human pathogens. Requires validation for your specific cell type and process. |
| Cell Separation Filters(e.g., 100μm strainer) | Removes tissue debris and cell clumps after digestion to obtain a single-cell suspension. | Sterile, single-use, and certified non-pyrogenic. |
| Validated Assay Kits(e.g., Flow Cytometry, Endotoxin) | Characterizing cell identity (ISCT markers), purity, and safety. | Assays must be validated for accuracy, precision, and specificity. Kits should be sourced from reliable manufacturers. |
Sterility Assurance Decision Pathway
Sterility testing is a critical release criterion. The following pathway outlines the decision process for implementing modern sterility assurance methods.
Integrating isolator technology is a key advancement for sterility testing. By 2025, these systems are expected to incorporate AI-driven environmental controls and automation, reducing human intervention by up to 50% and significantly lowering the risk of false-positive results due to laboratory contamination [24]. This aligns with the CGMP principle of using current, modern systems to assure product quality [27].
Advanced GMP-Compliant Expansion Systems and Media Optimization Strategies
Transitioning from Research-Grade to GMP-Compliant, Animal Component-Free Culture Media
What is the fundamental difference between research-grade and GMP-compliant media? Research-grade media are optimized for experimental feasibility and cost-effectiveness, with acceptable batch-to-batch variability. GMP (Good Manufacturing Practice) compliant media are manufactured under stringent, documented quality controls to ensure consistency, purity, and safety for therapeutic applications [28]. GMP validation provides documented evidence that a manufacturing process will consistently produce products meeting predefined quality standards [28].
What exactly constitutes "Animal Component-Free" (ACF) and related media types?
- Animal Component-Free (ACF): Products that do not contain any primary raw materials directly derived from animal tissue and do not use animal components in their manufacturing process [29].
- Non-Animal Origin (NAO): Products that contain no animal origin components in the synthesis steps or in the final chemical structure [29].
- Xeno-Free (XF): Formulations without any components from non-human animal sources, ensuring human cell cultures are maintained in completely human-compatible environments [30].
- Chemically Defined: Media containing only components with known chemical structure and concentration, with no animal origin primary raw materials used in manufacturing [29].
Advantages and Regulatory Drivers for Transition
Why are regulatory bodies pushing for animal component-free media in clinical applications? Global regulatory agencies are driving the shift to animal-origin-free solutions to enhance product safety, standardize quality, and align with ethical considerations [31]. Key concerns with animal-derived components include:
- Contamination Risks: Animal-derived materials can harbor viruses, prions, endotoxins, or other harmful contaminants that compromise the safety of cell therapies and vaccines [31].
- Batch-to-Batch Variability: The inherent inconsistency of animal-derived components challenges manufacturing reproducibility and reliability [31].
- Regulatory Compliance: Agencies including the FDA, EMA, and PMDA enforce stricter GMP guidelines that limit animal-origin materials, requiring extensive additional testing [31].
What are the primary scientific benefits of transitioning to ACF media for MSC research? Adopting ACF media enhances experimental consistency by reducing variability from unidentified culture components and unknown levels of vitamins, hormones, and growth factors [32]. This is particularly crucial for extended passage studies with MSCs, where maintaining proliferation capacity and differentiation potential across multiple passages is essential for generating clinically relevant cell numbers [33].
Transition Methodology and Protocol Implementation
Systematic Transition Workflow
The following diagram illustrates the recommended workflow for transitioning from research-grade to GMP-compliant, animal component-free media:
Experimental Protocol for Validating MSC Performance in ACF Media
Objective: To systematically evaluate the proliferation capacity and differentiation potential of Mesenchymal Stem Cells (MSCs) during extended passage culture in GMP-compliant, animal component-free media compared to research-grade media.
Materials and Reagents:
- Early passage MSCs (P2-P4)
- Research-grade FBS-containing media (control)
- Selected GMP-compliant ACF media [34] [30]
- Trypsinization reagents or Non-Enzymatic Cell Dissociation Solution [34]
- Differentiation induction media (osteogenic, chondrogenic, adipogenic)
- Serum-Free Cell Freezing Medium [34]
Methodology:
- Parallel Culture Setup: Split MSC stock into two groups - one maintained in research-grade media (control) and one in GMP-compliant ACF media (test). Use identical seeding densities (e.g., 1,000-4,000 cells/cm²) and culture conditions [33].
Extended Passage Study Design:
- Subculture cells at preconfluent densities (70-80% confluence)
- For test group, consider both standard passage frequency (every 7 days) and extended first passage (up to 53 days without subculturing) to assess proliferation dynamics [33]
- Record population doublings at each passage
- Monitor viability and morphology regularly
Performance Metrics Assessment:
- Proliferation Capacity: Calculate population doublings and cumulative cell numbers through multiple passages (e.g., up to passage 7) [33]
- Differentiation Potential: At passages 3, 5, and 7, assess tri-lineage differentiation capacity using standardized protocols:
- Osteogenic: Mineralization assessment via Alizarin Red staining
- Chondrogenic: Pellet culture system with sulfated proteoglycan detection
- Adipogenic: Lipid droplet formation via Oil Red O staining [33]
- Phenotype Stability: Analyze surface marker expression (CD73, CD90, CD105) via flow cytometry at critical passages
In Vivo Function Validation (if applicable): Assess bone formation capacity in ceramic cube implantation models in immunocompromised mice [33].
Troubleshooting Common Transition Challenges
Media Preparation and Quality Issues
| Issue | Possible Cause | Solution |
|---|---|---|
| Poor Cell Growth in New ACF Media | Missing critical components from serum | Supplement with recombinant proteins (transferrin, insulin, albumin) [31] |
| Reduced Attachment | Lack of attachment factors | Incorporate recombinant attachment peptides or use ECM-coated surfaces [35] |
| Media pH Deviation | Contamination or improper storage | Check storage conditions, perform pH testing before use, discard deviated batches [36] |
| Precipitation/Crystallization | Improper dissolution or component interaction | Ensure thorough mixing, follow preparation protocols exactly, prepare fresh batches [36] |
MSC Performance and Differentiation Challenges
| Issue | Possible Cause | Solution |
|---|---|---|
| Reduced Proliferation in Extended Culture | Cumulative adaptation stress | Implement gradual adaptation strategy (25:75, 50:50, 75:25 blending over passages) |
| Diminished Adipogenic Differentiation | Missing differentiation co-factors | Supplement with PPARγ agonists or optimize differentiation protocol for ACF conditions [33] |
| Altered Morphology | Suboptimal growth environment | Verify that ACF media is specifically formulated for MSC culture [30] |
| Increased Senescence | Cumulative population doublings | Monitor senescence markers, consider using ECM-coated surfaces to improve proliferation [35] |
Contamination and Quality Control Issues
| Issue | Possible Cause | Solution |
|---|---|---|
| Mycoplasma Contamination | Compromised reagents or technique | Implement routine mycoplasma testing, use quarantined reagents, review aseptic technique [37] |
| Bacterial/Fungal Growth | Non-sterile handling or contaminated reagents | Practice proper aseptic technique, sterilize all equipment, inspect media before use [36] [37] |
| Batch-to-Batch Variability | Despite ACF claims | Request certificate of analysis for each lot, perform incoming quality control testing |
MSC-Specific Considerations for Extended Passage Studies
How does extended passage culture affect MSC proliferation and differentiation in ACF systems? Research indicates that MSC cultured under extended first passage (EFP) conditions achieved approximately 16 population doublings after 34 days with minimal increase thereafter, while standard passage conditions yielded approximately 27 population doublings through seven passages [33]. Importantly, EFP cells maintained osteogenic and chondrogenic differentiation capacity equivalent to standard passage cells, though adipogenic potential was somewhat diminished [33].
What strategies can enhance MSC proliferation in ACF systems? Utilizing basement membrane extracellular matrix proteins (ECMP) such as laminin-1, laminin-5, fibronectin, and collagen IV can significantly improve MSC proliferation capacity [35]. Studies demonstrate that MSC cultured on ECM-gel surfaces yielded 250-fold higher cumulative cell numbers compared to plastic-adherent MSC after 50 days of culture [35].
Essential Research Reagent Solutions for ACF MSC Culture
| Reagent Category | Specific Products | Function in MSC Culture |
|---|---|---|
| Basal Media | Mesenchymal Stem Cell Growth Medium XF [30] | Supports proliferation while maintaining differentiation potential |
| Dissociation Reagents | Non-Enzymatic Cell Dissociation Solution [34] | Gentle cell detachment while maintaining surface marker integrity |
| Cryopreservation Media | Serum-Free Cell Freezing Medium [34], Cryo-SFM Plus [30] | Maintains high post-thaw viability without animal components |
| Growth Supplements | Recombinant Transferrin (Optiferrin) [31], ITS Animal-Free [31] | Provides essential iron transport and micronutrients |
| Extracellular Matrix | ECM-gel, Laminin-1, Laminin-5, Fibronectin, Collagen IV [35] | Enhances proliferation capacity and maintains stemness |
| Cell Survival Enhancers | CEPT Cocktail [32] | Improves viability during passaging, cryopreservation, and differentiation |
Regulatory Documentation and Quality Assurance
What documentation is required for GMP compliance? GMP validation requires comprehensive documentation including protocols, standard operating procedures (SOPs), and validation reports that provide transparency and accountability [28]. This includes:
- Risk Assessment: Identifying potential sources of variability or failure before validation [28]
- Change Control Procedures: Rigorous evaluation of any changes to equipment, processes, or materials [28]
- Batch Records: Detailed documentation of each media lot's production and quality testing
- Training Records: Documentation that personnel are adequately trained to execute procedures accurately [28]
How does ACF media simplify regulatory approvals? Products developed with ACF materials are easier to license and market globally, especially in regions with strict restrictions on animal-derived ingredients [31]. This streamlined approval process results from reduced contamination risks, eliminated donor-dependent variability, and compliance with evolving international regulatory standards [31].
Frequently Asked Questions (FAQs)
Do I need to completely change my protocols when transitioning to GMP-compliant ACF media? Not necessarily. Many animal-free media and reagents can be seamlessly integrated into existing experiments without protocol changes [29]. However, for optimal results with sensitive cell types like MSCs, some protocol optimization may be required, particularly for extended passage studies and differentiation assays.
Can I replace all my media and reagents with animal-free products for MSC culture? Most critical cell culture workflow products now have animal-free alternatives suitable for MSC culture, including basal media, growth factors, dissociation reagents, and cryopreservation media [29] [30]. The continuously evolving ACF portfolio now supports most 2D and 3D cell culture workflows [29].
Are all recombinant products automatically animal-free? Not necessarily. Although the primary raw material may not be of animal origin, some recombinant products may use animal-derived components during manufacturing or be lyophilized from buffers containing animal-derived stabilizers such as BSA [29]. It is essential to verify with manufacturers that products are truly animal-free throughout the entire production process.
How can I enhance MSC survival during challenging processes in ACF systems? Small molecule cocktails like the CEPT cocktail (Chroman 1, Emricasan, Polyamine supplement, Trans-ISRIB) can significantly improve stem cell viability during passaging, single cell cloning, cryopreservation, and differentiation in ACF conditions [32].
Transitioning from research-grade to GMP-compliant, animal component-free culture media for MSC research requires systematic planning and validation. By understanding the regulatory landscape, implementing robust testing protocols, and utilizing appropriate troubleshooting strategies, researchers can successfully maintain MSC proliferation capacity and differentiation potential while meeting the stringent requirements for therapeutic applications. The key success factors include gradual adaptation, comprehensive performance testing across extended passages, proper documentation, and selection of high-quality, specifically formulated ACF reagents designed for MSC culture systems.
The following tables summarize key quantitative findings from studies comparing MSC-Brew GMP Medium with standard MSC media formulations.
| Performance Parameter | MSC-Brew GMP Medium | Standard MSC Media | Notes |
|---|---|---|---|
| Cell Doubling Time | Lower across passages | Higher across passages | Indicates enhanced proliferation rates |
| Colony Forming Unit (CFU) Capacity | Higher | Lower | Supports enhanced cell potency |
| Post-Thaw Viability (after 180 days) | >95% | Not Reported | Exceeds the >70% minimum requirement for product release |
| Quality Attribute | Result for GMP-FPMSC | Release Requirement |
|---|---|---|
| Viability | >95% | >70% |
| Sterility | Maintained | Maintained |
| Stem Cell Marker Expression | Maintained post-thaw | Maintained |
Detailed Experimental Protocols
This protocol details the initial steps for obtaining FPMSCs from patient tissue.
- Tissue Acquisition & Preparation: Infrapatellar fat pad (IFP) tissue is acquired as waste tissue from procedures like ACL reconstructive surgery. The tissue is then cut into small pieces of approximately 1 mm³.
- Enzymatic Digestion: The tissue pieces are digested using 0.1% collagenase in serum-free media. This process is carried out for 2 hours at 37°C.
- Cell Pellet Isolation: The digested tissue is centrifuged at 300 ×g for 10 minutes. The resulting supernatant and surfactant are carefully removed.
- Washing and Filtration: The cell pellet is washed with Phosphate-Buffered Saline (PBS) and passed through a 100 μm filter to remove debris.
- Initial Resuspension: After a final centrifugation step, the cell pellet is resuspended in a standard MSC media (e.g., MEM α supplemented with 10% FBS and gentamicin) for initial culture.
This method is used to directly compare the effects of different culture media on MSC proliferation and potency.
- Cell Seeding and Culture: Isolated FPMSCs are thawed and seeded. Cells are passaged at 80-90% confluency and consistently seeded at a density of 5 × 10³ cells/cm².
- Media Comparison: The behavior of cells is assessed using two animal component-free media formulations (e.g., MesenCult-ACF Plus Medium and MSC-Brew GMP Medium) and compared to a standard MSC media control.
- Doubling Time Calculation: Cell doubling time is evaluated over multiple passages. Cells are counted at each passage, and the doubling time is calculated using the formula: Doubling Time = (Duration of Culture × log(2)) / (log(Final Cell Count) - log(Initial Cell Count))
- Colony Forming Unit (CFU) Assay: Colony formation capacity is assessed by seeding cells at low densities (e.g., 20, 50, 100, and 500 cells) in a culture dish. After 10 days of growth, colonies are fixed with formalin, stained with Crystal Violet, and counted.
Diagram: Experimental Workflow for Media Performance Evaluation
Frequently Asked Questions & Troubleshooting
FAQ 1: What are the primary advantages of switching to an animal component-free, GMP-compliant medium like MSC-Brew GMP Medium?
Using a GMP-compliant, animal component-free medium offers several critical advantages for translational research [5]:
- Enhanced Safety Profile: It eliminates the risks associated with animal-derived components (like FBS), such as potential contamination, immunogenicity, and batch-to-batch variability.
- Regulatory Compliance: It is specifically designed to meet Good Manufacturing Practice (GMP) standards, which is a necessary step for progressing from preclinical studies to clinical trials.
- Improved Performance: Studies have demonstrated that MSC-Brew GMP Medium can support enhanced MSC proliferation rates and higher colony-forming potential compared to some standard media formulations.
FAQ 2: Our MSCs are showing decreased proliferation and differentiation potential after extended passages. Could the culture medium be a factor?
Yes, the culture medium is a significant factor. It is well-documented that prolonged in vitro expansion can cause MSCs to gradually lose their progenitor properties, including a diminished proliferation rate and a reduction in multiple differentiation potential [38]. Furthermore, the "mechanobiological memory" of MSCs is influenced by their culture environment. Using a consistent, high-quality GMP medium helps provide a stable environment, but it is also crucial to monitor passage number and avoid over-confluent cultures to maintain stemness [39].
FAQ 3: What are the critical quality control checks for MSCs intended for clinical use, and how does MSC-Brew GMP Medium support them?
For clinical-grade MSCs, key product release specifications include [5]:
- Viability: Typically required to be >70%, with >95% achievable using optimized GMP protocols.
- Sterility: Must be free from microbial contamination (e.g., tested via BacT/Alert).
- Purity/Identity: Confirmed via flow cytometry for standard MSC surface markers (e.g., positive for CD73, CD90, CD105 and negative for CD34, CD45, etc.).
- Potency: Assessed through functional assays like the CFU assay or differentiation potential. Using a GMP-compliant medium like MSC-Brew is part of a robust manufacturing process that ensures these quality attributes are met and maintained, even after cryostorage.
Troubleshooting Guide: Common Issues in MSC Culture
| Problem | Potential Causes | Recommended Solutions |
|---|---|---|
| Low Cell Attachment After Passaging | Over-digestion with enzymatic reagent; excessive pipetting; low initial seeding density. | Reduce incubation time with passaging reagent; minimize pipetting to avoid single-cell suspension; plate 2-3 times higher number of cell aggregates initially [40]. |
| Excessive Spontaneous Differentiation in Culture | Overgrown colonies; old or degraded culture medium; plates kept out of incubator for too long. | Passage cultures when colonies are large but not over-confluent; ensure medium is fresh; limit time culture plate is outside the incubator to under 15 minutes [40]. |
| Inconsistent or Poor Proliferation Across Passages | Suboptimal culture medium; high passage number; inconsistent passaging techniques. | Transition to a high-performance GMP medium like MSC-Brew; standardize seeding density and passaging schedule; monitor population doublings and avoid using high-passage cells [5] [38]. |
| Loss of Differentiation Potential | Accumulated "mechanobiological memory" from long-term culture on stiff plastic substrates; high passage number. | Consider culture surfaces that prevent lineage bias; use lower passage cells; validate differentiation potential at regular intervals [39]. |
The Scientist's Toolkit: Essential Research Reagents
The table below lists key materials used in the featured experiments for evaluating MSC media [5].
| Reagent / Material | Function / Application | Example Product |
|---|---|---|
| MSC-Brew GMP Medium | Animal component-free, GMP-compliant medium for clinical-grade MSC expansion. | Miltenyi Biotec, Cat# 170-076-325 |
| MesenCult-ACF Plus Medium | Animal component-free medium used for comparison in culture media studies. | StemCell Technologies, Cat# 05447 |
| Fetal Bovine Serum (FBS) | Serum supplement for standard (non-GMP) MSC culture media. | Atlas, Cat# F-0500-A |
| Collagenase | Enzyme for digesting tissue to isolate primary MSCs. | 0.1% Collagenase solution |
| BD Stemflow Human MSC Analysis Kit | Antibody panel for flow cytometric analysis of MSC surface markers. | BD Biosciences, Cat# 562245 |
| Gentamicin | Antibiotic added to culture media to prevent bacterial contamination. | Thermofisher, Cat# 15750060 |
| Crystal Violet | Stain used to visualize and quantify colonies in the CFU assay. | MilliporeSigma, Cat# V5265 |
Conceptual Framework: Maintaining Stemness
Diagram: The Concept of Frustrated Differentiation to Maintain Stemness
This concept illustrates that culturing MSCs on a substrate with a heterogeneous distribution of elasticity (microelastically patterned) causes the cells to migrate between stiff and soft regions. This nomadic migration results in an oscillating input of mechanical signals, which prevents the sustained activation of mechanosensitive transcription factors like YAP/TAZ that drive lineage specification. This state, known as "frustrated differentiation," helps maintain MSCs in an undifferentiated, multipotent state, which is crucial for their therapeutic efficacy [39].
The manufacturing of Mesenchymal Stem/Stromal Cells (MSCs) for clinical applications as Advanced Therapy Medicinal Products (ATMPs) requires large-scale expansion under stringent Good Manufacturing Practice (GMP) standards. Automated bioreactor systems address critical challenges in traditional manual culture, including labor intensity, process variability, and contamination risks, while enabling the production of clinically relevant cell numbers (millions to hundreds of millions of cells) [41]. This technical support center focuses on two prominent automated platforms: the Quantum Cell Expansion System (Terumo BCT) and the CliniMACS Prodigy (Miltenyi Biotec). Within the context of extended passage studies for GMP research, understanding the operation, performance, and troubleshooting of these systems is paramount for ensuring the quality, safety, and efficacy of the final MSC product [41] [42].
The following table summarizes the core characteristics and documented performance of the Quantum and CliniMACS Prodigy systems for MSC expansion.
Table 1: Key Features and Performance of Automated MSC Expansion Systems
| Feature | Quantum Cell Expansion System | CliniMACS Prodigy |
|---|---|---|
| Technology Type | Hollow fiber bioreactor [41] | Integrated, closed system with Adherent Cell Culture (ACC) process and tubing sets [41] [43] |
| Culture Surface Area | 21,000 cm² (equivalent to ~120 T-175 flasks) [41] | Varies with culture chamber (e.g., 1-layer CellSTACK) [41] |
| Level of Automation | Closed, automated system with continuous medium exchange [41] | Fully integrated automation from cell isolation/inoculation to cultivation, harvesting, and final formulation [41] [43] |
| Reported MSC Yield | 100–276 × 10⁶ BM-MSCs from a 7-day expansion starting from 20 × 10⁶ cells [41] | >29 × 10⁶ equine MSCs at Passage 0, significantly higher than manual protocols [41] |
| Typical Process Duration | ~7 days for expansion [41] | ~10 days for a complete process from isolation to harvest [41] |
| Key Advantages | Reduced passages & open manipulation; controlled gas environment (normoxia/hypoxia) [41] | Reduced cleanroom requirements; parallel manufacturing capability; easy technology transfer [43] |
Frequently Asked Questions (FAQs) and Troubleshooting Guides
Quantum Cell Expansion System
Q1: Our MSCs are not achieving the expected expansion yield in the Quantum system. What could be the cause?
- A: Suboptimal yield can stem from several factors. First, ensure the hollow fibers are properly coated with an adhesive substrate like fibronectin, vimentin, or cryoprecipitate prior to cell seeding, as this is critical for cell attachment [41]. Second, consider substituting fetal bovine serum (FBS) with human platelet lysate (hPL) as a growth supplement, which has been shown to significantly enhance the expansion of adipose tissue-derived MSCs (AT-MSCs) in the Quantum system [41]. Finally, monitor glucose and lactate levels closely, as the system's continuous supply of fresh media is designed to support superior survival and growth [41].
Q2: How does the Quantum system manage gas exchange for creating specialized microenvironments?
- A: The Quantum system can be directly connected to any gases and their combinations. This allows researchers to provide either a standard normoxic or a low-oxygen hypoxic microenvironment, which has been shown to enhance the productivity of bone marrow-derived MSCs (BM-MSCs) in some studies [41].
CliniMACS Prodigy Platform
Q1: What is the recommended medium for GMP-compliant MSC expansion on the CliniMACS Prodigy?
- A: The system has been successfully used with specific GMP-grade media, such as MSC-Brew GMP medium, to generate clinical-grade cells [41]. For a fully closed-system process, xeno-free, serum-free media formulations are available that are compatible with automated hollow-fiber and other bioreactor platforms [44].
Q2: Can the CliniMACS Prodigy isolate and expand MSCs from different tissue sources?
- A: Yes. The platform is versatile and can process primary tissue-isolated BM-MSCs, as well as single-cell suspensions of adipose tissue-derived MSCs (AT-MSCs) and umbilical cord-derived MSCs (UC-MSCs) [41]. Its automation covers the initial isolation step, often via density gradient centrifugation for bone marrow samples [41].
General MSC Expansion and Quality Control
Q1: How does extended passaging in these bioreactors affect MSC quality and functionality?
- A: Extended in vitro expansion impacts MSC characteristics. While surface marker expression and immunogenic properties may remain stable, immunosuppressive capacity can be reduced at higher passages (e.g., passage 8 and 12) for both BM-MSCs and UC-MSCs [42]. Furthermore, while osteogenic and chondrogenic potential may be maintained, adipogenic differentiation capacity can diminish with extended culture [45] [42]. It is crucial to define an optimal passage number that balances yield with functional potency for your specific therapeutic application.
Q2: What are the critical quality control checks for the final MSC product?
- A: According to International Society for Cellular Therapy (ISCT) standards, MSCs must demonstrate: (I) plastic adherence; (II) specific surface marker expression (positive for CD105, CD73, CD90; negative for CD45, CD34, CD14, CD11b, CD79α, HLA-DR); and (III) ability to differentiate into adipocytes, chondrocytes, and osteoblasts in vitro [41]. Additional testing for immunomodulatory activity, genome stability, and absence of microbial contamination is essential to ensure safety and functionality [41].
Experimental Protocols for System Evaluation
Protocol: Assessing the Impact of Extended Culture on MSC Potency
Objective: To evaluate the proliferation capacity and immunomodulatory function of MSCs expanded in an automated bioreactor over multiple passages, within a GMP research framework [42].
Materials:
- Automated bioreactor (e.g., Quantum or CliniMACS Prodigy)
- GMP-compliant, xeno-free medium (e.g., PRIME-XV MSC XSFM [44])
- Trypsin-EDTA or equivalent dissociation reagent
- Flow cytometry antibodies against CD105, CD73, CD90, CD45, CD34, CD14
- T-cell suppression assay kit
- Materials for trilineage differentiation (osteogenic, chondrogenic, adipogenic)
Method:
- Cell Expansion: Isolate and expand MSCs in the automated bioreactor according to the manufacturer's instructions. Use a standardized seeding density.
- Serial Passaging: At the end of each passage (e.g., P2, P4, P6, P8), harvest a representative sample of cells for analysis. Continue expanding the remaining cells.
- Growth Kinetics: Count cells at each passage to calculate population doublings and cumulative population doublings over time [45].
- Phenotypic Analysis (Identity/Purity): Analyze the expression of characteristic MSC surface markers (e.g., CD105, CD73, CD90) and absence of hematopoietic markers (e.g., CD45, CD34) by flow cytometry at each passage [41] [42].
- Functional Potency Assay:
- T-cell Suppression Assay: Co-culture irradiated MSCs from different passages with activated peripheral blood mononuclear cells (PBMCs). Measure T-cell proliferation via 3H-thymidine incorporation or CFSE dilution. A reduction in suppression capacity at higher passages indicates functional decline [42].
- Trilineage Differentiation: Subject MSCs from early and late passages to osteogenic, chondrogenic, and adipogenic induction media. Use appropriate staining (Alizarin Red, Alcian Blue, Oil Red O) to quantify differentiation potential [45].
Protocol: Comparing Manual vs. Automated Expansion
Objective: To quantitatively demonstrate the efficiency and consistency gains of automated bioreactor expansion over traditional flask-based culture.
Method:
- Parallel Culture: Split a single donor MSC source. Expand one fraction in a T-flask stack (manual process) and the other in the automated bioreactor.
- Process Metrics: Record the total hands-on time, number of open manipulations, and incubator space used for each method [41].
- Output Metrics: At the end of the culture period, compare the final cell yield, viability, and population doubling time.
- Quality Metrics: Assess the phenotypic and functional characteristics (as described in Protocol 4.1) of the cells from both systems to ensure quality is maintained.
Signaling Pathways and Experimental Workflows
The following diagram illustrates a generalized workflow for planning and executing an MSC expansion study in an automated bioreactor, incorporating key process steps and quality control checks.
The Scientist's Toolkit: Essential Research Reagents and Materials
The table below lists key reagents and materials critical for successful and GMP-compliant MSC expansion in automated bioreactors.
Table 2: Essential Reagents for Automated MSC Expansion
| Reagent/Material | Function & Importance | GMP-Compliant Example / Note |
|---|---|---|
| Xeno-Free, Serum-Free Medium | Provides nutrients and growth factors without animal-derived components, minimizing risk of adventitious agents and lot-to-lot variability [44]. | PRIME-XV MSC XSFM [44] |
| Human Platelet Lysate (hPL) | A human-derived growth supplement that can replace FBS to enhance MSC expansion and meet GMP standards [41]. | Must be sourced from approved donors and qualified for use. |
| Cell Dissociation Reagent | For detaching adherent MSCs from microcarriers or culture surfaces during harvesting or passaging. | GMP-grade trypsin-EDTA or enzyme-free alternatives. |
| Bioreactor-Specific Consumables | Single-use cultivation chambers, tubing sets, and separation columns that ensure a closed, sterile processing environment [41] [43]. | Quantum cartridge, CliniMACS Prodigy TS730 tubing set [41]. |
| Cryopreservation Medium | Formulated to maintain high cell viability and functionality during frozen storage for off-the-shelf therapies. | Contains DMSO and cryoprotectants in an optimized buffer [41]. |
Optimizing Seeding Density and Passage Protocols to Maintain Stemness During Scale-Up
Frequently Asked Questions
FAQ 1: What is the most critical factor to monitor during MSC scale-up to prevent premature senescence? Maintaining optimal cell density at each passage is paramount. Excessive cell density accelerates senescence and reduces differentiation capacity, while overly low density can diminish the concentration of beneficial autocrine factors [46] [47]. Key indicators of successful expansion include lower population doubling times and high colony-forming unit (CFU) capacity, which are associated with maintained "stemness" [5].
FAQ 2: How does the choice of culture medium impact the quality of scaled-up MSCs? The culture medium formulation directly impacts cell proliferation, potency, and stability. Research-grade media containing fetal bovine serum (FBS) are prone to lot-to-lot variability and risk of introducing xenogenic contaminants [48]. For GMP compliance, animal component-free media, such as those supplemented with human platelet lysate (hPL), are recommended. Studies show that MSCs cultured in specific GMP media (e.g., MSC-Brew GMP Medium) exhibited enhanced proliferation rates and higher colony formation compared to those in standard media [5].
FAQ 3: What are the key quality control checkpoints for a scaled-up MSC process? A robust manufacturing process requires consistent quality control from cell isolation to final product release [48]. Essential checkpoints include:
- Identity: Confirmation of MSC surface markers (CD105, CD73, CD90) and absence of hematopoietic markers [46].
- Viability: Post-thaw viability should typically exceed a minimum of 70%, with many protocols achieving >95% [5].
- Potency: Functional assays such as tri-lineage differentiation (osteogenic, adipogenic, chondrogenic) and immunomodulatory capability tests [48].
- Safety: Sterility, mycoplasma, and endotoxin testing [5] [48].
Troubleshooting Guide
| Problem | Potential Causes | Recommended Solutions |
|---|---|---|
| Reduced Proliferation Rate | Over-confluence at passage, suboptimal seeding density, serum lot variability, cumulative population doublings leading to senescence [46] [49]. | Standardize seeding density (e.g., 5x10³ cells/cm² [5]); Qualify and pre-test serum/hPL lots; Monitor population doublings and use early-passage cells; Switch to defined, xeno-free media [48]. |
| Loss of Differentiation Potential | Inconsistent passaging protocol, enzymatic over-exposure during cell detachment, extended culture time [46]. | Standardize passage schedule based on confluence (e.g., 80-90%); Standardize detachment reagent contact time and temperature; Use quality-controlled reagents and defined differentiation kits. |
| High Variability Between Batches | Donor-to-donor heterogeneity, inconsistent handling techniques, raw material lot-to-lot variability [46] [49]. | Implement donor screening and cell banking; Adopt closed, automated systems where possible [48]; Establish rigorous raw material qualification programs [5] [48]. |
| Low Post-Thaw Viability/Recovery | Suboptimal cryopreservation medium, uncontrolled freezing rate, damaged cold chain. | Use controlled-rate freezing; Validate cryopreservation formula (e.g., with DMSO); Perform stability studies to determine shelf-life [5]. |
Experimental Protocols & Data
Protocol 1: Optimizing Seeding Density for Expansion
This protocol is adapted from studies on optimizing culture conditions for primary human MSCs [5].
Objective: To determine the seeding density that maximizes proliferation while maintaining stem cell markers.
Materials:
- MesenCult-ACF Plus Medium or MSC-Brew GMP Medium [5]
- Trypsin/versene solution
- Phosphate-Buffered Saline (PBS)
- T-75 or T-175 culture flasks
- Hemacytometer or automated cell counter
Method:
- Cell Thawing & Acclimation: Thaw a vial of early-passage MSCs (e.g., P2) rapidly and seed at a standard density. Culture for 3-5 days to allow acclimation.
- Experimental Seeding: After acclimation, detach cells and seed them at different densities (e.g., 2.5x10³, 5x10³, 1x10⁴ cells/cm²) in triplicate flasks.
- Monitoring & Passaging: Culture cells and monitor confluence daily. Passage all cultures when the fastest-growing group reaches 80-90% confluence.
- Data Collection: At each passage, record:
- Cell Count: Use a hemacytometer.
- Population Doubling Time (PDT): Calculate using the formula:
PDT = (T * ln(2)) / ln(FCI), where T is culture time and FCI is the fold change in cell number [5]. - Viability: Assess via Trypan Blue exclusion.
- Flow Cytometry: Analyze for MSC markers (CD73, CD90, CD105) and absence of hematopoietic markers.
- CFU Assay: Seed a low density of cells (e.g., 100 cells/dish) and culture for 10-14 days. Fix, stain with Crystal Violet, and count colonies (>50 cells) [5].
Quantitative Data from Media Comparison Studies
The table below summarizes key performance metrics from a study comparing culture media for infrapatellar fat pad-derived MSCs (FPMSCs) [5].
| Culture Medium | Average Doubling Time | Colony Forming Unit (CFU) Capacity | Key Findings |
|---|---|---|---|
| Standard MSC Media | Reference (higher time) | Reference | Serves as a baseline for comparison. |
| MSC-Brew GMP Medium | Lower across passages | Higher | Enhanced proliferation and potency, suitable for GMP-compliant scale-up [5]. |
| MesenCult-ACF Plus Medium | Intermediate | Intermediate | Animal component-free alternative with good performance [5]. |
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function & Rationale |
|---|---|
| Xeno-Free, GMP-Compliant Media (e.g., MSC-Brew) | Provides a defined, animal-free environment for expansion, reducing variability and safety risks associated with FBS, and supporting enhanced proliferation [5] [48]. |
| Human Platelet Lysate (hPL) | A xeno-free supplement alternative to FBS; rich in growth factors that promote MSC adhesion and mitogenesis [48]. |
| Recombinant Trypsin-like Enzymes | Animal-origin-free enzymes for cell detachment, reducing risk of contamination and immunogenic responses compared to porcine trypsin. |
| Stem Cell Markers Analysis Kit (e.g., BD Stemflow) | Validated antibody panels for flow cytometry to confirm MSC identity (CD105, CD73, CD90) and purity, ensuring product consistency [5] [46]. |
| Cell Dissociation Reagents (e.g., Versen) | Used for gentle cell harvesting during passaging, helping to maintain cell viability and function [50]. |
Process Optimization Diagrams
Diagram 1: Optimized MSC Scale-Up Workflow
Diagram 2: Culture Parameters Impact on Stemness
Implementing Human Platelet Lysate as a Superior Alternative to Fetal Bovine Serum
Troubleshooting Guide: Common Challenges in Transitioning from FBS to HPL
Problem 1: Reduced Cell Proliferation After Switching to HPL
- Potential Cause: Suboptimal HPL concentration or lot-to-lot variability.
- Solution: Perform a dose-response curve to determine the optimal HPL concentration for your specific cell type, typically between 5-10% [51] [52]. Use pooled HPL from multiple donors to minimize variability [53] [54].
- Preventive Measure: Establish a quality control protocol to pre-test each new HPL batch for growth promotion and ensure it meets predefined specifications for key growth factors (PDGF, TGF-β, VEGF) [53].
Problem 2: Poor Cell Attachment and Spreading
- Potential Cause: Lack of attachment factors in HPL compared to FBS.
- Solution: Pre-coat culture vessels with human fibronectin or other human-derived adhesion proteins [54]. Ensure HPL supplementation includes necessary cell adhesion molecules.
- Alternative Approach: Use human platelet lysate serum (PLS), which may provide better attachment properties, as some studies show PLS performs comparably to FBS in cryopreservation and cell attachment [51] [52].
Problem 3: Inconsistent Differentiation Capacity
- Potential Cause: Inadequate characterization of HPL impact on differentiation pathways.
- Solution: Systematically validate trilineage differentiation potential (adiopogenic, osteogenic, chondrogenic) of MSCs expanded in HPL [53] [4]. Ensure differentiation media are optimized for HPL-based cultures.
- Validation Method: Conduct parallel differentiation experiments with HPL and FBS to confirm equivalent or superior performance [53].
Problem 4: Heparin-Induced Cytotoxicity
- Potential Cause: Sensitivity to heparin, which is often added to HPL to prevent gelation.
- Solution: Use heparin-free HPL formulations [55]. Alternatively, determine the minimal effective heparin concentration required, or use alternative anticoagulants.
- Testing Protocol: Perform viability assays with different heparin concentrations to establish a safe threshold for your cell system [54].
Frequently Asked Questions (FAQs)
Q1: What are the primary advantages of HPL over FBS in GMP-compliant MSC manufacturing? HPL offers several critical advantages for GMP applications: (1) eliminates xenogenic risks and ethical concerns associated with FBS [51] [53]; (2) provides higher concentrations of human-specific growth factors leading to enhanced proliferation rates [51] [52]; (3) reduces batch-to-batch variability when using pooled products [54]; and (4) aligns with regulatory preferences for animal-component-free manufacturing in clinical therapies [5] [56].
Q2: How does HPL impact MSC immunophenotype and differentiation capacity? Multiple studies confirm that MSCs expanded in HPL maintain standard immunophenotype (CD73+, CD90+, CD105+, CD34-, CD45-) and trilineage differentiation capacity, with no significant differences compared to FBS-cultured cells [53] [4] [52]. Some studies report enhanced proliferation while preserving stem cell characteristics [51] [54].
Q3: What is the recommended protocol for transitioning from FBS to HPL? Implement a phased transition: (1) Begin with parallel cultures of HPL and FBS for comparative analysis; (2) Use 5-10% HPL concentration, typically lower than the 10% FBS standard [51] [52]; (3) Monitor cell morphology, doubling time, and viability over at least 3 passages; (4) Validate critical quality attributes including immunophenotype and differentiation potential; (5) Establish acceptance criteria for HPL batches based on growth factor content and performance [53] [54].
Q4: How can HPL quality and consistency be ensured for GMP manufacturing? Source HPL from reputable suppliers providing pathogen-inactivated products [57]. Implement rigorous testing for endotoxins, mycoplasma, and sterility [56]. Establish manufacturer-specific specifications for growth factor content (PDGF, TGF-β, VEGF) [53]. Use pooled HPL from multiple donors to minimize individual donor variation [54]. Maintain comprehensive traceability and documentation for regulatory compliance [5] [56].
Quantitative Comparison: HPL vs. FBS Performance Metrics
Table 1: Biochemical Composition Comparison
| Parameter | HPL | FBS | Significance for Cell Culture |
|---|---|---|---|
| Growth Factors | Higher concentration [51] | Lower concentration [51] | Enhanced proliferation in HPL |
| Fibrinogen | Present [51] [52] | Minimal [51] [52] | May require anticoagulants in HPL |
| Calcium | Lower concentration [51] [52] | Higher concentration [51] [52] | Affects coagulation pathways |
| Immunoglobulins | Higher (mainly IgG) [53] | Lower [53] | Human-specific immune components |
Table 2: Functional Performance in MSC Culture
| Performance Metric | HPL | FBS | Research Findings |
|---|---|---|---|
| Proliferation Rate | Higher [51] [54] | Lower [51] [54] | Shorter doubling times with HPL |
| Cell Morphology | Spindle-shaped, elongated [53] | Typical fibroblastoid [53] | No significant functional differences |
| Cryopreservation | Good recovery with PLS [51] [52] | Good recovery [51] [52] | PLS and FBS show better results than PL |
| Immunomodulatory Properties | Maintained or enhanced [4] | Maintained [4] | HPL does not impair immunomodulation |
Experimental Protocols for HPL Implementation
Protocol 1: Dose Optimization for HPL in MSC Expansion
- Prepare media supplemented with HPL at concentrations of 2.5%, 5%, 7.5%, and 10%.
- Seed MSCs at density of 5 × 10³ cells/cm² [5].
- Culture for 5-7 days, monitoring cell morphology daily.
- Harvest and count cells at 80-90% confluency.
- Calculate population doubling time using the formula: Doubling Time = (Duration × log(2)) / (log(Final Concentration) - log(Initial Concentration)) [5].
- Select the concentration yielding optimal doubling time while maintaining characteristic morphology.
Protocol 2: Validation of MSC Properties in HPL
- Expand MSCs for 3 passages in optimized HPL concentration.
- Analyze immunophenotype by flow cytometry for CD73, CD90, CD105, CD34, CD45 [4].
- Evaluate differentiation potential using standardized osteogenic, adipogenic, and chondrogenic induction protocols [53] [4].
- Assess immunomodulatory capacity through T-cell suppression assays [4].
- Compare results with FBS-expanded controls to confirm equivalent or superior functionality.
Signaling Pathways Activated by HPL
Experimental Workflow for HPL Implementation Study
Research Reagent Solutions for HPL Implementation
Table 3: Essential Materials for HPL Transition Studies
| Reagent/Category | Specific Examples | Function & Application Notes |
|---|---|---|
| HPL Types | Pooled HPL, Pathogen-inactivated HPL, Heparin-free HPL [57] [55] | Provides growth factors; pathogen-inactivated versions enhance safety; heparin-free alternatives prevent cytotoxicity |
| Basal Media | DMEM, α-MEM, MSC-Brew GMP Medium [5] | Foundation for culture media; MSC-Brew shows enhanced proliferation in comparative studies |
| Supplemental Factors | Heparin (as needed), L-glutamine, antibiotics | Prevents coagulation of HPL; maintains cell health and prevents contamination |
| Characterization Antibodies | CD73, CD90, CD105, CD34, CD45 [4] | Validation of MSC immunophenotype according to ISCT criteria |
| Differentiation Kits | Osteogenic, Adipogenic, Chondrogenic induction media | Validation of trilineage differentiation capacity post-HPL adaptation |
| Quality Control Assays | Growth factor ELISAs (PDGF, TGF-β, VEGF), endotoxin testing | Ensures HPL batch consistency and quality for GMP applications |
Troubleshooting Guide: Doubling Time and CFU Assays in MSC Research
Troubleshooting Doubling Time Calculations
| Problem | Possible Cause | Solution |
|---|---|---|
| Increasing Doubling Time | Nutrient depletion, metabolic byproduct accumulation, or suboptimal culture conditions (e.g., pH, temperature fluctuations) [58]. | Review and adjust media replacement schedules. Ensure consistent incubator conditions (stable temperature and CO₂) [58]. |
| High Variability in Doubling Time Between Replicates | Inconsistent seeding density, uneven sampling, or manual cell counting errors [58]. | Standardize seeding protocols. Use automated cell counters (e.g., LUNA-FX7) to improve accuracy and reproducibility [58]. |
| Unexpectedly Short Doubling Time | Contamination or an error in the starting cell count calculation. | Check cultures for microbial contamination. Verify the accuracy of the initial cell count and dilution factors [59]. |
| Gradual Increase in Doubling Time over Multiple Passages | Natural decline in proliferation kinetics and early progenitor properties due to extended in vitro expansion [33] [38]. | Monitor differentiation potential. Limit the number of passages used for critical experiments or therapeutic applications [38]. |
Troubleshooting Colony Forming Unit (CFU) Assays
| Problem | Possible Cause | Solution |
|---|---|---|
| Too Many Colonies to Count | Insufficient dilution of the cell suspension before plating [59]. | Perform a serial dilution series to ensure a countable plate (30-300 colonies) [59]. |
| Too Few or No Colonies | Over-dilution of the cell suspension, non-viable cells, or harsh plating conditions [59]. | Reduce the dilution factor. Verify cell viability and ensure the use of appropriate, pre-warmed culture media. |
| Colonies Merged Together | Cells were plated at too high a density, or were not spread evenly across the plate [59]. | Ensure proper technique when spreading the cell suspension and plate at an appropriate density. |
| Inconsistent CFU Results Between Assays | Inconsistent cell dissociation, inaccurate pipetting during serial dilution, or variations in culture media batches. | Standardize the trypsinization and neutralization process. Use calibrated pipettes and maintain consistent reagent quality. |
Frequently Asked Questions (FAQs)
Q1: Why is my MSC doubling time increasing consistently across different time points? A consistent increase in doubling time is a key indicator that cell health may be declining. This is often due to nutrient depletion, pH fluctuations, or the accumulation of metabolic byproducts in the culture medium. In the context of extended passage studies, a gradual increase is expected as MSCs naturally lose proliferative capacity with in vitro expansion [33] [58] [38].
Q2: How often should I measure cells to build an accurate growth curve for doubling time calculation? For most mammalian cells like MSCs, measuring every 12–24 hours provides reliable data. The frequency should be adjusted based on the cell line's known doubling time; faster-growing cultures may require shorter intervals to accurately capture the exponential (log) growth phase [58].
Q3: My CFU results are highly variable. What are the critical steps to improve reproducibility? The two most critical steps are achieving a single-cell suspension during trypsinization and performing an accurate serial dilution series. Incomplete dissociation will lead to clumps that form oversized colonies, while inaccurate dilutions will prevent you from obtaining plates with a countable number of colonies (30-300) [59]. Using automated cell counters for initial counts can significantly improve reproducibility.
Q4: At what passage should I conduct proliferation and CFU assays for a GMP-compliant study? The choice of passage number is critical. Early passages (e.g., P2-P5) are typically used to establish a baseline for proliferation capacity and differentiation potential. However, since extended culture leads to senescence and loss of functionality, it is essential to also monitor these parameters at later passages to define the safe and effective "cell age" limit for your specific GMP application [33] [38].
Q5: What GMP documentation is required for a doubling time or CFU assay? In a GMP environment, all methods must be defined in approved documents. This includes a Standard Operating Procedure (SOP) for the assay, a test method detailing specific steps, and forms for recording raw data (cell counts, colony counts, calculations). All equipment used must have calibration and maintenance records, and analysts must be trained on the procedures [60].
Experimental Protocols & Data Presentation
Protocol 1: Calculating Population Doubling Time
Detailed Methodology:
- Seed cells at a defined density in appropriate culture vessels.
- Harvest and count cells at regular intervals (e.g., every 24 hours). For accurate and consistent counts, use an automated cell counter [58].
- Plot a growth curve with time on the x-axis and the log of the total live cell concentration on the y-axis.
- Identify the exponential (log) phase of growth from the curve.
- Calculate the doubling time (Td) using two time points (t1 and t2) within the log phase and their corresponding cell numbers (N1 and N2):
- Doubling Time (Td) = (t2 - t1) × log(2) / log(N2 / N1) [58].
Quantitative Data: Table of Growth Kinetics
The following table summarizes population doublings and key findings from extended passage studies on human MSCs:
| Study Focus | Passage / Condition | Total Population Doublings | Key Finding on Differentiation Potential |
|---|---|---|---|
| Extended First Passage Culture [33] | Standard Conditions (SC) through P7 | ~27 doublings | Osteogenic and chondrogenic potential maintained; adipogenic potential diminished relative to SC. |
| Extended First Passage Culture [33] | Extended First Passage (EFP) for 34 days | ~16 doublings | Bone formation and chondrogenesis at least equivalent to SC cells through 7 passages. |
| Ex Vivo Expansion of BMSC [38] | After First Passage | Markedly diminished rate | Gradual loss of multiple differentiation potential upon continued culture expansion. |
Protocol 2: Colony Forming Unit (CFU) Assay
Detailed Methodology:
- Perform Serial Dilutions:
- Aseptically prepare a series of tubes (e.g., 1:10 dilutions) by transferring 1 mL of cell suspension into 9 mL of sterile diluent (e.g., culture medium or PBS) [59].
- Plate Cells:
- Pipette a specific volume (e.g., 100 µL) from selected dilutions onto labeled agar plates containing the appropriate growth medium [59].
- Spread Cells:
- Using a sterile cell spreader, gently spread the liquid evenly across the entire surface of the agar. Allow the liquid to be fully absorbed.
- Incubate:
- Incubate plates under optimal growth conditions for the required time (e.g., overnight for bacteria, 1-2 weeks for MSCs).
- Count and Calculate:
- After incubation, select a plate with 30-300 distinct colonies for counting [59].
- Calculate the CFU/mL using the formula: CFU/mL = (Number of colonies counted) / (Dilution Factor × Volume plated in mL).
- Example: For 150 colonies from a plate with a 1:1000 dilution where 100 µL was plated: CFU/mL = 150 / (0.001 × 0.1) = 1,500,000 [59].
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function in Process Monitoring |
|---|---|
| Automated Cell Counter | Provides accurate, reproducible, and rapid cell counts for generating reliable growth curves and calculating doubling time, minimizing human error [58]. |
| Cell Culture Growth Media | Formulated to support MSC growth and maintain pluripotency. Specific media and supplements (e.g., FBS, growth factors) are critical for consistent proliferation and CFU assay results. |
| Trypsin/Enzymatic Dissociation Reagent | Used to detach adherent MSCs into a single-cell suspension, which is a critical first step for both accurate cell counting and CFU assays. |
| Sterile PBS (Phosphate Buffered Saline) | Used for washing cells to remove serum and residual media before trypsinization, and as a diluent for creating serial dilutions in CFU assays. |
| Culture-Tested Agar | Forms a solid, gel-like surface in Petri dishes that allows individual cells to grow into isolated colonies for quantification in the CFU assay [59]. |
| Stains (e.g., Crystal Violet) | Used to stain colonies for easier visualization and counting. Some stains are specific for viable cells. |
| Quality Control Logs & SOPs | GMP-mandated documents that ensure all procedures (from reagent preparation to equipment calibration) are performed consistently and traceably [60]. |
Process Monitoring Workflows
Experimental Workflow for MSC Growth Monitoring
Decision Tree for Doubling Time Anomalies
Addressing Critical Challenges in GMP MSC Manufacturing: Senescence, Contamination, and Variability
Overcoming Donor-to-Donor Variability in Proliferation Capacity Through Protocol Standardization
Troubleshooting Guides
Common Experimental Challenges and Solutions
Issue: Low Cell Yield or Proliferation After Thawing
- Potential Cause 1: Inefficient cryopreservation or thawing process.
- Solution: Standardize the cryopreservation protocol using defined, GMP-compliant reagents like CryoStor10, which is optimized to preserve cellular quality and functionality at the peak of viability [61]. Ensure consistent cooling rates and use controlled-rate freezers.
- Potential Cause 2: Donor-specific biological variability.
- Solution: Implement rigorous donor pre-screening. Utilize services that provide pre-characterized donors with extensive data on immune cell phenotyping and proliferation history to select optimal starting material [61].
- Potential Cause 3: Suboptimal culture medium.
- Solution: Transition to animal component-free, GMP-compliant media formulations. Validation studies show that media like MSC-Brew GMP Medium can significantly enhance proliferation rates and colony-forming unit (CFU) capacity compared to standard media [5].
Issue: Inconsistent Experimental Results Between Batches
- Potential Cause 1: Drift in standard operating procedures (SOPs).
- Solution: Manage protocol drift through regular training, auditing, and proficiency testing for all laboratory personnel. Minor alterations can have a large impact on outcomes [62].
- Potential Cause 2: Variability in raw materials, including donor tissue.
- Solution: Source starting materials from suppliers with a robust regulatory compliance framework for GMP products. This ensures consistency in collection and processing methods, such as using standardized apheresis systems and immediate processing in controlled environments [61].
- Potential Cause 3: Inconsistent cell characterization post-thaw.
Issue: Poor Potency or Immunomodulatory Function in Assays
- Potential Cause: Lack of cellular priming or activation.
- Solution: Implement a cytokine licensing step, such as Interferon-gamma (IFN-γ) priming. Studies on GMP-grade Wharton's jelly MSCs show that IFN-γ priming enhances immunosuppressive properties by inducing IDO activity, leading to a higher potency to inhibit T-cell proliferation both in vitro and in vivo [63].
Frequently Asked Questions (FAQs)
Q1: What are the most critical factors to control when moving from research-grade to GMP-compliant MSC cultures? The most critical factors are the standardization of all raw materials and protocols. This includes the adoption of animal component-free media, the use of enzymatic digestion methods that are GMP-compliant, and the implementation of rigorous quality control checks at every stage—from donor selection and cell isolation to expansion, cryopreservation, and final product release [5] [19] [61].
Q2: How can we objectively quantify and compare proliferation capacity across different donors? Standardized assays must be performed and quantified consistently. Key metrics include:
- Population Doubling Time: Calculated at each passage during expansion [5].
- Colony-Forming Unit (CFU) Assay: A measure of clonogenic potency, where cells are seeded at low density and the number of colonies formed is counted after a set period [5]. These quantitative data allow for direct comparison between donors and culture conditions.
Q3: Beyond media, what other protocol aspects can be standardized to reduce variability? Standardization should extend to the complete workflow:
- Donor Selection: Use pre-characterized donors with known attributes [61].
- Cell Isolation: Adhere to validated methods like enzymatic digestion or density gradient centrifugation with strict parameters [5] [19].
- Passaging: Standardize seeding density, confluence at passage, and the enzymes used for detachment [5].
- Cryopreservation: Use uniform freezing media, controlled-rate freezing, and storage conditions [5] [61].
- Characterization: Employ validated, high-sensitivity flow cytometry panels to ensure consistent phenotyping and purity checks [5] [64].
Experimental Data and Protocols
The following table summarizes key quantitative findings from a study optimizing culture conditions for infrapatellar fat pad-derived MSCs (FPMSCs), highlighting the impact of standardized, GMP-compliant media on proliferation and potency [5].
Table 1: Impact of Culture Media on FPMSC Proliferation and Potency
| Parameter | Standard MSC Media | MesenCult-ACF Plus Medium | MSC-Brew GMP Medium |
|---|---|---|---|
| Doubling Time | Higher across passages | Lower than standard media | Lowest across passages |
| Colony Formation | Baseline | Enhanced compared to standard | Highest observed |
| Viability Post-Thaw | Not specified in results | Not specified in results | >95% (requirement: >70%) |
| Sterility | Not specified in results | Not specified in results | Maintained after 180 days of storage |
Detailed Methodologies
Protocol 1: GMP-Compliant Isolation and Expansion of Adipose-derived MSCs
- Tissue Acquisition: Infrapatellar fat pad (IFP) tissue is acquired as waste tissue during reconstructive surgery and collected in a sterile in-line collection chamber [5].
- Digestion: The tissue is minced and digested with 0.1% collagenase in serum-free media for 2 hours at 37°C [5].
- Cell Isolation: The digested tissue is centrifuged. The cell pellet is washed with PBS, filtered through a 100μm filter, and centrifuged again before being resuspended in culture medium [5].
- Culture & Expansion: Cells are cultured in GMP-compliant, animal component-free media (e.g., MSC-Brew GMP Medium). They are passaged at 80-90% confluency and seeded at a standardized density of 5 × 10³ cells/cm² [5].
- Cryopreservation: Cells are frozen in a cryoprotectant like DMSO and stored in liquid nitrogen vapor phase. Viability and sterility are checked post-thaw [5].
Protocol 2: IFN-γ Priming to Enhance MSC Potency
- Cell Preparation: Culture GMP-grade MSCs (e.g., Wharton's jelly MSCs) to the desired passage [63].
- Priming: Treat the cells with a defined concentration of IFN-γ (e.g., 50 ng/mL) for a set period (e.g., 24-48 hours) prior to harvesting them for experiments or administration [63].
- Mechanism: This priming licenses the MSCs, potentiating their immunosuppressive activity primarily through the induction of Indoleamine 2,3-dioxygenase (IDO) activity, which depletes tryptophan and generates immunosuppressive kynurenines [63].
Diagrams and Workflows
Experimental Workflow for Standardized MSC Processing
The following diagram outlines the core steps in a standardized GMP-compliant workflow for processing MSCs, from donor to final product, incorporating critical quality control checkpoints.
Strategic Approach to Managing Donor Variability
This diagram illustrates the multi-faceted strategy required to mitigate the impact of inherent donor variability on final MSC product quality.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for GMP-Compliant MSC Research
| Item | Function | Example Product / Method |
|---|---|---|
| Animal Component-Free Medium | Provides a consistent, xeno-free environment for MSC expansion, eliminating batch variability and contamination risks. | MSC-Brew GMP Medium [5] |
| GMP-Grade Enzymes | Used for the isolation of MSCs from tissue sources (e.g., collagenase) in a controlled and reproducible manner. | 0.1% Collagenase for tissue digestion [5] |
| Defined Cryopreservation Medium | Protects cell viability and functionality during freezing and storage, ensuring high post-thaw recovery. | CryoStor10 [61] |
| Flow Cytometry Antibody Panels | Critical for cell characterization, identity confirmation (ISCT criteria), and monitoring purity throughout culture. | BD Stemflow Human MSC Analysis Kit [5] |
| Priming Cytokines | Enhances the immunosuppressive potency of MSCs for therapeutic applications prior to use. | Recombinant Interferon-gamma (IFN-γ) [63] |
Preventing Cellular Senescence and Maintaining Differentiation Potential During Extended Culture
For researchers conducting extended passage studies on Mesenchymal Stem Cells (MSCs), two fundamental challenges emerge: the inevitable onset of cellular senescence and the progressive loss of differentiation potential. Cellular senescence is a highly stable cell cycle arrest triggered by various stresses, including DNA damage, oxidative stress, and telomere shortening [65] [66]. In the context of Good Manufacturing Practice (GMP) research for cell therapy, these processes pose a significant barrier to producing sufficient quantities of high-quality, functional cells. This technical support center provides targeted guidance to help you identify, troubleshoot, and prevent these issues in your experiments, ensuring your extended culture studies yield reliable and clinically relevant data.
FAQ: Understanding Senescence and Differentiation Loss
Q1: What are the key hallmarks of cellular senescence I should monitor in my MSC cultures?
You should monitor a combination of morphological, biochemical, and functional hallmarks. Key indicators include:
- Morphological Changes: Cells become significantly enlarged and flattened [66].
- Proliferation Arrest: An irreversible cell cycle arrest occurs, which is non-responsive to mitogenic stimuli [67] [65].
- Senescence-Associated β-Galactosidase (SA-β-Gal): Increased SA-β-Gal activity at pH 6.0 is a widely used biomarker [67].
- Senescence-Associated Secretory Phenotype (SASP): Secretion of pro-inflammatory factors like IL-8, CXCL1/GROα, and various cytokines is a major functional hallmark [67] [66].
- DNA Damage Response (DDR): Persistent DNA damage, marked by DNA double-strand breaks and senescence-associated heterochromatin foci (SAHF), is a common trigger [67] [65].
Q2: How quickly can I expect to see a loss of differentiation potential during extended culture?
The loss of differentiation potential can occur surprisingly early. Research on bone marrow-derived MSCs (BM-MSCs) indicates that after the first passage, cells exhibit a markedly diminished proliferation rate and gradually lose their multiple differentiation potential [38]. One study found that the bone-forming efficiency in vivo of first-passage BM-MSCs was reduced by about 36 times compared to fresh bone marrow [38]. This underscores that both the duration and conditions of culture are critical for maintaining progenitor properties.
Q3: What are the main signaling pathways involved in driving MSC senescence?
The two primary tumor suppressor pathways governing senescence are the p53/p21Cip1 and p16INK4a/Rb pathways [65] [66]. These pathways can be activated by various stresses. Furthermore, hyper-activation of the Ras-MAPK (Mitogen-Activated Protein Kinase) signaling pathway has been directly linked to senescence in human adipose stem/progenitor cells (ASCs). Sprouty1 (SPRY1), a negative regulator of MAPK signaling, has been identified as a key factor; its loss-of-function leads to MAPK hyper-activation, triggering a full senescence phenotype and abrogating adipogenesis [67] [68].
Q4: Are there GMP-compliant culture strategies to delay senescence and maintain functionality?
Yes, optimizing culture conditions is a primary strategy. A 2025 study demonstrated that using specific animal component-free, GMP-compliant media (e.g., MSC-Brew GMP Medium) enhanced the proliferation rates of infrapatellar fat pad-derived MSCs (FPMSCs) compared to standard media, as shown by lower doubling times [5]. Replacing fetal bovine serum (FBS) with human serum (HS) has also shown promise, enhancing the proliferative capacity of placental and umbilical cord MSCs without altering their immunophenotype or immunosuppressive properties, moving towards a xeno-free GMP protocol [4].
Troubleshooting Guides
Guide 1: Addressing Premature Senescence in MSC Cultures
| Observation | Potential Cause | Recommended Action |
|---|---|---|
| Rapid decrease in proliferation rate | Suboptimal culture medium / serum lot | Transition to GMP-compliant, animal component-free media (e.g., MSC-Brew GMP Medium) [5] or test different batches/lots of serum. |
| High SA-β-Gal staining | Hyper-activation of MAPK signaling; Persistent DNA damage | Monitor MAPK pathway activity. Consider strategies to maintain negative regulators like Sprouty1 [67]. Ensure culture conditions minimize oxidative stress. |
| Increased cell size & flat morphology | Onset of senescence program | Isolate and use cells at earlier passages. Implement regular morphological assessment as a quality control check. |
| Presence of DNA double-strand breaks | Oxidative or replicative stress | Use antioxidants where appropriate (though efficacy varies [66]) and avoid over-confluency to reduce replicative stress. |
Guide 2: Mitigating Loss of Differentiation Potential
| Observation | Potential Cause | Recommended Action |
|---|---|---|
| Poor adipogenic/osteogenic yield after induction | Exhaustion of progenitor cells during expansion | Limit the number of population doublings. One study showed MSCs in extended first passage (EFP) maintained osteogenic and chondrogenic capacity but had diminished adipogenic potential relative to frequently passaged cells [33]. |
| Failure to form colonies in CFU assays | Loss of stemness / clonogenic progenitors | Optimize seeding density. Use culture media that supports stemness, as some GMP-media have been shown to enhance colony-forming units [5]. |
| Inconsistent differentiation between batches | Lot-to-lot variability of culture supplements | Switch to defined, xeno-free media formulations or thoroughly pre-test and pool human serum batches to ensure consistency [5] [4]. |
Key Experimental Protocols for Monitoring Senescence and Potency
Protocol 1: Detecting Senescence-Associated β-Galactosidase (SA-β-Gal) Activity
This is a standard cytochemical assay for identifying senescent cells in a population [67].
- Cell Seeding: Seed MSCs at a density of 2,600 cells/cm² on sterile cover slips placed in a multi-well plate.
- Culture: Culture cells in growth medium for 3 days.
- Fixation: Wash cells twice with ice-cold PBS. Fix with 4% w/v Paraformaldehyde/PBS for 20 minutes at room temperature.
- Staining: After fixation and permeabilization, incubate cells with the SA-β-Gal staining solution (pH 6.0) overnight at 37°C in a dry incubator (without CO₂).
- Analysis: Examine cells under a standard light microscope. Senescent cells will display blue cytoplasmic staining.
Protocol 2: Assessing Multilineage Differentiation Potential
This protocol verifies the trilineage differentiation capacity, a core defining property of MSCs.
Adipogenic Differentiation:
- Seed MSCs at a density of 2 × 10⁴ cells/cm² [67] or 7.5 × 10⁴ cells in a 35-mm² dish [4].
- After adherence, replace growth medium with adipogenic induction medium (containing IBMX, indomethacin, dexamethasone, and insulin).
- Maintain cultures for 2-4 weeks, replacing the medium twice weekly.
- Fix cells and stain lipids with Oil Red O. Quantify by eluting the stain with isopropanol and measuring absorbance at 570 nm [67] [4].
Osteogenic Differentiation:
Table 1: Molecular and Phenotypic Markers of Senescent MSCs
| Marker Category | Specific Marker / Assay | Observation in Senescent MSCs | Reference |
|---|---|---|---|
| Cell Cycle Regulators | p53 / p21Cip1 | Induced / Activated | [67] [65] |
| p16INK4a | May or may not be induced (context-dependent) | [67] [66] | |
| Hypo-phosphorylated Rb protein | Increased | [67] | |
| SASP Factors | IL-8, CXCL1/GROα | mRNA expression and secretion significantly increased | [67] |
| DNA Integrity | DNA double-strand breaks | Increased number | [67] |
| Senescence-associated heterochromatin foci (SAHF) | Considerably increased number | [67] | |
| Functional Assays | SA-β-Gal Activity | Positive staining | [67] |
| Adipogenesis | Abrogated | [67] |
Table 2: Impact of Culture Media on MSC Proliferation and Potency
| Culture Condition | Cell Type | Key Outcome vs. Control | Reference |
|---|---|---|---|
| MSC-Brew GMP Medium | FPMSCs | Lower doubling time across passages; higher colony formation | [5] |
| Human Serum (HS) | PL-MSCs, UC-MSCs | Faster proliferation; similar immunophenotype and immunosuppressive capacity | [4] |
| Extended First Passage (EFP) | BM-MSCs | ~16 population doublings then plateau; maintained bone formation in vivo and chondrogenesis; diminished adipogenesis | [33] |
Signaling Pathways and Experimental Workflows
Diagram Title: Core Signaling Pathways Driving Cellular Senescence
Diagram Title: GMP Workflow for Extended MSC Culture
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Senescence and Potency Research
| Reagent / Material | Function / Application | Example & Note |
|---|---|---|
| GMP-compliant, Animal-free Media | Base medium for clinical-grade expansion; reduces batch variability and safety risks. | MSC-Brew GMP Medium [5]; MesenCult-ACF Plus Medium [5]. |
| Human Serum (HS) | Xeno-free supplement for cell growth; can enhance proliferation over FBS. | Pooled allogenic human serum, filtered and tested [4]. |
| SA-β-Gal Staining Kit | Histochemical detection of senescent cells in culture. | Commercial kits available based on standard protocols [67]. |
| Antibodies for WB/Flow Cytometry | Protein-level detection of senescence and stemness markers. | Targets: p53, p21Cip1, p16INK4a, CD73, CD90, CD105 [67] [4]. |
| Differentiation Induction Kits | Standardized assessment of multilineage differentiation potential. | Adipogenic (IBMX, Indomethacin, Insulin); Osteogenic (Dexamethasone, Ascorbate, β-Glycerophosphate) [4]. |
| Collagenase IV | Enzymatic digestion for primary isolation of MSCs from tissue. | Used for isolating MSCs from placenta, umbilical cord, and adipose tissue [67] [4]. |
In Good Manufacturing Practice (GMP) research, particularly in extended passage studies of Mesenchymal Stem Cell (MSC) proliferation capacity, ensuring sterility is not merely a regulatory requirement but a fundamental determinant of product quality, patient safety, and data integrity. The transition from research-scale to clinically applicable manufacturing necessitates robust, closed-system technologies and comprehensive environmental monitoring (EM) protocols. These systems minimize human intervention and prevent adventitious contamination during critical processes like cell isolation, expansion, and passage. This technical support center provides targeted troubleshooting guides and FAQs to help researchers and drug development professionals address specific sterility challenges encountered during GMP-compliant MSC research and manufacturing.
Core Principles of Sterility Assurance
The Role of Closed Systems
A "closed system" in bioprocessing is one where the product-contact components are sealed from the external environment, preventing the introduction of contaminants. For extended MSC culture, this is vital for maintaining sterility across multiple passages, as the risk of contamination increases with every manipulation, media change, or subculturing step.
Fundamentals of Environmental Monitoring
Environmental monitoring is an essential program for contamination control in pharmaceutical manufacturing and cell therapy production. It enables the early detection of risks through the continuous monitoring of air quality, surface cleanliness, and water systems, allowing for swift corrective actions to prevent product contamination [69]. A robust EM program is a cornerstone of GMP, providing data to demonstrate environmental control throughout the lengthy process of MSC expansion.
Troubleshooting Common Sterility Challenges
FAQ 1: Our MSC cultures are consistently showing microbial contamination after the third passage. What could be the source?
Contamination that appears consistently at a later passage typically indicates a systematic process flaw rather than a single isolated incident.
Potential Source & Investigation:
- Raw Materials: Test all culture media, supplements, and reagents for sterility, including performing growth promotion tests on new media lots [69].
- Process-Related: The contamination source is likely introduced during a routine, repeated manipulation, such as media feeding or passaging. Review your aseptic technique and validate that all closed-system transfer connections are secure and sterile.
- Environmental: Intensify your EM in the immediate vicinity of the incubator and biosafety cabinet where these processes occur. Use active air samplers and contact plates on critical surfaces to identify any resident microbial flora [69].
Corrective Action:
- Implement a closed-system processing platform that reduces or eliminates open manipulations.
- Review and retrain staff on aseptic techniques, including the proper use of disinfectants and gowning procedures.
- Introduce more rigorous pre-filtration sterility testing of all media and reagents before use in MSC cultures.
FAQ 2: We are experiencing failed sterility tests on our final MSC product, but our in-process environmental monitoring data shows all results within acceptable limits. How is this possible?
This discrepancy suggests a breach in the container-closure system of the final product or a flaw in the sterility test itself.
Potential Source & Investigation:
- Container-Closure Integrity (CCI): The sterility test itself has limitations; it only detects viable microorganisms present at the time of testing and can be subject to false positives from adventitious contamination during testing [70]. A more reliable approach is to replace sterility testing as a stability-indicating test with a validated CCI test [70].
- Investigate CCI using validated physical methods like vacuum decay, high-voltage leak detection, or dye ingress tests on the filled product containers [70].
- Last Manipulation: Consider the final manufacturing step, such as filling into cryobags or vials. This is a high-risk step where a transient loss of environmental control could occur without being captured by your routine EM schedule.
Corrective Action:
FAQ 3: Our autoclave cycles are aborting when sterilizing large liquid loads, disrupting our production schedule. What is the cause and solution?
This is a common autoclave problem related to the physics of heat transfer.
- Cause: Large volumes of liquid, especially in single large containers, take a very long time to heat to the sterilization temperature (e.g., 121°C). The autoclave's cycle timer may abort the cycle because the setpoint temperature is not reached within the expected timeframe [71].
- Corrective Action:
- Load Configuration: Distribute the liquid load into smaller, standardized containers. This dramatically increases the surface-area-to-volume ratio, allowing for faster and more uniform heating.
- Cycle Selection: Use a specialized "Liquids Cycle" that includes a slow exhaust phase to prevent boil-over, and if available, an F₀ Cycle. An F₀ cycle enables the autoclave to count the time spent heating the liquid toward the total sterilization time, preventing cycle abortion [71].
Essential Experimental Protocols
Protocol: Designing an Effective Environmental Monitoring Program
A scientifically sound EM program is critical for demonstrating environmental control during MSC manufacturing.
- 1. Risk-Based Location Selection: Identify critical control points (CCPs) and high-risk areas susceptible to contamination or with direct product exposure. These include [69]:
- ISO 5 (Grade A) biosafety cabinets or laminar airflow hoods where open processing occurs.
- Areas adjacent to open processing zones.
- Incubator interiors and door handles.
- Pass-through hatches and staging areas for materials.
- 2. Sampling Methods and Frequency:
- Active Air Sampling: Use calibrated volumetric air samplers to quantify airborne particles and microorganisms.
- Passive Air Sampling (Settle Plates): Use open agar plates to monitor microbial fallout over a set exposure time (e.g., 4 hours).
- Surface Sampling: Use contact plates or swabs on equipment and personnel (e.g., gloves) after critical interventions.
- Frequency: Sample at a frequency that reflects the risk and activity level of the operation, typically during every production run.
- 3. Incubation Regime: The incubation protocol must support the recovery of both bacteria and fungi. A dual-temperature incubation is standard.
- A typical evidence-based regime involves initial incubation of Tryptone Soya Agar (TSA) plates at 30–35°C for 2-3 days, followed by incubation at 20–25°C for an additional 3-4 days [72]. This sequence optimizes the recovery of mesophilic bacteria and fungi commonly associated with human activity.
- 4. Data Management and Response:
- Establish scientifically justified Alert and Action Limits based on historical data and risk assessment [69].
- An Alert limit indicates a potential drift from normal conditions and should prompt investigation.
- An Action limit represents a significant deviation requiring immediate corrective action and impact assessment on the product.
Protocol: Validating a Closed System for MSC Expansion
Validating that your system remains "closed" during processing is essential for sterility assurance.
- Objective: To demonstrate that the system integrity is maintained throughout the entire MSC culture process, from inoculation to harvest, preventing microbial ingress.
- Methodology:
- System Integrity Test: Perform a physical test on the system (e.g., pressure hold test, helium leak test) before and after the process simulation to verify the integrity of all seals, tubing, and connectors.
- Process Simulation (Media Hold): Aseptically introduce a sterile growth medium (like TSB) into the system. Perform all typical process operations—mixing, sampling, media additions (via closed transfer), and temperature changes—over the full duration of a typical MSC production run.
- Incubation and Analysis: Incubate the medium within the system for 14 days at 20-25°C and 30-35°C, examining it for turbidity at days 3, 7, and 14.
- Acceptance Criteria: The system maintains physical integrity, and the growth medium remains clear throughout the incubation period, with no evidence of microbial growth.
The Scientist's Toolkit: Essential Reagents and Materials
Table 1: Key Reagents and Materials for GMP-Compliant MSC Research and Sterility Assurance.
| Item | Function in Sterility/EM | GMP-Compliant Example(s) |
|---|---|---|
| Animal Component-Free Cell Culture Media | Eliminates risk of immunogenicity and contamination from animal-derived components (e.g., FBS); essential for clinical-grade MSCs. | MSC-Brew GMP Medium [5], MesenCult-ACF Plus Medium [5], StemPro MSC SFM CTS [73]. |
| Human Platelet Lysate (hPL) | A xeno-free supplement for MSC culture that supports high proliferation rates and is GMP-compliant. | Used at 2%-5% concentration in serum-free media [74]. |
| General Recovery Culture Media | Used in EM settle plates, contact plates, and air samplers to support the growth of environmental bacteria and fungi. | Tryptone Soya Agar (TSA) [72]. |
| Quality Control Microorganisms | Used for Growth Promotion Testing (GPT) to ensure EM and sterility test media support growth. | Characterized strains like Bacillus subtilis and Candida albicans [69]. |
| GMP-Grade Enzymes | For the sterile, controlled isolation of MSCs from tissue sources like the infrapatellar fat pad or Wharton's jelly. | Collagenase NB6 GMP Grade [74]. |
| Container-Closure Integrity Test Systems | Validated physical methods to replace sterility testing for demonstrating continued sterility throughout product shelf-life. | Dye Penetration, High-Voltage Leak Detection, or Vacuum Decay test systems [70]. |
Visualizing Workflows and Systems
Environmental Monitoring Data Response Workflow
The following diagram outlines the logical decision-making process when EM data exceeds established limits, which is critical for maintaining control in a GMP environment.
Closed System MSC Expansion Pathway
This diagram contrasts the contamination risk profiles of open and closed system approaches for a multi-passage MSC expansion process, highlighting where critical risks are mitigated.
Optimizing Cryopreservation and Post-Thaw Recovery to Maintain Viability and Function
Troubleshooting Guides
Common Post-Thaw Cell Recovery Challenges
Problem: Low Cell Viability Immediately Post-Thaw
- Potential Causes: Intracellular ice crystal formation during freezing; osmotic shock during thawing; cytotoxic effects of cryoprotectant (e.g., DMSO); improper cooling rate.
- Solutions:
- Ensure use of a controlled-rate freezer set to an optimal cooling rate (commonly -1°C/min for MSCs) to minimize intracellular ice formation [75] [76].
- For thawing, use a rapid method (e.g., 37°C water bath) to minimize exposure to potentially harmful concentrated solutes [76].
- Include a protein source (e.g., 2% Human Serum Albumin) in the thawing and wash solution to mitigate cell loss and improve viability [77].
- Consider evaluating DMSO-free cryopreservation formulations that have shown improved post-thaw function and reduced toxicity in some studies [78].
Problem: Poor Cell Attachment and Spreading After Plating
- Potential Causes: Cryopreservation-induced disruption of the actin cytoskeleton; downregulation of surface adhesion molecules; ongoing apoptosis.
- Solutions:
- Implement a post-thaw "acclimation" or "recovery" period of 24 hours in standard culture conditions before using the cells. This allows cells to regain normal morphology, re-establish their cytoskeleton, and upregulate key regenerative genes [75] [79].
- Verify that culture surfaces are appropriately coated and that post-thaw media contain necessary supplements to support attachment.
Problem: Reduced Proliferation and Clonogenic Capacity
- Potential Causes: Cryopreservation-induced cellular stress and senescence; metabolic disruption.
- Solutions:
- Use cells at lower passage numbers for cryopreservation, as extensive in vitro expansion can increase susceptibility to freezing-induced senescence [80].
- Allow a 24-hour post-thaw recovery period. Studies show this reactivates metabolic activity and restores proliferation and clonogenic capacity significantly compared to immediately using freshly thawed cells [75].
Problem: Loss of Specific MSC Functionality (e.g., Immunomodulation)
- Potential Causes: Altered surface marker expression (e.g., CD44, CD105); transient reduction in gene expression related to therapeutic paracrine functions.
- Solutions:
- A 24-hour acclimation period post-thaw has been demonstrated to upregulate angiogenic and anti-inflammatory genes and restore potent immunomodulatory function, such as the ability to arrest T-cell proliferation [75] [79].
- Perform potency assays after the post-thaw recovery period to ensure functionality meets pre-defined release criteria for GMP manufacturing.
Problem: Significant Cell Loss During Thawing and Reconstitution
- Potential Causes: Thawing or reconstituting cells in protein-free solutions; diluting cells to excessively low concentrations during washing steps.
- Solutions:
- Always thaw and reconstitute cryopreserved MSCs in a solution containing a protein component, such as Human Serum Albumin (HSA). Up to 50% of MSCs can be lost when using protein-free vehicles [77].
- Avoid diluting MSCs to concentrations below 100,000 cells/mL in protein-free solutions. For post-thaw storage and transport, reconstitution in simple isotonic saline with HSA ensures high viability and stability for several hours [77].
Frequently Asked Questions (FAQs)
Q1: What is the single most critical factor for improving post-thaw MSC function in a GMP setting? Multiple studies consistently identify a 24-hour post-thaw acclimation period in standard culture conditions as critical for restoring functional potency. While freshly thawed (FT) MSCs maintain basic immunomodulatory properties, they exhibit increased apoptosis and reduced metabolic and clonogenic activity. After 24 hours of recovery, these cells show significantly reduced apoptosis and regain their full functional profile, including enhanced expression of key regenerative genes [75] [79].
Q2: Is it better to use freshly cultured MSCs or cryopreserved "off-the-shelf" MSCs for therapies? Cryopreserved "off-the-shelf" MSCs are logistically favorable for GMP processes and acute treatments, allowing for completion of quality control and providing immediate product availability [77] [81]. Evidence shows that with proper handling, thawed MSCs can demonstrate comparable in vitro and in vivo immunomodulatory potency to freshly cultured cells [82]. The key is to optimize the cryopreservation and post-thaw protocols to minimize functional losses.
Q3: What are the best practices for thawing and reconstituting MSCs prior to administration? A clinically compatible and robust method involves:
- Thawing Rapidly: Use a 37°C water bath until only a small ice crystal remains [78].
- Using Protein-Containing Solution: Immediately dilute the thawed cell suspension in a solution like saline or Ringer's acetate supplemented with 2% Human Serum Albumin (HSA). This step is crucial to prevent massive cell loss [77].
- Maintaining Adequate Concentration: During subsequent washing or formulation steps, keep cell concentrations above 100,000 cells/mL to prevent dilution-induced cell death [77].
- Short-Term Storage: For temporary storage (up to 4 hours) before administration, reconstituted MSCs in isotonic saline with HSA maintain >90% viability with minimal cell loss [77].
Q4: How does cryopreservation impact the critical quality attributes (CQAs) of MSCs in extended passage studies? Cryopreservation can temporarily impact several CQAs, especially in cells from higher passages that may be nearing senescence [80]. Key impacts include:
- Phenotype: Transient decrease in surface markers like CD44 and CD105 immediately post-thaw, which can recover after 24 hours [75].
- Viability and Apoptosis: Increased early and late apoptosis immediately post-thaw [75] [82].
- Function: Reduced metabolic activity, proliferation, and clonogenic capacity immediately after thawing. A recovery period is essential to restore these functions [75].
- Genomics and Epigenetics: Cryopreservation, particularly with DMSO, has been associated with epigenetic changes and alterations in the transcription of cytoprotective genes [78]. Monitoring genomic stability during extended passage studies is critical.
Q5: What are the alternatives to DMSO for GMP-compliant cryopreservation? While DMSO remains the most common cryoprotectant, concerns about cytotoxicity and side effects in patients drive the search for alternatives. Novel DMSO-free formulations using combinations of sugars (e.g., sucrose), sugar alcohols (e.g., trehalose, mannitol), and other small molecules (e.g., creatine) are being developed [75] [78]. These have shown promise in improving post-thaw attachment, promoting a more normal cytoskeleton alignment, and upregulating beneficial cytoprotective genes in MSCs [78].
Experimental Protocols for Key Assays
Protocol 1: Assessing Post-Thaw Viability and Apoptosis
Method: Flow cytometry using Annexin V/Propidium Iodide (PI) staining.
- Procedure:
- Thaw MSCs and process according to experimental groups (e.g., Freshly Thawed vs. 24-hour post-thaw recovery).
- Wash cells with PBS containing 1% BSA.
- Resuspend 1.5 x 10^6 cells/mL in 1x Annexin V binding buffer.
- Incubate with Annexin V-FITC for 10 minutes in the dark.
- Add PI immediately before analysis on a flow cytometer.
- Analysis: Gate cells as follows: Viable (Annexin V-/PI-), Early Apoptotic (Annexin V+/PI-), Late Apoptotic/Necrotic (Annexin V+/PI+) [75] [82].
Protocol 2: Evaluating Functional Potency via T-cell Suppression Assay
Method: Co-culture of MSCs with activated peripheral blood mononuclear cells (PBMCs).
- Procedure:
- Isolate PBMCs from donor blood using a Ficoll density gradient.
- Label PBMCs with a cell proliferation dye (e.g., CFSE).
- Activate PBMCs using anti-CD3/CD28 antibodies.
- Co-culture activated PBMCs with test MSCs (e.g., Freshly Thawed, 24-hour recovered) at various MSC:PBMC ratios (e.g., 1:10) for 5 days.
- Analyze PBMCs by flow cytometry to measure the reduction in CFSE dilution (proliferation) in T-cell populations compared to PBMCs cultured alone [75] [82].
Quantitative Data on Post-Thaw MSC Recovery
Table 1: Functional Recovery of MSCs After a 24-Hour Post-Thaw Acclimation Period
| Cellular Parameter | Freshly Thawed (FT) MSCs | 24-Hour Post-Thaw (TT) MSCs | Measurement Method |
|---|---|---|---|
| Viable Cells (Annexin V-/PI-) | Significantly reduced | Significantly increased | Flow Cytometry [75] |
| Metabolic Activity | Significantly decreased | Recovered to higher levels | Resazurin Reduction Assay [75] |
| Clonogenic Capacity | Decreased | Significantly improved | Colony-Forming Unit Assay [75] |
| T-cell Suppression | Maintained, but less potent | Significantly more potent | Co-culture with activated PBMCs [75] |
| Anti-inflammatory Gene Expression | Diminished (e.g., IFN-γ) | Upregulated | RNA Analysis [75] |
Table 2: Impact of Thawing and Reconstitution Solutions on MSC Yield and Viability
| Solution Used for Thawing/Reconstitution | Cell Loss | Viability After 1h | Key Finding |
|---|---|---|---|
| Protein-Free Solution (e.g., Saline alone) | Up to 50% | <80% | Induces significant cell loss during thawing [77] |
| Isotonic Saline + 2% HSA | Minimal (No observed loss for 4h) | >90% | Optimal for post-thaw storage and stability [77] |
| Culture Medium / PBS | >40% | <80% | Poor MSC stability at room temperature [77] |
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagents for MSC Cryopreservation and Post-Thaw Analysis in GMP Research
| Reagent / Material | Function | GMP & Research Considerations |
|---|---|---|
| Dimethyl Sulfoxide (DMSO) | Penetrating cryoprotectant that reduces ice crystal formation. | Cytotoxic at high concentrations/temperatures. Use clinical grade. Requires careful washing or dilution post-thaw [75] [78]. |
| Human Serum Albumin (HSA) | Protein source in freezing or thawing media. Mitigates osmotic stress, improves cell yield and viability. | Preferred over animal serums (e.g., FBS) for clinical applications to reduce xenogenic reactions [77]. |
| Controlled-Rate Freezer | Equipment that precisely controls cooling rate (e.g., -1°C/min). | Critical for process standardization and reproducibility in GMP. Superior to passive freezing for controlling Critical Process Parameters [76]. |
| Annexin V / Propidium Iodide Kit | Fluorescent stains for distinguishing viable, early apoptotic, and dead cells via flow cytometry. | Essential for detailed assessment of post-thaw health beyond simple membrane integrity [75] [82]. |
| Cryopreservation Bags/Vials | Containers for sterile long-term storage in liquid nitrogen vapor phase. | Must be leak-proof, medically graded polypropylene, and DNase/RNase-free to ensure sample integrity [83]. |
| DMSO-Free Cryopreservation Media | Formulations using sugars, sugar alcohols, and amino acids as cryoprotectants. | Emerging alternative to reduce DMSO-related toxicity and potential epigenetic effects [78]. |
Workflow and Signaling Pathway Visualizations
Post-Thaw MSC Recovery Workflow
Cryopreservation Stress and Recovery Signaling
Troubleshooting Guides and FAQs
Common Problem: Inconsistent Proliferation and Yield
Q: My MSC cultures from different donors are growing at vastly different rates, leading to inconsistent cell yields. What is the cause and how can I manage this?
- Primary Cause: Donor-specific factors, particularly age and underlying health conditions, significantly impact MSC proliferation capacity [84] [85].
- Troubleshooting Steps:
- Donor Screening: Review donor metadata. Cells from older or diseased donors (e.g., with critical limb ischemia) inherently exhibit longer population doubling times and reduced final cell yield [85].
- Quality Control: Perform a Colony-Forming Unit (CFU) Assay immediately after isolation. This assesses the clonogenic potential and fitness of the initial cell population.
- Process Adjustment: For donors with known proliferative limitations (e.g., elderly), plan for a longer expansion phase or consider pooling cells from multiple donors to achieve the required therapeutic dose [86].
Experimental Protocol: Colony-Forming Unit (CFU) Assay
- Method: Seed cells at a very low density (e.g., 20, 50, 100 cells) in a culture dish [5].
- Culture: Maintain cells in standard culture conditions for 10-14 days without disturbance.
- Staining and Analysis: Fix cells with 10% neutral buffered formalin and stain with 0.5% Crystal Violet solution. Count colonies containing >50 cells [5]. A lower CFU frequency indicates reduced proliferative capacity.
Common Problem: Poorly Defined MSC Population
Q: I am observing significant functional variability in my MSC batches, even from the same tissue source. How can I better characterize my cell population?
- Primary Cause: MSCs are inherently a heterogeneous mix of cells, including stem/progenitor cells, fibroblasts, and myofibroblasts [86]. Lack of precise characterization exacerbates this issue.
- Troubleshooting Steps:
- Adhere to ISCT Standards: Strictly follow the International Society for Cell & Gene Therapy (ISCT) minimal criteria for defining MSCs: plastic adherence, specific surface marker expression (≥95% positive for CD105, CD73, CD90; ≤2% positive for hematopoietic markers), and trilineage differentiation potential [84] [86].
- Functional Potency Assays: Move beyond basic characterization. Develop disease-relevant functional assays to measure immunomodulatory (e.g., T-cell suppression assay) or tissue repair potency.
- Advanced Characterization: Employ single-cell RNA sequencing (scRNA-seq) to identify and understand distinct functional subpopulations within your MSC cultures [86].
Experimental Protocol: Trilineage Differentiation
- Osteogenic Differentiation: Culture MSCs in media supplemented with dexamethasone, ascorbate-2-phosphate, and β-glycerophosphate for 2-3 weeks. Confirm differentiation by staining for mineral deposits with Alizarin Red.
- Adipogenic Differentiation: Culture MSCs in media with dexamethasone, indomethacin, and insulin for 1-2 weeks. Confirm differentiation by staining lipid vacuoles with Oil Red O.
- Chondrogenic Differentiation: Pellet MSCs and culture in a defined medium with TGF-β for 3-4 weeks. Confirm differentiation by staining for proteoglycans with Alcian Blue or Safranin O.
Common Problem: Variable Performance in Animal Models
Q: My MSC-based therapy shows efficacy in some animal models but fails in others, with no clear reason. What factors should I investigate?
- Primary Cause: Inconsistent therapeutic outcomes often stem from unstandardized manufacturing and administration protocols, which can mask the true efficacy of the cells [86].
- Troubleshooting Steps:
- Standardize Manufacturing: Use defined, xeno-free (animal-free) culture media to eliminate batch-to-batch variability introduced by fetal bovine serum (FBS) [5].
- Cell Source Selection: Choose your MSC tissue source based on scientific rationale. For example, infrapatellar fat pad-derived MSCs (FPMSCs) may be more relevant for osteoarthritis studies than umbilical cord MSCs [5] [86].
- Control Administration Protocols: Standardize the cell concentration, delivery solution (e.g., saline, hyaluronic acid), and post-thaw handling procedures across all experiments [86].
The following tables summarize key quantitative data relevant to managing MSC heterogeneity in a GMP-compliant extended passage study.
Table 1: Impact of Culture Media on MSC Proliferation and Potency This data is adapted from a study comparing animal-free media for culturing infrapatellar fat pad-derived MSCs (FPMSCs) [5].
| Media Formulation | Average Doubling Time | Colony Formation Capacity (CFU) | Key Characteristics |
|---|---|---|---|
| Standard MSC Media (with FBS) | Higher doubling time | Lower colony formation | Introduces variability, not GMP-compliant |
| MSC-Brew GMP Medium | Lower doubling time | Higher colony formation | Defined, xeno-free, supports proliferation & potency |
| MesenCult-ACF Plus | Intermediate doubling time | Intermediate colony formation | Defined, xeno-free |
Table 2: Impact of Donor Health Status on MSC Proliferative Capacity This data is derived from a comparison of bone marrow-derived MSCs from patients with Critical Limb Ischemia (CLI) versus young healthy donors [85].
| Donor Population | Marrow Mononuclear Cells | Colony-Forming Units (CFU-F) | Population Doubling Time | Final Cell Yield |
|---|---|---|---|---|
| CLI Patients | Significantly lower | No significant difference | Significantly longer | Significantly reduced |
| Young Healthy Donors | Higher | No significant difference | Shorter | Higher |
Experimental Workflow and Signaling Pathways
MSC Heterogeneity Management Workflow
This diagram outlines a core workflow for managing MSC heterogeneity from isolation to characterization in a GMP-compliant research setting.
Immunomodulatory Signaling Pathways of MSCs
This diagram visualizes the key immunomodulatory mechanisms of MSCs, which are a primary source of their therapeutic effect and subject to heterogeneity.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for GMP-Compliant MSC Research
| Reagent / Material | Function & Role in Managing Heterogeneity | Example Products (from search) |
|---|---|---|
| Defined, Xeno-Free Media | Eliminates batch-to-batch variability from animal sera; ensures consistent, GMP-compliant cell expansion. | MSC-Brew GMP Medium [5], MesenCult-ACF Plus Medium [5] |
| Flow Cytometry Antibodies | Confirms cell identity and purity per ISCT criteria (CD105, CD73, CD90 positive; CD45, CD34, CD14 negative). | BD Stemflow Human MSC Analysis Kit [5] |
| Trilineage Differentiation Kits | Validates multipotent differentiation potential, a core defining property of MSCs. | Various commercial osteogenic, adipogenic, chondrogenic kits. |
| GMP-Compliant Dissociation Enzymes | For tissue dissociation and cell passaging without introducing animal-derived contaminants. | 0.1% Collagenase (used in FPMSC isolation) [5] |
FAQ: Key Challenges in Late-Passage MSC Cultures
Why does my MSC proliferation rate decrease in later passages?
A decline in proliferation is a common challenge in extended MSC cultures and is often indicative of the onset of senescence. This can be caused by several factors related to culture conditions and handling. Key contributors include the accumulation of metabolic waste products from infrequent medium changes, suboptimal passaging techniques that select for less robust cells, and inconsistent seeding densities that disrupt normal cell-cell contact signaling. Over time, these factors can trigger stress responses and epigenetic changes that lead to irreversible growth arrest [87] [88].
How can I ensure my MSC culture process is aligned with GMP standards for drug development?
Adherence to Current Good Manufacturing Practice (cGMP) regulations is fundamental. This requires a scientific, risk-based approach to your control strategy. Per 21 CFR § 211.110, you must identify and monitor critical quality attributes (CQAs) of your cells and the in-process materials [89] [90]. This involves defining and justifying where and when in-process controls and tests occur during significant phases of your culture process, such as at passaging or before cryopreservation. All processes, including the methods and reagents for passaging and media formulation, must be thoroughly documented and validated to ensure batch-to-batch uniformity and product integrity [90].
Troubleshooting Guide: Diagnosis and Solutions
Use the following table to diagnose and address common issues leading to reduced proliferation.
| Observation | Potential Cause | Recommended Action | GMP Compliance Consideration |
|---|---|---|---|
| Gradual decrease in growth rate and changes in cell morphology | Onset of replicative senescence; Accumulation of metabolic waste. | Increase feeding frequency; Optimize seeding density; Use fresh reagent aliquots. | Document all process parameters and reagent lot numbers for traceability [90]. |
| Sudden drop in viability and proliferation post-passaging | Over-exposure to enzymatic passaging reagents (e.g., Accutase); Mechanical damage during cell detachment. | Protocol: Switch to or optimize use of enzyme-free dissociation reagents (e.g., ReLeSR). Standardize incubation time and quenching. | Validate any new passaging method to ensure it consistently yields viable cells with required critical quality attributes [87] [88]. |
| High variability in proliferation between flasks | Inconsistent seeding densities; Poor distribution of cells during plating. | Protocol: Ensure a homogeneous single-cell suspension or consistent aggregate size before seeding. Use a systematic plate motion (back-and-forth, side-to-side) for even distribution. | Define and justify acceptable ranges for in-process controls like seeding density and cell aggregate size [87] [90]. |
| Increased expression of senescence markers | Culture stress from suboptimal base medium or growth factor concentration. | Systemically optimize basal medium and growth factor supplementation (see Media Optimization Table). | Any change in medium formulation constitutes a process change and must be approved by the quality unit per established control strategy [90]. |
Experimental Protocol: Media Optimization and Evaluation
This protocol provides a detailed methodology for testing and optimizing media components to rescue proliferation in late-passage MSCs.
A. Preparations Before You Begin
- Institutional Permissions: All cell culture work must comply with local institutional guidelines for laboratory safety and ethics.
- Cell Lines: Use late-passage MSCs (e.g., Passage 8+) with documented reduced proliferation.
- Materials:
- Basal media (e.g., DMEM/F12).
- Pre-selected growth factor supplements (e.g., FGF-2, PDGF).
- Attachment substrates (e.g., Vitronectin, Matrigel, Synthemax).
- Enzyme-free passaging reagent (e.g., ReLeSR) or enzymatic alternative (e.g., Accutase).
- Rock inhibitor (Y-27632 2HCI) for enhancing survival after passaging [88].
- Equipment: Humidified incubator at 37°C with 5% CO₂, plate vortexer, pre-chilled labware for matrix handling.
B. Step-by-Step Optimization Procedure
- Coating Plates: At least 1 hour before passaging, coat new tissue culture plates with your selected substrate (e.g., Vitronectin or Matrigel). Keep Matrigel on ice at all times during handling to prevent premature gelling [88].
- Preparation of Media Conditions: Aliquot the basal medium and prepare at least 3-5 different test media by supplementing with different combinations and concentrations of growth factors. Always include a standard/control medium for baseline comparison.
- Passaging MSCs:
- Wash cells with D-PBS (without Ca++ and Mg++) [87].
- For enzymatic passaging, add enough Accutase to cover the monolayer and incubate at 37°C until cells detach. Neutralize with serum-containing medium or inhibitor.
- For non-enzymatic passaging with ReLeSR:
- Add ReLeSR and aspirate within 1 minute, leaving a thin film.
- Incubate at 37°C for 6-8 minutes (optimize time for your cell line).
- Add culture medium and detach cells by firmly tapping the plate or using a plate vortexer at 1200 rpm for 2-3 minutes [87].
- Transfer the cell suspension (either single cells or aggregates) and centrifuge to form a pellet.
- Reseeding and Culture:
- Resuspend the cell pellet in control medium.
- Seed cells at a consistent, optimized density (e.g., 10,000 cells/cm²) into the pre-coated plates containing the different test media. Include Rock inhibitor (Y-27632 2HCI) in the medium for the first 24 hours post-passaging to improve cell survival [88].
- Move the plate in quick, short, back-and-forth and side-to-side motions to distribute cells evenly. Do not disturb for 24 hours.
- Monitoring and Analysis:
- Change medium after 24 hours and then every other day, or as required by the experiment.
- Monitor cultures daily for morphology and confluence.
- At a pre-defined endpoint (e.g., 3-5 days), harvest cells and quantify outcomes:
- Proliferation Rate: Use cell counting or metabolic activity assays.
- Viability: Perform trypan blue exclusion staining.
- Senescence: Conduct β-galactosidase staining.
- Identity/Potency: Perform immunostaining for MSC markers (e.g., CD73, CD90, CD105) and differentiation assays.
The following table summarizes key components to test during media optimization.
| Media Component | Function | Typical Concentration Range | Optimization Consideration |
|---|---|---|---|
| Basic Fibroblast Growth Factor (FGF-2) | Promotes self-renewal, inhibits spontaneous differentiation. | 1 - 20 ng/mL | Essential for maintaining proliferation; titrate to counteract senescence. |
| Platelet-Derived Growth Factor (PDGF) | Potent mitogen for mesenchymal cells. | 1 - 20 ng/mL | Can synergize with FGF-2 to enhance long-term growth. |
| Vitamin C (Vc) | Antioxidant; promotes collagen synthesis and genomic stability. | 50 - 100 µg/mL | Can reduce oxidative stress-induced senescence in late passages [88]. |
| Transforming Growth Factor-β (TGF-β) | Regulates cell cycle and extracellular matrix production. | 0.5 - 5 ng/mL | Biphasic effect; low concentrations may support growth, high concentrations can induce growth arrest. |
| CHIR99021 | GSK-3 inhibitor; activates Wnt/β-catenin signaling. | 1 - 6 µM | Can boost proliferation but requires careful titration to prevent unwanted differentiation [88]. |
The Scientist's Toolkit: Research Reagent Solutions
| Reagent | Function | Key Consideration for GMP Compliance |
|---|---|---|
| ReLeSR | Enzyme-free, selective passaging reagent that enables cell detachment as aggregates. | Reduces exposure to enzymatic variability; requires validation for aggregate size and post-passaging viability [87]. |
| Vitronectin XF / Synthemax | Defined, xeno-free cell culture substrates for cell attachment. | Preferred over animal-derived matrices (e.g., Matrigel) for reduced lot-to-lot variability and better regulatory alignment [87] [88]. |
| TeSR-E8 / mTeSR Plus | Defined, serum-free medium specifically formulated for pluripotent stem cells. | While for PSCs, these exemplify the defined, high-quality media standards to emulate for MSC culture to ensure consistency and safety [87] [88]. |
| Y-27632 (Rock Inhibitor) | Improves cell survival after passaging and cryopreservation by inhibiting apoptosis. | Use should be documented and justified; its necessity may indicate suboptimal passaging conditions [88]. |
| Accutase | Enzymatic cell dissociation blend for gentle detachment into a single-cell suspension. | Enzymatic activity can vary; requires precise control of incubation time and temperature during the validated process [88]. |
Signaling Pathways and Experimental Workflow
Media Optimization Logic
This diagram outlines the systematic approach to troubleshooting and the key biological pathways that can be targeted through media optimization to improve MSC health and proliferation in extended cultures.
Quality Control, Potency Assays, and Comparative Analysis of GMP-Grade MSC Products
Troubleshooting Guides
Viability and Proliferation Capacity
Problem: Low Cell Viability Post-Thaw or During Extended Passage Question: Why are my Mesenchymal Stem Cells (MSCs) showing low viability after cryopreservation or failing to maintain proliferation capacity through extended passages in GMP-compliant media?
Investigation and Solution:
- Check Culture Medium Formulation: Suboptimal culture medium is a primary cause. Research shows that switching from standard media or fetal bovine serum (FBS) to specific GMP-compliant, xeno-free media can significantly enhance proliferation. A 2025 study found that MSCs cultured in
MSC-Brew GMP Mediumexhibited lower doubling times across passages compared to other media, indicating increased proliferation [5]. - Confirm Post-Thaw Viability Specifications: For clinical applications, your protocol must define minimum viability thresholds. A GMP validation study successfully established a specification of >95% viability post-thaw, far exceeding the minimum requirement of >70% [5]. Use Trypan Blue exclusion assay to track this parameter [5].
- Assess Proliferation Kinetics: Calculate population doubling time throughout your extended passage study. The formula is: Doubling Time = (Duration of Culture × log(2)) / (log(Final Cell Count) - log(Initial Cell Count)) [5]. Monitor for significant increases in doubling time, which indicate reduced proliferative capacity.
- Verify Colony Forming Unit (CFU) Potential: Reduced CFU capacity indicates diminished stem cell potency. Seed cells at low densities (e.g., 20, 50, 100 cells per dish), culture for 10-14 days, fix with formalin, and stain with Crystal Violet to count colonies [5]. Studies correlate enhanced proliferation media with higher colony formation [5].
Problem: Inconsistent Proliferation Rates Between Donors Question: How do I address significant donor-to-donor variability in MSC proliferation rates while maintaining GMP compliance?
Investigation and Solution:
- Systematic Media Comparison: This is a recognized challenge in GMP manufacturing [73]. Evaluate multiple GMP-compliant media in parallel using cells from different donors. A comparative assessment of xeno-free media like
MSC-Brew GMP Medium,MesenCult-ACF Plus Medium, andHuman Platelet Lysate (HPL)-based mediacan identify the most robust formulation for your specific MSC source [5] [73]. - Quality All Raw Materials: Ensure that all media components, including HPL, are sourced from approved suppliers and tested for GMP-compliant quality attributes, including growth promotion capacity and sterility [73]. Document all raw material lot numbers.
- Establish Donor-Specific Ranges: For critical quality attributes like doubling time, it may be necessary to establish acceptance criteria ranges specific to each donor, rather than applying a single, rigid limit across all donors.
Sterility and Purity
Problem: Positive Sterility Test Result Question: My batch of MSCs has failed a sterility test. What is the correct investigation procedure according to GMP regulations?
Investigation and Solution:
- Immediate Laboratory Investigation: The FDA requires a thorough investigation by the quality control unit to definitively determine if the microbial presence is due to a laboratory error or faulty materials used in the sterility test itself [91] [92] [93]. This includes:
- Reviewing the analyst's technique and calculations.
- Examining instrumentation and maintenance records.
- Checking the quality of culture media and other test materials [91].
- Follow Repeat Test Protocol: A sterility test may be repeated only once if the investigation conclusively attributes the initial positive result to a laboratory error or faulty materials [92] [93]. The repeat test must use the same method and a comparable product sample from the same stage of manufacturing [93]. If the repeat test is negative, the product meets sterility requirements; if positive, the batch must be rejected [92] [93].
- Formal Out-of-Specification (OOS) Investigation: If the laboratory investigation is inconclusive, a formal OOS investigation extending beyond the lab must be initiated. This involves quality assurance personnel and must outline:
- The reason for the investigation.
- A summation of process sequences that may have caused the problem.
- Corrective actions to save the batch and prevent recurrence.
- An assessment of other batches or products possibly affected [91].
Problem: High Endotoxin Levels in Final Product Question: Endotoxin testing results are out-of-specification for my MSC-based product. What are the potential sources and corrective actions?
Investigation and Solution:
- Audit Raw Materials and Reagents: Endotoxins are pyrogenic substances introduced from bacterial contaminants. Test all incoming raw materials, including culture media, supplements, and reagents, for endotoxin levels against established specifications [94].
- Verify Cleaning and Sterilization Procedures: Review and validate cleaning procedures for all bioreactors, closed system sets, and tubing. Ensure that sterilization cycles (e.g., autoclaving, sterile filtration) are performed correctly and documented.
- Review Environmental Monitoring Data: Check data from your cleanroom environmental monitoring program. excursions in viable particle counts could indicate a breach in aseptic processing conditions that introduced contaminants.
Identity and Characterization
Problem: Loss of MSC Marker Expression During Extended Passage Question: Flow cytometry analysis shows diminished expression of classic MSC surface markers (e.g., CD73, CD90, CD105) after several passages. What does this mean and how should I proceed?
Investigation and Solution:
- Confirm Immunophenotype with Controls: Adhere to the International Society for Cellular Therapy (ISCT) guidelines. Use a standardized kit like the
BD Stemflow Human MSC Analysis Kitand include appropriate isotype controls [5]. The typical immunophenotype is CD73+, CD90+, CD105+, CD34-, CD45- [5] [4]. - Correlate with Functional Potency Assays: Loss of marker expression may indicate senescence or differentiation. Correlate flow cytometry data with functional potency assays:
- Trilineage Differentiation: Confirm ability to differentiate into adipocytes (Oil Red O staining), osteoblasts (Alizarin Red S staining), and chondrocytes [5] [4].
- Immunomodulatory Function: Perform a suppression assay, such as the inhibition of peripheral blood mononuclear cell (PBMC) proliferation, to assess therapeutic potency [4].
- Review Culture Conditions: Some GMP-compliant media are better at maintaining immunophenotype over long-term culture. If marker loss is observed, re-evaluate your culture medium. Studies show that MSCs expanded in
MSC-Brew GMP MediumorHuman Serum-supplemented mediacan maintain stem cell marker expression even after cryopreservation and through passages [5] [4].
Frequently Asked Questions (FAQs)
Q1: What are the minimum required quality control tests for releasing an MSC batch for clinical use? A1: At a minimum, each lot must be tested for:
- Viability: Must meet a predefined threshold, often >70% as a minimum, with many GMP protocols achieving >95% [5].
- Sterility: Must test negative for bacterial and fungal contamination using a validated method like the BacT/Alert system [92] [5].
- Identity: Must express characteristic MSC surface markers (CD73, CD90, CD105) and lack hematopoietic markers (CD34, CD45) via flow cytometry [5] [4].
- Purity: Must test negative for Mycoplasma and have endotoxin levels below the specified limit [5].
- Potency: Must demonstrate a relevant biological function, such as differentiation capacity or immunomodulatory activity [4].
Q2: Can I use an "outlier test" to discard an initial OOS viability result? A2: The use of outlier tests for chemical (and by extension, biological) test results is highly restricted. An FDA guide states that "outlier tests cannot be used for chemical testing results" and are "never appropriate for statistically based tests" [91]. The focus must be on conducting a thorough laboratory investigation to find the root cause, not on statistically discarding unfavorable data.
Q3: What is the difference between a laboratory investigation and a formal OOS investigation? A3:
- Laboratory Investigation: An informal, initial inquiry conducted by the analyst and supervisor. It focuses on the testing procedure, calculation, equipment, and reagents used in the specific test [91].
- Formal OOS Investigation: A comprehensive, documented investigation triggered when the lab investigation is inconclusive. It extends beyond the lab to include manufacturing and quality assurance, examines the entire process, and requires a definitive conclusion and corrective actions [91].
The following tables summarize key quantitative data from recent GMP-focused MSC research to aid in establishing your own specifications and protocols.
Table 1: Performance of GMP-Compliant Culture Media in MSC Expansion
| Media Name | Key Characteristics | Impact on Doubling Time | Impact on CFU Capacity | Viability Post-Thaw | Reference |
|---|---|---|---|---|---|
| MSC-Brew GMP Medium | Animal component-free | Lower doubling times across passages | Higher colony formation | >95% | [5] |
| MesenCult-ACF Plus | Animal component-free | Not specified in results | Not specified in results | Not specified | [5] |
| StemPro MSC SFM CTS | Xeno-free, for clinical applications | Highest proliferation rate | Not specified | Not specified | [73] |
| Human Platelet Lysate (HPL)-based media | Xeno-free, cost-effective | Lower than StemPro, higher than standard FBS media | Maintained | Not specified | [73] |
Table 2: Key Release Specifications for Clinical-Grade MSCs from a GMP Validation Study
| Quality Attribute | Test Method | Release Specification | Result Achieved in GMP Study | |
|---|---|---|---|---|
| Viability | Trypan Blue Exclusion | >70% | >95% | |
| Sterility | BacT/Alert System | No growth of microorganisms | No growth detected | |
| Mycoplasma | Mycoplasma Assay | Negative | Negative | |
| Endotoxin | Endotoxin Assay | Below specified limit | Below specified limit | |
| Identity | Flow Cytometry | Expression of CD73, CD90, CD105 | Marker expression maintained post-thaw | |
| Stability | Viability assessment after storage | Maintain viability over shelf-life | Maintained for up to 180 days | [5] |
Experimental Protocols for Key QC Tests
Protocol 1: Colony Forming Unit (CFU) Assay for Potency Assessment
- Cell Seeding: Trypsinize, count, and seed MSCs at very low densities (e.g., 20, 50, 100, and 500 cells) in large (e.g., 15 mm) cell culture dishes containing 15 mL of your GMP-compliant culture medium [5].
- Incubation: Incubate the cells for 10-14 days, replacing the medium twice a week [5].
- Staining and Fixing:
- Aspirate the medium and wash the cell layer gently with PBS.
- Fix the cells with 10% neutral buffered formalin for 30 minutes.
- Aspirate the formalin and wash twice with PBS.
- Stain with 10% Crystal Violet solution for 20-30 minutes [5].
- Analysis: Rinse with water to remove excess stain, air dry, and count the number of colonies (aggregates of >50 cells) manually or using imaging software.
Protocol 2: Flow Cytometry for MSC Immunophenotype Identity
- Cell Preparation: Harvest MSCs at approximately 70-80% confluency (e.g., passage 3-5) using a trypsin-EDTA solution. Wash the cells with PBS [5] [4].
- Staining: Resuspend the cell pellet and divide into aliquots for each antibody. Stain with fluorescently conjugated antibodies against CD73, CD90, CD105, CD34, and CD45, along with appropriate isotype controls. Incubate for 30 minutes at 4°C in the dark [5] [4].
- Analysis: Wash the cells to remove unbound antibody, resuspend in fixation buffer, and acquire at least 20,000 events per sample using a flow cytometer (e.g., FACScalibur) [5] [4]. Analyze the data to confirm a positive profile for CD73, CD90, CD105 and a negative profile for CD34 and CD45.
Experimental Workflow and Decision Pathways
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for GMP-Compliant MSC QC Testing
| Reagent / Solution | Function in QC Testing | GMP-Compliant Consideration |
|---|---|---|
| Trypan Blue Solution | Viability staining for dye exclusion assay. | Must be sterile-filtered and qualified for use. Document source and lot number [5]. |
| BacT/Alert Culture Media | Sterility testing using automated microbial detection system. | Use validated test kits. Growth promotion tests must be performed for each lot [92] [5]. |
| Mycoplasma Testing Kit | Detection of Mycoplasma contamination. | Use PCR-based or culture-based kits approved for lot release testing [5]. |
| Endotoxin Testing Kit (LAL) | Quantification of bacterial endotoxins. | Must be validated for the specific product (MSCs). Use FDA-approved methods [5]. |
| Flow Cytometry Antibody Cocktail (e.g., CD73, CD90, CD105, CD34, CD45) | Immunophenotype identity verification. | Use pre-configured GMP-grade kits where possible (e.g., BD Stemflow). Validate antibody panels [5]. |
| Primary Reference Standards (e.g., for endotoxin) | Calibration and qualification of test methods. | Source from official pharmacopoeias (USP, EP) or recognized standards bodies (WHO). Maintain a traceable inventory [94]. |
| Differentiation Induction Kits (Adipo, Osteo, Chondo) | Functional potency testing via trilineage differentiation. | Use xeno-free or GMP-compliant kits. Validate staining outcomes (Oil Red O, Alizarin Red) [5] [4]. |
Why is potency testing critical for MSC-based therapies? Mesenchymal Stromal Cells (MSCs) are advanced therapy medicinal products with inherent complexity and heterogeneity. Their biological activity can vary between donors and be altered during manufacturing and storage. Potency assays are essential quality control measures that confirm a batch can exert its specific desired biological effect, directly linking to clinical efficacy. These assays move beyond simple identity, purity, and viability tests to functionally demonstrate the MSC's therapeutic mechanism of action [95].
In the context of extended passage studies and GMP research, documenting potency becomes paramount. As MSCs expand in vitro, they undergo morphological, phenotypic, and genetic changes that can compromise their fitness and functionality. A robust potency testing strategy ensures that MSC products maintain their critical quality attributes throughout their defined shelf-life and manufacturing scale-up [96].
Assessing Immunomodulatory Capacity
The immunomodulatory function of MSCs is a primary therapeutic mechanism, but it involves multiple pathways and immune cell interactions. Selecting a potency assay must therefore be guided by the intended clinical indication and mechanism of action.
Key Assay Principles
What are the main approaches to test immunomodulatory potency? Two main approaches exist, targeting different arms of the immune system:
- Innate Immunity Focus (Macrophage-based assays): For conditions dominated by innate immunity (e.g., chronic wounds), assays measuring MSC effects on macrophages are relevant. A key readout is the secretion of Interleukin-1 Receptor Antagonist (IL-1RA), which induces a switch from pro-inflammatory M1 to pro-regenerative M2 macrophages [95].
- Adaptive Immunity Focus (Lymphocyte-based assays): For T-cell-mediated diseases (e.g., GvHD), assays measuring inhibition of T-cell proliferation are standard. These use stimuli like phytohemagglutinin (PHA) or antibody-mediated CD3/CD28 activation to trigger proliferation, which is then suppressed by MSCs [97].
Detailed Protocol: IL-1RA Secretion Assay
This protocol quantifies anti-inflammatory potency in an M1 macrophage-driven environment [95].
Step 1: Macrophage Differentiation
- Differentiate human THP-1 monocytic cells by incubating with 150 nmol/mL Phorbol 12-myristate 13-acetate (PMA) for 48 hours.
- Confirm differentiation by flow cytometry for marker CD36 (≥50% expression required).
Step 2: Co-culture and M1 Polarization
- Co-culture ABCB5+ MSCs (or your MSC product) with the differentiated macrophages at an optimized ratio.
- Induce M1 polarization by adding 50 IU/mL recombinant human IFN-γ at co-culture start, followed by a second dose with 20 ng/mL LPS from E. coli O111:B4 at 24 hours.
- Include controls: unstimulated co-cultures and macrophage-only cultures.
Step 3: Quantification and Analysis
- After 48 hours, collect culture supernatants.
- Measure IL-1RA concentration using a validated, quantitative sandwich ELISA (e.g., Human IL-1RA/IL-1F3 Quantikine ELISA Kit).
- Compare results against pre-defined release specifications.
The workflow for this assay is standardized as follows:
Detailed Protocol: T-cell Proliferation Inhibition Assay
This protocol measures the suppression of adaptive immune responses [97].
Step 1: PBMC Preparation
- Isolate PBMCs from healthy donor buffy coats using density gradient centrifugation (e.g., LymphoPrep).
- Cryopreserve PBMCs in standardized cryomedium (e.g., 90% FBS + 10% DMSO) for consistent, long-term use.
Step 2: CFSE Staining and Stimulation
- Thaw and recover PBMCs. Label with 5 µM Carboxyfluorescein succinimidyl ester (CFSE) in PBS with 2.5% FBS for 10 minutes at 37°C.
- Seed CFSE-labelled PBMCs in a 96-well round-bottom plate.
- Activate T-cells by adding a mitogen. PHA-L (5 µg/mL) is a robust and recommended stimulus.
- Co-culture with MSCs at a predefined ratio (e.g., 1:10 MSC:PBMC).
Step 3: Flow Cytometric Analysis
- After 3-5 days, analyze cells by flow cytometry.
- The CFSE dye dilutes with each cell division. Measure the percentage of proliferated (CFSE-low) cells in the lymphocyte gate.
- Calculate percentage inhibition relative to stimulated PBMCs without MSCs.
Assessing Differentiation Potential
Multilineage differentiation is a defining characteristic of MSCs. Quantifying this potential confirms cellular "fitness" and is especially sensitive to culture-induced senescence.
Impact of In-Vitro Aging on Differentiation
How does extended passage affect MSC differentiation potential? Studies show that prolonged in vitro expansion significantly impacts differentiation capacity, but not all lineages are affected equally.
- Osteogenic Potential: This is highly susceptible to culture-induced senescence. Osteogenesis of aged MSCs (e.g., passage 8) is significantly compromised compared to early-passage cells (e.g., passage 4) [96].
- Adipogenic Potential: This potential appears to be better preserved with in vitro aging. MSCs at later passages can still differentiate into adipocyte-like cells, though quality may vary [96].
- Culture Conditions Matter: The expansion medium (e.g., DMEM-based vs. αMEM-based) can influence the rate of senescence and the quality of differentiated cells [96].
Table 1: Impact of Extended Passage on MSC Differentiation Potential
| Differentiation Lineage | Impact of In-Vitro Aging (Extended Passage) | Notes and Experimental Considerations |
|---|---|---|
| Osteogenesis | Significantly compromised [96] | Use early-passage cells (e.g., P4) for bone formation studies. αMEM-based medium may support better osteogenic outcomes [96]. |
| Adipogenesis | Better preserved [96] | Adipocyte-like cells can form at late passages (e.g., P8), but lipid accumulation and gene expression should be quantified. |
| Chondrogenesis | Not explicitly covered in results | A standard trilineage potential; consult other sources for age-related effects. |
Strategies for Monitoring Differentiation
What are alternatives to endpoint differentiation stains? Traditional endpoint assays (Oil Red O for adipocytes, Alizarin Red for osteocytes) are destructive and provide limited dynamic information. Advanced methods offer richer data:
- Real-time, Non-invasive Monitoring: Live-cell imaging systems allow continuous observation of morphological changes throughout differentiation without fixing cells [98].
- High-throughput Cytometry: Multiplexed, rapid analysis provides a comprehensive view of the differentiation process, increasing efficiency and preserving cellular material for further analysis [98].
Troubleshooting Common Potency Assay Problems
FAQ: Our potency assay results are inconsistent between replicates. What could be wrong?
- Problem: High variability in immunomodulatory potency readings.
- Solution:
- Standardize Responder Cells: Use cryopreserved PBMCs from a characterized master bank to minimize donor-to-donor variability. Test different mitogens (PHA, CD3/CD28) to find the most robust stimulus for your system [97].
- Optimize Cell Ratios: Titrate the MSC-to-immune cell ratio. For macrophage co-cultures, a specific ratio is needed for near-maximal IL-1RA stimulation [95].
- Control Immune Cell Activation: For macrophage assays, ensure successful M1 polarization by confirming increased expression of surface markers like CD80 and secretion of TNF-α [95].
FAQ: Our late-passage MSCs are failing the osteogenic differentiation potency test. What should we do?
- Problem: Loss of osteogenic potential upon extended expansion.
- Solution:
- Establish a Passage Limit: Data suggests using early-passage cells (e.g., P4) for osteogenesis. Determine the maximum population doubling level (PDL) where your cells retain acceptable osteogenic capacity [96].
- Optimize Expansion Medium: The choice of base medium (DMEM vs. αMEM) can affect the retention of osteogenic potential. Validate which medium better preserves your MSC's differentiation capacity [96].
- Monitor Senescence Markers: Implement checks for senescence-associated markers (e.g., p53, p21, p16) as part of your quality control. Increased expression may predict loss of differentiation potency [99].
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for MSC Potency Assays
| Reagent / Assay Kit | Function in Potency Assessment | Specific Example / Catalog Number |
|---|---|---|
| THP-1 Human Monocytic Cell Line | Source for generating M1 macrophages in co-culture models of innate immunity [95]. | Catalog number 300356 (Cell Lines Service) [95]. |
| Recombinant Human IFN-γ & LPS | Critical stimuli for polarizing macrophages toward a pro-inflammatory M1 phenotype in co-culture [95]. | Imukin (IFN-γ); LPS from E. coli O111:B4 (Sigma-Aldrich) [95]. |
| Human IL-1RA/IL-1F3 Quantikine ELISA Kit | Quantifies secretion of IL-1RA, a key anti-inflammatory mediator, in macrophage co-culture potency assays [95]. | Available from Bio-Techne [95]. |
| Carboxyfluorescein succinimidyl ester (CFSE) | Fluorescent cell dye for tracking and quantifying lymphocyte proliferation in inhibition assays [97]. | Available from BD Biosciences [97]. |
| Phytohemagglutinin-L (PHA-L) | Unspecific mitogen used to robustly activate T-cells in lymphocyte proliferation inhibition assays [97]. | Available from Sigma-Aldrich [97]. |
| CD3/CD28 Activators (TransAct/Dynabeads) | Specific antibody-mediated activators of T-cell receptor and co-stimulatory signaling for proliferation assays [97]. | TransAct (Miltenyi Biotec); Dynabeads (Thermo Fisher) [97]. |
| LymphoPrep Density Gradient Medium | Used for isolation of peripheral blood mononuclear cells (PBMCs) from donor blood [97]. | Available from Alere Technologies [97]. |
| GMP-compliant Cryomedium (CryoStor CS10) | Defined, xeno-free medium for cryopreserving PBMCs or MSC products, ensuring GMP compliance [97]. | Available from Biolife Solutions [97]. |
The relationship between assay selection and the therapeutic target is logical and direct, as shown below:
Performance and Scalability Comparison
The selection between automated bioreactors and traditional flask-based systems significantly impacts the outcomes of extended passage studies on Mesenchymal Stromal Cell (MSC) proliferation capacity under Good Manufacturing Practice (GMP) standards. The table below summarizes the key quantitative and qualitative differences.
| Performance Characteristic | Traditional Flask-Based Culture | Automated Bioreactor Systems |
|---|---|---|
| Typical MSC Expansion Yield | Limited by surface area; time-consuming for clinical-scale doses (hundreds of millions of cells) [41]. | High-yield; systems like Quantum can produce 100–276 × 10⁶ BM-MSCs in a 7-day run [41]. |
| Process Control & Monitoring | Limited to no real-time monitoring. Parameters like pH and pO₂ require external equipment and are not controlled [100]. | Precise, automated control and real-time monitoring of pH, pO₂, temperature, and more [100]. |
| Hands-on Time & Manipulation | High; one study reported ~54,400 manual manipulations for flask-based propagation [41]. | Significantly reduced; the same study noted only ~133 steps with a bioreactor system [41]. |
| Risk of Contamination | Higher risk due to frequent open manipulations (e.g., media changes, harvesting) [101]. | Lower risk through the use of closed-system processing and automated, sterile fluid pathways [41] [101]. |
| Scalability for GMP Production | Limited scalability; expanding to clinical-relevant doses requires numerous flasks and immense incubator space [41]. | Highly scalable; designed for large-scale, reproducible production in a single, integrated system [41] [101]. |
| Process Reproducibility | Lower, due to manual operations and difficulty in maintaining consistent conditions across batches [102]. | High batch-to-batch consistency enabled by automation, predefined protocols, and comprehensive data logging [100]. |
| Cost & Time Efficiency for Clinical Doses | Lower efficiency; higher hands-on time and lower yields per run increase the cost per dose [101]. | Higher efficiency; one study reported a reduction of ~326.6 hours of hands-on time and a significantly lower cost per dose [101]. |
Experimental Protocols for MSC Expansion
Protocol 1: Expansion of MSCs in an Automated Hollow Fiber Bioreactor
This methodology outlines the use of a system like the Quantum Cell Expansion System for the large-scale production of clinical-grade MSCs [41].
- Objective: To achieve high-yield, GMP-compliant expansion of MSCs with minimal manual intervention and maximal reproducibility.
- Materials:
- Quantum Cell Expansion System or equivalent hollow fiber bioreactor.
- GMP-grade MSC source (e.g., Bone Marrow Aspirate).
- GMP-grade culture media (e.g., MSC-Brew GMP medium) supplemented with human Platelet Lysate (hPL).
- Bioreactor set with integrated hollow fiber membrane.
- Coating substrate (e.g., fibronectin, cryoprecipitate).
- Procedure:
- Bioreactor Preparation: Aseptically install the bioreactor set and coat the hollow fibers with the chosen substrate to facilitate cell adhesion [41].
- Cell Seeding: Load a thawed vial of passage 2 MSCs at a high density (e.g., 20 × 10⁶ cells) into the bioreactor [41].
- Expansion Phase: Set the system for continuous or fed-batch media exchange. Culture parameters (dissolved oxygen, pH) can be set to specific setpoints. For example, a hypoxic microenvironment can be established if required by the research protocol [41].
- Harvesting: After approximately 7 days, or when target yields are met, initiate the automated harvest procedure to collect the expanded MSCs [41].
- Key Outcome: This protocol can reliably generate hundreds of millions of MSCs, with studies showing preserved immunomodulatory function and therapeutic efficacy in animal models [41].
Protocol 2: Manual Flask-Based Culture for MSC Expansion
This protocol describes the traditional, multi-flask method for expanding MSCs, often used as a baseline for comparison.
- Objective: To expand MSCs in a 2D, planar culture system for research or small-scale applications.
- Materials:
- Multilayer flasks (e.g., CellSTACK) or T-flasks.
- GMP-grade culture media, typically supplemented with FBS or hPL.
- Enzymatic cell detachment agent (e.g., trypsin-EDTA).
- Centrifuge tubes, serological pipettes, and a biosafety cabinet.
- Procedure:
- Seeding: Aseptically seed the isolated MSCs into flasks at the recommended density.
- Media Changes: Manually change the media every 2-3 days by removing the spent medium from open flasks and adding fresh, pre-warmed medium. This open step is a critical contamination risk point [101].
- Passaging: Once cells reach 70-80% confluence, manually dissociate them using a detachment agent, neutralize the enzyme, and centrifuge the cell suspension. Count the cells and reseed a portion into new flasks for further expansion [103].
- Harvesting: Repeat the passaging steps until the required cell number is achieved for the final harvest.
- Key Outcome: Generates MSCs for basic research but is challenging to scale for clinical doses due to extensive hands-on time, high contamination risk, and phenotypic changes potentially induced by repeated trypsinization and confluency [41].
Frequently Asked Questions (FAQs)
Q1: Our MSC proliferation rate declines significantly after several passages in flasks. How can a bioreactor mitigate this? Proliferation decline can result from replicative senescence, suboptimal culture conditions, or over-confluence during manual passaging. Automated bioreactors maintain a more consistent and optimal environment (e.g., constant nutrient supply, waste removal, controlled pH/pO₂), reducing metabolic stress. Some systems allow perfusion, which mimics a more physiological state and may help maintain proliferation capacity over more passages [41] [100].
Q2: We observe high batch-to-batch variability in our flask-grown MSCs. Can bioreactors improve consistency? Yes. The primary advantage of automated bioreactors is enhanced process control and reproducibility. Every critical parameter is precisely controlled and logged, eliminating the variability introduced by manual media changes, passaging, and environmental fluctuations inherent in flask-based systems. This leads to highly consistent cell products between batches [100] [101].
Q3: For a GMP-compliant process, what are the key advantages of a closed-system bioreactor over multilayer flasks? While multilayer flasks (e.g., CellSTACK) offer more surface area than T-flasks, they still require open manipulations for media changes and harvesting, posing a contamination risk. Closed-system bioreactors automate all fluid handling within a sterile, sealed pathway. This drastically reduces contamination risk, simplifies the environmental control burden (e.g., cleanroom classification), and provides comprehensive data tracing for regulatory filings [41] [101].
Q4: How does oxygen control in a bioreactor differ from a shake flask, and why does it matter for MSCs? In shake flasks, oxygen transfer is passive and limited to the surface of the medium, leading to gradients and insufficient supply at high cell densities. Bioreactors actively control dissolved oxygen via sparging and agitation, ensuring homogenous and optimal levels throughout the culture. Precise oxygen control is crucial for managing MSC metabolism, improving yield, and even directing differentiation or paracrine function, which is not achievable in flasks [102] [100].
Experimental Workflow and Decision-Making
The following diagram illustrates the logical workflow for selecting a culture system and the key steps in an automated bioreactor process, crucial for planning GMP-compliant MSC studies.
The Scientist's Toolkit: Essential Research Reagent Solutions
The table below lists key materials and reagents critical for successful and reproducible MSC expansion in GMP-focused research.
| Reagent / Material | Function in MSC Culture | GMP & Scalability Considerations |
|---|---|---|
| Human Platelet Lysate (hPL) | A xeno-free growth supplement that enhances MSC expansion and proliferation, serving as a replacement for Fetal Bovine Serum (FBS) [41]. | Critical for clinical-grade manufacturing. Sourced from approved human donors to reduce immunogenicity and pathogen risk [41]. |
| GMP-Grade Culture Media | A defined, serum-free or human supplement-based formulation (e.g., MSC-Brew GMP) that supports robust MSC growth [41]. | Ensures product consistency, safety, and compliance. Eliminates variability and safety concerns associated with non-GMP reagents [41]. |
| Cell Detachment Agents | Enzymatic (e.g., trypsin) or non-enzymatic solutions used to dissociate adherent MSCs from the culture surface for passaging and harvesting [103]. | For GMP, use recombinant, animal-origin-free enzymes to minimize contamination risk and ensure traceability [103]. |
| Cryopreservation Medium | A solution containing cryoprotectants (e.g., DMSO) and base medium to preserve MSC viability and functionality during frozen storage [103]. | Use of defined, GMP-grade formulations is essential for creating master cell banks. Controlled-rate freezing is required for viability [41]. |
| Bioreactor Coating Substrate | Proteins (e.g., fibronectin) used to coat the internal surface of hollow fiber or other bioreactors to facilitate MSC adhesion and growth [41]. | Must be sourced as a GMP-grade material. Cryoprecipitate from multiple human donors has been used, but defined, recombinant proteins are ideal [41]. |
Troubleshooting Guide: Common Issues in MSC Stability Studies
Problem 1: Low Post-Thaw Cell Viability Low viability after thawing cryopreserved MSC batches can jeopardize product release and clinical efficacy.
- Potential Cause & Solution: The cryopreservation protocol or formulation may be suboptimal. Solution: Ensure the use of a standardized cryopreservation medium, such as one containing 10% dimethyl sulfoxide (DMSO), and control the freezing rate using a controlled-rate freezer [5].
- Potential Cause & Solution: The cells were stored for an extended period beyond their validated shelf-life. Solution: Conduct real-time stability monitoring to establish the maximum safe storage duration. One GMP-validation study demonstrated that MSCs can maintain >95% viability after 180 days of storage [5].
Problem 2: Slow Proliferation or Reduced Cell Yield After Storage MSCs exhibit longer population doubling times and lower final cell yields after recovery from storage, particularly when derived from certain patient populations.
- Potential Cause & Solution: The source of the MSCs impacts their intrinsic proliferative capacity. Solution: Be aware that MSCs from patients with certain diseases, such as critical limb ischemia (CLI), may have inherently reduced expansion potential. For such cases, increasing the initial bone marrow aspirate volume (e.g., from 30 mL to 100 mL) may not mitigate the poor growth kinetics, making allogeneic sources a more reliable option [85].
- Potential Cause & Solution: The culture medium used for post-thaw expansion is not optimal. Solution: Use a GMP-compliant, animal component-free medium, such as MSC-Brew GMP Medium, which has been shown to enhance proliferation rates and maintain stem cell characteristics compared to standard media [5].
Problem 3: Inconsistent Stability Results Between Batches Data from stability studies show high variability between different production batches, making it difficult to establish a reliable shelf-life.
- Potential Cause & Solution: An insufficient number of batches were used for the stability study. Solution: Follow regulatory guidance, such as ICH Q1A, which recommends including at least three batches of the drug substance or product in stability studies to account for batch-to-batch variability [104].
- Potential Cause & Solution: The testing intervals are too far apart to create a robust stability profile. Solution: For products with a proposed shelf life longer than 12 months, testing frequency should be every three months in the first year, every six months in the second year, and annually thereafter [104].
Frequently Asked Questions (FAQs)
Q1: What is the primary goal of a stability study for MSC-based therapies? The primary goal is to determine the shelf life of the cell therapy product by demonstrating how long it maintains its safety, potency, purity, and identity under specified storage conditions. This involves monitoring critical quality attributes like viability, sterility, immunophenotype, and differentiation capacity over time [5] [104].
Q2: What is the difference between real-time and accelerated stability studies?
- Real-Time Stability Studies monitor the product under its intended storage conditions throughout its proposed shelf life. This provides direct evidence of product stability but is time-consuming [105] [106].
- Accelerated Stability Studies expose the product to elevated stress conditions (e.g., higher temperatures) to rapidly predict degradation and estimate shelf life. These studies are valuable for early product development but are often supplemented with real-time data for regulatory submissions [105] [104].
Q3: How long can MSCs typically be stored and still meet release criteria? With optimized protocols, MSCs can maintain compliance with strict release criteria for extended periods. One GMP-compliant study on infrapatellar fat pad-derived MSCs (FPMSCs) showed that cells maintained >95% viability, sterility, and correct immunophenotype for up to 180 days (approximately 6 months) in storage [5].
Q4: What are the key regulatory guidelines for designing a stability study? Stability studies for cell-based products are governed by several key regulations:
- ICH Q1A (R2): Provides the core guidance on stability testing for new drug substances and products [104].
- Good Manufacturing Practice (GMP): Requires that ATMPs, including MSCs, are manufactured and controlled under strict quality standards, which includes rigorous stability testing [56].
- FDA and EMA Guidance: Both agencies provide specific guidelines for the Chemistry, Manufacturing, and Controls (CMC) of cell therapy products, which encompass stability data requirements [56].
Experimental Protocols for Key Assays
Protocol: Stability Study Sampling and Post-Thaw Viability Assessment
This protocol is critical for establishing the shelf-life of your final MSC product.
Methodology:
- Storage: Store the final cryopreserved MSC product (in its final container and formulation) at the intended long-term storage temperature (e.g., vapor phase of liquid nitrogen, typically below -150°C).
- Sampling Timepoints: Withdraw samples for analysis at predefined intervals. A recommended frequency for a proposed shelf life of over 12 months is [104]:
- Every 3 months during the first year.
- Every 6 months during the second year.
- Annually thereafter.
- Thawing: Rapidly thaw the vial in a 37°C water bath with gentle agitation until only a small ice crystal remains.
- Viability Assessment:
- Dilute the cell suspension in a pre-warmed medium.
- Mix a sample of the cell suspension with Trypan Blue dye at a defined ratio (e.g., 1:1).
- Load the mixture onto a hemacytometer and count the viable (unstained) and non-viable (blue) cells under a microscope.
- Calculate the percentage viability: (Number of viable cells / Total number of cells) × 100%. A viability of >95% has been achieved in GMP studies [5].
- Additional Testing: At each timepoint, also perform sterility testing (e.g., using the Bact/Alert system), endotoxin and mycoplasma assays, and flow cytometry for MSC marker expression to ensure functional integrity is maintained [5].
Protocol: Colony-Forming Unit (CFU) Assay to Assess Clonogenic Potency
The CFU assay measures the proliferative and clonogenic capacity of MSCs, which is a key indicator of their functional potency after storage.
Methodology:
- Cell Seeding: After thawing and viability assessment, seed MSCs at low densities in culture dishes. Typical densities are 20, 50, 100, and 500 cells per dish to determine the optimal seeding density for colony formation [5].
- Culture: Culture the cells for 10-14 days in an animal component-free GMP medium, such as MSC-Brew GMP Medium, which has been shown to support higher colony formation [5]. Replace the medium twice a week.
- Staining: After the culture period, remove the medium and wash the cells with phosphate-buffered saline (PBS).
- Fix the cells with 10% neutral buffered formalin for 30 minutes.
- Wash twice with PBS.
- Stain with 10% Crystal Violet solution for 20-30 minutes to visualize the colonies [5].
- Analysis: Wash off the excess stain and allow the dish to dry. Image the entire dish using a microscope and count the colonies (clusters of >50 cells). A higher number of colonies indicates better-preserved clonogenic potential.
The Scientist's Toolkit: Essential Research Reagents
The table below lists key reagents used in the isolation, expansion, and stability testing of MSCs under GMP-compliant conditions.
| Reagent/Solution | Function in MSC Research | GMP-Compliant Example(s) |
|---|---|---|
| Animal Component-Free Medium | Provides nutrients for cell growth and expansion while eliminating the risk of xenogenic contamination and immunogenicity. | MSC-Brew GMP Medium [5], MesenCult-ACF Plus Medium [5] |
| Human Serum (HS) | An alternative to fetal bovine serum (FBS) that avoids animal-derived components; can enhance the proliferative and immunomodulatory capacity of MSCs [4]. | Pooled allogeneic human serum [4] |
| Cryopreservation Medium | Protects cells from ice-crystal damage during freezing and storage. Typically contains a cryoprotectant like DMSO. | FBS with 10% DMSO [5] |
| Collagenase | An enzyme used to digest tissue (e.g., fat pad, placenta) to isolate MSCs from the extracellular matrix. | Collagenase IV [5] [4] |
| Flow Cytometry Antibody Panel | Used to confirm MSC identity (positive for CD73, CD90, CD105) and purity (negative for CD34, CD45) as per International Society for Cell & Gene Therapy (ISCT) criteria. | BD Stemflow Human MSC Analysis Kit [5] |
Experimental Workflow and Decision Pathway
The following diagram illustrates the logical workflow for designing and conducting a stability study for an MSC-based therapy.
Stability Study Design Workflow
The pathway below outlines a systematic approach for troubleshooting stability failures based on experimental data.
Troubleshooting Stability Failures
This technical support center provides troubleshooting guides and FAQs to help researchers address key challenges in the development and manufacturing of clinical-grade Mesenchymal Stromal Cell (MSC) products, framed within the context of extended passage study and GMP compliance.
Troubleshooting Common MSC Manufacturing Challenges
Issue 1: High Heterogeneity and Inconsistent Product Quality
Problem Description: Significant batch-to-batch variability in MSC characteristics, including proliferation capacity, marker expression, and functional potency, particularly noticeable across extended passages [107] [108].
Root Causes:
- Donor-related Variability: Genetic profile, disease state, lifestyle, and age of the donor can significantly impact both the final product and in-process performance [108].
- Source Tissue Differences: Biopsies from bone marrow, adipose, or umbilical cord tissue contain different fractions and starting numbers of true progenitor cells [108].
- Culture-Induced Variability: Donor-dependent cell responses to culture plastic, media formulations, and bioprocess operating conditions (e.g., seeding density, dissolved oxygen) [108].
Solutions:
- Adopt Robust Process Analytics: Implement a combination of automated production processes and robust process monitoring to gain quantitative process understanding [108].
- Standardize Assays: Develop and use standardized assays that enable comparison or benchmarking across manufacturers, processes, and cell sources [107].
- Control Culture Conditions: Migrate to xeno-free, chemically defined media to reduce batch-dependent variability and eliminate risks associated with animal-derived components like FBS [5] [109] [110].
Issue 2: Failure to Meet Release Specifications Post-Cryopreservation
Problem Description: Reduced cell viability or altered cell functionality after thawing cryopreserved MSC products, jeopardizing product release.
Root Causes:
- Suboptimal cryopreservation media formulation.
- Inconsistent freezing or thawing protocols.
- Extended or improper storage conditions.
Solutions:
- Validate the Entire Process: A GMP validation study demonstrated that FPMSCs (Infrapatellar Fat Pad-derived MSCs) maintained >95% viability and sterility even after 180 days of storage when using a defined protocol [5].
- Use Qualified Reagents: Implement GMP-compliant, animal-component-free cryopreservation media.
- Establish Strict Protocols: Define and adhere to standardized procedures for freezing rates, storage temperature monitoring, and thawing.
Issue 3: Lack of Standardized Potency Assays
Problem Description: Inability to demonstrate product comparability after process changes due to the lack of discriminative and clinically relevant potency assays, a major bottleneck for late-phase clinical trials [108] [109].
Root Causes:
- Over-reliance on the minimal ISCT criteria (plastic adhesion, CD marker expression, in vitro differentiation), which do not fully reflect cell identity or in vivo potency [108].
- No single surface marker exists for unambiguous MSC identification [108].
Solutions:
- Develop Functional Immune Assays: Explore assays that measure immunomodulatory capacity, which is central to the therapeutic mechanism of many MSC products [109].
- Investigate Advanced Analytics: Utilize new technologies like epigenetic analyses to generate signatures that reflect functional properties and therapeutic potential [108].
- Correlate with Clinical Outcomes: Use AI/ML models to correlate multi-omic, imaging, and functional readouts with clinical outcomes to define more predictive release criteria [109].
Frequently Asked Questions (FAQs)
Q1: What are the critical quality attributes (CQAs) that should be monitored during extended passage of MSCs? Beyond the standard ISCT criteria (morphology, CD73+/CD90+/CD105+ expression, CD34-/CD45-/HLA-DR- absence, and trilineage differentiation), CQAs must include functional potency assays. These should be linked to the intended mechanism of action (e.g., immunomodulation via T-cell suppression) [108] [111]. Population doubling time, genomic stability, and secretome analysis (e.g., VEGF secretion) are also critical, as these can change with prolonged culture [110].
Q2: Our MSC proliferation capacity has decreased significantly at later passages. What could be the cause? This is a common challenge in extended passage studies. Key factors to investigate include:
- Culture Medium: The choice of basal medium and supplements greatly impacts long-term health. Research shows that switching from FBS to human serum (HS) or specific GMP-compliant, serum-free media (SFM) like
MSC-Brew GMP Mediumcan enhance proliferation rates and maintain characteristics over passages [5] [111] [110]. - Seeding Density: An optimal seeding density (e.g., 5,000 cells/cm²) is crucial for efficient expansion and can vary with different media formulations [110].
- Donor Senescence: The biological age of the donor and the inherent replicative senescence of MSCs will ultimately limit proliferation capacity.
Q3: How can we ensure our MSC manufacturing process is GMP-compliant? GMP compliance requires a holistic approach [5]:
- Raw Materials: Use xeno-free, chemically defined media and reagents.
- Documentation: Establish and follow strict Standard Operating Procedures (SOPs) for every process step.
- Quality Control: Implement rigorous in-process and release testing, including sterility, mycoplasma, endotoxin, viability, identity, and potency.
- Facility and Equipment: Use qualified equipment and maintain a controlled cleanroom environment.
- Traceability: Ensure full traceability of all materials and the cell product itself.
Experimental Data & Protocols
Table 1: Comparison of Serum-Free Media for BM-MSC Expansion
Data adapted from a study testing six commercially available media for expanding Bone Marrow MSCs, showing variability in performance characteristics [110].
| Media Name | Population Doubling Time | Cell Morphology | Surface Marker Expression (CD73/90/105+) | Immunosuppressive Properties |
|---|---|---|---|---|
| StemMACS MSC XF | Comparable to FBS controls | Spindle-shaped, slender, mat-like | Maintained | Maintained |
| MSC NutriStem XF | Comparable to FBS controls | Spindle-shaped, shorter and thicker | Maintained | Maintained |
| PLTMax hPL | Favorable | Spindle-shaped, elongated, comparable to FBS | Maintained | Maintained |
| RoosterNourish | Favorable | Spindle-shaped, elongated, showed aggregation | Maintained | Maintained |
| StemXVivo SFM | Less favorable at later passages | Highly elongated, aggregation at late passage | Maintained (where testable) | Not determined |
Table 2: Key Process Parameters for GMP-Compliant FPMSC Isolation and Culture
Summary of a validated protocol for deriving MSCs from the infrapatellar fat pad, demonstrating feasibility under GMP standards [5].
| Process Parameter | Specification | Result / Release Criterion |
|---|---|---|
| Tissue Digestion | 0.1% collagenase for 2 hours at 37°C | N/A |
| Culture Medium | MSC-Brew GMP Medium (animal component-free) | Enhanced proliferation & colony formation |
| Seeding Density | 5 x 10³ cells/cm² | N/A |
| Post-Thaw Viability | >70% required | >95% achieved |
| Sterility (Bact/Alert) | No contamination | Sterile |
| Endotoxin/Mycoplasma | Negative | Negative |
| Marker Expression | Positive for CD73, CD90, CD105 | Maintained |
| Stability | Up to 180 days at cryogenic conditions | Viability and sterility maintained |
Objective: To establish a reproducible, GMP-compliant protocol for the isolation, expansion, and cryopreservation of human Infrapatellar Fat Pad-derived MSCs (FPMSCs) for clinical use.
Reagents:
- Infrapatellar fat pad tissue (surgical waste from ACL reconstruction)
- Collagenase solution (0.1% in serum-free media)
- GMP-compliant, animal component-free medium (e.g., MSC-Brew GMP Medium)
- Phosphate-Buffered Saline (PBS)
- Trypsin-EDTA for subculture
- Cryopreservation solution (e.g., with 10% DMSO)
Methodology:
- Tissue Processing: Minced fat pad tissue is digested with 0.1% collagenase for 2 hours at 37°C.
- Cell Isolation: The digested tissue is centrifuged, the supernatant is removed, and the cell pellet is washed with PBS and filtered through a 100μm filter.
- Primary Culture: The pellet is resuspended in the chosen GMP medium and cultured at 37°C, 5% CO₂. The medium is replaced twice weekly.
- Subculture: At 80-90% confluency, cells are harvested using trypsin-EDTA and re-seeded at a density of 5 x 10³ cells/cm² for further expansion.
- Cryopreservation: At the end of the expansion phase, cells are frozen in a controlled-rate freezer in a cryopreservation solution, then transferred to long-term liquid nitrogen storage.
- Quality Control:
- Viability: Assessed post-thaw using Trypan Blue exclusion. >95% viability was consistently achieved.
- Sterility: Tested using the Bact/Alert system.
- Purity/Identity: Confirmed via flow cytometry for MSC markers (CD73, CD90, CD105) and lack of hematopoietic markers.
- Potency: Assessed via Colony Forming Unit (CFU) assays, which showed higher colony formation in the optimized GMP medium.
The Scientist's Toolkit: Key Research Reagent Solutions
| Item / Reagent | Function in MSC Manufacturing | Example & Note |
|---|---|---|
| Chemically Defined, Xeno-Free Media | Provides a consistent, safe, and GMP-compliant environment for cell expansion, eliminating batch variability and risks of animal-derived components. | MSC-Brew GMP Medium [5], StemMACS MSC XF [110]. |
| Human Platelet Lysate (hPL) | A human-derived alternative to FBS, rich in growth factors, used to supplement media to enhance MSC proliferation. | PLTMax hPL supported BM-MSC growth with a morphology comparable to FBS-cultured cells [110]. |
| GMP-Grade Enzymes | For the gentle dissociation of tissues during isolation and for passaging adherent cells without damaging surface proteins. | Collagenase IV [111], Trypsin-EDTA [5]. |
| Flow Cytometry Antibody Panels | Critical for quality control to verify MSC identity (positive for CD73, CD90, CD105) and purity (negative for CD34, CD45, HLA-DR). | BD Stemflow Human MSC Analysis Kit [5]. |
| Functional Potency Assay Kits | To measure the biological activity of the MSC product, which is a key release specification. Examples include immunosuppression and VEGF secretion assays. | T-cell suppression assays are promoted as release criteria in advanced trials [109]. VEGF secretion was used as a potency marker in SFM studies [110]. |
Workflow and Process Diagrams
MSC Characterization Workflow
Troubleshooting Decision Tree
Troubleshooting Guides
Guide 1: Resolving Data Integrity and ALCOA++ violations
Problem: During an internal audit, it was found that electronic records for MSC passage counts lack contemporaneous entries and some entries are inconsistent between different log sheets.
Investigation Steps:
- Verify the Scope: Identify all affected records, including electronic batch records, laboratory notebooks, and environmental monitoring data logs. Check the time-stamps and user metadata for discrepancies [112].
- Assess Against ALCOA++: Systematically check each data point for Attributability, Legibility, Contemporaneity, Originality, Accuracy, Completeness, Consistency, Endurance, and the new requirement of Traceability [113].
- Review System Validation: For electronic systems, ensure the computerized system validation includes testing of audit trails and electronic signature functionality as per the revised Annex 11 [114].
Solution:
- Immediate Action: Quarantine all questionable records. Implement a procedure that mandates real-time data entry, disabling the ability to back-date entries in electronic systems [112].
- Corrective Action: Upgrade the Laboratory Information Management System (LIMS) to include a secure, time-stamped audit trail that logs all user actions. Train all personnel on the updated SOP for data recording, emphasizing the importance of the "C" (Contemporaneous) in ALCOA++ [113].
- Preventive Action: Incorporate a data integrity review as a standard step in the batch record review process. Perform periodic audits of electronic system logs [114].
Guide 2: Addressing Inadequate Raw Data Definition in MSC Proliferation Studies
Problem: A regulatory submission for an MSC-based product receives questions about the definition of "raw data" from a long-term proliferation study, creating uncertainty about what constitutes the original record.
Investigation Steps:
- Define Raw Data: Consult the glossary of the new Chapter 4 draft, which provides a clearer definition of raw data. In the context of an MSC study, this includes original printouts from analytical scales, cell counters, and electronic files from flow cytometers, not just transcribed values [112].
- Map the Data Lifecycle: Document the entire flow of data from generation (e.g., microscope image) to reporting (e.g., final calculation of population doublings in the study report) [113].
- Identify Gaps: Find steps where the "original" data is replaced by a transcribed version without a clear, traceable link.
Solution:
- Immediate Action: Compile a complete data package for the regulator that includes all source data as defined.
- Corrective Action: Establish an SOP titled "Definition and Retention of Raw Data for Preclinical Studies." The SOP must provide a definitive list of what constitutes raw data for each piece of equipment used in the laboratory [112].
- Preventive Action: Configure instruments to automatically save raw data files to a validated, secure server with restricted access to prevent manipulation. Use software that creates "Verified Copies" for routine use, preserving the original data [112].
Guide 3: Managing Hybrid Documentation Systems
Problem: A laboratory uses a hybrid of paper worksheets for experiment observations and an electronic system for final results, leading to confusion about which document is the definitive GMP record.
Investigation Steps:
- Process Mapping: Flowchart all steps where data is recorded or transferred between paper and electronic formats [113].
- Gap Analysis: Check for discrepancies between the paper and electronic records for the same data set. Look for missing signatures or unsigned changes on paper.
- Regulatory Check: Review the draft Chapter 4 guidance, which states that for hybrid systems, it must be defined which signature (paper or electronic) is the relevant one for regulatory purposes [112].
Solution:
- Immediate Action: For current records, clearly annotate in the quality system which version (paper or electronic) is the master GMP record.
- Corrective Action: Redesign the process to be fully electronic where possible. If paper is unavoidable, implement a procedure where the paper sheet is formally scanned and certified as a "True Copy" immediately upon completion, with the original sent to archives [112].
- Preventive Action: When implementing new equipment or systems, prioritize those with fully electronic data capture capabilities that comply with Annex 11 requirements for computerised systems [114].
Frequently Asked Questions (FAQs)
Q1: What is the most significant change in the 2025 EU GMP Chapter 4 revision regarding documentation? The most transformative change is the mandated, risk-based Data Governance System, fully integrated into the Pharmaceutical Quality System. This shifts data management from a procedural task to a strategic function. The principles of ALCOA+ have been expanded to ALCOA++, formally adding "Traceability" as a core requirement for all GMP records [113].
Q2: Our MSC research uses AI-based image analysis for counting cells. How will the new Annex 22 affect us? Annex 22 establishes the first GMP framework for AI/ML. If your AI model is used for a GMP-critical step (e.g., final cell count for batch release), you must define its intended use, establish performance metrics, and demonstrate the suitability of all training data. Crucially, Annex 22 currently excludes adaptive, self-learning models for critical GMP applications, requiring the use of static, validated models with continuous human oversight and accountability [114] [113].
Q3: What are the new requirements for electronic signatures? The revised guidelines require electronic signatures to be legally binding, uniquely traceable to an individual, and permanent for the entire lifetime of the document. The use of hybrid (wet-ink and digital) signatures is discouraged; if used, you must explicitly define which signature is the legally relevant one. Systems must ensure non-repudiation, meaning a user cannot deny having signed the record [112] [114].
Q4: How long must we retain GMP documentation for an advanced therapy medicinal product (ATMP) like an MSC therapy? The new draft of Chapter 4 provides clearer definitions for retention periods. For ATMPs involved in clinical trials, sections 4.76 to 4.79 specify the retention times, also with a specific reference to Advanced Therapy Medicinal Products (ATMPs). You must consult the final guidance, but it mandates retention for the duration of the product's shelf life and a defined period after batch release, which must be justified within your Pharmaceutical Quality System [112].
Q5: What is the best approach for a root cause analysis when a documentation error is found? A disciplined approach using basic tools is most effective. Start with the "Five Whys" to drill down to the underlying cause. Use a fishbone (Ishikawa) diagram to visually map potential causes related to people, processes, equipment, and materials. For errors involving human factors, employ human error reduction techniques to identify the specific error type and implement process changes to prevent recurrence [115] [116].
Table 1: Key Updates in EU GMP Guidelines (2025-2026)
| Document | Title | Status (as of 2025) | Key Focus Areas | Expected Finalization |
|---|---|---|---|---|
| Chapter 4 | Documentation | Draft | Data Governance, ALCOA++, Lifecycle Approach, Hybrid Systems Control [112] [113] | 2026 [112] |
| Annex 11 | Computerised Systems | Draft | AI/ML, Cloud Services, Agile, Advanced Digital Workflows, Audit Trails [114] | 2026 [114] |
| Annex 22 | Artificial Intelligence | Draft | First dedicated framework for static, deterministic AI/ML in manufacturing, excluding generative AI [114] [113] | 2026 [114] |
Table 2: ALCOA++ Principles for Data Integrity
| Principle | Description | Application in MSC Research |
|---|---|---|
| Attributable | Who acquired the data or performed an action? | Electronic signatures on logbooks; user-specific login to instruments. |
| Legible | Can the data be read? | Permanent ink; secure electronic data preventing corruption. |
| Contemporaneous | Recorded at the time of the activity. | Real-time entry into electronic notebooks; date/time stamps on records. |
| Original | The first capture of data. | Raw data file from analyzer, not a transcribed printout. |
| Accurate | No errors edited or removed. | Audit trails to track changes; no use of white-out on paper. |
| Complete | All data including repeats. | All data points preserved, not just those that "fit." |
| Consistent | Chronological and sequential. | Consistent use of units; sequential dating. |
| Enduring | Lasting for the retention period. | Archived on permanent media (e.g., validated server). |
| Traceable++ | New: Able to trace the data's lifecycle and relationships. | Linking raw proliferation data to the final report and batch record. |
Experimental Protocol: Documentation for Extended Passage of MSCs
This protocol outlines the documentation requirements for a study investigating the proliferation capacity of Human Marrow-derived Mesenchymal Stem Cells (hMSCs) through extended passages, ensuring GMP-compliance for regulatory submissions.
1.0 Objective To generate and maintain complete, accurate, and traceable records for the extended passage culture of hMSCs, enabling reliable assessment of proliferation capacity and differentiation potential over time.
2.0 Materials and Reagents
- Cell Source: Human marrow-derived MSCs, Passage 0 [33].
- Culture Vessels: Validated, sterile tissue culture flasks/plates.
- Culture Medium: Pre-released GMP-grade MSC expansion medium, with batch number recorded.
- Dissociation Agent: GMP-grade Trypsin or validated enzymatic alternative.
3.0 Methodology 3.1 Primary Culture and Seeding
- Record the receipt of the primary cell stock, including donor identifier, passage number, and Certificate of Analysis.
- Document the thawing procedure and initial cell count/viability using an automated cell counter, saving the raw data file.
- Record the seeding density (cells/cm²), medium volume, and flask identifier in the culture log.
3.2 Passage Procedure
- Standard Conditions (SC): Subculture preconfluent cultures (e.g., every 7 days) [33]. Record passage number, date, and time.
- Extended First Passage (EFP): Maintain first-passage cultures without subculturing for an extended period (e.g., up to 53 days) [33]. Justify the EFP duration in the study protocol.
- Documentation at Each Passage:
- Pre-harvest: Record microscope image (raw data) of cell morphology and confluence.
- Harvest: Record the dissociation reagent, incubation time, and neutralization method.
- Cell Count and Viability: Perform in duplicate. The original file from the cell counter is raw data and must be retained. Transcribed values must be verified and initialed.
- Calculation: Document the formula used to calculate total yield, population doublings, and seeding density for the next passage.
- Electronic Signature: The scientist must electronically sign the record upon completion.
3.3 Differentiation Assays (at designated endpoints)
- Perform osteogenic, chondrogenic, and adipogenic differentiation assays as per validated methods [33].
- Retain all raw data from these assays (e.g., micrographs of stained pellets, PCR data, flow cytometry files) and link them traceably to the specific culture flask and passage they were derived from.
4.0 Data Management and Records Retention
- All data must be recorded following the ALCOA++ principles.
- The final study report must reference all raw data, allowing a clear audit trail from the conclusion back to the original observations.
- Retain all documentation, including electronic raw data, for the period specified in your PQS, guided by the new Chapter 4 retention clauses (4.76-4.79), especially for clinical trial materials [112].
The Scientist's Toolkit: Essential Research Reagents & Materials
| Item | Function in MSC Proliferation Studies | GMP Consideration |
|---|---|---|
| GMP-grade MSC Medium | Supports cell growth and maintains stemness. | Must be sourced with a full traceability matrix and Certificate of Analysis. |
| Validated Trypsin Solution | Detaches adherent cells for passaging and counting. | The validation package must confirm it is free of adventitious agents. |
| Cell Counting Instrument | Provides accurate cell count and viability data. | Requires installation/operational qualification; raw data file export is essential. |
| Defined Fetal Bovine Serum | Supplements medium to promote growth. | Sourcing must address TSE/BSE compliance; full traceability to origin is required. |
| Differentiation Induction Kits | Induces lineage-specific differentiation for potency assays. | Each kit component must be qualified; use according to a predefined protocol. |
Diagrams and Workflows
Documentation Lifecycle for MSC Data
GMP Documentation Troubleshooting Workflow
Conclusion
Successful clinical translation of MSC therapies requires a holistic approach that integrates optimized proliferation protocols with stringent GMP compliance. The convergence of xeno-free culture media, automated expansion systems, and robust quality control frameworks enables reproducible manufacturing of clinically viable MSCs with enhanced proliferative capacity. Future directions include further refinement of serum-free media formulations, implementation of advanced process analytical technologies for real-time monitoring, and development of potency assays that better predict clinical efficacy. As the field evolves, standardized GMP-compliant manufacturing protocols will be crucial for unlocking the full therapeutic potential of MSCs across diverse clinical applications, from autoimmune diseases to tissue regeneration, ensuring both safety and consistent therapeutic outcomes for patients.
References
- 1. Mesenchymal stromal cell therapy: Progress to date and ... [https://www.sciencedirect.com/science/article/pii/S1525001625000930]
- 2. Donor age and breed determine mesenchymal stromal cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11871689/]
- 3. Understanding the molecular basis of mesenchymal stem ... [https://www.nature.com/articles/s41419-025-08094-x]
- 4. Human serum enhances the proliferative capacity and ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-019-1175-3]
- 5. Optimizing mesenchymal stem cell therapy: from isolation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12054297/]
- 6. Recombinant Proteins for Stem Cell Research [https://www.thermofisher.com/us/en/home/life-science/cell-culture/mammalian-cell-culture/recombinant-proteins/stem-cell.html]
- 7. Optimizing mesenchymal stem cell therapy: from isolation to ... [https://bmcmolcellbiol.biomedcentral.com/articles/10.1186/s12860-025-00539-7]
- 8. Manufacturing & Controls for Advanced Therapy Medical ... [https://www.eurogct.org/research-pathways/research-and-innovation/fundamental-research/manufacturing-phase/manufacturing]
- 9. Immunomodulatory performance of GMP-compliant, clinical ... [https://www.sciencedirect.com/science/article/pii/S2405844024009794]
- 10. Current good manufacturing practice considerations for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9934414/]
- 11. Good Manufacturing Practice for ATMPs [https://www.eurogct.org/research-pathways/manufacturing/good-manufacturing-practice-atmps]
- 12. Support for advanced-therapy developers [https://www.ema.europa.eu/en/human-regulatory-overview/advanced-therapy-medicinal-products-overview/support-advanced-therapy-developers]
- 13. Comparative analysis of mesenchymal stem cells from ... [https://pubmed.ncbi.nlm.nih.gov/16410387/]
- 14. Comparison of human mesenchymal stem cells derived ... [https://www.sciencedirect.com/science/article/abs/pii/S1465324912000394]
- 15. Good manufacturing practice-compliant isolation and culture ... [https://translational-medicine.biomedcentral.com/articles/10.1186/1479-5876-12-56]
- 16. Culturing Human Mesenchymal Stem Cells (MSCs) under ... [https://www.thermofisher.com/us/en/home/references/protocols/cell-culture/stem-cell-protocols/ipsc-protocols/culturing-human-mesenchymal-stem-cells-mscs-under-serum-free-and-xeno-free-conditions.html]
- 17. Considerations When Deriving and Expanding MSCs [https://www.stemcell.com/considerations-when-deriving-and-expanding-primary-mouse-mesenchymal-stem-and-progenitors-cells-mscs.html]
- 18. Isolation and adipogenic differentiation of murine ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11174124/]
- 19. Validated methods for isolation and qualification of ... [https://translational-medicine.biomedcentral.com/articles/10.1186/s12967-025-06972-8]
- 20. Comparative advantages of infrapatellar fat pad: an emerging ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6256334/]
- 21. Infra-patellar fat pad-derived mesenchymal stem cells ... [https://link.springer.com/article/10.1007/s00264-023-05930-7]
- 22. Potential of Using Infrapatellar–Fat–Pad–Derived ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8945217/]
- 23. Mesenchymal stem cells in treating human diseases [https://www.nature.com/articles/s41392-025-02313-9]
- 24. GMP Sterility Tests: Isolator Technology in 2025 - QUALIA [https://qualia-bio.com/blog/gmp-sterility-tests-isolator-technology-in-2025/]
- 25. Healthcare Provider Guide [https://www.isctglobal.org/education/ecgt-healthcare-provider-guide]
- 26. FDA Good Manufacturing Practices (GMP): 2025 Guide [https://pharmuni.com/2025/11/06/fda-good-manufacturing-practices-complete-year-guide-for-compliance-and-quality/]
- 27. Facts About the Current Good Manufacturing Practice ... [https://www.fda.gov/drugs/pharmaceutical-quality-resources/facts-about-current-good-manufacturing-practice-cgmp]
- 28. A Comprehensive Guide About GMP Validation [https://safetyculture.com/topics/gmp/gmp-validation]
- 29. Animal Component Free Media & Reagents Frequently ... [https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/cell-culture-and-cell-culture-analysis/cell-culture-media-preparation/animal-component-free-frequently-asked-questions?srsltid=AfmBOorW5iJIUYl3GrJJnzIfegnJyol8aeEW55YrpIS2jANxq5CS9CkS]
- 30. Supporting the shift to animal component-free and xeno ... [https://www.bio-connect.nl/news/supporting-the-shift-to-animal-component-free-and-xeno-free-research-in-cell-culture/]
- 31. How Regulatory Bodies Are Driving the Shift to Animal- ... [https://invitria.com/resources/regulatory-bodies-shift-animal-origin-free-solutions/]
- 32. Serum-Free and Animal-Free Cell Culture Media [https://www.bio-techne.com/reagents/cell-culture-reagents/serum-free-animal-free]
- 33. The effect of extended first passage culture on ... [https://pubmed.ncbi.nlm.nih.gov/23197807/]
- 34. Animal Component–Free Cell Culture Media [https://www.atcc.org/blogs/2024/animal-component-free-cell-culture-media]
- 35. Improved proliferation and differentiation capacity of ... [https://www.sciencedirect.com/science/article/abs/pii/S1465324910704680]
- 36. Troubleshooting Guide: Ensuring Optimal Performance [https://www.tmmedia.in/troubleshooting-guide-ensuring-optimal-performance/]
- 37. Troubleshooting Common Cell Culture Contamination Issues [https://cellculturecompany.com/troubleshooting-common-cell-culture-contamination-issues/]
- 38. Proliferation kinetics and differentiation potential of ex vivo ... [https://www.sciencedirect.com/science/article/pii/S0301472X00001600]
- 39. Frustrated differentiation of mesenchymal stem cells - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6558097/]
- 40. Troubleshooting Guide for hPSC Culture [https://www.stemcell.com/troubleshooting-tips-for-hpsc-culture.html]
- 41. Automated Manufacturing Processes and Platforms for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11872983/]
- 42. Mesenchymal Stromal Cells Aging of bone marrow– and ... [https://www.sciencedirect.com/science/article/abs/pii/S146532491730525X]
- 43. GMP cell therapy manufacturing | CliniMACS Prodigy® [https://www.miltenyibioindustry.com/en/platforms/clinimacs-prodigy-platform.html]
- 44. Optimize, Automate, and Scale Closed-System MSC ... [https://fujifilmbiosciences.fujifilm.com/us/blog/optimize-automate-and-scale-closed-system-msc-expansion.html]
- 45. The Effect of Extended First Passage Culture on the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3659698/]
- 46. Challenges and advances in clinical applications of ... [https://jhoonline.biomedcentral.com/articles/10.1186/s13045-021-01037-x]
- 47. Optimization of Human T Cell Expansion Protocol [https://www.stemcell.com/optimization-of-human-t-cell-expansion-protocol-effects-of-early-cell-dilution.html]
- 48. Acceleration of Translational Mesenchymal Stromal Cell ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2021.648472/full]
- 49. Mesenchymal Stem Cell-Based Therapies: Challenges ... [https://link.springer.com/article/10.1007/s12013-025-01895-z]
- 50. The Optimization of a Protocol for the Directed ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12189164/]
- 51. Comparison between Platelet Lysate ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10813006/]
- 52. Comparison between Platelet Lysate, ... [https://pubmed.ncbi.nlm.nih.gov/38255245/]
- 53. Human platelet lysate to substitute fetal bovine serum in ... [https://translational-medicine.biomedcentral.com/articles/10.1186/s12967-020-02489-4]
- 54. Evaluation of human platelet lysate versus fetal bovine ... [https://www.sciencedirect.com/science/article/abs/pii/S1465324913007779]
- 55. Human Platelet Lysate Market Size and Forecast, 2025-2032 [https://www.coherentmarketinsights.com/market-insight/human-platelet-lysate-market-5613]
- 56. Optimization of Mesenchymal Stromal Cell (MSC ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2022.918565/full]
- 57. Human Platelet Lysate Market to Grow at 15.19% CAGR ... [https://www.towardshealthcare.com/insights/human-platelet-lysate-market-sizing]
- 58. Cell Culture Growth Curves and Doubling Time with LUNA- ... [https://logosbio.com/essential-metrics-in-cell-culture-understanding-growth-curves-and-doubling-time/]
- 59. How to quantify bacterial cultures - From CFU and OD ... [https://www.eppendorf.com/us-en/lab-academy/life-science/microbiology/how-to-quantify-bacterial-cultures/]
- 60. Typical GMP documentation in a quality control laboratory [https://www.gmpsop.com/typical-gmp-documentation-in-a-quality-control-laboratory/]
- 61. Overcoming the Donor Selection Challenges to Drive ... [https://allcells.com/blog-overcome-gmp-donor-selection-challenges/]
- 62. Addressing Variability in Donor Tissues and Cells - NCBI - NIH [https://www.ncbi.nlm.nih.gov/books/NBK544028/]
- 63. Optimized GMP-grade Wharton's jelly's mesenchymal ... [https://www.sciencedirect.com/science/article/abs/pii/S2452318625000522]
- 64. Validation of High-sensitivity Flow Cytometry for Reliable ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10345173/]
- 65. Mechanisms and functions of cellular senescence - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5873888/]
- 66. Hallmarks of cellular senescence: biology, mechanisms, ... [https://www.nature.com/articles/s12276-025-01480-7]
- 67. Sprouty1 Prevents Cellular Senescence Maintaining ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7662188/]
- 68. Sprouty1 Prevents Cellular Senescence Maintaining ... [https://pubmed.ncbi.nlm.nih.gov/32304210/]
- 69. Environmental Monitoring - Applications [https://www.thermofisher.com/us/en/home/industrial/microbiology/pharmaceutical-microbiology/environmental-monitoring/applications.html]
- 70. Guidance: Container and Closure System Integrity Testing [https://www.fda.gov/regulatory-information/search-fda-guidance-documents/container-and-closure-system-integrity-testing-lieu-sterility-testing-component-stability-protocol]
- 71. Watch out for These 7 Common Autoclave Problems [https://consteril.com/common-autoclave-problems/]
- 72. Incubation Times in Environmental Monitoring [https://www.bioprocessintl.com/qa-qc/a-case-study-in-environmental-monitoring-reviewing-incubation-times-upon-recovery-of-microorganisms]
- 73. Evaluation of GMP-compliant culture media for in vitro ... [https://www.sciencedirect.com/science/article/pii/S0301472X16000618]
- 74. A GMP-compliant manufacturing method for Wharton's jelly ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11069138/]
- 75. Cryopreserved mesenchymal stem cells regain functional ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6716839/]
- 76. Cryopreservation - Key Survey Findings from the ISCT ... [https://www.isctglobal.org/telegrafthub/blogs/ken-ip1/2025/05/28/cryopreservation-key-survey-findings-from-the-isct]
- 77. Reconstitution and post-thaw storage of cryopreserved human ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10154666/]
- 78. Improved Post-Thaw Function and Epigenetic Changes in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5466057/]
- 79. Cryopreserved mesenchymal stem cells regain functional ... [https://translational-medicine.biomedcentral.com/articles/10.1186/s12967-019-2038-5]
- 80. Clinical mesenchymal stromal cell products undergo ... [https://www.sciencedirect.com/science/article/abs/pii/S1465324914007865]
- 81. Impact of Cryopreservation and Freeze-Thawing on ... [https://link.springer.com/article/10.1007/s40778-022-00212-1]
- 82. Thawed Mesenchymal Stem Cell Product Shows ... [https://www.nature.com/articles/s41598-019-54462-x]
- 83. Guide to Cryopreservation (2023) [https://www.accumaxlab.com/guide-to-cryopreservation/]
- 84. The heterogeneity of mesenchymal stem cells [https://pmc.ncbi.nlm.nih.gov/articles/PMC10734083/]
- 85. Marrow changes and reduced proliferative capacity of ... [https://www.sciencedirect.com/science/article/pii/S1465324922007319]
- 86. The issue of heterogeneity of MSC-based advanced ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2024.1400347/full]
- 87. Protocol for Passaging Human Pluripotent Stem Cells Non ... [https://www.stemcell.com/enzyme-free-passaging-of-hpscs-using-relesr.html]
- 88. Protocol for differentiation of human pluripotent stem cells into ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12205342/]
- 89. Current Good Manufacturing Practice (CGMP) Regulations [https://www.fda.gov/drugs/pharmaceutical-quality-resources/current-good-manufacturing-practice-cgmp-regulations]
- 90. FDA Issues New Guidance on Current Good Manufacturing ... [https://www.arnoldporter.com/en/perspectives/advisories/2025/01/fda-guidance-good-manufacturing-practices-for-drugs]
- 91. Pharmaceutical Quality Control Labs (7/93) [https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/inspection-guides/pharmaceutical-quality-control-labs-793]
- 92. 21 CFR Part 610 -- General Biological Products Standards [https://www.ecfr.gov/current/title-21/chapter-I/subchapter-F/part-610]
- 93. 21 CFR 610.12 - Sterility. [https://www.customsmobile.com/regulations/expand/title21_chapterI-i6_part610_subpartB_section610.12]
- 94. Quality Control Testing Requirements of Finished Products [https://www.gmpsop.com/quality-control-testing-requirements-of-finished-products/]
- 95. Potency assay to predict the anti-inflammatory capacity of a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11065629/]
- 96. Changes in phenotype and differentiation potential of human ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-018-0876-3]
- 97. Optimizing an immunomodulatory potency assay for ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2022.1085312/full]
- 98. Application Note: MSC Differentiation with Non-Invasive ... [https://www.sartorius.com/en/products/live-cell-imaging-analysis/live-cell-analysis-resources/msc-differentiation-application-note]
- 99. Mesenchymal stem cells: opening a new chapter in the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12465317/]
- 100. 5 reasons to move from shake flasks to a bioreactor [https://infors-ht.com/en/blog/5-reasons-to-move-from-shake-flasks-to-a-bioreactor]
- 101. Generation of Current Good Manufacturing Practices ... [https://www.mdpi.com/2079-7737/14/3/313]
- 102. Shaker Flasks vs Bioreactors [https://gmpplastic.com/blogs/useful-articles-on-lab-supplies-faq-section/shaker-flasks-vs-bioreactors?srsltid=AfmBOorj2mWHXL7HKdb16EMi4cSvwm4nWj5i0fsusW7-mznhR2dllRnw]
- 103. Cell culture and maintenance protocol [https://www.abcam.com/en-us/technical-resources/protocols/cell-culture-and-maintenance-protocol]
- 104. Statistical Tools for Biologics Stability Testing [https://www.bioprocessintl.com/qa-qc/comprehensive-stability-assessment-of-biotechnological-products-degradation-mechanisms-statistical-tools-and-regulatory-compliance-part-1]
- 105. Shelf Life and Stability Testing [https://www.invisionbiomedical.com/shelf-life-and-stability-testing/]
- 106. Understanding Shelf Life, Stability, and Storage Trials [https://customercare.asurequality.com/hc/en-nz/articles/48950355753753-Understanding-Shelf-Life-Stability-and-Storage-Trials]
- 107. Attitudes Towards Standardization of Mesenchymal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10630078/]
- 108. Bioprocess engineering strategies for autologous human ... [https://insights.bio/immuno-oncology-insights/journal/article/404/bioprocess-engineering-strategies-for-autologous-human-msc-based-therapies-one-size-does-not-fit-all]
- 109. $3.6B to $6.5B: Mesenchymal Stem Cells (MSCs) Market ... [https://www.strategicmarketresearch.com/market-report/mesenchymal-stem-cells-market]
- 110. Expansion and characterization of bone marrow derived ... [https://www.nature.com/articles/s41598-021-83088-1]
- 111. Human serum enhances the proliferative capacity and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6407186/]
- 112. Chapter 4 - new Requirements for GMP Documentation? [https://www.gmp-compliance.org/gmp-news/ema-chapter-4-new-requirements-for-gmp-documentation]
- 113. Analysis of the 2025 EU GMP Chapter 4 Revision [https://www.linkedin.com/pulse/navigating-new-frontier-gmp-analysis-2025-eu-chapter-4-uday-shetty-qtkxf]
- 114. Regulatory Changes I am Watching – July 2025 [https://investigationsquality.com/2025/07/31/regulatory-change-is-here-are-you-ready/]
- 115. Troubleshooting in pharmaceutical manufacturing processes [https://www.hwi-group.de/en/blog/details/troubleshooting-in-pharmaceutical-manufacturing-processes-root-cause-analysis-of-quality-defects]
- 116. Troubleshooting Lab Operations: Be Proactive, Not Reactive [https://www.biopharminternational.com/view/troubleshooting-lab-operations-be-proactive-not-reactive]