PureCap Method: Revolutionizing mRNA Purification for Safer and More Effective Therapeutics
This article details the PureCap method, a groundbreaking technological advancement that enables the purification of completely capped mRNA with near-100% efficiency.
PureCap Method: Revolutionizing mRNA Purification for Safer and More Effective Therapeutics
Abstract
This article details the PureCap method, a groundbreaking technological advancement that enables the purification of completely capped mRNA with near-100% efficiency. Developed to overcome critical limitations in conventional mRNA production, this technique utilizes hydrophobic, photocleavable cap analogs to facilitate the physical separation of desired capped mRNA from immunogenic uncapped byproducts via reversed-phase HPLC. We explore the foundational science behind the method, its step-by-step application, and its significant implications for drug development. Validation data demonstrates that mRNAs purified with PureCap, particularly those with Cap-2 structures, exhibit dramatically enhanced translational efficiency (3-4 fold increases) and reduced immunogenicity, paving the way for more potent and safer mRNA vaccines and therapeutics for researchers, scientists, and drug development professionals.
The Critical Need for Pure Capped mRNA in Therapeutics
The Essential Role of the 5' Cap in mRNA Stability and Translation
The 5' cap is a crucial modification found at the terminus of eukaryotic messenger RNA (mRNA), characterized by a 7-methylguanosine (m7G) residue linked to the initial RNA nucleotide via an unusual 5'-to-5' triphosphate bridge (ppp) [1]. This structure is not merely a molecular adornment but serves as a master regulator of mRNA function, influencing virtually every aspect of the mRNA life cycle. In its most basic form (Cap-0), the guanosine is methylated at the N7 position, while higher-order structures include additional methylations at the 2'-O position of the first (Cap-1) or first and second (Cap-2) transcribed nucleotides, which are particularly effective at reducing immune recognition by pattern recognition receptors [2] [1].
The biological significance of the 5' cap extends across multiple cellular processes. It facilitates nuclear export of mature mRNA through interactions with the cap-binding complex (CBC), protects against 5' exonucleolytic degradation by creating a structural mimic of the 3' end, promotes translation initiation via recruitment of eukaryotic initiation factors, and enhances 5' proximal intron excision during splicing [1]. These functions collectively ensure that mRNA molecules are properly processed, protected during transit, and efficiently translated into functional proteins. For therapeutic applications, achieving complete and homogenous capping is paramount, as uncapped mRNA species not only translate poorly but also trigger undesirable immune responses through receptors such as RIG-I and MDA5 that recognize 5' triphosphate groups [2].
Molecular Mechanisms of Cap-Dependent Regulation
Cap-Dependent Translation Initiation
The 5' cap serves as the primary landing platform for the assembly of the translation initiation machinery. The key initial event is the recognition of the m7G cap by the eukaryotic initiation factor 4E (eIF4E), which then recruits other components including eIF4G and eIF4A to form the eIF4F complex [3] [1]. This complex acts as a bridge between the mRNA and the small ribosomal subunit (40S), facilitating ribosomal scanning along the 5' untranslated region (UTR) until an appropriate start codon is encountered [4]. The efficiency of this process is significantly enhanced by the closed-loop model of translation, where the cap-associated eIF4F complex interacts with poly(A)-binding proteins (PABP) bound to the 3' poly(A) tail, effectively circularizing the mRNA and promoting ribosomal recycling [4] [3].
Structural studies reveal that the binding between eIF4E and the cap involves specific molecular contacts, particularly hydrogen bonding to the N7-methylated guanine, which positions the cap within a conserved pocket on the eIF4E surface [3]. This precise molecular interaction explains why modifications to the cap structure, such as the introduction of bulky photocaging groups at the N2 position of guanosine, can effectively abrogate eIF4E binding and consequently inhibit translation initiation until the modification is removed [3].
Cap-Dependent mRNA Stabilization
The 5' cap provides critical protection against mRNA degradation through multiple mechanisms. First, the unusual 5'-5' triphosphate linkage creates a topological configuration that resists 5'→3' exonucleases by functionally mimicking the 3' end of RNA molecules [1]. Second, the cap-binding proteins (CBC and later eIF4E) physically shield the cap from decapping enzymes, thereby extending mRNA half-life [1].
Recent research has identified YB-1 (p50) as a potent cap-dependent mRNA stabilizer that operates in a complementary pathway to translation initiation [5]. YB-1 demonstrates a concentration-dependent stabilization effect on mRNA, dramatically increasing mRNA half-life in various cell-free systems including rabbit reticulocyte lysate, Krebs-2 ascites, and HeLa cell extracts [5]. The stabilization mechanism involves competition with eIF4E for cap access; YB-1 addition destabilizes the interaction between eIF4E and the cap structure, while sequestration of eIF4E enhances YB-1 association with the mRNA 5' end and significantly prolongs mRNA stability [5]. This suggests the existence of a regulatory switch where conditions that limit translation initiation (such as stress) may activate mRNA stabilization through YB-1, particularly in stored mRNPs where YB-1 is the predominant protein component [5].
Table 1: Cap-Dependent Regulatory Proteins and Their Functions
| Protein Factor | Primary Function | Effect on mRNA Stability | Effect on Translation |
|---|---|---|---|
| eIF4E | Cap-binding translation initiation factor | Moderate stabilization via decapping protection | Strong activation |
| YB-1/p50 | mRNA-associated protein | Potent stabilization via cap protection | Repression (competes with eIF4E) |
| CBC (Cap-Binding Complex) | Nuclear cap recognition | Stabilization during nuclear export | Minimal direct role |
| PABP (Poly(A)-Binding Protein) | Poly(A) tail binding | Synergistic stabilization with cap | Activation (closed-loop formation) |
The PureCap Method: Revolutionizing mRNA Capping for Therapeutic Applications
Limitations of Conventional Capping Approaches
Traditional methods for generating capped mRNA for therapeutic and research applications face significant challenges in achieving complete capping efficiency. Co-transcriptional capping using cap analogs like anti-reverse cap analog (ARCA) typically reaches only 80-90% capping efficiency, leaving a substantial fraction of transcripts uncapped [2]. These uncapped byproducts contain 5' triphosphate groups that are recognized as pathogen-associated molecular patterns by intracellular immune receptors such as RIG-I and MDA5, triggering undesirable innate immune responses and inflammation [2] [6]. Furthermore, uncapped mRNA is translationally incompetent and susceptible to rapid degradation, reducing overall protein expression yield.
Enzymatic capping methods, while generally more efficient, involve multiple enzymatic steps (RNA triphosphatase, guanylyltransferase, and methyltransferase activities) that increase process complexity and cost [4]. Additionally, both conventional approaches struggle with physical separation of capped and uncapped mRNA species due to their nearly identical physicochemical properties, making quality control challenging for therapeutic applications where purity is critical [2].
Principles and Workflow of the PureCap Technology
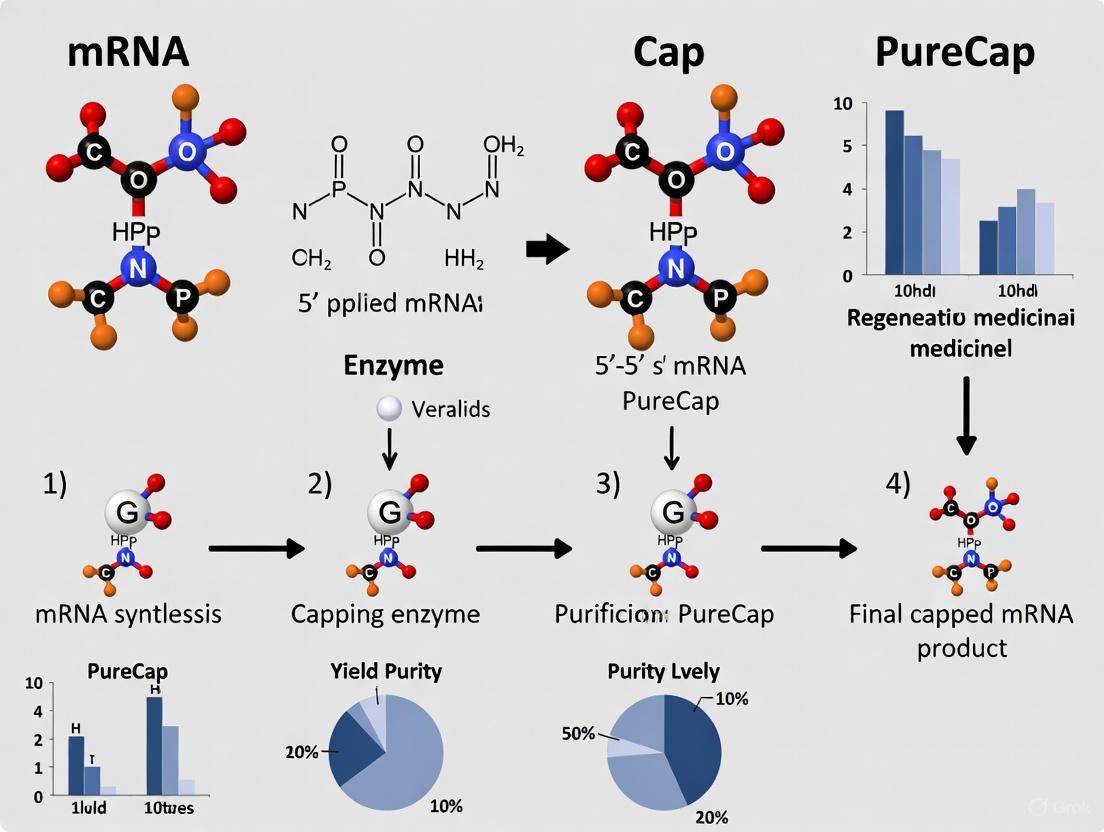
The PureCap method represents a transformative approach that overcomes the limitations of conventional capping strategies through the innovative use of hydrophobic photocaged cap analogs [2] [6]. These custom-designed analogs incorporate a photocleavable 2-nitrobenzyl derivative with a tert-butyl group that confers significant hydrophobicity to the capped mRNA product [2]. This strategic modification enables efficient separation based on hydrophobicity using reversed-phase high-performance liquid chromatography (RP-HPLC), followed by restoration of the native cap structure through gentle photoirradiation.
The PureCap workflow consists of four key stages:
- In vitro transcription incorporating PureCap analogs during initiation
- RP-HPLC purification exploiting hydrophobicity differences to resolve capped mRNA
- Photoirradiation to remove the hydrophobic tag
- Recovery of footprint-free native capped mRNA with near-100% capping efficiency
This method has demonstrated remarkable success with diverse mRNA constructs, including a 4,247-nucleotide coronavirus mRNA, achieving capping purity exceeding 98% without the need for enzymatic post-processing [2] [6]. The technology is versatile, supporting the production of various cap structures including Cap-0, Cap-1, and the more advanced Cap-2 type, which contains 2'-O-methylations on the first two nucleotides [2].
PureCap Method Workflow: From hydrophobic cap analog to pure capped mRNA
Functional Advantages of PureCap-Generated mRNA
The exceptional purity achieved by the PureCap method translates directly to enhanced functional performance of the resulting mRNA therapeutics. Comparative studies have revealed that Cap-2-type mRNA synthesized using PureCap technology demonstrates 3-4 times higher protein expression in both cultured cells and animal models compared to conventional Cap-1-type mRNA prepared by standard capping methods [2] [6]. This dramatic improvement in translational efficiency likely stems from multiple factors, including reduced competition for translation machinery by non-functional uncapped mRNA, enhanced stability, and potentially superior engagement with the translation initiation complex.
Additionally, PureCap-generated mRNAs exhibit significantly reduced immunostimulatory activity due to the absence of immunogenic uncapped byproducts [6]. This attribute is particularly valuable for therapeutic applications where minimizing inflammation at the injection site or systemic immune activation is desirable. The combination of high purity, superior translation capacity, and reduced immunogenicity positions PureCap as a revolutionary advancement in mRNA therapeutic production, with potential applications spanning vaccines, protein replacement therapies, and gene editing approaches [2] [6].
Table 2: Performance Comparison of mRNA Prepared by Different Capping Methods
| Capping Method | Capping Efficiency | Protein Expression | Immunogenicity | Process Complexity |
|---|---|---|---|---|
| Standard Co-transcriptional | 80-90% | Baseline | High (due to uncapped mRNA) | Low |
| Enzymatic Capping | >95% | Moderate improvement | Moderate | High (multiple enzymes) |
| PureCap Method | 98-100% | 3-4x higher (Cap-2) | Low | Moderate (chromatography + irradiation) |
Experimental Protocols for Analyzing Cap Function and mRNA Integrity
Protocol: Assessing mRNA Stability in Cell-Free Systems
Objective: Evaluate the protective function of the 5' cap on mRNA stability using cell-free extracts.
Materials and Reagents:
- Test mRNAs: Capped and uncapped versions of target mRNA (e.g., CAT mRNA, CAT-3'TNFα mRNA with AU-rich elements)
- Cell-free extracts: Rabbit reticulocyte lysate, Krebs-2 ascites extract, or HeLa cell extract
- YB-1 protein: Recombinantly expressed and purified
- Stop solution: Proteinase K with SDS
- Northern blot equipment or quantitative RT-PCR system
Procedure:
- Reaction Setup: Prepare 50μL reactions containing 70% (v/v) cell extract, 10mM creatine phosphate, 50μg/mL creatine phosphokinase, 50μM amino acids, 2mM DTT, and 0.5μg target mRNA.
- Experimental Conditions: Include reactions with:
- Capped mRNA only
- Uncapped mRNA only
- Capped mRNA + YB-1 (0.1-0.6μg)
- Appropriate controls (e.g., other RNA-binding proteins like La autoantigen, PABP, or NCp7)
- Incubation: Conduct at 30°C for time intervals (0, 15, 30, 60, 120 minutes).
- Termination: At each timepoint, remove aliquots and add stop solution. Incubate at 37°C for 30 minutes.
- RNA Extraction: Purify RNA using phenol-chloroform extraction and ethanol precipitation.
- Analysis: Separate RNA by denaturing gel electrophoresis, transfer to membrane, and detect target mRNA by Northern blotting using specific probes. Alternatively, use quantitative RT-PCR.
- Quantification: Determine mRNA half-life from decay curves. Compare stabilization effects across conditions.
Expected Results: Capped mRNA should demonstrate significantly extended half-life compared to uncapped counterparts. YB-1 addition should further stabilize capped mRNA in a concentration-dependent manner, while having minimal effect on uncapped mRNA [5].
Protocol: Functional Validation of Capping Efficiency via Translation Assay
Objective: Quantitatively assess the impact of capping efficiency on protein synthesis.
Materials and Reagents:
- mRNA samples: PureCap-prepared mRNA vs. conventionally capped mRNA
- In vitro translation system: Rabbit reticulocyte lysate or wheat germ extract
- Amino acid mixture: Including radiolabeled ^35^S-methionine or fluorescent non-canonical amino acids
- Cell culture models: As needed for in vivo validation
- Detection reagents: Antibodies for specific antigens, luciferase assay reagents, or other appropriate detection systems
Procedure:
- Sample Preparation: Dilute mRNA samples to equal concentrations (e.g., 100ng/μL) in nuclease-free water.
- Translation Reactions: Set up 25μL reactions containing:
- 50% (v/v) translation extract
- Complete amino acid mixture
- 0.5-1μg mRNA template
- Energy regeneration system
- Incubation: Conduct at 30°C (mammalian systems) or 25°C (wheat germ) for 60-90 minutes.
- Analysis:
- For radiolabeled incorporation: Separate proteins by SDS-PAGE, visualize by autoradiography, and quantify band intensity.
- For luciferase reporters: Measure luminescence at regular intervals.
- For specific antigens: Use Western blot or ELISA for quantification.
- Cell-based Validation: Transfert cultured cells (e.g., HEK293, HeLa) with equal mRNA amounts using appropriate transfection reagents. Harvest cells at 6, 12, 24, and 48 hours post-transfection for protein quantification.
Expected Results: PureCap-prepared mRNA with higher capping efficiency should demonstrate superior protein production compared to conventionally capped mRNA, with Cap-2 structures potentially showing 3-4-fold enhancement over Cap-1 [2] [6].
Research Reagent Solutions for Cap Studies
Table 3: Essential Research Tools for mRNA Cap Studies
| Reagent/Tool | Function | Application Examples | Key Features |
|---|---|---|---|
| PureCap Analogs | Co-transcriptional capping with purification handle | Production of high-purity capped mRNA; Cap-2 mRNA synthesis | Hydrophobic tag enables RP-HPLC separation; Photocleavable |
| FlashCaps (Photocaged Cap Analogs) | Optical control of mRNA translation | Spatiotemporal control of protein expression; mRNA functional studies | N2-position photocaging groups (DMNB, NPM); Carbamate linkage |
| YB-1/p50 Protein | mRNA-associated protein for stability studies | Investigation of cap-dependent mRNA stabilization; Translation repression studies | Cold shock domain mediates stabilization; Competes with eIF4E |
| Vaccinia Capping System | Enzymatic capping post-transcription | Conventional capped mRNA production; Cap-0 to Cap-1 conversion | Multi-enzyme system; Includes 2'-O-methyltransferase |
| Anti-Reverse Cap Analog (ARCA) | Standard co-transcriptional capping | Baseline capped mRNA production; Comparison studies | Prevents reverse incorporation; Standard Cap-0 structure |
| Cap-Specific Antibodies | Immunological detection of cap structures | Capping efficiency assessment; mRNA purification validation | Specific recognition of m7G cap; Not sequence-dependent |
The 5' cap represents a cornerstone of mRNA biology, serving as a multi-functional module that governs mRNA stability, translation efficiency, and immune recognition. The development of innovative technologies like the PureCap method addresses long-standing challenges in therapeutic mRNA production by enabling the generation of homogenously capped mRNA populations with near-perfect efficiency. This advancement, coupled with growing understanding of cap-binding proteins like YB-1 and their regulatory roles, provides powerful tools for optimizing mRNA-based therapeutics.
Future directions in cap biology and technology will likely focus on further refining cap structures for enhanced functionality, developing increasingly precise methods for temporal and spatial control of translation, and engineering cap analogs with customized properties for specific therapeutic applications. As these technologies mature, they will undoubtedly accelerate the transition of mRNA therapeutics from primarily vaccination applications to broader uses in protein replacement, gene editing, and regenerative medicine, ultimately fulfilling the promise of mRNA as a versatile and powerful therapeutic modality.
Cap-Dependent Functional Outcomes: Dual pathways regulating translation and stability
In mRNA therapeutics, the 5' cap structure is not merely a molecular accessory but a fundamental determinant of therapeutic efficacy and safety. This modified guanine nucleotide, linked to the mRNA's 5' end via a 5'-5' triphosphate bridge, serves critical functions: protecting the transcript from exonuclease degradation, facilitating nuclear export, and enabling efficient translation initiation through recruitment to the ribosome [7] [2]. More critically, the cap structure acts as a molecular signature distinguishing self from non-self RNA, thereby playing a pivotal role in modulating the innate immune response [7] [2].
The maturation of mRNA vaccines during the COVID-19 pandemic highlighted the limitations of conventional capping methods. These methods persistently generate significant amounts of uncapped mRNA impurities that escape detection in standard analytical assessments but exert profound biological consequences in therapeutic applications [2] [8]. This application note examines the limitations of conventional capping technologies, details how uncapped mRNA drives undesirable immune activation, and presents the PureCap method as a robust solution for producing highly pure, fully capped mRNA with enhanced translational performance and reduced immunogenicity.
Limitations of Conventional mRNA Capping Methods
Current methods for producing capped mRNA in vitro fall into two primary categories: co-transcriptional capping and enzymatic capping. Both approaches suffer from inherent inefficiencies that result in the persistent presence of uncapped and improperly capped mRNA species.
Co-transcriptional Capping
Co-transcriptional capping employs synthetic cap analogs (e.g., CleanCap AG, Anti-Reverse Cap Analogs - ARCAs) added to the in vitro transcription (IVT) reaction. While simpler, this method faces fundamental limitations:
- Competition with Initiating Nucleotides: During transcription initiation, cap analogs compete with the natural initiating nucleotide, GTP. This competition inherently limits capping efficiency to approximately 80-90% even under optimized conditions [2] [9].
- Reverse Capping: First-generation cap analogs (e.g., m7G(5′)ppp(5′)G) were incorporated in both forward and reverse orientations, with 30-50% of mRNA containing reverse caps that are translationally incompetent [2]. While Anti-Reverse Cap Analogs (ARCAs) addressed this issue by incorporating a 3′-O-methyl group on m7G to prevent incorportation in the reverse orientation, the problem of incomplete capping efficiency persists [7] [2].
- Structural Limitations: Standard cap analogs produce Cap-0 structures (m7GpppN...) that retain higher immunogenicity than Cap-1 (m7GpppN2'-O-methyl...) and Cap-2 structures, which more closely resemble mature eukaryotic mRNA [7] [2].
Enzymatic Capping
Enzymatic capping utilizes capping enzymes (e.g., Vaccinia Virus Capping Enzyme - VCE) to modify the 5' end of transcribed mRNA in a multi-step process:
- Theoretical Efficiency: This method can achieve near-100% capping efficiency in theory as it acts on all transcribed mRNA molecules [9].
- Practical Challenges: Enzymatic capping introduces additional steps (capping reaction, enzymatic purification), increasing process complexity, cost, and the risk of mRNA degradation during handling [2] [9]. Batch-to-batch variability in enzyme activity can also affect final capping efficiency [7].
Table 1: Comparison of Conventional mRNA Capping Methods
| Capping Method | Theoretical Efficiency | Practical Efficiency | Key Advantages | Key Limitations |
|---|---|---|---|---|
| Co-transcriptional (ARCA) | ~90% | 80-90% | Simple one-step process; No additional enzymes needed | Reverse capping issue; GTP competition limits efficiency |
| Co-transcriptional (CleanCap) | ~94% | 90-95% | Higher efficiency; Direct Cap-1 formation; Reduced immunogenicity | Still generates 5-10% uncapped mRNA; Specialized analogs required |
| Enzymatic (VCE) | ~100% | 90-98% (varies) | Potentially higher efficiency; Directionally correct capping | Multi-step process; Enzyme cost; Batch variability |
| Enzymatic (FCE) | ~100% | >95% | Higher activity than VCE; Broader temperature tolerance; Cost-effective at scale | Relatively new technology; Less established in manufacturing |
Uncapped mRNA and Immune System Activation
The presence of uncapped mRNA impurities—even at low levels—triggers potent innate immune responses that significantly compromise therapeutic efficacy and safety.
Molecular Recognition of Uncapped mRNA
Uncapped mRNA molecules terminate in a 5' triphosphate (5' ppp), a molecular pattern associated with viral RNA that is recognized as pathogen-associated molecular pattern (PAMP) by specific innate immune sensors [2]:
- RIG-I Recognition: Retinoic acid-inducible gene I (RIG-I) specifically detects 5' triphosphate RNA in the cytoplasm [2]. Upon binding, RIG-I undergoes conformational changes that trigger a signaling cascade culminating in type I interferon (IFN-α/β) production [8].
- MDA5 Activation: Melanoma differentiation-associated protein 5 (MDA5) can also sense certain uncapped RNA structures, contributing to interferon responses [2].
Consequences of Immune Activation
The type I interferon response initiated by uncapped mRNA impurities has several detrimental effects on mRNA therapeutic performance:
- Inhibition of Protein Translation: IFN-α/β signaling activates protein kinase R (PKR) and other antiviral effectors that globally suppress cellular protein synthesis, directly counteracting the translational intent of therapeutic mRNA [7] [8].
- Reduced Antigen Expression: The translational shutdown markedly decreases the production and expression of the encoded antigen, diminishing the vaccine's immunogenicity [8].
- Impaired Humoral Immunity: Recent studies demonstrate that type I interferon responses directly inhibit the development of robust antibody responses. PureCap mRNA generated 26-fold higher antibody titers compared to ARCA-capped mRNA in mouse models, and antibody blockade of IFN-α/β receptor restored responses to ARCA-capped mRNA to PureCap levels [8].
- Increased Reactogenicity: Unwanted immune activation contributes to inflammatory side effects, including local injection site reactions and systemic symptoms such as fever and fatigue [10] [11].
Diagram Title: Uncapped mRNA Immune Activation Pathway
The PureCap Method: A Solution for Complete mRNA Capping
The PureCap technology represents a paradigm shift in mRNA capping by integrating a purification-based approach that ensures 100% capping efficiency while simultaneously removing other immunostimulatory impurities.
The PureCap method employs specially designed cap analogs modified with a hydrophobic, photocleavable tag (2-nitrobenzyl derivative with tert-butyl group) [2]:
- Hydrophobic Tagging: The PureCap analog incorporates during co-transcriptional capping, imparting distinct hydrophobicity to successfully capped mRNA molecules [2].
- Chromatographic Separation: The hydrophobicity difference enables efficient separation of capped from uncapped mRNA using reversed-phase high-performance liquid chromatography (RP-HPLC), a standard, scalable purification technique [2].
- Photocleavage: Brief UV irradiation cleaves the hydrophobic tag after purification, yielding native, footprint-free capped mRNA without additional enzymatic treatments [2].
Key Advantages of PureCap mRNA
- 100% Capping Efficiency: Complete elimination of uncapped mRNA impurities and their associated immunostimulatory effects [2] [8].
- Simultaneous dsRNA Removal: The RP-HPLC purification also effectively removes double-stranded RNA (dsRNA) contaminants, another potent activator of innate immunity [2] [8].
- Versatile Cap Structure Incorporation: The technology supports various cap structures (Cap-0, Cap-1, Cap-2) with equal efficiency, enabling investigation of structure-activity relationships [2].
- Superior Translational Performance: PureCap-generated Cap-2 mRNA demonstrates 3- to 4-fold higher protein expression in cultured cells and animal models compared to standard Cap-1 mRNA [2].
- Enhanced Therapeutic Efficacy: In vaccination models, PureCap mRNA elicited 26-fold higher antibody titers and provided superior protection against Pseudomonas aeruginosa challenge compared to ARCA-capped mRNA [8].
Diagram Title: PureCap Method Workflow
Experimental Protocols
Protocol: PureCap mRNA Production and Purification
Materials:
- PureCap analog (e.g., DiPure, DiPure/2'OMe, DiPure/3'OMe)
- T7 RNA Polymerase
- RNase-free water and reagents
- RP-HPLC system with C18 column
- UV light source (365 nm)
Procedure:
In Vitro Transcription Setup
- Assemble IVT reaction containing:
- 1 µg DNA template
- 5 µL 5× Transcription Buffer (400 mM Tris-HCl pH 8.0, 40 mM MgCl₂, 50 mM NaCl, 10 mM spermidine)
- 4 µL 10 mM rNTPs mix
- 2.5 µL 100 mM DTT
- 2 µL T7 RNA Polymerase
- 2 µL 10 mM PureCap analog
- RNase-free water to 25 µL final volume
- Incubate at 37°C for 2-4 hours depending on transcript length [2]
- Assemble IVT reaction containing:
RP-HPLC Purification
- Dilute transcription reaction with equal volume of 0.1 M triethylammonium acetate (TEAA) buffer, pH 7.0
- Inject onto C18 RP-HPLC column equilibrated with 0.1 M TEAA
- Apply linear gradient of 5-50% acetonitrile over 30 minutes
- Monitor absorbance at 260 nm
- Collect hydrophobic peak corresponding to capped mRNA [2]
Photocleavage
- Place collected fractions in quartz cuvette
- Irradiate with 365 nm UV light for 15-30 minutes
- Confirm tag removal by analytical RP-HPLC [2]
Concentration and Storage
- Desalt using ethanol precipitation or size exclusion chromatography
- Resuspend in RNase-free water or storage buffer
- Quantify by UV spectrophotometry
- Store at -80°C until use
Protocol: Assessing mRNA Purity and Immunogenicity
Methods for Detection of Uncapped mRNA:
LC-MS/MS Cap Analysis
- Hybridize mRNA with DNA probe complementary to 5' region
- Digest with RNase 4 (NEB #M1284) to generate 5' fragment
- Analyze by LC-MS/MS to verify cap structure and detect uncapped species [12]
RIG-I Binding Assay
- Immobilize recombinant RIG-I protein on biosensor chips
- Flow mRNA samples over chip surface
- Measure binding response; uncapped mRNA shows significantly higher RIG-I binding [2]
Type I Interferon Reporter Assay
- Transfert HEK-293 cells with interferon-stimulated response element (ISRE) luciferase reporter
- Treat with mRNA preparations (100 ng/well)
- Measure luciferase activity after 24 hours
- PureCap mRNA should show >10-fold reduction in IFN activation vs. standard capped mRNA [8]
Table 2: Quantitative Comparison of mRNA Performance Metrics
| Performance Metric | ARCA-Capped mRNA | Enzymatically Capped mRNA | PureCap mRNA |
|---|---|---|---|
| Capping Efficiency | 80-90% | 90-98% | 100% |
| Protein Expression | 1.0× (Reference) | 1.5-2.0× | 3-4× higher [2] |
| IFN-α Production | High | Moderate | Undetectable [8] |
| Antibody Titer | 1.0× (Reference) | 1.5-2.0× | 26× higher [8] |
| dsRNA Contamination | Present | Reduced | Eliminated [2] [8] |
| Therapeutic Protection | Partial (40% survival) | Improved (60% survival) | Robust (100% survival) [8] |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for mRNA Capping Research
| Reagent/Kit | Supplier | Function/Application | Key Features |
|---|---|---|---|
| PureCap Analogs | Crafton Biotechnology | Co-transcriptional capping with purification handle | Hydrophobic photocleavable tag; Enables RP-HPLC purification |
| Faustovirus Capping Enzyme (FCE) | New England Biolabs (M2081) | Enzymatic capping | Higher activity than VCE; Broad temperature tolerance; Cost-effective scaling [12] |
| Vaccinia Capping Enzyme (VCE) | New England Biolabs (M2080) | Enzymatic capping | Established standard; Compatible with 2'-O-methyltransferase [12] |
| HiScribe T7 mRNA Kit with CleanCap | New England Biolabs (E2080) | Co-transcriptional capping | Single-reaction Cap-1 capping; ~94% efficiency; Simplified workflow [12] |
| RNase 4 | New England Biolabs (M1284) | mRNA cap analysis | Simplified LC-MS sample prep; Tolerates nucleotide modifications [12] |
| mRNA Cap 2'-O-methyltransferase | New England Biolabs (M0366) | Cap-1 formation | Converts Cap-0 to Cap-1; Reduces immunogenicity [12] |
The presence of uncapped mRNA in therapeutic preparations represents a critical, often underestimated source of immunogenicity that substantially compromises efficacy and safety. Conventional capping methods, with their inherent efficiency limitations, consistently generate these immunostimulatory impurities that activate RIG-I and trigger type I interferon responses, ultimately inhibiting protein translation and impairing protective immunity.
The PureCap method addresses this fundamental limitation through a novel purification-based approach that ensures 100% capping efficiency while simultaneously removing other potent immune activators like dsRNA. The resulting mRNA demonstrates markedly enhanced translational capacity, reduced reactogenicity, and superior therapeutic performance, as evidenced by 26-fold improvements in antibody titers and complete protection in challenge models where conventional mRNA fails.
For researchers and drug developers, adopting stringent capping quality control and considering next-generation capping technologies like PureCap is essential for advancing mRNA therapeutics beyond current limitations, particularly for applications requiring high potency, repeat dosing, or delivery to immune-sensitive tissues.
The 5′ cap is a fundamental modification found at the beginning of eukaryotic messenger RNA (mRNA) molecules, playing an indispensable role in mRNA stability, translation efficiency, and immune recognition. This critical structure consists of a methylated guanine nucleotide attached to the 5′ end of the mRNA via an unusual 5′-5′ triphosphate bridge [2] [13] [14]. Since its initial discovery in the 1970s, understanding of the cap structure has evolved significantly, revealing multiple cap variants with distinct biological properties [13] [14]. The cap structure protects mRNA from exonucleolytic degradation and serves as a recognition signal for the translation initiation machinery, thereby enhancing the protein synthesis capability of the mRNA [14]. In therapeutic applications, particularly mRNA vaccines, the cap structure plays an additional crucial role in minimizing unintended immune activation by distinguishing self-RNA from non-self RNA [2] [15].
Cap structures are categorized based on their methylation patterns into Cap-0, Cap-1, and Cap-2 configurations. While Cap-0 and Cap-1 have been extensively characterized and utilized in biomedical applications, Cap-2 has remained largely elusive due to challenges in its production and purification [2] [16]. Recent methodological advances, particularly the development of the PureCap purification method, have now enabled researchers to produce Cap-2 mRNA with high purity, facilitating proper evaluation of its functional properties for the first time [2] [17] [18]. This technological breakthrough has revealed that Cap-2 mRNA exhibits superior translational efficiency and reduced immunostimulatory activity compared to its Cap-1 counterpart, opening new possibilities for therapeutic mRNA optimization [2] [16] [19].
Biochemical Definition of Cap Structures
Structural Characteristics and Methylation Patterns
The foundational structure common to all eukaryotic mRNA caps is N7-methylguanosine (m7G) linked to the first nucleotide of the mRNA via a 5′-5′ triphosphate bridge (m7GpppN, where N represents any nucleotide) [2] [13]. This basic structure, known as Cap-0, is characterized by a single methyl group exclusively at the N7 position of the terminal guanine [13]. Cap-0 is predominantly found in lower eukaryotes and serves as the substrate for additional methylation events that produce more complex cap structures [13] [14].
The Cap-1 structure represents an evolutionary advancement in cap complexity, featuring an additional methyl group at the 2′-O position of the ribose of the first transcribed nucleotide (m7GpppN2′-OMe) [13]. This structure is common in higher eukaryotes, including mammals, and provides enhanced protection from immune recognition compared to Cap-0 [2] [13]. The 2′-O-methylation of the first nucleotide prevents recognition by innate immune sensors such as RIG-I and MDA5, which would otherwise trigger antiviral immune responses [2].
The most complex cap structure, Cap-2, contains methyl groups at the 2′-O positions of both the first and second nucleotides following the m7G cap (m7GpppN2′-OMepN2′-OMe) [2] [13] [14]. While Cap-2 is present in some viral and higher eukaryotic RNAs, its precise functions remained poorly characterized until recently due to difficulties in obtaining pure Cap-2 mRNA for study [2] [16]. In mammals, an additional modification occurs when the first transcribed base is adenosine; in this case, the N6 position is frequently methylated to form m6A, which further increases resistance to mRNA-decapping enzymes [2].
Table 1: Comparative Structural Features of mRNA 5′ Cap Types
| Cap Type | M7G Methylation | First Nucleotide 2′-O-Methylation | Second Nucleotide 2′-O-Methylation | Predominant Occurrence |
|---|---|---|---|---|
| Cap-0 | Yes | No | No | Lower eukaryotes, some viruses |
| Cap-1 | Yes | Yes | No | Higher eukaryotes, mammals |
| Cap-2 | Yes | Yes | Yes | Some viral and higher eukaryotic RNAs |
Functional Significance in mRNA Biology
The 5′ cap structure serves multiple essential functions throughout the mRNA life cycle. During translation initiation, the cap structure is recognized by eukaryotic initiation factor 4E (eIF4E), which recruits additional translation factors and the ribosomal subunits to the 5′ end of the mRNA [14]. This cap-dependent translation mechanism significantly enhances the efficiency of protein synthesis. Additionally, the cap structure protects mRNA from degradation by 5′-3′ exonucleases, thereby extending mRNA half-life [14]. The interaction between the 5′ cap and the poly-A tail at the 3′ end of mRNA further enhances translation activity and stability through a circularization mechanism facilitated by protein complexes [2].
Beyond these fundamental roles, cap structures participate in pre-mRNA splicing, mRNA export from the nucleus, and nuclear transport [2] [14]. The methylation status of the cap structure serves as a molecular signature that enables the immune system to distinguish between self and non-self RNA. Innate immune receptors such as retinoic acid-inducible gene-I (RIG-I) recognize uncapped 5′ triphosphate RNA as viral in origin, triggering interferon responses [2]. The Cap-1 structure, commonly found in mammalian mRNA, evades this recognition, while Cap-2 provides even greater reduction in immunostimulatory activity [2] [18]. Recent research has demonstrated that Cap-2 structure drastically reduces mRNA affinity to RIG-I compared to Cap-1 structure, while simultaneously increasing mRNA stability and translation activity [2].
Conventional mRNA Capping Methods and Limitations
Enzymatic Capping Approaches
Enzymatic capping represents one of the two primary methods for adding cap structures to in vitro transcribed (IVT) mRNA. This approach utilizes enzymes derived from the vaccinia virus capping system, which includes RNA triphosphatase, RNA guanylyltransferase, and RNA guanine-N7-methyltransferase activities [14]. These enzymes work sequentially to first remove the γ-phosphate from the 5′ triphosphate of the nascent RNA, then add GMP from GTP to form the GpppN structure, and finally methylate the N7 position of guanine to generate the Cap-0 structure [14]. To produce Cap-1 structures, an additional 2′-O-methyltransferase enzyme (such as VP39) must be employed [14].
The vaccinia capping enzyme method provides high capping efficiency but involves multiple enzymatic steps and requires two purification procedures [13]. While this method can be scaled up in a cost-efficient manner, the requirement for multiple enzymes increases production complexity and expense [14]. Additionally, the yields of capped RNA depend entirely on the amount of available uncapped RNA target, and the process can be time-consuming compared to co-transcriptional capping methods [14].
Co-transcriptional Capping with Cap Analogs
Co-transcriptional capping involves incorporating cap analogs directly during the in vitro transcription reaction, simplifying the production process by combining transcription and capping into a single step [13]. The first-generation cap analog, m7G(5′)ppp(5′)G (mCap), suffers from a significant limitation: its symmetrical nature results in incorporation in both correct and reverse orientations, with approximately 50% of capped mRNAs becoming untranslatable due to reverse incorporation [2] [13].
The development of Anti-Reverse Cap Analog (ARCA) addressed this issue through methylation of the 3′-hydroxyl group of m7G, preventing reverse incorporation and ensuring all capped mRNAs remain translatable [2] [13]. Despite this improvement, ARCA-based capping still requires high cap analog to GTP ratios (typically 4:1), which reduces overall transcription yield and provides capping efficiencies of only 80-90% [2] [13]. This limitation means that 10-20% of mRNAs remain uncapped, retaining 5′ triphosphates that can trigger undesirable immune responses [2].
More recently, novel cap analogs such as CleanCap have been developed, utilizing trinucleotide and tetranucleotide analogs that enable direct co-transcriptional synthesis of Cap-1 structures with efficiencies exceeding 95% [13]. However, even these advanced systems cannot achieve 100% capping efficiency, leaving uncapped immunostimulatory impurities that must be removed through additional purification steps [2].
Table 2: Performance Comparison of Conventional Capping Methods
| Capping Method | Cap Structure | Maximum Efficiency | Key Advantages | Key Limitations |
|---|---|---|---|---|
| Enzymatic Capping | Cap-0 or Cap-1 | >95% | High fidelity, natural cap structure | Multiple steps, high cost, complex purification |
| mCap Analog | Cap-0 | ~70% | Simple one-step process | ~50% reverse orientation, low yield |
| ARCA Analog | Cap-0 | 80-90% | Correct orientation only | High cap:GTP ratio reduces yield |
| CleanCap Analog | Cap-1 | >95% | High efficiency, correct orientation | Requires specific initiation sequence |
The Challenge of Uncapped mRNA Impurities
A fundamental limitation shared by all conventional capping methods is their inability to achieve 100% capping efficiency [2]. The remaining uncapped mRNA molecules possess 5′ triphosphate groups (5′ ppp-RNA), a molecular pattern associated with viral RNA that is recognized by innate immune receptors including RIG-I and MDA5 [2]. Even small amounts of uncapped mRNA impurities can trigger substantial immune responses, potentially leading to increased reactogenicity and inflammation at the injection site for therapeutic applications [2] [17].
The physicochemical similarity between capped and uncapped mRNA poses significant challenges for their physical separation [2]. Traditional purification approaches have relied on enzymatic treatment to remove uncapped impurities, using RNA 5′ polyphosphatase to convert 5′ triphosphates to monophosphates followed by degradation with XRN I, a 5′→3′ exoribonuclease that specifically targets monophosphorylated RNA [2]. However, these enzymatic treatments increase production complexity and cost while potentially leading to non-specific mRNA degradation [2]. The need for multiple purification processes represents a significant hurdle for commercial-scale production of therapeutic mRNA, necessitating the development of more efficient separation methods [2].
The PureCap Method: Revolutionizing mRNA Cap Purification
Principle and Design of PureCap Analogs
The PureCap method, developed by researchers at Nagoya University, represents a revolutionary approach to mRNA capping that enables complete separation of capped mRNA from uncapped impurities through a physical purification process [2] [16]. This innovative technology centers on specially designed cap analogs, termed PureCap analogs, which incorporate a hydrophobic photocleavable tag at various positions within the cap structure [2]. The hydrophobic tag consists of a 2-nitrobenzyl (Nb) derivative with a tert-butyl group that enhances both hydrophobicity and chemical stability [2].
Four distinct dinucleotide PureCap analogs were designed with the hydrophobic tag positioned at different locations: DiPure (1) with the Nb group linked via an acetal group at the 2′ position of m7G; DiPure/2′OMe (2) and DiPure/3′OMe (3) with O-methyl and O-Nb modifications at the 2′/3′ positions of m7G; and DiPure/N2 (4) with the Nb tag attached to the exocyclic amino group of m7G [2]. These strategic modifications ensure that the analogs maintain anti-reverse cap activity while introducing sufficient hydrophobicity to enable separation based on this physicochemical property [2].
A key innovation of the PureCap system is the use of a photodegradable 2-nitrobenzyl derivative as the hydrophobic tag, which allows for subsequent removal under mild conditions via light irradiation after the purification step [2]. This photocleavage process recovers the native capped mRNA without any residual modifications, making the PureCap method suitable for producing therapeutic-grade mRNA [2].
Synthetic Strategy for PureCap Analogs
The synthesis of PureCap analogs presented significant challenges due to the introduction of lipophilic moieties that decrease solubility in aqueous solvents used in traditional purification methods [2]. To address this issue, researchers developed a streamlined one-pot synthesis approach that enables direct production of diphosphates from nucleoside precursors, avoiding the solubility problems associated with multiple aqueous purification steps [2].
Two primary synthetic routes were employed: the first approach uses phosphoryl chloride to generate a phosphorodichloridate intermediate, which is then reacted with an alkylammonium phosphate salt to produce the diphosphate [2]. The second method involves direct conversion of 5′-tosylated guanosine to the diphosphate through reaction with tetrabutylammonium pyrophosphate salt [2]. The resulting diphosphate is subsequently methylated at the N7 position and condensed with guanosine monophosphate imidazolide in the presence of zinc chloride to yield the final dinucleotide PureCap analog [2].
This optimized synthetic protocol simplifies and shortens the production process for PureCap analogs while overcoming the solubility limitations imposed by the hydrophobic tags, making large-scale synthesis feasible [2].
Diagram Title: PureCap mRNA Purification Workflow
Experimental Protocol for PureCap mRNA Production
Step 1: In Vitro Transcription with PureCap Analogs
- Set up a standard T7 RNA polymerase-based in vitro transcription reaction including:
- DNA template containing appropriate promoter sequence
- Nucleotide triphosphates (ATP, CTP, GTP, UTP)
- PureCap analog (replace conventional cap analog)
- Transcription buffer with appropriate magnesium concentration
- T7 RNA polymerase
- Incubate at 37°C for 2-4 hours to allow mRNA synthesis [2]
Step 2: Reversed-Phase HPLC Purification
- Prepare the transcribed mRNA mixture for RP-HPLC
- Use a C18 or similar reversed-phase column
- Employ a gradient elution with buffer A (e.g., 0.1 M ammonium acetate) and buffer B (e.g., 0.1 M ammonium acetate in 25% acetonitrile)
- Monitor separation at 260 nm; capped mRNA with hydrophobic tag will elute later than uncapped mRNA due to increased hydrophobicity
- Collect the peak corresponding to capped mRNA [2] [18]
Step 3: Photocleavage of Hydrophobic Tag
- Expose the collected capped mRNA fraction to UV light at 365 nm for 15-30 minutes
- Confirm tag removal by analytical RP-HPLC or mass spectrometry
- Precipitate and concentrate the pure capped mRNA using standard ethanol precipitation or tangential flow filtration [2]
Step 4: Quality Control and Characterization
- Analyze capping efficiency by LC-MS or enzymatic methods
- Assess mRNA integrity by agarose or capillary electrophoresis
- Quantify mRNA concentration by spectrophotometry
- Test for dsRNA contaminants by specific immunoassays [2] [15]
This protocol enables production of fully capped mRNA with 98-100% capping efficiency, effectively eliminating both uncapped mRNA and double-stranded RNA impurities in a single purification process [2] [18].
Comparative Analysis of Cap Structures Using PureCap Technology
Translation Efficiency Across Cap Types
The availability of pure, fully capped mRNA samples prepared using the PureCap method has enabled systematic comparison of the functional properties of different cap structures without the confounding factor of variable capping efficiency [2]. These studies have revealed significant differences in translational capacity between Cap-0, Cap-1, and Cap-2 structures.
Notably, Cap-2 mRNA demonstrated substantially enhanced protein production compared to Cap-1 mRNA across multiple experimental systems. In cultured cells, Cap-2 mRNA showed 3-4 times higher translation activity than Cap-1 mRNA prepared by standard capping methods [2] [17]. This enhanced translational efficiency was confirmed in animal models, where Cap-2 mRNA again exhibited 3-4 fold greater protein expression compared to conventional Cap-1 mRNA [2] [16].
The superior performance of Cap-2 structures extends beyond translation efficiency to include enhanced mRNA stability. The additional 2′-O-methyl group on the second nucleotide provides increased resistance to ribonucleases and decapping enzymes, thereby extending the functional half-life of the mRNA [2]. This combination of improved stability and enhanced translation makes Cap-2 mRNA particularly valuable for therapeutic applications where sustained protein expression is desired.
Table 3: Functional Characterization of Cap Structures Produced via PureCap Method
| Cap Type | Translation Efficiency | Immunostimulatory Activity | Stability | Therapeutic Potential |
|---|---|---|---|---|
| Cap-0 | Baseline | High | Standard | Limited due to immunogenicity |
| Cap-1 | Moderate improvement | Significantly reduced | Moderate improvement | Established in current vaccines |
| Cap-2 | 3-4 fold increase vs Cap-1 | Lowest | Highest | High potential for next-generation therapeutics |
Immunogenic Properties of Different Cap Structures
The immunogenic profiles of Cap-0, Cap-1, and Cap-2 mRNAs have been rigorously evaluated using the highly pure mRNA preparations enabled by the PureCap technology [2]. These studies demonstrate that Cap-2 structure drastically reduces mRNA affinity to retinoic acid-inducible gene-I (RIG-I), a key innate immune receptor that recognizes viral RNA, compared to Cap-1 structure [2]. Since viruses predominantly produce Cap-1 mRNA, the immune system is less stimulated by Cap-2 structures, making them less likely to trigger inflammatory responses [18] [16].
Highly purified Cap-0, Cap-1, and Cap-2-type mRNAs synthesized using the PureCap method all show lower immunostimulatory activity compared to mRNAs synthesized using conventional techniques, highlighting the importance of eliminating uncapped impurities [17] [18]. However, even among these pure preparations, Cap-2 mRNA causes the lowest stimulation of inflammatory responses, suggesting intrinsic immunological advantages beyond simply avoiding immune activation by uncapped impurities [2] [19].
The reduced immunogenicity of Cap-2 mRNA has important implications for therapeutic applications. In vaccine development, lower intrinsic immunogenicity of the mRNA vector allows the immune response to focus more specifically on the encoded antigen, potentially improving vaccine efficacy while reducing side effects such as inflammation at the injection site [17] [18]. For protein replacement therapies, reduced immunogenicity enables repeated administration without generating neutralizing antibodies against the mRNA vehicle itself [2].
Research Reagent Solutions for Cap Studies
Table 4: Essential Research Reagents for mRNA Cap Structure Investigations
| Reagent / Method | Function / Application | Key Features |
|---|---|---|
| PureCap Analogs | Production of pure capped mRNA for functional studies | Enables 100% capping efficiency; photocleavable hydrophobic tag permits RP-HPLC purification |
| Vaccinia Capping Enzyme | Enzymatic capping of in vitro transcribed mRNA | Generates natural cap structures; suitable for various RNA lengths and sequences |
| ARCA Cap Analogs | Co-transcriptional capping with correct orientation | Prevents reverse incorporation; produces Cap-0 structure |
| CleanCap Analogs | Co-transcriptional capping for Cap-1 structures | High capping efficiency (>95%); does not require high cap:GTP ratio |
| RP-HPLC Systems | Separation of capped and uncapped mRNA based on hydrophobicity | Critical for purification of PureCap-tagged mRNA; also removes dsRNA contaminants |
| mMESSAGE mMACHINE T7 Kits | Commercial systems for in vitro mRNA transcription | Include optimized cap analogs (mCap, ARCA, or CleanCap) and transcription reagents |
The development of PureCap technology represents a significant advancement in mRNA therapeutics by enabling the production of fully capped mRNA with 100% efficiency [2]. This method successfully addresses the long-standing challenge of separating capped mRNA from uncapped impurities through the innovative use of hydrophobically tagged cap analogs and RP-HPLC purification [2] [18]. The subsequent photocleavage step efficiently removes the hydrophobic tag under mild conditions, yielding native, footprint-free capped mRNA suitable for therapeutic applications [2].
The ability to produce pure Cap-2 mRNA using the PureCap method has revealed the remarkable functional advantages of this cap structure, including 3-4 fold higher translation activity and reduced immunostimulatory potential compared to conventional Cap-1 mRNA [2] [17] [16]. These properties make Cap-2 mRNA an attractive candidate for next-generation mRNA therapeutics and vaccines, potentially enabling lower dosing and reduced reactogenicity [18] [19].
Looking forward, the PureCap method provides an unbiased platform for studying structure-activity relationships among various cap modifications by eliminating the confounding effects of variable capping efficiency [2]. This capability will facilitate the rational design of optimized cap structures tailored to specific therapeutic applications. As mRNA therapeutics continue to expand beyond infectious disease vaccines to include cancer immunotherapies, protein replacement therapies, and gene editing applications, the availability of highly pure, fully capped mRNA with enhanced translational efficiency and reduced immunogenicity will play an increasingly important role in advancing these promising treatments toward clinical use [2] [17] [18].
The efficacy and safety of messenger RNA (mRNA) vaccines are fundamentally dependent on the purity of the final pharmaceutical product. Impurities introduced during manufacturing can significantly compromise vaccine performance by triggering unwanted immune responses and reducing the desired therapeutic effect. These impurities predominantly originate from two critical stages of production: the in vitro transcription (IVT) process, which can generate incomplete or truncated mRNA sequences and double-stranded RNA (dsRNA) byproducts, and the capping step, where uncapped mRNA molecules with 5'-triphosphates may persist [2] [20]. These process-related impurities possess distinct molecular patterns that the human innate immune system recognizes as pathogenic, activating signaling pathways that ultimately hinder vaccine effectiveness.
The 5' cap structure is particularly crucial for mRNA function and immunological neutrality. Beyond its essential roles in promoting translation initiation and protecting mRNA from degradation, the cap structure serves as a key molecular identifier that allows the host cell to distinguish between "self" and "non-self" RNA [2]. Uncapped mRNA species with 5'-triphosphates mimic viral RNA patterns, triggering potent type I interferon responses through recognition by cytoplasmic innate immune receptors such as retinoic acid-inducible gene-I (RIG-I) and melanoma differentiation-associated protein 5 (MDA-5) [2]. Similarly, dsRNA byproducts formed during IVT activate additional interferon-induced pathways, creating an antiviral cellular state that profoundly inhibits translation of the encoded antigen [20]. This unintended immunostimulation not only reduces antigen production and compromises immunogenicity but also contributes to inflammatory side effects, including injection site reactions and systemic symptoms [6] [17].
Conventional capping methods, whether enzymatic or co-transcriptional using cap analogs, face significant limitations in achieving complete capping efficiency, typically reaching only 80-90% for widely used Cap-0 and Cap-1 type mRNAs [2]. The remaining 10-20% uncapped mRNA impurities are sufficient to trigger substantial immune responses, as demonstrated by Moradian et al., who reported that even small amounts of 5' triphosphate RNA byproducts from standard capping methods induce significant innate immune activation [2]. This purity problem has necessitated the development of innovative solutions to achieve complete capping and effective separation of capped from uncapped mRNA species.
The PureCap Method: A Revolutionary Approach to mRNA Purification
Technology Foundation and Working Mechanism
The PureCap method represents a paradigm shift in mRNA production technology, addressing the fundamental purity problem through a novel chemical and purification approach. Developed by researchers at Nagoya University in collaboration with Tokyo Medical and Dental University, this innovative strategy enables the selective purification of exclusively capped mRNA, effectively eliminating both uncapped mRNA and dsRNA impurities in a single process [2] [6]. The technology centers on specially engineered hydrophobic cap analogs that incorporate a photocleavable tag, creating a temporary physicochemical distinction between capped and uncapped mRNA species that can be exploited for purification.
The core innovation lies in the design of hydrophobic photocaged tag-modified cap analogs that are incorporated into the mRNA during the IVT reaction [2]. These custom cap analogs feature a 2-nitrobenzyl (Nb) derivative as a hydrophobic photocaging molecule, enhanced with a tert-butyl (tBu) group to increase hydrophobicity and chemical stability [2]. This strategic modification enables the separation of capped mRNA from uncapped counterparts using reversed-phase high-performance liquid chromatography (RP-HPLC), a standard purification technique in nucleic acid pharmaceutical production. Following chromatographic separation, brief photo-irradiation cleaves the hydrophobic tag under mild conditions, yielding footprint-free native capped mRNA without residual modifications [2].
The PureCap platform synthesizes four distinct dinucleotide analogs with the hydrophobic Nb tag positioned at different molecular locations: DiPure (1) with the Nb group at the 2'-position via an acetal group; DiPure/2′OMe (2) and DiPure/3′OMe (3) with O-methyl and O-Nb modifications at the 2′/3′ positions; and DiPure/N2 (4) with Nb modification at the exocyclic amino group of m7G [2]. This strategic diversity enables the production of various cap structures while maintaining anti-reverse cap analog (ARCA) activity to ensure proper orientation during transcription initiation.
Key Advantages Over Conventional Methods
The PureCap method delivers substantial improvements over traditional capping approaches across multiple critical parameters:
Maximized Capping Efficiency: The technology achieves 100% capping efficiency, completely eliminating immunostimulatory uncapped mRNA impurities that plague conventional methods limited to 80-90% efficiency [2].
Simultaneous Impurity Removal: RP-HPLC purification effectively removes both uncapped mRNA and dsRNA contaminants in a single process, addressing the two most significant impurity classes that impact vaccine safety and performance [2].
Structural Versatility: The platform supports the production of Cap-0, Cap-1, and Cap-2 type mRNA with equally high efficiency, enabling precise structure-activity relationship studies that were previously impossible due to varying capping efficiencies between cap types [2] [17].
Non-Enzymatic Process: By eliminating the need for multiple enzymatic treatments (e.g., RNA 5' polyphosphatase and XRN I exoribonuclease), the method reduces production costs, minimizes non-specific mRNA degradation, and streamlines manufacturing [2].
Scalability: The technology has been successfully demonstrated across mRNA constructs of varying lengths, from 650 nt to 4,247 nt (coronavirus mRNA), maintaining over 98% purity even for the longest sequences [6].
Table 1: Comparison of Capping Methods for mRNA Vaccine Production
| Parameter | Conventional Co-transcriptional Capping | Enzymatic Capping | PureCap Method |
|---|---|---|---|
| Maximum Capping Efficiency | 80-90% | >95% (with optimization) | 100% |
| Uncapped mRNA Removal | Requires additional enzymatic treatment | Built-in but incomplete | Physical separation via RP-HPLC |
| dsRNA Removal | Separate purification needed | Separate purification needed | Simultaneous removal with RP-HPLC |
| Cap Structure Flexibility | Limited by analog availability | Limited by enzyme specificity | Cap-0, Cap-1, Cap-2 with equal efficiency |
| Production Complexity | Moderate | High (multiple enzymatic steps) | Simplified (physical separation) |
| Cost Considerations | Lower initial cost, higher purification cost | High enzyme costs | Higher analog cost, lower purification cost |
Experimental Evidence and Performance Data
Enhanced Translational Efficiency
The exceptional purity achieved through the PureCap method translates directly into superior biological performance. Comparative studies demonstrate that fully capped Cap-2-type mRNA synthesized using PureCap technology exhibits 3- to 4-fold higher translation activity in both cultured cells and animal models compared to Cap-1-type mRNA prepared by standard capping methods [2]. This dramatic enhancement reflects the combined effects of complete capping and the intrinsic properties of the Cap-2 structure, which includes methylation at the first two transcribed nucleotides.
The Cap-2 structure specifically reduces mRNA affinity for RIG-I, an innate immune receptor that recognizes viral RNA patterns, while simultaneously enhancing mRNA stability and translational efficiency [2]. Unlike conventional methods that cannot produce pure Cap-2 mRNA at scale, the PureCap platform enables unbiased evaluation of this cap structure, revealing its significant functional advantages. The technology also facilitates precise comparison of different cap configurations by eliminating the confounding variable of varying capping efficiency, demonstrating that Cap-2 mRNA produces 3-5 times more protein than Cap-1 mRNA while eliciting lower immunostimulation [17].
Reduced Immunostimulation
The exceptional purity of PureCap-synthesized mRNA directly correlates with reduced activation of innate immune pathways. Highly purified Cap-0, Cap-1, and Cap-2-type mRNAs synthesized using the PureCap method all demonstrate significantly lower immunostimulatory activity compared to mRNAs synthesized using conventional techniques [6]. This reduction in intrinsic immunogenicity is particularly pronounced for Cap-2 structures, which are naturally occurring in mammalian cells but have been largely inaccessible for therapeutic applications until now.
Since viruses predominantly produce Cap-1 mRNA, the immune system is less stimulated by Cap-2 structures, suggesting that vaccines incorporating this cap variant would be less likely to cause unwanted inflammatory side effects while maintaining robust antigen production [6]. This combination of high translational efficiency and low immunostimulation represents a fundamental advance in mRNA vaccine technology, addressing one of the most significant challenges in the field—balancing potency with tolerability.
Table 2: Performance Characteristics of PureCap-Derived mRNA
| Performance Metric | Cap-0 mRNA | Cap-1 mRNA | Cap-2 mRNA |
|---|---|---|---|
| Protein Production Level | Baseline | 1.5-2x Cap-0 | 3-4x Cap-0 |
| RIG-I Binding Affinity | High | Moderate | Low |
| Type I Interferon Induction | High | Moderate | Low |
| Immunostimulatory Activity | High | Moderate | Low |
| mRNA Stability | Baseline | Moderate improvement | Significant improvement |
| Capping Efficiency | 100% | 100% | 100% |
Implementation Protocols
PureCap mRNA Synthesis and Purification Workflow
Materials Required:
- PureCap analog (DiPure/2'OMe recommended for Cap-1 mRNA)
- T7 RNA polymerase and transcription buffer
- NTP mixture (ATP, CTP, UTP, GTP)
- DNA template with target sequence
- Reversed-phase HPLC system with C18 column
- Photocleavage apparatus (365 nm UV source)
- Purification buffers (aqueous and organic phases)
Procedure:
In Vitro Transcription Setup:
- Prepare transcription reaction containing:
- 1 μg linearized DNA template
- 10 μL 5× transcription buffer
- 7.5 mM each NTP
- 2 mM PureCap analog
- 2 μL T7 RNA polymerase
- Nuclease-free water to 50 μL final volume
- Incubate at 37°C for 2-4 hours
- Prepare transcription reaction containing:
RP-HPLC Purification:
- Dilute transcription reaction 1:1 with 0.1 M TEAA buffer, pH 7.0
- Inject onto C18 RP-HPLC column equilibrated with 5% buffer B
- Apply gradient: 5% B to 18% B over 19 minutes, then to 70% B over 1 minute
- Monitor at 260 nm; capped mRNA elutes later due to hydrophobicity
- Collect capped mRNA fraction
Photocleavage and Recovery:
- Exclude collected fraction to 365 nm UV light for 15-30 minutes
- Confirm tag removal by analytical RP-HPLC
- Precipitate mRNA with 0.1 volume 3M sodium acetate and 2.5 volumes ethanol
- Resuspend purified mRNA in nuclease-free water
- Quantify by spectrophotometry and assess integrity by capillary gel electrophoresis
Quality Assessment and Analytical Methods
Purity Verification:
- IP-RPLC-UV: Use ion-pair reversed-phase liquid chromatography with UV detection to assess mRNA purity and profile product-related impurities [20].
- Capillary Gel Electrophoresis (CGE): Determine mRNA integrity and size distribution using the SCIEX RNA 9000 Purity and Integrity kit with SYBR Green II RNA Gel Stain [20].
- Mass Photometry: Characterize mRNA molecular weight and detect size variants, including aggregates and partial degradation products [20].
Functional Assessment:
- In vitro translation: Compare protein production levels in relevant cell lines (e.g., HEK293) using ELISA or western blot.
- Immunostimulation assay: Quantify interferon-beta and proinflammatory cytokine production in human peripheral blood mononuclear cells (PBMCs).
- Stability testing: Monitor mRNA integrity under appropriate storage conditions over time.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for PureCap mRNA Synthesis and Characterization
| Reagent/Material | Function | Application Notes |
|---|---|---|
| PureCap Analogs | Co-transcriptional capping with hydrophobic tag | DiPure/2'OMe recommended for Cap-1; DiPure for Cap-0 |
| T7 RNA Polymerase | High-yield in vitro transcription | Pharma-grade recommended for therapeutic applications |
| RP-HPLC System | Separation of capped vs. uncapped mRNA | C18 column with wide pores optimal for long mRNAs |
| Photocleavage Apparatus | Removal of hydrophobic tag post-purification | 365 nm UV source with cooling to prevent mRNA degradation |
| DNAPac RP Column | Analytical assessment of mRNA purity | Thermo Fisher Scientific; compatible with long mRNA sequences |
| RNA 9000 Purity Kit | Integrity analysis by capillary gel electrophoresis | SCIEX; includes standards and staining reagents |
| SYBR Green II Stain | Detection of dsRNA impurities | Specific binding to double-stranded regions |
Visualizing the PureCap Workflow and Technology Mechanism
Diagram 1: PureCap mRNA Synthesis and Purification Workflow. The process begins with in vitro transcription (IVT) using hydrophobic PureCap analogs, followed by reversed-phase HPLC separation that effectively resolves capped from uncapped mRNA. The collected capped mRNA fraction undergoes photocleavage to remove the temporary hydrophobic tag, yielding pure native capped mRNA with 100% capping efficiency.
Diagram 2: Impact of mRNA Impurities on Vaccine Efficacy and Safety. Uncapped mRNA with 5'-triphosphates and double-stranded RNA (dsRNA) byproducts activate cytoplasmic innate immune receptors (RIG-I and MDA-5), triggering type I interferon responses that inhibit translation of the encoded antigen and promote inflammatory side effects, ultimately compromising vaccine performance.
The PureCap technology represents a transformative approach to addressing the fundamental purity problem in mRNA vaccine production. By enabling the synthesis and purification of completely capped mRNA with 100% efficiency, this method directly targets the key impurities that compromise both the safety and efficacy of mRNA-based therapeutics. The platform's unique ability to produce Cap-2 mRNA with superior translational capacity and reduced immunostimulation positions it as a enabling technology for next-generation mRNA vaccines, particularly for applications requiring exceptional tolerability profiles, such as cancer vaccines and prophylactic vaccines for vulnerable populations.
Future developments in mRNA purity will likely build upon the foundation established by PureCap technology, potentially integrating continuous manufacturing approaches [21] and advancing analytical characterization methods [20] to further enhance product quality and production efficiency. As the field progresses toward more sophisticated mRNA applications, including self-amplifying platforms [22] and combination therapies, the critical importance of mRNA purity will only intensify, making solutions like the PureCap method essential components of the therapeutic mRNA toolkit.
A Step-by-Step Guide to the PureCap mRNA Purification Workflow
The PureCap method represents a transformative approach in the synthesis of messenger RNA (mRNA) therapeutics by addressing a fundamental challenge in production: the separation of functionally capped mRNA from immunogenic uncapped byproducts. Conventional mRNA capping techniques, whether enzymatic or co-transcriptional, achieve maximum capping efficiencies of only 80-90%, leaving significant quantities of uncapped mRNA with a 5' triphosphate group that triggers undesirable immune responses via innate immune receptors such as RIG-I and MDA5 [2]. The PureCap technology overcomes this limitation through the strategic design of hydrophobic photocaged cap analogs that enable physical separation of capped mRNA, achieving nearly 100% capping efficiency and significantly enhancing the safety and efficacy profile of therapeutic mRNA [2] [6] [16].
This application note details the core chemistry behind designing these hydrophobic photocleavable cap analogs, provides optimized protocols for their implementation, and presents comprehensive data on the performance characteristics of mRNAs produced using the PureCap system. The methodology is particularly valuable for advancing mRNA vaccine development and therapeutic applications where reduced immunogenicity and enhanced translational capacity are critical for clinical success [6] [18].
Core Chemistry & Molecular Design
Strategic Rationale
The molecular design of PureCap analogs addresses a fundamental limitation in mRNA therapeutics: the nearly identical physicochemical properties of capped and uncapped mRNA, which previously prevented their physical separation [2]. By incorporating a strategically positioned hydrophobic tag and photocleavable linker, these analogs enable purification based on hydrophobicity differences while ensuring the final product retains a native cap structure after tag removal [2] [23].
The design capitalizes on the well-established reverse-phase high-performance liquid chromatography (RP-HPLC) platform already used in nucleic acid therapeutic manufacturing for removing double-stranded RNA impurities [2]. This integration with existing purification infrastructure facilitates straightforward implementation in current mRNA production workflows without requiring entirely new process development [6] [16].
Molecular Architectures
The PureCap platform encompasses several dinucleotide cap analog designs, each incorporating a hydrophobic photocleavable tag at distinct positions on the N7-methylguanosine (m⁷G) moiety:
- DiPure (1): Features a 2-nitrobenzyl (Nb) derivative connected via an acetal group at the 2'-O position, maintaining a free 3'-OH group to prevent reverse incorporation during transcription [2].
- DiPure/2′OMe (2): Contains both O-methyl and O-Nb modifications at the 2′/3′ positions of m⁷G [2].
- DiPure/3′OMe (3): Analogous to DiPure/2'OMe but with alternative positioning of the O-methyl and O-Nb modifications [2].
- DiPure/N2 (4): Incorporates the Nb tag at the exocyclic amino group of m⁷G via a carbamate linkage [2].
The hydrophobic tag incorporates a tert-butyl (tBu) group within a 2-nitrobenzyl (Nb) photocaging molecule to enhance both hydrophobicity for effective RP-HPLC separation and chemical stability during synthesis and transcription [2]. Upon exposure to specific light wavelengths, the photocleavable Nb group is removed, regenerating the native cap structure without molecular footprints that could alter biological activity [2] [6].
Table 1: PureCap Analog Structures and Modifications
| Analog Name | Tag Position | Structural Modifications | Key Features |
|---|---|---|---|
| DiPure (1) | 2'-O position | Nb tag via acetal group, free 3'-OH | Prevents reverse incorporation; footprint-free after deprotection |
| DiPure/2′OMe (2) | 2′/3′ positions | Combination of O-methyl and O-Nb modifications | Anti-reverse activity; precise methylation patterning |
| DiPure/3′OMe (3) | 2′/3′ positions | Alternative O-methyl/O-Nb configuration | Anti-reverse activity; controlled methylation |
| DiPure/N2 (4) | Exocyclic amino group | Nb tag via carbamate linkage | Alternative positioning for HPLC separation |
Synthetic Methodology
The synthesis of PureCap analogs employs innovative chemistry to overcome solubility challenges presented by the introduced hydrophobic tags. Traditional stepwise phosphorylation approaches using aqueous purification methods proved problematic for these lipophilic compounds [2]. The PureCap synthesis instead utilizes:
- One-pot diphosphate synthesis directly from guanosine derivatives, avoiding intermediate isolations in aqueous solvents [2].
- Two complementary approaches:
- Subsequent N7 methylation and zinc chloride-catalyzed condensation with guanosine monophosphate imidazolide to form the final dinucleotide cap analogs [2].
This streamlined synthesis achieves improved yields of hydrophobic cap analogs while reducing purification steps, facilitating broader implementation of the technology [2].
Experimental Protocols
mRNA Transcription with PureCap Analogs
Objective: Incorporate PureCap analogs during in vitro transcription (IVT) to produce mRNA with hydrophobic tags enabling subsequent purification.
Materials:
- PureCap analog (e.g., DiPure, DiPure/2'OMe, DiPure/3'OMe, DiPure/N2)
- T7 RNA Polymerase and 10X transcription buffer
- NTP mix (ATP, CTP, GTP, UTP)
- DNA template with appropriate promoter
- RNase inhibitor
- MgCl₂ solution
- Dithiothreitol (DTT)
Procedure:
- Prepare IVT master mix on ice:
- 10 µL 10X transcription buffer
- 10 µL 25 mM NTP mix (2.5 mM each NTP)
- 5 µL PureCap analog (10 mM stock)
- 5 µL T7 RNA Polymerase
- 2 µL RNase inhibitor
- 3 µL 100 mM DTT
- 15 µL nuclease-free water
- 50 µL total volume
Add 50 µL DNA template (0.5-1 µg/µL in nuclease-free water)
Mix gently by pipetting and incubate at 37°C for 2-4 hours
Optional: Add DNase I (2 µL) and incubate at 37°C for 15 minutes to digest template DNA
Proceed to purification or store at -20°C for short-term storage
Critical Parameters:
- Maintain 5:1 to 10:1 molar ratio of PureCap analog to GTP for efficient initiation [2]
- Adjust MgCl₂ concentration to 6-8 mM final concentration for optimal yield
- Include DTT to maintain enzyme stability during extended incubation
RP-HPLC Purification of Capped mRNA
Objective: Separate capped from uncapped mRNA species based on hydrophobicity differences imparted by the PureCap tag.
Materials:
- RP-HPLC system with C18 column (4.6 × 250 mm, 5 µm particle size)
- Mobile phase A: 100 mM triethylammonium acetate (TEAA), pH 7.0
- Mobile phase B: acetonitrile
- mRNA transcription reaction mixture
- Nuclease-free water
- Collection tubes
Procedure:
- Dilute mRNA transcription reaction with equal volume of nuclease-free water
- Set column temperature to 60°C
- Program binary gradient:
- 0-5 min: 5% B
- 5-40 min: linear gradient from 5% to 25% B
- 40-45 min: 25% B
- 45-50 min: return to 5% B
- 50-60 min: re-equilibration at 5% B
- Set flow rate to 0.8 mL/min
- Monitor detection at 254 nm and 260 nm
- Inject 50-100 µg mRNA per run
- Collect capped mRNA fraction (typically eluting at higher acetonitrile concentration)
- Precipitate collected mRNA with 2.5 volumes ethanol and 0.1 volume 3M sodium acetate, pH 5.2
- Wash pellet with 70% ethanol and resuspend in nuclease-free water
Critical Parameters:
- Maintain column temperature at 60°C to optimize separation efficiency
- Identify capped mRNA fraction by increased retention time relative to uncapped species
- Scale injection volume according to column capacity to maintain resolution
- For large-scale preparations, implement analytical RP-HPLC to confirm separation before preparative runs
Photocleavage and Tag Removal
Objective: Remove hydrophobic tag from purified mRNA to regenerate native cap structure.
Materials:
- Purified mRNA with hydrophobic tag
- Light source (LED lamp, 365-420 nm)
- UV-transparent reaction vessel
- Cooling apparatus (if using high-intensity light source)
- Nuclease-free water
Procedure:
- Adjust mRNA concentration to 0.1-0.5 mg/mL in nuclease-free water
- Transfer to UV-transparent reaction vessel
- For DMNB- or NPM-tagged analogs: Irradiate at 365 nm for 5-15 seconds at intensity of 140 mW/cm² [3]
- For redshifted tags (NP, NPM): Alternative irradiation at 405 nm for 60 seconds or 420 nm for 120 seconds [3]
- Maintain sample temperature below 25°C during irradiation using cooling apparatus if necessary
- Confirm complete deprotection by analytical RP-HPLC (shift to earlier retention time)
- Precipitate mRNA if necessary to remove any photolysis byproducts
Critical Parameters:
- Optimize irradiation time to achieve complete deprotection while minimizing mRNA degradation
- For large volumes, ensure uniform irradiation with stirring or circulation
- Verify tag removal by retention time shift in analytical RP-HPLC
- Process samples promptly after purification to prevent premature tag cleavage
Performance Data & Validation
Capping Efficiency and Purity
The PureCap method achieves exceptional capping efficiency across diverse mRNA lengths and cap structures, substantially outperforming conventional capping methods.
Table 2: Capping Efficiency and Purity of PureCap mRNAs
| mRNA Length | Cap Structure | Purity After RP-HPLC | Capping Efficiency | Comparison to Conventional Methods |
|---|---|---|---|---|
| 650 nt | Cap-0 | >99% | ~100% | Conventional: 80-90% [2] |
| 650 nt | Cap-1 | >99% | ~100% | Conventional: 80-90% [2] |
| 650 nt | Cap-2 | >99% | ~100% | Not achievable conventionally [2] |
| 4,247 nt | Cap-1 | >98% | ~100% | Conventional: 80-90% [2] [16] |
| 4,247 nt | Cap-2 | >98% | ~100% | Not achievable conventionally [2] [16] |
Functional Characterization
Translation Efficiency:
- Cap-2-type mRNA synthesized using PureCap technology demonstrates 3- to 4-fold higher translation activity in cultured cells compared to Cap-1-type mRNA prepared by standard capping methods [2] [23].
- In animal studies, Cap-2-type mRNA shows 3- to 5-fold greater protein production than conventional Cap-1 mRNA, significantly enhancing potential immune response for vaccines [6] [16] [18].
Immunogenicity Profile:
- PureCap-synthesized mRNAs demonstrate significantly lower immunostimulatory activity across all cap structures (Cap-0, Cap-1, Cap-2) compared to mRNAs synthesized using conventional techniques [16] [18].
- The near-complete removal of uncapped mRNA impurities substantially reduces activation of innate immune receptors (RIG-I, MDA5), potentially mitigating injection site inflammation and other immune-related adverse effects [2] [6].
Structural Versatility:
- The PureCap platform successfully produces mRNAs with Cap-0, Cap-1, and Cap-2 structures at high purity, enabling previously impossible direct comparison of structure-function relationships [2] [16].
- Cap-2 structure, difficult to evaluate with conventional methods due to purity limitations, shows particularly favorable characteristics for therapeutic applications with enhanced translation and reduced immunogenicity [2] [18].
The Scientist's Toolkit
Table 3: Essential Research Reagents for PureCap Implementation
| Reagent/Category | Specific Examples | Function/Application |
|---|---|---|
| PureCap Analogs | DiPure, DiPure/2'OMe, DiPure/3'OMe, DiPure/N2 | Co-transcriptional capping with hydrophobic tags for purification |
| Transcription Components | T7 RNA Polymerase, NTPs, RNase inhibitor, transcription buffer | mRNA synthesis with cap analog incorporation |
| Purification Materials | RP-HPLC with C18 column, triethylammonium acetate, acetonitrile | Separation of capped and uncapped mRNA based on hydrophobicity |
| Photocleavage Equipment | LED lamp (365-420 nm), UV-transparent reaction vessels | Removal of hydrophobic tag to regenerate native cap |
| Analysis & QC Tools | Analytical RP-HPLC, LC-MS, capillary gel electrophoresis | Verification of capping efficiency, purity, and mRNA integrity |
Implementation Workflow
Troubleshooting Guide
Table 4: Common Implementation Challenges and Solutions
| Issue | Potential Causes | Recommended Solutions |
|---|---|---|
| Poor RP-HPLC separation | Incorrect column temperature, inadequate mobile phase pH | Maintain column at 60°C; ensure TEAA buffer at pH 7.0; optimize gradient |
| Incomplete photocleavage | Insufficient light intensity, inappropriate wavelength | Verify light source output; optimize irradiation time; consider alternative PureCap analog with redshifted tag |
| Low transcription yield | Suboptimal PureCap:GTP ratio, enzyme inhibition | Adjust PureCap analog to GTP ratio (5:1 to 10:1); ensure DTT present in reaction; check Mg²⁺ concentration |
| Residual immunogenicity | Incomplete removal of uncapped mRNA | Verify RP-HPLC fraction collection; confirm photocleavage efficiency; implement analytical QC methods |
| mRNA degradation | RNase contamination, excessive irradiation | Use RNase-free reagents; optimize irradiation time to balance tag removal and mRNA integrity |
Integration via Co-Transcriptional Capping during IVT
The production of messenger RNA (mRNA) for therapeutic applications, such as vaccines and gene therapies, relies heavily on in vitro transcription (IVT). A critical step in this process is 5' capping, which is essential for mRNA stability, efficient translation, and minimizing unwanted immune responses [24] [1]. Co-transcriptional capping, where cap analogs are incorporated into the mRNA during the IVT reaction itself, streamlines production by eliminating extra enzymatic steps [25]. However, a significant challenge persists: traditional co-transcriptional capping methods, such as those using Anti-Reverse Cap Analogs (ARCA), achieve capping efficiencies of only 80-90%, leaving immunogenic uncapped mRNA impurities that are difficult to remove due to their nearly identical physicochemical properties to capped mRNA [2] [25].
The PureCap method represents a revolutionary advancement by integrating a unique, hydrophobic tag into the cap analog structure. This tag enables the simple and effective purification of fully capped mRNA from uncapped impurities using standard reversed-phase high-performance liquid chromatography (RP-HPLC). Subsequent irradiation with light removes the tag, yielding native, footprint-free mRNA with up to 100% capping efficiency [2] [6]. This application note details the protocol for implementing the PureCap technology to achieve superior mRNA purity and performance.
Quantitative Comparison of Capping Technologies
The table below summarizes the key performance metrics of current capping technologies, highlighting the quantitative advantages of the PureCap method.
Table 1: Performance Comparison of mRNA Capping Technologies
| Technology | Capping Efficiency | Final Cap Structure | Key Impurities | Protein Expression (Relative to ARCA Cap-0) |
|---|---|---|---|---|
| ARCA [25] | 50-80% (Cap-0) | Cap-0 (requires additional step for Cap-1) | ~20-50% uncapped mRNA | 1x (Baseline) |
| CleanCap [25] | >95% (Cap-1) | Cap-1 in a single step | ~5% uncapped mRNA | Data not available |
| PureCap [2] [6] | 98-100% (Cap-0, Cap-1, or Cap-2) | Cap-0, Cap-1, or Cap-2 | <2% uncapped mRNA | Cap-2 mRNA: 3-4x higher |
Experimental Protocol for PureCap mRNA Synthesis and Purification
Principle and Workflow
The PureCap method utilizes specially synthesized cap analogs (e.g., DiPure (1), DiPure/2'OMe (2), DiPure/3'OMe (3)) modified with a hydrophobic photocleavable tag, such as a 2-nitrobenzyl derivative with a tert-butyl group [2]. During the IVT reaction, this analog is incorporated at the 5' end of the mRNA. The incorporated hydrophobic tag then facilitates the separation of capped mRNA from uncapped mRNA based on differential affinity to the hydrophobic stationary phase in RP-HPLC. A final photoirradiation step removes the tag to yield the pure, native cap structure.
The following diagram illustrates the core workflow of the PureCap method:
Step-by-Step Procedure
Step 1: In Vitro Transcription with PureCap Analog
- Reaction Setup: Prepare a standard IVT reaction mixture containing:
- T7 RNA Polymerase (and buffer)
- DNA template (linearized plasmid)
- Nucleotide Triphosphates (NTPs)
- PureCap analog (e.g., 4-6 mM, replacing GTP or other cap analogs) [2].
- RNase-free water.
- Incubation: Incubate the reaction at 37°C for 2-4 hours to allow for RNA synthesis and capping.
- Termination: After incubation, degrade the DNA template by adding DNase I and incubating further for 15-30 minutes.
Step 2: Purification by Reversed-Phase HPLC
- Sample Preparation: Dilute the IVT reaction mixture with an equal volume of an RP-HPLC compatible buffer, such as 0.1 M Triethylammonium acetate (TEAA), and filter through a 0.22 µm membrane.
- Chromatography: Inject the sample onto a C18 or C8 reversed-phase column.
- Mobile Phase A: 0.1 M TEAA in water.
- Mobile Phase B: 0.1 M TEAA in acetonitrile.
- Use a gradient of 5% to 25% Mobile Phase B over 20-40 minutes at a flow rate of 1 mL/min.
- Collection: Monitor the elution at 260 nm. The hydrophobic, capped mRNA will elute later than the uncapped mRNA due to the presence of the photocleavable tag [2]. Precisely collect the peak corresponding to the capped mRNA.
Step 3: Removal of the Hydrophobic Tag
- Photoirradiation: Place the collected HPLC fraction in a quartz cuvette or a suitable transparent container. Irradiate the sample with long-wavelength UV light (e.g., 365 nm) for a defined period (e.g., 30-60 minutes) to cleave the 2-nitrobenzyl linker [2] [6].
- Desalting/Buffer Exchange: After irradiation, use ethanol precipitation or a desalting column to remove the cleavage byproducts and exchange the buffer into a suitable storage buffer (e.g., nuclease-free water or Tris-EDTA buffer).
- Quality Control: Analyze the final mRNA product by analytical RP-HPLC or LC-MS to confirm cap integrity and purity. Quantify the mRNA by spectrophotometry.
The Scientist's Toolkit: Essential Reagents for PureCap Experiments
Table 2: Key Research Reagent Solutions for PureCap mRNA Synthesis
| Item | Function/Description | Example/Catalog Context |
|---|---|---|
| PureCap Analogs | Hydrophobic, photocleavable cap analogs (e.g., DiPure series) for incorporation during IVT. | Synthesized cap analogs like DiPure (1), DiPure/2'OMe (2) [2]. |
| T7 RNA Polymerase | Enzyme for catalyzing the in vitro transcription of mRNA from a DNA template. | Included in kits like Takara IVTpro mRNA Synthesis Kit [25]. |
| RP-HPLC System | Chromatography system for purifying tagged, capped mRNA from uncapped impurities. | Standard HPLC systems with C8 or C18 columns [2] [6]. |
| Long-Wave UV Lamp | Light source for the photocleavage of the hydrophobic tag from the purified mRNA. | UVP UV Lamps (365 nm wavelength) [2]. |
Analytical and Functional Validation
Assessment of Capping Efficiency and Purity
Analytical RP-HPLC is the primary method for assessing the success of the purification. The chromatogram of the final product post-photoirradiation should show a single, sharp peak, with the earlier-eluting peak corresponding to uncapped mRNA being absent, confirming >98% capping efficiency [2] [6]. Cap structure identity can be further confirmed by mass spectrometry.
Functional Performance: Translation and Immunogenicity
Functionally, mRNA produced with the PureCap method demonstrates superior performance:
- Enhanced Translation: Cap-2-type mRNA synthesized using PureCap showed a 3- to 4-fold increase in protein production in both cultured cells and animal models compared to Cap-1-type mRNA produced by standard methods [2].
- Reduced Immunogenicity: Due to the near-complete removal of uncapped mRNA impurities, PureCap-synthesized mRNAs (including Cap-0, Cap-1, and Cap-2) exhibit significantly lower immunostimulatory activity, minimizing the risk of inflammation caused by impurity-triggered immune responses [6].
Reversed-phase high-performance liquid chromatography (RP-HPLC) is the most common HPLC separation technique, used for the analysis of compounds with accessible hydrophobic moieties [26]. The separation mechanism is primarily governed by hydrophobic interactions between non-polar regions of analyte molecules and the hydrophobic ligands (e.g., C8 or C18 chains) attached to the stationary phase [27]. The solvophobic theory provides a reliable basis for explaining this process, wherein hydrophobic molecules or regions in a polar solvent tend to associate with the hydrophobic stationary phase [26].
In RP-HPLC, the relative hydrophobicity of an analyte determines its retention on the column. More hydrophobic analytes exhibit stronger interaction with the stationary phase and thus longer retention times. Elution is typically achieved using a gradient of an organic solvent (e.g., acetonitrile or methanol) in water, which gradually reduces the interaction between the analytes and the stationary phase [26]. Based on the octanol/water partition coefficient (log Kow), compounds with log Kow values ranging from approximately -1.75 to +5.55 can be effectively analyzed using RP-HPLC [26].
The Hydrophobic-Subtraction Model of RP-HPLC Selectivity
The hydrophobic-subtraction (H-S) model quantitatively describes the five major interactions that govern selectivity in RP-HPLC [27]. While hydrophobic interaction is the dominant force, the model accounts for four other secondary—but selectivity-critical—interactions.
Table 1: The Five Interaction Terms of the Hydrophobic-Subtraction Model [27]
| Interaction Term | Symbol | Description | Molecular Basis |
|---|---|---|---|
| Hydrophobic Interaction | H | The dominant contributor to retention. | Interaction between hydrophobic solute regions and the hydrophobic stationary phase. |
| Steric Interaction | S* | Resistance to penetration by bulky molecules. | Dependent on bonded phase density and ligand length; akin to size exclusion. |
| Hydrogen-Bonding (Acidic) | A | H-B interaction between basic solutes and acidic silanols. | Solute basic groups with column acidic sites (e.g., silanols). |
| Hydrogen-Bonding (Basic) | B | H-B interaction between acidic solutes and a vicinal-silanol pair. | Solute acidic groups with a paired basic site on the column surface. |
| Ionic Interaction | C | Cation-exchange between ionized bases and ionized silanols. | Interaction at neutral to high pH between protonated bases and deprotonated silanols. |
The H-S model allows for the quantitative comparison of columns to identify those with equivalent or orthogonal selectivity, which is crucial for method development and validation [27].
Application in mRNA Therapeutics: The PureCap Method
The purification of in vitro transcribed (IVT) mRNA presents a significant challenge due to the need to separate functionally critical capped mRNA from immunogenic uncapped mRNA impurities, which have nearly identical physicochemical properties [2]. The PureCap method leverages a unique RP-HPLC-based approach to achieve this separation.
The Scientific Principle
The method employs specially designed hydrophobic cap analogs (PureCap analogs) that are co-transcriptionally incorporated into the 5' end of the mRNA strand [2]. These analogs are modified with a hydrophobic tag, such as a photocleavable 2-nitrobenzyl (Nb) derivative featuring a tert-butyl (tBu) group to enhance hydrophobicity [2]. This tag introduces a significant difference in hydrophobicity between capped and uncapped mRNA species, enabling their physical separation by RP-HPLC. Following purification, brief photo-irradiation cleaves the hydrophobic tag, yielding a footprint-free, native capped mRNA [2].
Table 2: Key Outcomes of mRNA Purification via the PureCap RP-HPLC Method [2]
| Parameter | Standard Capping Method | PureCap RP-HPLC Method |
|---|---|---|
| Capping Efficiency | 80–90% | 100% |
| Cap Structures Accessible | Primarily Cap-0 and Cap-1 | Cap-0, Cap-1, and Cap-2 |
| Separation of Uncapped mRNA | Not physically separable; requires enzymatic treatment | Direct physical separation achieved |
| Co-removal of dsRNA Impurity | Not achieved by capping method alone | Yes, simultaneous removal during RP-HPLC |
| Translation Activity | Baseline (Cap-1) | Up to 3- to 4-fold higher (for Cap-2) |
Experimental Protocol: mRNA Purification via PureCap RP-HPLC
A. In Vitro Transcription with PureCap Analog
- Reaction Setup: Perform a standard IVT reaction containing DNA template, T7 RNA polymerase, NTPs, and MgCl₂.
- Cap Incorporation: Substitute the conventional cap analog (e.g., ARCA) with an equivalent concentration of a PureCap analog (e.g., DiPure (1), DiPure/2'OMe (2), or DiPure/3'OMe (3)) [2].
- Incubation: Incubate the reaction at 37°C for 2–4 hours [2] [28].
B. RP-HPLC Purification
- Sample Preparation: Dilute the IVT reaction mixture with an equal volume of RNase-free water. Centrifuge at >12,000 × g for 2 minutes and pass through a 0.45 μm filter.
- Chromatography System: Use an HPLC system equipped with a binary pump, autosampler, and UV detector (monitoring at 260 nm).
- Column: A C18 column with 300 Å pore size is recommended (e.g., 150 mm length × 4.6 mm diameter, 5 μm particle size) [26].
- Mobile Phase:
- Buffer A: 0.1 M Triethylammonium Acetate (TEAA), pH 7.0.
- Buffer B: 0.1 M TEAA in 70% Acetonitrile.
- Elution Gradient:
- Time: 0 min | %B: 25
- Time: 5 min | %B: 25
- Time: 35 min | %B: 55
- Time: 36 min | %B: 100
- Time: 40 min | %B: 100
- Time: 41 min | %B: 25
- Time: 50 min | %B: 25
- Flow Rate: 1.0 mL/min.
- Collection: Collect the peak corresponding to the hydrophobic-tagged, capped mRNA, which elutes later than the untagged uncapped mRNA and other IVT components [2].
C. Photocleavage and Recovery
- Irradiation: Expose the collected fraction to 365 nm UV light for 15–30 minutes to cleave the 2-nitrobenzyl tag [2].
- Desalting/Buffer Exchange: Use ethanol precipitation, size-exclusion chromatography, or tangential flow filtration to remove cleavage by-products and exchange the mRNA into a storage buffer (e.g., 10 mM Tris-HCl, pH 7.4) [2] [28].
- Quality Control: Determine mRNA concentration by A260 measurement and analyze integrity by capillary electrophoresis or analytical AEX-HPLC [28].
Diagram 1: PureCap mRNA Purification Workflow. This diagram outlines the key steps from in vitro transcription to the final purified product.
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for RP-HPLC-based mRNA Purification
| Item | Function/Description | Example Application/Note |
|---|---|---|
| PureCap Analogs | Hydrophobic, photocleavable cap analogs (e.g., DiPure series). | Enables RP-HPLC separation of capped mRNA; introduced at IVT start [2]. |
| C18 RP-HPLC Column | Stationary phase with C18 ligands; pore size >200 Å. | Large pores accommodate biomolecules; 300 Å pore size is suitable for mRNA [26]. |
| Ion-Pairing Reagents | Buffers like Triethylammonium Acetate (TEAA). | Enhances separation of nucleic acids in RP-HPLC by masking phosphate charge [2]. |
| T7 RNA Polymerase | DNA-dependent RNA polymerase for in vitro transcription. | Highly processive enzyme for mRNA synthesis; a standard for IVT [28]. |
| Nucleoside Triphosphates (NTPs) | ATP, CTP, GTP, UTP; building blocks for RNA synthesis. | Used in equimolar ratios (e.g., 10 mM each) in the IVT reaction [28]. |
| Photocleavage UV Lamp | Light source emitting at 365 nm. | Required for removal of the 2-nitrobenzyl tag from PureCap mRNA [2]. |
Comparison with Related Chromatographic Techniques
It is important to distinguish RP-HPLC from other chromatographic methods that also utilize hydrophobicity, such as Hydrophobic Interaction Chromatography (HIC).
Table 4: RP-HPLC vs. Hydrophobic Interaction Chromatography (HIC)
| Characteristic | RP-HPLC | HIC |
|---|---|---|
| Stationary Phase | Densely covered with long alkyl chains (C8, C18). | Mildly hydrophobic ligands (short alkyl, phenyl) on hydrophilic base [29] [26]. |
| Mobile Phase | Water-miscible organic solvent (acetonitrile, methanol). | High-salt aqueous buffer (e.g., ammonium sulfate); no organic solvent [29] [30]. |
| Elution Mechanism | Decrease in mobile phase polarity (inverse gradient). | Decrease in salt concentration (reverse salt gradient) [29] [30]. |
| Typical Application | Small molecules, peptides, and nucleic acids like mRNA. | Separation of proteins while maintaining native conformation [29] [30]. |
| Impact on Biomolecules | Can denature proteins due to organic solvents. | Considered a "native" technique; preserves 3D structure [29]. |
Diagram 2: RP-HPLC vs. HIC Separation Mechanisms. This diagram contrasts the fundamental drivers of separation and elution in these two hydrophobic techniques.
The purity of messenger RNA (mRNA), particularly the completeness of the 5' cap structure, is a critical determinant for the efficacy and safety of mRNA-based therapeutics. Impurities, such as uncapped mRNA species, can trigger unwanted innate immune responses and reduce protein expression levels, compromising therapeutic outcomes [31] [32]. The PureCap method, developed by researchers at Nagoya University, represents a revolutionary approach to obtaining mRNA with near-perfect capping efficiency [6] [2]. This application note details the final and crucial step in this protocol: the use of photocleavage to release the purification tag and recover a native, footprint-free, fully capped mRNA molecule.
The core innovation of the PureCap method lies in its use of synthetic cap analogs modified with a hydrophobic, photocleavable tag. This tag enables the physical separation of capped mRNA from uncapped impurities using reversed-phase high-performance liquid chromatography (RP-HPLC). Following purification, the tag is removed under mild conditions via light irradiation, yielding a pure, functional mRNA product without any residual chemical modifications that could interfere with its biological activity [2]. This process is summarized in the workflow below.
Key Advantages of the PureCap-Photocleavage Workflow
The combination of the PureCap method with a photocleavage final activation step offers several distinct advantages over traditional mRNA capping and purification techniques.
Table 1: Advantages of the PureCap-Photocleavage Method over Traditional Techniques
| Feature | Traditional Capping Methods (e.g., Co-transcriptional) | PureCap Method with Photocleavage |
|---|---|---|
| Capping Efficiency | 80-90% maximum [2] | 98-100% [6] [2] |
| Separation of Capped mRNA | Not feasible; capped and uncapped mRNA have nearly identical properties [2] | Highly efficient via RP-HPLC, leveraging the hydrophobic tag [2] |
| Final Product | May contain immunogenic uncapped mRNA impurities [2] | Footprint-free native capped mRNA; the tag is completely removed [2] |
| Cap Structure Versatility | Limited by enzymatic specificity and efficiency | Enables production of high-purity Cap0, Cap1, and Cap2 mRNAs for functional comparison [2] |
| Immunogenicity Profile | Higher risk due to uncapped ppp-RNA and dsRNA contaminants [2] [31] | Significantly lower immunostimulatory activity due to high purity [6] [2] |
Materials and Reagents
Research Reagent Solutions
The following table lists the essential materials required for the final activation and overall PureCap protocol.
Table 2: Essential Research Reagents for the PureCap and Photocleavage Workflow
| Item | Function / Description |
|---|---|
| PureCap Analogs | Synthetic cap analogs (e.g., DiPure(1), DiPure/2′OMe(2)) featuring a hydrophobic photocleavable tag (e.g., 2-nitrobenzyl derivative with a tert-butyl group) [2]. |
| In Vitro Transcription (IVT) System | Standard components: T7 RNA Polymerase, ribonucleotide triphosphates (NTPs), DNA template, and reaction buffer. |
| RP-HPLC System | For chromatographic separation; equipped with a C18 or similar reversed-phase column and a UV detector. |
| UV Light Source | Controlled wavelength source (e.g., 365 nm); can be a handheld lamp or chamber system for consistent irradiation [2]. |
| Buffers for RP-HPLC | Mobile phase A: aqueous buffer (e.g., 0.1 M Triethylammonium acetate). Mobile phase B: organic solvent (e.g., acetonitrile). |
| Magnetic Beads (Optional) | For alternative solid-phase mRNA purification, simplifying template removal and reducing DNase I usage [33]. |
Experimental Protocol
mRNA Synthesis and Initial Purification
- DNA Template Preparation: Linearize a plasmid DNA template containing the gene of interest downstream of a T7 promoter. Alternatively, use a PCR-amplified template. For solid-phase transcription, the template may be biotinylated and immobilized on streptavidin-coupled magnetic beads [33].
- In Vitro Transcription (IVT): Perform the IVT reaction using a standard kit. Include the desired PureCap analog (e.g., for Cap1 or Cap2 structures) in the reaction mixture instead of a traditional cap analog. The transcription reaction can be scaled as needed [2] [33].
- RP-HPLC Purification:
- Prepare the IVT reaction mixture for injection into the RP-HPLC system.
- Use a gradient elution with Mobile Phase A (aqueous buffer) and Mobile Phase B (organic solvent, e.g., acetonitrile).
- The hydrophobic tag on the PureCap analog causes the capped mRNA to be retained longer on the column than uncapped mRNA and other impurities like dsRNA.
- Collect the peak corresponding to the tagged, capped mRNA. The purity can be confirmed by analytical RP-HPLC, achieving separations where "the RP-HPLC process had separated completely the capped and uncapped RNAs" [6].
Final Activation: Photocleavage Protocol
This is the critical step to recover the native mRNA.
- Sample Preparation: Place the collected, tagged mRNA fraction from RP-HPLC in a quartz cuvette or a suitable UV-transparent container.
- Photoirradiation:
- Irradiate the sample with UV light at 365 nm [2].
- The required irradiation time and energy dose should be optimized for the specific setup. The reaction is typically performed at room temperature.
- Mechanism of Cleavage: The 2-nitrobenzyl-based photocleavable linker absorbs the UV light, leading to a chemical reaction that severs the bond between the hydrophobic tag and the cap analog. This process leaves behind the native cap structure without any residual chemical "footprint" [2].
- Confirmation of Cleavage: The success of the photocleavage can be confirmed by a shift in retention time on a subsequent RP-HPLC analysis, indicating the removal of the hydrophobic tag. The final product is a solution of highly pure, footprint-free, fully capped mRNA.
Results and Discussion
Quantitative Outcomes of the PureCap Method
Implementation of the PureCap method followed by photocleavage consistently yields superior results compared to standard protocols, as quantified by several key metrics.
Table 3: Quantitative Performance Data of PureCap-Generated mRNA
| Parameter | Result from PureCap Method | Note / Comparative Baseline |
|---|---|---|
| Capping Efficiency | 98% - 100% [6] [2] | Traditional methods: 80-90% [2] |
| Protein Synthesis Capacity (Cap2 mRNA) | 3 to 4-fold higher than Cap1 mRNA from standard methods [6] [2] | Measured in cultured cells and animal models. |
| Immunostimulatory Activity | Lower than mRNAs from conventional techniques [6] [2] | Reduced activation of innate immune receptors (e.g., RIG-I) due to high purity and advanced cap structures. |
| mRNA Length Versatility | Successful purification of mRNAs from 650 nt to 4,247 nt [2] | Demonstrates the method's applicability for a wide range of therapeutic constructs. |
Functional Significance of a Footprint-Free Product
The photocleavage step is essential for translating high purity into high functionality. Leaving any part of the purification tag on the mRNA could potentially:
- Hinder Recognition by Translation Machinery: The eukaryotic translation initiation factor 4E (eIF4E) must bind the 5' cap to initiate translation. A residual tag could sterically block this interaction, reducing protein yield.
- Alter Immunogenic Properties: An unnatural chemical moiety on the cap could be recognized as a non-self element by the immune system, potentially leading to increased reactogenicity or accelerated clearance of the therapeutic.
The PureCap method's use of a meticulously designed photocleavable linker ensures that the cap structure returned to its native state is functionally identical to endogenous mRNA, enabling optimal translation and minimal immune activation [2]. This results in vaccines and therapeutics with higher efficacy and a better safety profile.
The final activation via photocleavage is the pivotal step that unlocks the full potential of the PureCap method. It seamlessly bridges the gap between a highly efficient purification process and the delivery of a biologically pristine, fully functional mRNA product. By ensuring the recovery of a footprint-free native cap, this protocol guarantees that the superior purity achieved through RP-HPLC directly translates into enhanced translational efficiency and reduced immunogenicity. This integrated approach provides researchers and drug developers with a robust, scalable, and revolutionary tool for producing next-generation mRNA therapeutics.
The 5' cap is a critical modification for messenger RNA (mRNA), essential for its stability, efficient translation, and ability to evade the innate immune system. This structure consists of a 7-methylguanosine (m7G) linked to the first transcribed nucleotide of the mRNA via a 5'-5' triphosphate bridge [2] [34]. The extent of methylation on the ribose of the subsequent nucleotides defines the major cap variants found in eukaryotic cells. Cap 0 (m7GpppN) possesses no additional ribose methylations, while Cap 1 (m7GpppNm) features a 2'-O-methyl group on the first transcribed nucleotide. The Cap 2 (m7GpppNmpNm) structure carries this 2'-O-methylation on both the first and second nucleotides [13] [35]. The Cap 1 structure is predominant in higher eukaryotes and is crucial for distinguishing "self" RNA from "non-self" viral RNA, thereby preventing an undesirable immune response [34] [35]. Although less studied, Cap 2 is present in some viral and higher eukaryotic RNAs and recent evidence suggests it may further enhance translation and reduce immunogenicity [2] [35].
The purification of completely capped mRNA populations remains a significant challenge in the production of therapeutic mRNA. Standard capping methods, whether enzymatic or co-transcriptional, typically achieve capping efficiencies of 80-95%, leaving immunogenic uncapped mRNA species as byproducts [2] [13]. This application note details methodologies for synthesizing Cap-0, Cap-1, and Cap-2 mRNA constructs, with a specific focus on the innovative PureCap method, which enables the purification of fully capped mRNA with 100% capping efficiency.
Established mRNA Capping Methodologies
Co-transcriptional Capping with Cap Analogs
Co-transcriptional capping involves incorporating synthetic cap analogs during the in vitro transcription (IVT) reaction. This approach streamlines mRNA production by combining synthesis and capping into a single step.
- mCap (m7GpppG): This first-generation Cap-0 analog is incorporated in both correct and reverse orientations during transcription, resulting in approximately 50% of capped mRNA being untranslatable. It requires a high cap:GTP ratio (~4:1), yielding capping efficiencies around 70% but reducing overall full-length mRNA yield [13].
- Anti-Reverse Cap Analog (ARCA): Designed to prevent reverse incorporation, ARCA (m7(3'-O-Me)GpppG) ensures all capped mRNAs are translatable. However, it still requires a high cap:GTP ratio, which suppresses transcription yield, and typically achieves Cap-0 capping efficiencies between 50-80% [2] [13] [34].
- CleanCap Reagent: This newer technology uses trinucleotide cap analogs to co-transcriptionally produce Cap-1 mRNA with high efficiency (>95%) without requiring an unfavorable cap:GTP ratio, thus maintaining high mRNA yields. A limitation is its requirement for an AG initiation sequence instead of the standard GG [13].
- Tetranucleotide Cap Analogs: Recent advances have enabled the synthesis of tetranucleotide cap analogs, such as m7GpppNmpGmpG, which allow for the direct co-transcriptional synthesis of Cap-2 mRNA. These tools provide a novel means to study the impact of cap methylation status on mRNA functionality [35].
Post-transcriptional Enzymatic Capping
Enzymatic capping occurs after the IVT reaction is complete. The process typically uses the Vaccinia Virus Capping Enzyme (VCE), which possesses both guanylyltransferase and methyltransferase activities, to add a Cap-0 structure. To achieve a Cap-1 structure, a subsequent methylation step with a dedicated 2'-O-methyltransferase (2'-O-MTase) is required [34]. While this method offers high capping efficiency and precise control over the cap structure, it introduces additional enzymatic steps and purification processes, increasing production time and cost [2] [34].
Table 1: Comparison of Common mRNA Capping Methods
| Capping Method | Cap Structure | Typical Efficiency | Key Advantages | Key Limitations |
|---|---|---|---|---|
| mCap | Cap 0 | ~70% | Simple, widely available | Low efficiency, reverse incorporation |
| ARCA | Cap 0 | 50-80% | Prevents reverse incorporation | High cap:GTP ratio lowers yield |
| CleanCap | Cap 1 | >95% | High efficiency & yield, simple workflow | Requires AG start codon |
| Enzymatic Capping | Cap 0 or Cap 1 | High (>90%) | High precision, flexible sequence | Multi-step, time-consuming, costly |
| Tetranucleotide Analogs | Cap 2 | Variable (tool-dependent) | Enables direct Cap-2 synthesis | Complex synthesis, not yet widespread |
The PureCap Method for Complete Capping and Purification
Principle and Workflow
The PureCap method is an innovative co-transcriptional capping approach designed to overcome the fundamental limitation of incomplete capping efficiency. The core innovation involves cap analogs modified with a hydrophobic, photocleavable tag [2]. These custom analogs, such as DiPure (1), DiPure/2'OMe (2), and DiPure/3'OMe (3), are incorporated into the mRNA during IVT. The incorporated hydrophobic tag then enables the physical separation of capped mRNA from uncapped mRNA impurities using Reversed-Phase High-Performance Liquid Chromatography (RP-HPLC), a standard and scalable purification process. Following purification, brief photo-irradiation cleaves the tag, yielding footprint-free, natively capped mRNA with 100% capping efficiency [2].
The following workflow diagram illustrates the key steps of the PureCap method:
Advantages of the PureCap Technology
The PureCap method offers several distinct advantages for research and therapeutic applications:
- 100% Capping Efficiency: It achieves complete separation of capped mRNA from uncapped byproducts, resulting in a perfectly capped final product. This eliminates the immunogenic risk posed by 5'-triphosphate uncapped mRNA [2].
- Versatility across Cap Structures and Lengths: The technology has been successfully applied to produce Cap-0, Cap-1, and Cap-2 mRNAs, as well as mRNAs of varying lengths (e.g., 650 nt and 4,247 nt) [2].
- Simultaneous Impurity Removal: The RP-HPLC purification step also removes other significant impurities generated during IVT, such as double-stranded RNA (dsRNA), which are potent inducers of innate immune responses and inhibitors of translation [2].
- An Unbiased Platform for SAR Studies: By guaranteeing 100% capping efficiency, the PureCap method allows for a precise comparison of the structure-activity relationships of different cap variants (Cap-0, Cap-1, Cap-2) without the confounding variable of varying capping efficiencies [2].
Experimental Protocol: Synthesis of Cap-2 mRNA Using PureCap Analogs
This protocol provides a detailed methodology for synthesizing fully capped Cap-2 mRNA, utilizing a hydrophobic photocleavable cap analog, based on the procedure described by [2].
Materials and Reagents
Table 2: Research Reagent Solutions for PureCap mRNA Synthesis
| Item | Function/Description | Supplier Examples |
|---|---|---|
| PureCap Cap Analog (e.g., DiPure/2'OMe) | Co-transcriptional capping with hydrophobic tag for purification | Custom Synthesis [2] |
| T7 RNA Polymerase | Enzyme for in vitro transcription | Thermo Fisher Scientific, New England Biolabs |
| Ribonucleotide Triphosphates (NTPs) | Building blocks for RNA synthesis | Thermo Fisher Scientific, TriLink BioTechnologies |
| DNA Template | Linearized plasmid with promoter (T7) and gene of interest | - |
| RP-HPLC Column | Stationary phase for purification (e.g., C18) | Agilent, Waters |
| UV Light Source | For cleaving the photocleavable tag (∼350-365 nm) | - |
Step-by-Step Procedure
In Vitro Transcription (IVT) Setup
- Assemble the IVT reaction on ice in a nuclease-free microcentrifuge tube:
- 1 µg of linearized DNA template
- 10 µL of 10x Transcription Buffer
- 8 µL of 75 mM NTP Mix (ATP, CTP, UTP, GTP)
- 4 µL of 20 mM PureCap Cap Analog (e.g., DiPure/2'OMe for Cap-2)
- 2 µL (100 U) of T7 RNA Polymerase
- Nuclease-free water to a final volume of 100 µL
- Gently mix and centrifuge briefly. Incubate at 37°C for 2-4 hours.
- Assemble the IVT reaction on ice in a nuclease-free microcentrifuge tube:
RP-HPLC Purification of Capped mRNA
- Terminate the IVT reaction by adding 10 µL of 0.5 M EDTA, pH 8.0.
- Dilute the reaction mixture with an equal volume of RP-HPLC binding buffer.
- Load the sample onto a C18 RP-HPLC column equilibrated with the binding buffer.
- Elute the RNA using an acetonitrile gradient. The hydrophobic tag on the PureCap analog will cause the correctly capped mRNA to elute later than the uncapped mRNA.
- Collect the fraction containing the capped mRNA based on UV absorbance and retention time.
Photocleavage of the Hydrophobic Tag
- Place the collected fraction containing the capped mRNA in a transparent quartz cuvette or tube.
- Irradiate the sample with long-wave UV light (∼350-365 nm) for 15-30 minutes on ice to cleave the 2-nitrobenzyl-based tag.
- Confirm tag removal and cap integrity via LC-MS or other analytical methods.
mRNA Recovery and Quantification
- Precipitate the mRNA by adding 1/10 volume of 3 M sodium acetate (pH 5.2) and 2.5 volumes of cold ethanol. Incubate at -20°C for 1 hour.
- Centrifuge at >12,000 × g for 15 minutes at 4°C. Wash the pellet with 70% ethanol, air-dry, and resuspend in nuclease-free water.
- Quantify the mRNA concentration by measuring absorbance at 260 nm. Assess integrity and purity by capillary electrophoresis or agarose gel electrophoresis.
Analytical Techniques for Capping Efficiency and mRNA Integrity
Rigorous quality control is essential for characterizing capped mRNA products. The following table summarizes key analytical methods.
Table 3: Analytical Methods for Capped mRNA Quality Control
| Analytical Method | Parameter Measured | Principle | Considerations |
|---|---|---|---|
| Liquid Chromatography-Mass Spectrometry (LC-MS) | Capping efficiency, Cap structure identity | Separates and quantifies capped/uncapped RNA species following enzymatic digestion. | High precision; requires specialized equipment and expertise [36]. |
| 5' CapQ Assay | Intact mRNA with 5' cap and 3' poly(A) tail | Microarray immunoassay using anti-cap antibody and fluorescent poly(T) detection probe. | Rapid (2 hrs), benchtop-friendly; provides a single measure of intact mRNA [37] [36]. |
| Ribozyme Assay + PAGE/LC-MS | Capping efficiency | Ribozyme cleaves mRNA near 5' end; short 5' fragments are analyzed by denaturing PAGE or LC-MS. | Allows efficiency measurement in diverse mRNA constructs [34]. |
| Capillary Electrophoresis (CE) | mRNA Integrity | Separates RNA fragments by size-to-charge ratio in a capillary. Assesses RNA degradation. | High resolution; does not provide cap-specific information [36]. |
Functional and Comparative Analysis of Cap Constructs
The biological performance of mRNA is heavily influenced by its cap structure. Research utilizing precise capping methods like PureCap and tetranucleotide analogs has yielded key insights:
- Translation Efficiency: Cap-2 mRNA can demonstrate significantly enhanced protein production. Studies have shown that Cap-2 mRNA exhibits up to 3- to 4-fold higher translation activity in cultured cells and animals compared to Cap-1 mRNA prepared by standard capping methods [2]. The effect of Cap-2 on translation can be cell-line specific, sometimes showing no significant difference or even a slight reduction compared to Cap-1, highlighting the importance of cellular context [35].
- Immunogenicity: The primary role of Cap-1 is to evade the innate immune system by preventing recognition by proteins like IFIT1 [35]. Cap-2 structure appears to further reduce immunogenicity by drastically reducing mRNA affinity to RIG-I, an innate immune receptor, compared to Cap-1 [2].
- mRNA Stability: Cap-2 mRNA shows unique resistance to certain decay pathways. While it remains susceptible to decapping by DCP2, it is resistant to DXO (Decapping Exoribonuclease)-mediated decapping and degradation, potentially contributing to its extended half-life in some systems [35].
The synthesis of high-quality, properly capped mRNA is a cornerstone of effective mRNA therapeutics and vaccines. While established methods like ARCA, CleanCap, and enzymatic capping are capable of producing Cap-0 and Cap-1 mRNAs with good efficiency, the PureCap method represents a significant technological advance. Its ability to deliver mRNA with 100% capping efficiency, coupled with its versatility across different cap structures and transcript lengths, makes it an invaluable tool for both basic research and clinical applications. The precise synthesis of Cap-2 mRNA, enabled by technologies like PureCap and tetranucleotide analogs, opens new avenues for optimizing mRNA therapeutics, as Cap-2 constructs offer the potential for enhanced translation, reduced immunogenicity, and improved stability. As the field progresses, the choice of capping strategy and purification protocol will remain critical for maximizing the efficacy and safety of mRNA-based products.
Overcoming Production Hurdles and Maximizing PureCap Efficiency
Addressing Synthesis Challenges of Hydrophobic Cap Analogs
In mRNA therapeutic development, the 5' cap structure is critical for stability, translation efficiency, and immune evasion. Conventional co-transcriptional capping methods, which utilize cap analogs during in vitro transcription (IVT), are inherently inefficient, typically achieving capping rates of only 80-90% [38]. The remaining uncapped mRNA impurities possess nearly identical physicochemical properties to their capped counterparts, making physical separation extremely difficult. These uncapped species can trigger undesired immune responses via innate immune receptors like RIG-I, compromising therapeutic safety and efficacy [38] [6].
The PureCap method overcomes this fundamental limitation through a novel class of cap analogs engineered with a strategic hydrophobic modification. These PureCap analogs are designed with a photocleavable tag, featuring a hydrophobic 2-nitrobenzyl (Nb) group coupled with a tert-butyl (tBu) moiety to enhance hydrophobicity and stability [38]. During IVT, the analog is incorporated at the 5' end of the mRNA. The attached hydrophobic tag then enables the facile separation of capped from uncapped mRNA using standard reversed-phase high-performance liquid chromatography (RP-HPLC). A final photo-irradiation step removes the tag, yielding footprint-free, natively structured, fully capped mRNA with 100% efficiency [38] [39]. This application note details the protocols and considerations for synthesizing and applying these hydrophobic cap analogs.
Experimental Protocols
Design and Synthesis of PureCap Analogs
The synthesis of hydrophobic cap analogs addresses the solubility challenges of intermediates in aqueous solvents by employing a streamlined, one-pot phosphorylation approach [38].
Key Protocol Steps:
- Precursor Synthesis: Begin with the synthesis of guanosine derivatives modified at specific positions with the o-nitrobenzyl-based hydrophobic tag. For example, DiPure (1) features the Nb group introduced via an acetal at the 2'-O position, leaving the 3'-OH free to prevent inhibition of chain elongation [38].
- One-Pot Diphosphate Synthesis: To circumvent solubility issues and multiple purification steps, employ a direct one-pot synthesis of the diphosphate from the nucleoside precursor. This can be achieved via:
- Methylation and Coupling: The resulting diphosphate is subsequently methylated at the N7 position. The final dinucleotide PureCap analog is formed through a condensation reaction with a guanosine monophosphate imidazolide, catalyzed by zinc chloride in anhydrous DMF. The Lewis acid is crucial for coordinating the reactants and achieving high yields [38] [40].
- Purification: Purify the final cap analog using anion exchange chromatography (e.g., DEAE Sepharose) with a triethylammonium bicarbonate (TEAB) buffer gradient, followed by reversed-phase chromatography if necessary [38] [40].
mRNA Transcription and Purification Using PureCap Analogs
This protocol outlines the use of synthesized PureCap analogs for the production of highly pure, capped mRNA.
Key Protocol Steps:
- In Vitro Transcription (IVT): Perform a standard IVT reaction, substituting a PureCap analog (e.g., DiPure/2'OMe) for the conventional cap analog. The IVT reaction conditions (polymerase, NTP concentrations, buffer) remain largely unchanged [38].
- RP-HPLC Purification: After transcription, purify the mRNA using RP-HPLC.
- Principle: The hydrophobic tag on the PureCap analog increases the retention time of successfully capped mRNA, while uncapped mRNA and other impurities (like double-stranded RNA) elute earlier [38] [6].
- Procedure: Load the IVT reaction mixture onto a C18 or similar reversed-phase column. Employ a gradient of water and acetonitrile. The capped mRNA fraction will be the last major peak to elute. Collect this fraction separately.
- Photocleavage: Recover the native cap structure by removing the hydrophobic tag.
- Procedure: Expose the collected mRNA fraction to 365 nm UV light. The 2-nitrobenzyl group is photocleaved, efficiently removing the hydrophobic tag without leaving a molecular footprint on the mRNA [38] [39].
- Outcome: This step yields 100% pure, natively capped mRNA, as confirmed by analytical techniques [6].
Quantitative Data and Performance
The performance of mRNA synthesized via the PureCap method significantly surpasses that of mRNA produced with conventional capping methods.
Table 1: Comparative Performance of PureCap vs. Conventional Capping Methods
| Parameter | Conventional Co-Transcriptional Capping | PureCap Method |
|---|---|---|
| Capping Efficiency | ~80-90% [38] | ~100% [38] [6] |
| Applicable mRNA Length | Varies with method | Demonstrated for 650 nt and 4,247 nt mRNAs [38] |
| Separation of Capped/Uncapped mRNA | Not feasible by standard physical methods | Achieved via RP-HPLC [38] |
| Immunostimulatory Activity (of capped product) | Higher due to uncapped impurity | Significantly lower [6] |
Table 2: Translation Activity of mRNA with Different Cap Structures Purified via PureCap
| Cap Structure | Description | Relative Translation Activity |
|---|---|---|
| Cap-0 | m7GpppN... | Baseline |
| Cap-1 | m7GpppNm... (conventional high-grade) | Baseline |
| Cap-2 | m7GpppNmNm... (produced via PureCap) | 3- to 4-fold higher than Cap-1 [38] |
Visual Experimental Workflow
The following diagram illustrates the logical flow of the PureCap method, from synthesis to pure mRNA production.
The Scientist's Toolkit: Essential Research Reagents
This table lists key reagents and their functions for implementing the PureCap method.
Table 3: Key Reagents for the PureCap Method
| Reagent / Material | Function / Description |
|---|---|
| PureCap Analogs (e.g., DiPure) | Dinucleotide cap analogs (e.g., m7GpppG) modified with a hydrophobic, photocleavable 2-nitrobenzyl tag. Serves as the initiation nucleotide for IVT and the purification handle [38]. |
| RNA Polymerase (T7, SP6, T3) | Standard enzyme for in vitro transcription (IVT). Incorporates the PureCap analog at the 5' end of the mRNA transcript [41]. |
| Reversed-Phase HPLC System | Chromatography system equipped with a C18 or similar column. Essential for separating capped mRNA (hydrophobic) from uncapped mRNA and other impurities based on hydrophobicity [38] [6]. |
| UV Light Source (365 nm) | Light source for the photocleavage reaction. Removes the 2-nitrobenzyl tag from the purified mRNA after HPLC, restoring the native cap structure without leaving a footprint [38] [39]. |
| Anion Exchange Resin (e.g., DEAE) | Used for the purification of synthesized PureCap analogs, separating them from reaction byproducts based on charge [38] [40]. |
| Zinc Chloride (Anhydrous) | Lewis acid catalyst. Critical for the efficient coupling of the diphosphate and guanosine monophosphate imidazolide during the final stage of cap analog synthesis [38] [40]. |
Optimizing RP-HPLC Conditions for Different mRNA Lengths
The purification of completely capped mRNA is a critical determinant of the efficacy and safety of mRNA-based therapeutics and vaccines. The PureCap method offers a robust approach to obtaining highly pure capped mRNA, which has been demonstrated to enhance protein expression and reduce undesired immunogenicity by minimizing type I interferon responses [42]. Reversed-phase high-performance liquid chromatography (RP-HPLC) serves as a powerful analytical and preparative technique for assessing the quality of these products, enabling the separation of full-length mRNA from key impurities such as truncated transcripts and double-stranded RNA (dsRNA). This application note provides detailed protocols for the optimization of RP-HPLC conditions to achieve high-resolution separation of mRNA across a range of lengths, directly supporting the production and quality control of PureCap-generated mRNA.
The therapeutic application of messenger RNA (mRNA) necessitates rigorous purification and analysis to ensure the final product is free from impurities that can compromise efficacy and safety. Contaminants such as truncated mRNA, dsRNA, and incompletely capped species can stimulate innate immune responses and reduce translational efficiency [42] [43]. The PureCap method facilitates the production of mRNA with a defined, complete 5' cap structure, which is vital for stability and efficient translation. Verifying the integrity and purity of this product requires sophisticated analytical methods.
RP-HPLC is a cornerstone technique for the analysis of mRNA critical quality attributes (CQAs). Its effectiveness, however, depends heavily on the careful optimization of parameters such as column pore size, mobile phase composition, and operating temperature. This document outlines a systematic approach to method development, providing validated protocols for the analysis of mRNA, from short transcripts to species several thousand nucleotides in length.
Key Principles of RP-HPLC for mRNA Separation
The separation of mRNA via RP-HPLC relies on the hydrophobic interaction between the nucleic acid backbone and the stationary phase. The extensive negatively charged phosphodiester backbone of mRNA makes it highly polar, yet its single-stranded nature allows for dynamic secondary and tertiary structures that can complicate analysis [44]. The use of ion-pairing reagents in the mobile phase is essential to neutralize these charges and promote retention on reversed-phase columns [45]. The choice of column, particularly the pore size of the packing material, is a critical factor for resolving long RNA strands, as it must be large enough to permit full access of the macromolecule to the hydrophobic surface within the pores [46].
Optimized RP-HPLC Conditions for Different mRNA Lengths
General Considerations and Reagents
The following reagents and conditions form the basis for robust RP-HPLC analysis of mRNA. Optimization should be performed around this core set of parameters.
- Ion-Pairing Reagent: Triethylammonium acetate (TEAA) is the standard ion-pairing agent. A common concentration is 0.1 M, prepared from a 1 M stock solution [46] [47].
- Organic Modifier: Acetonitrile is typically used for gradient elution [47].
- Column Temperature: Elevated temperatures (e.g., 60°C) are recommended to minimize secondary structure and maintain mRNA in a denatured state, leading to more predictable chromatography [46].
- Detection: UV detection at 260 nm is standard for mRNA analysis.
Systematic Optimization Guide
The table below summarizes optimized RP-HPLC conditions for different mRNA length ranges, based on experimental data.
Table 1: Optimized RP-HPLC Conditions for Different mRNA Lengths
| mRNA Length Range | Recommended Column | Pore Size | Mobile Phase | Temperature | Key Application |
|---|---|---|---|---|---|
| 50 - 1,000 nt | Octadecyl (C18)-based column (e.g., RNA-RP1) | > 30 nm (super-wide pore) | 0.1 M TEAA / Acetonitrile | 60°C | Separation of single-stranded and double-stranded RNAs in non-denatured states [46]. |
| > 1,000 nt (several thousand) | Octadecyl (C18)-based column (e.g., RNA-RP1) | > 30 nm (super-wide pore) | 0.1 M TEAA / Acetonitrile | 60°C | Successful separation of full-length mRNA from impurities in IVT samples [46]. |
| Oligonucleotides (for mapping) | Acquity Premier Oligonucleotide BEH C18 | Not Specified | 0.1 M TEAA, HFIP / Acetonitrile | Not Specified | High-resolution separation of oligonucleotide fragments (e.g., 6-20 mers) for mRNA identity testing [45] [47]. |
Detailed Experimental Protocol
Protocol: Analysis of an IVT mRNA Sample via RP-HPLC
This protocol describes the steps to analyze and separate a long-stranded mRNA (e.g., ~4000 nt) from its impurities using an optimized RP-HPLC method.
I. Materials and Equipment
- HPLC System: UHPLC or HPLC system capable of stable gradients and column heating to 60°C.
- Column: Octadecyl-based column with super-wide pores (>30 nm), such as the RNA-RP1 column [46].
- Mobile Phase A: 0.1 M Triethylammonium Acetate (TEAA) in water. Filter and degas before use.
- Mobile Phase B: Acetonitrile (LC-MS grade). Filter and degas before use.
- mRNA Sample: In vitro transcribed mRNA, purified or crude, resuspended in RNase-free water.
- Vials: HPLC-compatible vials.
II. Method Parameters
- Column Temperature: 60°C
- Flow Rate: 0.2 - 0.5 mL/min (adjust based on column specifications and backpressure)
- Detection: UV at 260 nm
- Injection Volume: 1-10 µL (depending on sample concentration)
Gradient Program: A generic starting gradient for method development is suggested below. Optimal slopes and hold times must be determined empirically for each specific mRNA sample.
Time (min) % Mobile Phase A % Mobile Phase B 0 95 5 2 95 5 20 70 30 25 10 90 30 10 90 31 95 5 40 95 5
III. Procedure
- System Preparation: Equilibrate the HPLC system and the column with the starting mobile phase (95% A, 5% B) for at least 5 column volumes or until a stable baseline is achieved.
- Sample Preparation: Dilute the mRNA sample to an appropriate concentration (e.g., 0.1-1 mg/mL) in RNase-free water. Centrifuge briefly to remove any particulate matter.
- Sample Injection: Load the sample into an HPLC vial and place it in the autosampler. Inject the specified volume.
- Chromatographic Run: Initiate the method with the gradient program described above.
- Data Analysis: Identify peaks corresponding to the full-length mRNA, truncated RNA species, and other impurities based on retention time. The purity can be assessed by integrating the peak area of the main product relative to the total peak area.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Materials for mRNA Analysis by RP-HPLC
| Item | Function / Application |
|---|---|
| Octadecyl (C18) Column with >30 nm Pores | Stationary phase designed for large biomolecules; super-wide pores allow access and separation of long mRNA strands [46]. |
| Triethylammonium Acetate (TEAA) | Volatile ion-pairing reagent that masks the negative charge on the mRNA backbone, enabling retention on RP columns [45] [47]. |
| 1,1,1,3,3,3-Hexafluoro-2-propanol (HFIP) | Additive used in IP-RPLC to improve peak shape and resolution for oligonucleotide separations, particularly with MS detection [47]. |
| RNase T1 Enzyme | Specific endoribonuclease used to digest mRNA into defined oligonucleotide fragments for identity testing and poly(A) tail analysis [48] [47]. |
| Thermostatable Column Oven | Equipment to maintain consistent and elevated column temperature (e.g., 60°C), which is crucial for denaturing mRNA secondary structures [46]. |
Analytical Workflow for mRNA Characterization
The analysis of PureCap mRNA involves multiple techniques to fully characterize its critical quality attributes. RP-HPLC is a key component of a broader analytical workflow.
The optimization of RP-HPLC methods is indispensable for the development and quality control of next-generation mRNA therapeutics, particularly those produced using the PureCap method. The key to success lies in selecting a stationary phase with an appropriate pore size (>30 nm) and operating the system at an elevated temperature of 60°C with a volatile ion-pairing mobile phase. The protocols and conditions detailed herein provide a reliable foundation for researchers to separate mRNA based on length, assess the purity of their samples, and ensure the production of a safe and highly efficacious final product. As the field of mRNA therapeutics continues to expand, robust and reproducible analytical methods like these will be crucial for regulatory compliance and clinical success.
Ensuring Complete Photocleavage and mRNA Integrity
The production of messenger RNA (mRNA) for therapeutic applications requires stringent quality control to ensure efficacy and safety. A critical quality attribute is the presence of a complete 5' cap structure, which enhances translation and stability while reducing immunogenicity. The PureCap method represents a revolutionary approach to obtaining mRNA with 100% capping efficiency by incorporating a hydrophobic, photocleavable tag during transcription, enabling purification via reversed-phase high-performance liquid chromatography (RP-HPLC) and subsequent tag removal by light irradiation [2] [6]. This application note details protocols for ensuring complete photocleavage of this tag and for assessing the integrity of the final mRNA product, which are essential steps for generating high-quality therapeutic mRNA.
Experimental Protocols
Protocol 1: Optimized Photocleavage of PureCap mRNA
This protocol describes the process for removing the hydrophobic photocleavable tag from purified PureCap mRNA to yield native, functional mRNA.
Materials
- PureCap mRNA: mRNA synthesized using PureCap analogs (e.g., DiPure/2′OMe, DiPure/3′OMe) [2].
- Irradiation Equipment: Light source emitting at 365 nm (LED-based recommended) or 405 nm [2] [49].
- Reaction Vessel: Low-protein-binding microcentrifuge tubes or a quartz cuvette for bulk processing.
- Nuclease-free Water or Buffer.
Methodology
- Sample Preparation: Dilute the purified, tag-modified PureCap mRNA to a concentration of 0.1–0.5 mg/mL in nuclease-free water or a compatible aqueous buffer (e.g., Tris-EDTA, pH 7.0-7.5). Distribute the sample in a thin-walled vessel to ensure uniform light penetration. A recommended sample volume is 50-100 µL per 0.2 mL tube.
- Irradiation Parameters:
- Wavelength: 365 nm is highly effective. As reported for similar photocaged caps, irradiation at 405 nm or even 420 nm can also be used, though it may require longer exposure times [49].
- Power Intensity: A power density of 140 mW cm⁻² at 365 nm has been successfully used for analogous systems [49].
- Duration: For a 100 µL sample irradiated at 365 nm (140 mW cm⁻²), a duration of 5–15 seconds is typically sufficient for complete deprotection. When using 405 nm, extend the irradiation time to approximately 60 seconds [49].
- Irradiation Procedure: Place the sample tube uncapped directly under the light source. Initiate irradiation for the predetermined time. Gently agitate the tube halfway through the process if the setup does not include a built-in mixer.
- Post-Irradiation Processing: Confirm complete photocleavage using an analytical method such as RP-HPLC or capillary electrophoresis (CE). The purified mRNA is now ready for downstream applications or integrity assessment.
Table 1: Key Parameters for Photocleavage Optimization
| Parameter | Recommended Condition | Alternative/Note |
|---|---|---|
| Wavelength | 365 nm | 405 nm, 420 nm (for NPM-group) [49] |
| Power Density | 140 mW cm⁻² [49] | Adjust time accordingly if power varies |
| Irradiation Time | 5-15 sec (at 365 nm) | Up to 60-120 sec at longer wavelengths [49] |
| Sample Volume | 50-100 µL (in a 0.2 mL tube) | Scale vessel size for larger volumes |
| Purity Post-Cleavage | >98% (including 4247 nt mRNA) [6] | Confirm via RP-HPLC/CE |
Protocol 2: Assessment of mRNA Integrity and Purity
Following photocleavage, it is critical to verify that the mRNA backbone has not been degraded during the process. Capillary Electrophoresis (CE) is a high-resolution method ideal for this purpose.
Materials
- Capillary Electrophoresis Instrument: Agilent Bioanalyzer 2100 (with RNA 6000 Nano Kit), Fragment Analyzer (with Agilent HS RNA Kit), or equivalent systems like LabChip GXII or PA800 Plus [50].
- CE Kits: As listed in Table 2.
- Nuclease-free Water.
- RNA Marker/Ladder.
Methodology
- Sample Preparation: Dilute the photocleaved mRNA sample according to the specific CE kit's instructions. A typical concentration range is 25–500 ng/µL.
- Instrument Setup: Prime the capillary or microchip as per the manufacturer's protocol for the selected kit.
- Loading and Run: Load the RNA ladder and samples into the designated wells. Initiate the separation run. The entire process typically takes between 5 and 50 minutes, depending on the kit [50].
- Data Analysis:
- Integrity: A single, sharp peak corresponding to the expected length of the full-length mRNA indicates high integrity. The presence of multiple smaller peaks or a smear suggests degradation.
- Purity: The electropherogram should be inspected for the absence of significant peaks corresponding to impurities like uncapped mRNA or double-stranded RNA (dsRNA).
- Quantification: Software provided with the instrument (e.g., Bioanalyzer 2100 Expert software) automatically calculates the RNA Integrity Number (RIN) or similar metrics.
Table 2: Comparison of Capillary Electrophoresis Methods for mRNA Analysis [50]
| CE Method / Kit | Recommended Instrument | Key Strength | Typical Analysis Time |
|---|---|---|---|
| Sciex RNA 9000 Purity and Integrity | PA800 Plus | Highest selectivity and resolving power for in-depth characterization | Not Specified |
| Agilent RNA 6000 Nano | Bioanalyzer 2100 | Fast analysis, suitable for screening | Faster |
| Revvity RNA Reagent | LabChip GXII | Fast analysis, suitable for screening | Faster |
| Agilent HS RNA Kit | Fragment Analyzer | Fast analysis, suitable for screening | Faster |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Kits for PureCap Workflow and Analysis
| Item | Function/Description | Example/Note |
|---|---|---|
| PureCap Cap Analogs | Dinucleotide analogs (e.g., DiPure/2'OMe) with a hydrophobic, photocleavable tag for co-transcriptional capping and subsequent RP-HPLC purification [2]. | Synthesized to enable 100% capping efficiency [2]. |
| RP-HPLC System | For the primary purification step, separating capped mRNA (with hydrophobic tag) from uncapped mRNA based on hydrophobicity [2] [6]. | Critical for obtaining high-purity capped mRNA pre-photocleavage. |
| Controlled Light Source | LED-based irradiator for precise and efficient removal of the photocleavable tag from the PureCap mRNA. | Wavelengths: 365 nm, 405 nm [49]. |
| CE Instrument & Kits | High-resolution analytical tools for assessing mRNA integrity, purity, and stability post-photocleavage [50]. | Sciex RNA 9000 kit is noted for highest selectivity [50]. |
| In Vitro Transcription Kit | Kit optimized for use with cap analogs to produce high yields of full-length mRNA. | Can be used with PureCap analogs for synthesis [2]. |
Workflow and Pathway Visualizations
Diagram 1: PureCap mRNA Workflow
Diagram 2: Photocleavage Validation
Scaling Up the Process for Commercial mRNA Production
The commercial success of messenger RNA (mRNA) vaccines and therapeutics hinges on the development of robust, scalable production processes that deliver products with well-defined critical quality attributes. A central challenge in this endeavor is ensuring the complete capping of mRNA transcripts, as uncapped mRNA impurities can trigger undesirable immune responses and reduce translational efficiency [6] [51]. The PureCap method, developed by researchers at Nagoya University, represents a revolutionary advance by enabling the production of mRNA with capping efficiencies of 98-100% [6] [2]. This application note details the integration of the PureCap purification technology into a scalable, cGMP-compliant mRNA manufacturing process, providing a structured framework for researchers and process development scientists aiming to transition from research to commercial production.
The 5' cap structure is indispensable for mRNA function, enhancing translation, stabilizing the molecule, and reducing immunogenicity by marking the transcript as "self" [2] [51]. Conventional co-transcriptional capping methods, which use cap analogs like Anti-Reverse Cap Analog (ARCA), typically achieve capping efficiencies of only 80-90% [2]. The remaining uncapped mRNA, bearing a 5' triphosphate, can act as a pathogen-associated molecular pattern, stimulating innate immune receptors such as RIG-I and MDA5, which leads to inflammatory responses and potential side effects at the injection site [6] [2]. The PureCap method directly addresses this purity bottleneck, facilitating the manufacture of safer and more potent mRNA pharmaceuticals.
The PureCap Method: Principle and Advantages
The PureCap technology is a novel capping and purification platform that employs custom-synthesized hydrophobic cap analogs to facilitate the physical separation of fully capped mRNA from reaction impurities [2]. The core innovation involves a cap analog modified with a photocleavable hydrophobic tag, such as a tert-butyl-containing 2-nitrobenzyl (tBu-Nb) group [2]. This tag is incorporated into the mRNA during the in vitro transcription (IVT) reaction via T7 RNA polymerase.
The subsequent purification workflow leverages reversed-phase high-performance liquid chromatography (RP-HPLC), a standard and scalable unit operation. The hydrophobic tag causes the desired, capped mRNA to elute at a different retention time compared to the uncapped mRNA and other impurities like double-stranded RNA (dsRNA) [6] [2]. Following chromatography, the hydrophobic tag is cleanly removed by mild UV irradiation, yielding footprint-free, natively capped mRNA [2]. This process is versatile and can be used to produce various cap structures, including the advanced Cap-2 type, which is found in animal and plant cells but has been difficult to study due to purity challenges [6] [17].
Table 1: Key Advantages of the PureCap Method Over Conventional Capping
| Feature | PureCap Method | Conventional Co-Transcriptional Capping |
|---|---|---|
| Capping Efficiency | 98% - 100% [6] [2] | 80% - 90% [2] |
| Key Impurity Removal | Simultaneously removes uncapped mRNA and dsRNA [2] | Often requires additional enzymatic steps to remove uncapped mRNA [2] |
| Cap Structure Versatility | Enables high-purity production of Cap-0, Cap-1, and Cap-2 [6] | Typically limited to Cap-0 and Cap-1 |
| Impact on Protein Expression | Cap-2 mRNA shows 3- to 5-fold higher protein production vs. Cap-1 [6] [17] | Standard Cap-1 performance |
| Immunostimulation | Significantly lower inflammatory response [6] [2] | Higher risk due to uncapped mRNA impurities |
The following diagram illustrates the core workflow of the PureCap method, from IVT to purified mRNA.
Strategic Considerations for Scaling Up mRNA Production
Integrating a new technology like PureCap into a commercial manufacturing process requires careful strategic planning. Early decisions on process design and raw material selection are critical for ensuring scalability, consistency, and regulatory compliance.
Capping Strategy Selection
The choice between co-transcriptional capping (as used in PureCap) and post-transcriptional enzymatic capping is one of the most decisive factors in process design. This decision profoundly impacts yield, process speed, and the design of downstream purification steps [52] [53]. While enzymatic capping is highly efficient, the PureCap co-transcriptional approach simplifies the workflow by combining capping and impurity resolution into a single, streamlined process, potentially reducing the number of unit operations and overall cost [2].
Raw Materials and Template Supply
Secring a sufficient and consistent supply of critical raw materials is paramount for scale-up. This includes:
- PureCap Analogs: Must be sourced as GMP-grade materials to ensure quality and regulatory compliance [54].
- DNA Template: Plasmid DNA (pDNA) supply can be a bottleneck. It is crucial to ensure a steady supply of GMP-grade pDNA or consider synthetic alternatives like doggybone DNA (dbDNA), which can also serve as a template for IVT [54] [52].
- Enzymes and NTPs: Using GMP-suitable enzymes and nucleotides from the outset of process development facilitates a smoother transition to commercial manufacturing [54] [55]. Early supplier qualification for these materials mitigates supply chain risks.
Process Optimization and Scale-Up Approach
A platform process philosophy, where unit operations are scalable and interchangeable, is highly recommended [52]. Employing Design of Experiments (DoE) is a powerful tool for optimizing IVT conditions. One case study demonstrated that screening over 50 different IVT conditions led to a two- to threefold increase in mRNA yields [52] [53]. Furthermore, a modular approach to manufacturing, using the same technology across different scales (from microliters to liters), can increase flexibility and reduce costs [54].
Experimental Protocols for Process Development
Protocol 1: In Vitro Transcription with PureCap Analogs
This protocol describes the IVT reaction optimized for the incorporation of PureCap analogs.
- Reaction Setup: In a nuclease-free tube, assemble the following components on ice:
- Linearized DNA Template: 0.05 mg/mL (final concentration) [55].
- Nucleotide Triphosphates (ATP, GTP, CTP, UTP): 5 mM of each nucleotide (final concentration) [55]. Modified nucleotides like N1-Methylpseudouridine can be substituted to reduce immunogenicity [55].
- PureCap Analog: 4 mM (final concentration) [55].
- Transcription Buffer: As supplied by the enzyme manufacturer.
- T7 RNA Polymerase Mix: As per manufacturer's specifications.
- Incubation: Mix the reaction thoroughly and incubate at 37°C for 2-4 hours with shaking [55]. The reaction time and temperature can be optimized via DoE to maximize yield and full-length product [54].
- DNase Treatment: After transcription, add DNase I and incubate for an additional 15-30 minutes at 37°C to digest the DNA template [55].
- Reaction Quenching: Add EDTA to a final concentration of 10-20 mM to chelate divalent cations and stop the reaction [55].
Protocol 2: Purification of Capped mRNA via RP-HPLC
This protocol outlines the key steps for separating capped mRNA using the hydrophobic tag.
- Sample Preparation: The crude IVT reaction mixture may require dilution or buffer exchange into the RP-HPLC mobile phase. Centrifugation or filtration is recommended to remove any particulate matter.
- Chromatography Setup:
- Column: Use a suitable preparative-scale C18 or C8 reversed-phase column.
- Mobile Phase: A: 0.1 M Triethylammonium acetate (TEAA) buffer; B: Acetonitrile.
- Detection: Monitor absorbance at 260 nm.
- Elution: Employ a linear gradient of mobile phase B. The hydrophobic PureCap-tagged mRNA will elute at a higher acetonitrile concentration than uncapped mRNA and other impurities [2].
- Fraction Collection: Collect the peak fraction corresponding to the PureCap-tagged, capped mRNA. The identity of the peak can be confirmed by analytical RP-HPLC or mass spectrometry during method development.
Protocol 3: Photocleavage of the Hydrophobic Tag
This final step recovers the native mRNA structure.
- Sample Preparation: Pool the collected RP-HPLC fractions containing the PureCap-tagged mRNA.
- Irradiation: Expose the sample to long-wavelength UV light for a defined period. The 2-nitrobenzyl group is cleaved under these mild conditions, leaving no residual modification on the mRNA [2].
- Buffer Exchange and Concentration: The deprotected mRNA is then buffer-exchanged into an appropriate storage buffer and concentrated using tangential flow filtration (TFF) [55]. A case study on TFF demonstrated greater than 80% mRNA recovery across various feed rates and mRNA sizes [52].
Performance Data and Analytical Characterization
Robust analytical methods are essential for characterizing the purified mRNA and validating the process. The following table summarizes key quality attributes and the performance of mRNA produced using the PureCap method.
Table 2: Analytical Profile of PureCap-Purified mRNA
| Critical Quality Attribute (CQA) | Analytical Method | Performance of PureCap mRNA |
|---|---|---|
| Capping Efficiency | RP-HPLC / LC-MS | 98% - 100% [6] [2] |
| Capping Type | Mass Spectrometry | Cap-0, Cap-1, and Cap-2 structures [6] |
| Protein Expression Level | In vitro translation / in vivo models | Cap-2 mRNA: 3- to 4-fold higher than standard Cap-1 mRNA [6] [2] |
| Immunostimulatory Activity | Cytokine assay on immune cells | Significantly lower than conventionally capped mRNA [6] [2] |
| mRNA Integrity/Purity | Agarose Gel Electrophoresis / Bioanalyzer | High integrity, low dsRNA contamination [2] |
| Identity & Sequence | Sequencing | Confirmed for various lengths (650 nt to 4247 nt) [2] |
The superior performance of PureCap-derived mRNA, particularly the Cap-2 variant, is not merely a function of purity but also of its enhanced biological activity. The Cap-2 structure is more efficiently recognized by the cellular translation machinery, leading to a dramatic increase in protein synthesis. Concurrently, it is a poorer ligand for innate immune receptors like RIG-I, resulting in a much lower inflammatory profile [2]. This combination of high potency and low reactogenicity is the hallmark of a next-generation mRNA therapeutic.
The Scientist's Toolkit: Essential Reagents and Materials
Successful implementation of this scaled-up process requires carefully selected, high-quality materials. The following table lists key reagents and their functions.
Table 3: Research Reagent Solutions for Scaling Up PureCap mRNA Production
| Reagent / Material | Function | Critical Considerations for Scale-Up |
|---|---|---|
| PureCap Analogs | Co-transcriptional capping agent with hydrophobic tag for purification. | Transition to GMP-grade material early in development [54]. Ensure supplier can provide required quantities and documentation. |
| GMP-grade NTPs | Building blocks for mRNA synthesis. | Use GMP-grade NTPs to ensure low endotoxin levels and consistent performance [54] [55]. |
| T7 RNA Polymerase | Enzyme for catalyzing mRNA transcription from DNA template. | Supplier variability can cause 2-fold yield differences [52]. Quality, activity, and GMP-compliance are key. |
| Linearized DNA Template | Template for IVT reaction. | pDNA or dbDNA. Ensure high quality (e.g., >70% supercoiled ratio, correct sequence) and secure supply chain [54] [55]. |
| RP-HPLC System & Columns | Purification of capped mRNA from impurities. | Scalability from analytical to preparative and production columns is critical. |
| TFF System | Buffer exchange and concentration of final mRNA drug substance. | Optimize cross-flow feed rates to achieve >80% recovery [52]. |
The path to commercial mRNA production demands processes that are not only scalable but also designed to maximize product quality and efficacy. The integration of the PureCap method directly addresses a fundamental purity challenge, enabling the manufacture of completely capped mRNA with unprecedented efficiency. The resulting Cap-2 mRNA demonstrates a powerful combination of enhanced translational capacity and reduced immunogenicity, setting a new standard for therapeutic performance [6] [17]. By adopting a strategic approach that includes early process optimization, careful raw material selection, and the implementation of the detailed protocols herein, developers can de-risk their scale-up activities and accelerate the delivery of advanced mRNA vaccines and therapeutics to the clinic.
Proof of Performance: PureCap vs. Conventional Methods
Achieving 98-100% Capping Efficiency and Purity
The production of messenger RNA (mRNA) for therapeutic and vaccine applications requires a high degree of purity to ensure efficacy and minimize undesirable immune responses. A critical challenge in mRNA manufacturing has been the separation of desired capped mRNA from uncapped byproducts, which possess nearly identical physicochemical properties [56]. The PureCap method, developed by researchers at Nagoya University, represents a revolutionary approach to this problem, enabling the production of mRNA with 98-100% capping efficiency and purity [6] [18] [17]. This application note details the protocols for implementing this technology, framed within a broader thesis on purifying completely capped mRNA.
Traditional co-transcriptional capping methods, which use analogs like Anti-Reverse Cap Analogs (ARCA), typically achieve capping efficiencies of only 80-90% [56]. The remaining uncapped mRNA, with a 5' triphosphate group, can trigger innate immune receptors such as RIG-I and MDA5, potentially leading to inflammatory responses [56] [6]. The PureCap method overcomes this limitation through a strategic integration of hydrophobic tagging and photocleavable chemistry, facilitating physical separation based on hydrophobicity rather than relying solely on enzymatic degradation of impurities [56].
Core Principle and Design
The PureCap method utilizes specially engineered cap analogs modified with a hydrophobic, photocleavable tag. This tag, based on a 2-nitrobenzyl (Nb) derivative with a tert-butyl (tBu) group, is incorporated into the mRNA during the in vitro transcription (IVT) reaction [56]. The introduced hydrophobicity enables the separation of capped from uncapped mRNA using reversed-phase high-performance liquid chromatography (RP-HPLC), a standard and scalable purification technique. Following purification, the hydrophobic tag is completely removed under mild conditions via photoirradiation, yielding native, footprint-free capped mRNA [56] [18].
This process is summarized in the following workflow:
Performance and Advantages
The PureCap method achieves near-perfect capping efficiency and purity, significantly outperforming conventional techniques. The table below summarizes its key performance metrics and advantages.
Table 1: Performance Summary of the PureCap Method
| Parameter | PureCap Method Performance | Conventional Method Comparison |
|---|---|---|
| Capping Efficiency/Purity | 98-100% [6] [18] [17] | 80-95% [56] [57] |
| Cap Structures Accessible | Cap-0, Cap-1, and Cap-2 [56] [17] | Primarily Cap-0 and Cap-1 |
| Reported Protein Expression | 3-4x higher for Cap-2 mRNA vs. standard Cap-1 [56] [6] [18] | Baseline (Cap-1) |
| Immunogenicity Profile | Lower immunostimulatory activity [6] [17] | Higher due to uncapped mRNA and dsRNA impurities |
| Key Impurities Removed | Uncapped mRNA and double-stranded RNA (dsRNA) [56] | Requires additional enzymatic steps for impurity removal |
A major advantage of this technology is its ability to facilitate the study and production of Cap-2-type mRNA. Previously, obtaining pure Cap-2 mRNA for a fair functional evaluation was difficult [17]. Research using the PureCap method has demonstrated that Cap-2 mRNA exhibits significantly higher translation activity (3-4 fold increase) in both cultured cells and animal models, coupled with lower stimulation of the inflammatory response compared to mRNAs synthesized by conventional techniques [56] [6] [18].
Experimental Protocols
Protocol 1: In Vitro Transcription with PureCap Analogs
This protocol describes the synthesis of mRNA using hydrophobic photocleavable cap analogs.
Materials:
- Template DNA: Linearized plasmid DNA or PCR product with a T7 promoter sequence.
- Nucleotides: ATP, CTP, GTP, UTP.
- Enzyme: T7 RNA Polymerase.
- PureCap Analog: e.g., DiPure (1), DiPure/2′OMe (2), or DiPure/3′OMe (3) [56].
- Buffer: Commercially available T7 RNA polymerase reaction buffer.
Method:
- Prepare the IVT reaction mixture on ice as follows:
- Template DNA: 1 µg
- ATP, CTP, UTP: 7.5 mM each
- GTP: 1.5 mM
- PureCap Analog: 6 mM [56]
- T7 RNA Polymerase: 50 U
- Reaction Buffer: 1X
- Nuclease-free water to a final volume of 50 µL.
- Critical Step: The high ratio of PureCap analog to GTP is essential to maximize capping efficiency by outcompeting GTP for the initiation of transcription [56] [57].
- Incubate the reaction at 37°C for 2-4 hours.
- After incubation, the mRNA can be precipitated or proceed directly to purification (Protocol 2).
Protocol 2: RP-HPLC Purification of Capped mRNA
This protocol separates capped mRNA from uncapped mRNA and other impurities like dsRNA based on hydrophobicity.
Materials:
- HPLC System: Reversed-phase capable.
- HPLC Column: C18 or C8 column, suitable for RNA separation.
- Mobile Phase A: 0.1 M Triethylammonium acetate (TEAA) buffer, pH 7.0.
- Mobile Phase B: Acetonitrile.
- Sample: IVT reaction mixture, diluted if necessary.
Method:
- Equilibrate the HPLC column with a mixture of 95% Mobile Phase A and 5% Mobile Phase B.
- Inject the sample onto the column.
- Run a gradient elution. Example method:
- 0-5 min: Hold at 5% B.
- 5-30 min: Linear gradient from 5% B to 25% B.
- 30-35 min: Hold at 25% B.
- 35-40 min: Return to 5% B for column re-equilibration [56].
- Monitor the elution at 260 nm. The hydrophobic, capped mRNA will elute later than the uncapped mRNA due to its interaction with the stationary phase [56] [18].
- Collect the peak corresponding to the capped mRNA.
- Remove the acetonitrile by vacuum centrifugation or dialysis against nuclease-free water.
Protocol 3: Photocleavage of the Hydrophobic Tag
This protocol removes the hydrophobic tag to yield a native, functional mRNA.
Materials:
- Purified mRNA: From Protocol 2, in a transparent tube or plate.
- Light Source: UV lamp or LED light source, 365-370 nm [56].
Method:
- Transfer the collected mRNA solution to a quartz cuvette or UV-transparent microcentrifuge tube.
- Place the sample on ice to minimize potential heat damage from the light source.
- Irradiate the sample with 365-370 nm light for 15-30 minutes [56].
- Post-irradiation, the mRNA is now in its native form and can be stored at -80°C or used directly in downstream applications. Confirm complete tag removal and cap integrity using LC-MS/MS or other capping detection methods.
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of the PureCap method relies on specific reagents and tools. The following table details the essential components of the research toolkit.
Table 2: Key Research Reagent Solutions for the PureCap Method
| Reagent / Material | Function / Description | Examples / Specifications |
|---|---|---|
| PureCap Analogs | Hydrophobic, photocleavable cap analogs (e.g., DiPure derivatives) incorporated during IVT to enable purification. | DiPure (1), DiPure/2'OMe (2), DiPure/3'OMe (3) with a 2-nitrobenzyl-tert-butyl tag [56]. |
| RP-HPLC System | Chromatography system for separating capped and uncapped mRNA based on hydrophobicity. | System with C8 or C18 column; Mobile phases: 0.1M TEAA (pH 7.0) and Acetonitrile [56]. |
| Photocleavage Device | Light source for removing the hydrophobic tag after purification to yield native mRNA. | UV lamp or LED with output at 365-370 nm [56]. |
| Capping Analysis Tools | Methods to verify capping efficiency and structure after purification. | LC-MS/MS for direct cap identification; Cap-specific ELISA [57]. |
Analytical Verification and Workflow Integration
After purification and tag removal, it is crucial to verify the quality and functionality of the mRNA. The logical relationship between the PureCap process and downstream analytical verification is shown below.
Analytical methods such as LC-MS/MS are used to identify and quantify Cap-0, Cap-1, and Cap-2 structures directly, providing definitive proof of cap integrity [57]. Cell-based potency assays are then used to confirm the high translation efficiency of the purified mRNA, with PureCap-derived Cap-2 mRNA demonstrating 3- to 4-fold higher protein production compared to standard Cap-1 mRNA [56] [58]. This integrated workflow from synthesis to verification ensures the production of high-purity, highly active mRNA for research and therapeutic development.
This application note details the experimental evidence and methodologies for achieving a 3-to-4-fold enhancement in protein expression using messenger RNA (mRNA) with a Cap-2 5' cap structure, purified via the PureCap method. The data confirms that the superior purity of fully capped mRNA, attainable only through the PureCap platform, directly translates to significantly higher translational output in both in vitro and in vivo models, while concurrently reducing undesired immunostimulation. This provides researchers with a validated protocol to overcome the critical bottleneck of mRNA capping efficiency in therapeutic development.
The 5' cap is a fundamental modifier of mRNA function, influencing its stability, translational efficiency, and recognition by the innate immune system [2] [59]. Cap structures are categorized based on their methylation status: Cap-0 (m7GpppN...), Cap-1 (m7GpppNm...), and Cap-2 (m7GpppNmNm...) [2]. While Cap-1 is common in therapeutic mRNAs, Cap-2, prevalent in host cells, has been difficult to study due to the inability to produce it in pure form.
Traditional co-transcriptional capping methods using cap analogs (e.g., ARCA, CleanCap) suffer from a fundamental limitation: GTP competes with the cap analog for initiation of transcription. This results in a final product that is a mixture of capped and uncapped (5' triphosphate) mRNA [2] [59]. Uncapped mRNA is not only translated less efficiently but is also a potent trigger of innate immune responses via sensors like RIG-I, leading to increased immunogenicity and reduced overall protein yield [2].
The PureCap method revolutionizes this process by enabling the physical separation and purification of fully capped mRNA from uncapped byproducts, achieving capping efficiencies of up to 100% [2] [6]. This breakthrough finally allows for a direct and unbiased comparison of cap structure biology, revealing the superior performance of the Cap-2 structure.
The following tables summarize the core quantitative findings from the evaluation of PureCap-purified mRNAs.
Table 1: Comparative Analysis of Cap Structures and Their Properties
| Cap Type | 2'-O-Methylation | Traditional Capping Efficiency | PureCap Capping Efficiency | Relative Immunostimulatory Activity |
|---|---|---|---|---|
| Cap-0 | None | ~80-90% | ~100% | High |
| Cap-1 | First nucleotide | ~80-90% | ~100% | Medium |
| Cap-2 | First two nucleotides | Not achievable | ~100% | Low |
Data synthesized from [2] [6] [17].
Table 2: Direct Comparison of Protein Expression Output
| Experiment | mRNA Construct | Cap Type | Protein Expression Relative to Cap-1 |
|---|---|---|---|
| In vitro translation | Not specified | PureCap Cap-2 | 3 to 4-fold higher [2] |
| In vivo immunization | Not specified | PureCap Cap-2 | 3 to 5-fold higher [6] [17] |
| Cultured cells | 4247 nt coronavirus mRNA | PureCap Cap-2 | Significantly enhanced [2] [6] |
Detailed Experimental Protocols
Protocol 1: Synthesis and Purification of Fully Capped mRNA Using PureCap Analog
This protocol describes the production of pure Cap-2 mRNA via co-transcriptional capping and RP-HPLC purification.
I. Materials
- PureCap Cap-2 Analog: A dinucleotide trinosphate (e.g., m7G(5')ppp(5')GmUm) modified with a hydrophobic photocleavable tag (e.g., tert-butyl 2-nitrobenzyl) on the m7G moiety [2].
- In Vitro Transcription (IVT) Reagents: T7 RNA Polymerase, NTPs, reaction buffer, DNA template.
- Purification System: Reversed-Phase High-Performance Liquid Chromatography (RP-HPLC) system with a C18 column.
- Photocleavage Setup: UV light source (e.g., 365 nm).
II. Procedure
- IVT Reaction Setup: Perform a standard IVT reaction, substituting the conventional cap analog with the hydrophobic-tagged PureCap Cap-2 analog. The transcription reaction will initiate from this analog, incorporating it at the 5'-terminus of the RNA chain [2].
- RP-HPLC Purification:
- Load the crude IVT reaction mixture onto the RP-HPLC column.
- Elute the sample using a gradient of a hydrophobic solvent (e.g., acetonitrile) in an aqueous buffer.
- The hydrophobic tag on the capped mRNA will significantly increase its retention time compared to the untagged, uncapped mRNA and other impurities like dsRNA [2] [59].
- Collect the separated peak corresponding to the capped mRNA.
- Tag Removal via Photocleavage:
- Quality Control: Analyze the final mRNA product by analytical RP-HPLC and/or capillary electrophoresis to confirm >98% purity and the absence of uncapped species [6].
Protocol 2: Assessing Protein Expression in Cultured Cells
This protocol is used to quantitatively compare the translational efficiency of different capped mRNAs.
I. Materials
- Cell line relevant to the research (e.g., HEK-293, HeLa, or dendritic cells).
- PureCap-purified Cap-0, Cap-1, and Cap-2 mRNAs encoding a reporter protein (e.g., Firefly Luciferase or EGFP).
- Transfection reagent (e.g., lipid nanoparticles (LNPs) or a commercial transfection agent).
- Lysis buffer and reporter assay kit (e.g., luciferase assay system).
- Microplate reader.
II. Procedure
- Cell Seeding: Seed cells in a multi-well plate and culture until they reach 70-80% confluency.
- mRNA Transfection: Transfect cells with equal masses of the purified Cap-0, Cap-1, and Cap-2 mRNAs using the transfection reagent. Include an untransfected control.
- Incubation: Incubate the cells for a predetermined period (e.g., 24 hours) to allow for protein expression.
- Protein Quantification:
- Lyse the cells.
- Measure the reporter protein activity (e.g., luminescence for luciferase) or fluorescence (for EGFP) according to the assay kit's instructions.
- Normalize the readings to total protein concentration or cell viability.
- Data Analysis: Compare the normalized protein expression levels. The Cap-2 mRNA is expected to yield a 3-to-4-fold higher signal than the Cap-1 mRNA, as reported in prior studies [2] [17].
Workflow and Mechanism Visualization
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for PureCap mRNA Research
| Reagent / Material | Function in the Protocol | Key Feature / Rationale |
|---|---|---|
| Hydrophobic PureCap Analog | Serves as the transcription initiator for co-transcriptional capping. | Contains a photocleavable tag (e.g., 2-nitrobenzyl derivative) for RP-HPLC separation [2]. |
| RP-HPLC System with C18 Column | Physically separates capped mRNA from uncapped impurities based on hydrophobicity. | Critical for achieving near 100% capping purity; also removes dsRNA contaminants [2] [6]. |
| UV Illuminator (365 nm) | Removes the hydrophobic purification tag from the capped mRNA after purification. | Provides a mild, footprint-free method to recover the native mRNA structure [2]. |
| Lipid Nanoparticles (LNPs) | Delivery vehicle for transferring in vitro transcribed mRNA into cultured cells or animals. | Protects mRNA from degradation and facilitates endosomal escape for robust protein expression [59]. |
The purity of messenger RNA (mRNA), specifically the presence of a complete 5' cap structure, is a critical determinant of its immunogenic profile. Conventional methods for mRNA production result in a significant population of uncapped mRNA impurities, which contain a 5' triphosphate (ppp) moiety [2]. This ppp-RNA is recognized by innate immune sensors, such as retinoic acid-inducible gene-I (RIG-I) and melanoma differentiation-associated protein 5 (MDA5), triggering pro-inflammatory signaling pathways and unwanted immune activation [2] [60]. This impurity-derived inflammation is a common source of vaccine reactogenicity and can also inhibit the translation of the desired therapeutic protein [6] [18]. The PureCap method developed by researchers at Nagoya University addresses this fundamental problem by enabling the production of mRNA with nearly 100% capping efficiency, thereby providing a platform for therapeutics with inherently lower innate immune stimulation [6] [2] [17].
The PureCap Method: Mechanism and Workflow
The core innovation of the PureCap technology is the use of custom-synthesized cap analogs that incorporate a hydrophobic tag via a photolabile linker. This design allows for the physical separation of capped mRNA from uncapped impurities, a feat not possible with standard mRNA due to their nearly identical physicochemical properties [2].
Key Chemical Design and Separation Principle
The PureCap analogs are based on anti-reverse cap analog (ARCA) structures to ensure proper orientation during in vitro transcription (IVT) [2]. A critical modification is the introduction of a photocleavable hydrophobic tag (e.g., a 2-nitrobenzyl derivative with a tert-butyl group) at specific positions on the 7-methylguanosine (m7G) moiety [2] [61]. During the IVT reaction, this modified analog is incorporated exclusively at the 5' end of the mRNA transcript.
- Hydrophobic Tagging: The introduced tag makes the capped mRNA molecule significantly more hydrophobic than its uncapped counterpart.
- Chromatographic Separation: This difference in hydrophobicity allows for the highly efficient separation of the two species using reversed-phase high-performance liquid chromatography (RP-HPLC), a standard and scalable purification technique [6] [2] [18].
- Tag Removal: Following purification, the hydrophobic tag is cleanly removed by mild UV irradiation, which cleaves the photolabile linker, yielding native, footprint-free, fully capped mRNA [2] [61].
Table 1: Key Research Reagent Solutions for the PureCap Method
| Reagent/Material | Function | Key Characteristic |
|---|---|---|
| PureCap Analog | Cap analog for IVT; incorporates hydrophobic tag | Contains photocleavable 2-nitrobenzyl group with tert-butyl moiety [2]. |
| RP-HPLC System | Purification platform | Separates capped and uncapped mRNA based on hydrophobicity [2] [61]. |
| UV Light Source | Cleavage of hydrophobic tag | Provides specific wavelength for clean tag removal without mRNA damage [2]. |
| T7 RNA Polymerase | Enzymatic RNA synthesis | Standard enzyme for in vitro transcription [2]. |
Visual Workflow of the PureCap Method
The following diagram illustrates the streamlined, enzymatic treatment-free workflow of the PureCap method for producing pure, capped mRNA.
Experimental Assessment of Innate Immune Activation
A critical component of profiling reduced immunogenicity is the direct measurement of innate immune responses to PureCap-synthesized mRNA in relevant cellular models.
Protocol: Quantifying Immune Sensor Activation
Objective: To compare the innate immune stimulation triggered by PureCap-derived mRNA versus mRNA produced by conventional capping methods.
- Cell Line: Immortalized human macrophage cell lines (e.g., THP-1) or primary human peripheral blood mononuclear cells (PBMCs) are suitable models [60].
- mRNA Transfection:
- Culture cells under standard conditions.
- Transfect cells with equimolar amounts (e.g., 100 ng/well in a 24-well plate) of mRNA using a standard lipid nanoparticle (LNP) formulation or a commercial transfection reagent. Key samples include:
- Test Article: PureCap-synthesized mRNA (Cap0, Cap1, or Cap2).
- Control Article: mRNA with identical sequence but synthesized using a conventional cap analog (e.g., ARCA).
- Positive Control: Known immunostimulatory RNA (e.g., in vitro transcribed ppp-RNA).
- Negative Control: Buffer-only transfection.
- Incubate for 6-24 hours post-transfection [2].
- Downstream Analysis:
- Gene Expression: Harvest cells and extract total RNA. Perform quantitative RT-PCR (qRT-PCR) to measure the induction of type I interferons (e.g., IFN-β), interferon-stimulated genes (ISGs) like ISG15 or OAS1, and pro-inflammatory cytokines (e.g., TNF-α, IL-6) [2] [60].
- Protein Secretion: Collect cell culture supernatants. Use enzyme-linked immunosorbent assays (ELISAs) to quantify secreted IFN-β, TNF-α, and IL-6 proteins [60].
Key Findings on Immunogenicity Reduction
Studies using the protocol above have consistently demonstrated the superior profile of PureCap-derived mRNA. The high purity of the preparation means that ligands for innate immune receptors are drastically reduced.
- Reduced Inflammatory Signaling: Highly purified Cap0, Cap1, and Cap2-type mRNAs synthesized using the PureCap method showed lower immunostimulatory activity compared to mRNAs synthesized using conventional techniques [6] [17] [18]. This is directly attributed to the removal of uncapped ppp-RNA impurities.
- Cap2 as a "Self" Signal: The immune system is highly attuned to viral RNA patterns. Since viruses predominantly produce Cap1 mRNA, the less common Cap2 structure is interpreted more as "self." PureCap technology enables, for the first time, the production of pure Cap2 mRNA for evaluation. It was found that Cap2 mRNA caused lower stimulation of the inflammatory response than mRNAs synthesized using conventional techniques [6] [18]. This suggests Cap2 can further evade recognition by cytosolic immune receptors [61].
Table 2: Quantitative Comparison of Innate Immune Activation by mRNA Type
| mRNA Sample | Capping Efficiency | Relative IFN-β mRNA Induction | Relative IL-6 Secretion | Key Impurity |
|---|---|---|---|---|
| Conventional Cap1 | ~80-90% [2] | High | High | Uncapped ppp-RNA |
| PureCap Cap0 | ~98-100% [6] [2] | Low | Low | None detected |
| PureCap Cap1 | ~98-100% [6] [2] | Low | Low | None detected |
| PureCap Cap2 | ~98-100% [6] [2] | Lowest | Lowest | None detected |
Underlying Signaling Pathways in Innate Immune Evasion
The reduced immunogenicity of PureCap-purified mRNA is explained by its inability to activate specific cytoplasmic pattern recognition receptors. The following diagram outlines the key pathways involved.
As illustrated, the key mechanistic insight is that uncapped ppp-RNA is a potent ligand for RIG-I, while double-stranded RNA (dsRNA) impurities, which are also removed during RP-HPLC purification, can activate MDA5 [2] [60]. The PureCap method removes both these classes of immunostimulatory impurities. Furthermore, the Cap2 structure itself has a lower affinity for these immune receptors compared to Cap1, providing an additional layer of immune evasion [61].
The PureCap method represents a significant advancement in mRNA production technology by directly addressing the challenge of impurity-driven immunogenicity. By enabling the synthesis of mRNA with 98-100% capping efficiency, particularly with the novel Cap2 structure, it provides a powerful tool for developing safer and more effective mRNA-based vaccines and therapeutics. The documented reduction in innate immune activation, coupled with enhanced protein expression, positions this technology as a cornerstone for future applications in not only prophylactic vaccines but also in sensitive areas like cancer immunotherapy and regenerative medicine, where controlling inflammatory responses is paramount [6] [61] [62]. The provided protocols and data offer researchers a clear roadmap for implementing and validating this technology in their own work.
Superiority Over Enzymatic Capping and Standard Co-Transcriptional Methods
The 5' cap structure is a critical determinant of efficacy for messenger RNA (mRNA) vaccines and therapeutics, serving to enhance stability, promote translation initiation, and reduce immunogenicity by marking the transcript as "self" [13]. Conventional methods for adding this cap during in vitro transcription (IVT) – enzymatic capping and standard co-transcriptional capping with analogs – are fundamentally limited, with maximum capping efficiencies of 80-90% and 70-95%, respectively [13] [63] [2]. These processes inevitably leave residual uncapped mRNA impurities, which possess a 5' triphosphate that can trigger unwanted innate immune responses via receptors like RIG-I and MDA5, potentially compromising therapeutic safety and efficacy [2].
The PureCap method represents a paradigm shift in mRNA production. This novel approach utilizes custom cap analogs featuring a hydrophobic, photocleavable tag, enabling the physical separation of fully capped mRNA from uncapped impurities and other byproducts like double-stranded RNA (dsRNA) for the first time [17] [2]. This application note details how the PureCap method achieves superiority over existing techniques by guaranteeing 100% capping efficiency, facilitating the production of advanced cap structures, and ultimately yielding mRNA with enhanced translational capacity and reduced immunogenicity.
Performance Comparison of Capping Methods
The following tables summarize the key performance metrics of different capping methods, highlighting the quantitative advantages of the PureCap technology.
Table 1: Quantitative Comparison of Capping Method Efficiencies and Output
| Capping Method | Maximum Capping Efficiency | Key Characteristic | Reported Protein Expression vs. Standard Cap 1 |
|---|---|---|---|
| Standard Co-transcriptional (e.g., ARCA) | ~80-90% [2] | Cap 0 structure; prone to reverse incorporation [13] | Baseline |
| Advanced Co-transcriptional (e.g., CleanCap) | >95% [13] | Cap 1 structure; high-fidelity incorporation [13] | Not Specified |
| Enzymatic Capping | 80-100% [63] [9] | Cap 0 or Cap 1 structure; requires multiple steps [13] [63] | Not Specified |
| PureCap Method | 100% [17] [2] | Enables pure Cap 0, Cap 1, or Cap 2 production [17] [2] | Cap 2 mRNA: 3-4x higher [17] [2] |
Table 2: Process and Commercial Considerations for Capping Methods
| Capping Method | Process Steps | Immunogenic Byproducts | Intellectual Property (IP) Landscape |
|---|---|---|---|
| Standard Co-transcriptional | Single IVT reaction [13] | Uncapped mRNA, dsRNA [2] | Constrained by IP and licensing [64] |
| Enzymatic Capping | IVT + enzymatic reaction + purification [13] [63] | Uncapped mRNA (if inefficient) | More freedom to operate [64] |
| PureCap Method | IVT + RP-HPLC + photo-irradiation [2] | None (fully capped mRNA) [2] | New, versatile platform |
As the data indicates, the PureCap method's ability to achieve 100% capping efficiency and produce pure Cap 2 mRNA with significantly higher biological activity establishes a new benchmark for mRNA therapeutic quality.
Experimental Workflow for PureCap mRNA Production
The following diagram illustrates the streamlined workflow for producing fully capped mRNA using the PureCap method.
Diagram 1: PureCap mRNA Production Workflow
Protocol: In Vitro Transcription with PureCap Analog
This protocol is adapted from the methodology detailed by Hiroshi Abe et al. [17] [2].
- Step 1: Reaction Setup. Assemble the IVT reaction on ice in a nuclease-free microcentrifuge tube. A typical 25 μL reaction contains:
- 5 μL 5X Transcription Buffer (e.g., 400 mM Tris-HCl pH 8.0, 40 mM MgCl₂, 10 mM spermidine, 50 mM NaCl) [9]
- 4 μL 10 mM rNTP Mix (ATP, CTP, UTP, modified nucleotides)
- 2.5 μL 100 mM DTT
- 1-2 μg linearized DNA template
- PureCap Analog (concentration to be optimized, e.g., 4-8 mM)
- 2 μL T7 RNA Polymerase
- Nuclease-free water to 25 μL
- Step 2: Transcription. Incubate the reaction at 37°C for 2 hours. For longer mRNAs (>4,000 nt), extending the incubation time to 4-6 hours may improve yield.
- Step 3: DNase I Treatment. After transcription, add 1 μL of DNase I (RNase-free) to the 25 μL reaction and incubate at 37°C for 15 minutes to digest the DNA template.
Protocol: Purification and Tag Cleavage via RP-HPLC and Photo-irradiation
This purification step is the core of the PureCap method, enabling the physical separation of capped mRNA [17] [2].
- Step 1: RP-HPLC Setup. Use a reversed-phase HPLC system equipped with a C18 or similar hydrophobic interaction column. The mobile phases are typically:
- Mobile Phase A: 0.1 M Triethylammonium Acetate (TEAA) buffer, pH 7.0
- Mobile Phase B: Acetonitrile
- Step 2: Sample Preparation. Dilute the IVT reaction mixture with an equal volume of Mobile Phase A and inject onto the column.
- Step 3: Gradient Elution. Execute a linear gradient from 5% to 60% Mobile Phase B over 30-40 minutes. The hydrophobic tag on the PureCap analog will cause the correctly capped mRNA to elute later than uncapped mRNA and other impurities.
- Step 4: Fraction Collection. Monitor the elution at 260 nm and collect the peak corresponding to the tagged, capped mRNA.
- Step 5: Photo-irradiation. Place the collected fraction in a quartz cuvette or transparent tube. Irradiate with long-wavelength UV light (e.g., ~365 nm) for 15-30 minutes to cleave the hydrophobic 2-nitrobenzyl tag. This step restores the native cap structure without any chemical footprint.
- Step 6: Desalting and Concentration. The mRNA can now be desalted and concentrated using standard methods like ethanol precipitation or centrifugal filters, resulting in a preparation of 100% pure, natively capped mRNA [17].
The Scientist's Toolkit: Essential Research Reagents
The following table lists key reagents and their functions essential for implementing the PureCap method and related mRNA research.
Table 3: Key Research Reagent Solutions for mRNA Capping and Analysis
| Reagent / Solution | Function / Application | Key Characteristic |
|---|---|---|
| PureCap Analogs [2] | Co-transcriptional incorporation of a hydrophobic, cleavable cap. | Enables RP-HPLC separation of capped mRNA; versatile for Cap 0, 1, 2. |
| POROS Oligo(dT)25 Resin [65] | Affinity capture of mRNA via poly-A tail during purification. | Scalable purification platform for IVT mRNA. |
| Faustovirus Capping Enzyme (FCE) [63] [9] | Single-subunit enzyme for post-transcriptional enzymatic capping. | High activity for generating Cap-0 structure. |
| mRNA Cap 2´-O-Methyltransferase [63] | Converts Cap-0 to Cap-1 structure in enzymatic capping. | Used in conjunction with capping enzymes. |
| Codex HiCap RNA Polymerase [66] | Engineered T7 RNAP for superior co-transcriptional capping. | >95% capping efficiency with analogs like CleanCap. |
| 5' CapQ Assay Kit [37] | Rapid quantification of intact, capped, and tailed mRNA. | 2-hour benchtop assay for capping efficiency and integrity. |
The PureCap method directly addresses the fundamental limitation of all previous capping technologies: the inability to completely eliminate immunogenic uncapped mRNA impurities. By enabling the production of mRNA with 100% capping efficiency and providing access to superior cap structures like Cap 2, this technology offers researchers and drug developers a clear path to creating safer and more potent mRNA-based vaccines and therapeutics. The streamlined, non-enzymatic purification workflow positions the PureCap method as a robust and scalable solution for the next generation of mRNA medicines.
Conclusion
The PureCap method represents a paradigm shift in mRNA production, effectively solving the long-standing challenge of purifying completely capped mRNA. By enabling the physical separation of capped from uncapped mRNA, it ensures unprecedented purity levels that directly translate to enhanced therapeutic protein production and significantly reduced inflammatory side effects. The method's ability to reliably produce Cap-2 mRNA, which demonstrates superior translation and lower immunogenicity, opens new avenues for highly effective and well-tolerated vaccines and therapeutics. As a revolutionary advance, PureCap not only provides a robust platform for current mRNA applications but also deepens our fundamental understanding of mRNA biology, accelerating the development of next-generation treatments for infectious diseases, cancer, and regenerative medicine.
References
- 1. -prime Five - Wikipedia cap [https://en.wikipedia.org/wiki/Five-prime_cap]
- 2. Cap analogs with a hydrophobic photocleavable tag ... [https://www.nature.com/articles/s41467-023-38244-8]
- 3. Photocaged 5′ cap analogues for optical control of mRNA ... [https://www.nature.com/articles/s41557-022-00972-7]
- 4. , CDS, UTR & PolyA in Cap Sequence - mRNA / BOC Sciences RNA [https://rna.bocsci.com/support/introduction-to-mrna-sequence-elements-cap-cds-utr-polya.html]
- 5. The major mRNA -associated protein YB-1 is a potent... [https://pmc.ncbi.nlm.nih.gov/articles/PMC125650/]
- 6. Purecap method offers hope for purer and safer mRNA ... [https://www.news-medical.net/news/20230705/PureCap-method-offers-hope-for-purer-and-safer-mRNA-vaccines.aspx]
- 7. mRNA therapeutics: Transforming medicine through ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12590236/]
- 8. Highly pure mRNA vaccine provides robust immunization ... [https://pubmed.ncbi.nlm.nih.gov/40383159/]
- 9. A comparative exploration of mRNA capping enzymes - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11625350/]
- 10. Prospects and Challenges in Developing mRNA Vaccines ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11130901/]
- 11. Safety and immunogenicity of an optimized self-replicating ... [https://www.nature.com/articles/s41467-025-55843-9]
- 12. Optimizing mRNA Capping and Analysis [https://www.neb.com/en-us/nebinspired-blog/optimizing-rna-capping-and-analysis?srsltid=AfmBOop81lz3K0B2D6m1d0GNdjWm8z_BZ3TkhUx00C2X2EHcjWsQR2pq]
- 13. Methods of IVT mRNA capping - Behind the Bench [https://www.thermofisher.com/blog/behindthebench/methods-of-ivt-mrna-capping/]
- 14. A general method for rapid and cost-efficient large-scale ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4986899/]
- 15. Combining an optimized mRNA template with a double ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8523304/]
- 16. Pure capped mRNA vaccine opens the door to more ... [https://en.nagoya-u.ac.jp/news/articles/research_information_151/]
- 17. Pure capped mRNA vaccine encourages more effective ... [https://www.drugtargetreview.com/news/110685/pure-capped-mrna-vaccine-encourages-more-effective-vaccine-development/]
- 18. Pure capped mRNA vaccine opens the door to more ... [https://www.sciencedaily.com/releases/2023/07/230705105841.htm]
- 19. Pure capped mRNA vaccine opens the door to more ... [https://www.eurekalert.org/news-releases/994412]
- 20. Comprehensive Impurity Profiling of mRNA [https://pmc.ncbi.nlm.nih.gov/articles/PMC10918618/]
- 21. Advancing continuous encapsulation and purification of ... [https://pubmed.ncbi.nlm.nih.gov/40578454/]
- 22. Safety, immunogenicity and efficacy of the self-amplifying ... [https://www.nature.com/articles/s41467-024-47905-1]
- 23. Cap analogs with a hydrophobic photocleavable tag ... [https://pubmed.ncbi.nlm.nih.gov/37169757/]
- 24. mRNA capping: biological functions and applications - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5027499/]
- 25. Co-transcriptional capping [https://www.takarabio.com/learning-centers/mrna-and-cdna-synthesis/mrna-synthesis/5-prime-capping-of-mrna/co-transcriptional-capping?srsltid=AfmBOopjmtK_EABw98Z-MwCZQ_WlDmqb3p0xoILkIoKXoylt_34VacW2]
- 26. Reverse Phase High Performance Liquid Chromatography [https://www.sciencedirect.com/topics/chemistry/reverse-phase-high-performance-liquid-chromatography]
- 27. The Hydrophobic-Subtraction Model for Reversed-Phase ... [https://www.chromatographyonline.com/view/hydrophobic-subtraction-model-reversed-phase-liquid-chromatography-reprise]
- 28. Anion exchange HPLC monitoring of mRNA in vitro ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10955092/]
- 29. Hydrophobic interaction chromatography [https://www.sciencedirect.com/science/article/pii/B9780323999687000266]
- 30. Separation of mAbs molecular variants by analytical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4171020/]
- 31. Advancing mRNA technologies for therapies and vaccines [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2022.1018961/full]
- 32. Recent Advances and Innovations in the Preparation and ... [https://www.mdpi.com/1999-4923/15/9/2182]
- 33. How to Achieve More Sustainable mRNA Synthesis and ... [https://www.thermofisher.com/blog/behindthebench/how-to-achieve-more-sustainable-mrna-synthesis-and-purification/]
- 34. mRNA Capping Methods Explained [https://rna.bocsci.com/support/mrna-capping-techniques-cap-0-cap-1-and-co-transcriptional-methods.html]
- 35. 2′-O-Methylation of the second transcribed nucleotide ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9458431/]
- 36. Assay for rapid quantification of capped and tailed intact ... [https://www.sciencedirect.com/science/article/abs/pii/S0264410X2500636X]
- 37. Assay for rapid quantification of capped and tailed intact ... [https://pubmed.ncbi.nlm.nih.gov/40466481/]
- 38. with a Cap ... | Nature Communications analogs hydrophobic [https://www.nature.com/articles/s41467-023-38244-8?error=cookies_not_supported]
- 39. Research | Abe Group, Department of Chemistry, Graduate School of... [https://biochemistry.chem.nagoya-u.ac.jp/research-e/]
- 40. Recent Advances in Modified Cap Analogs: Synthesis ... - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9111249/]
- 41. RNA Cap Analog Selection Chart [https://www.neb.com/en-us/tools-and-resources/selection-charts/rna-cap-analog-selection-chart?srsltid=AfmBOoqsjyrxHsUTQuY_-zNimv-zT1X1j2ujvBnaXXxghvFGKnpv0h3q]
- 42. Highly pure mRNA vaccine provides robust immunization ... [https://www.sciencedirect.com/science/article/pii/S0168365925004808]
- 43. mRNA Purification Methods and Process [https://lifesciences.danaher.com/us/en/library/mrna-purification.html]
- 44. Characterisation and analysis of mRNA critical quality ... [https://www.sciencedirect.com/science/article/pii/S0021967325000731]
- 45. mRNA Characterization, from 5' Cap to Poly (A): What Ion- ... [https://www.chromatographyonline.com/view/mrna-characterization-from-5-cap-to-poly-a-what-ion-pair-reversed-phase-liquid-chromatography-ip-rplc-analysis-can-tell-you]
- 46. Separation of long-stranded RNAs by RP-HPLC using an ... [https://pubmed.ncbi.nlm.nih.gov/36566342/]
- 47. Liquid Chromatography Methods for Analysis of mRNA ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10535021/]
- 48. mRNA Analysis Using (U)HPLC Separation Methods [https://www.thermofisher.com/us/en/home/industrial/chromatography/chromatography-learning-center/chromatography-consumables-resources/biopharmaceutical-analysis-u-hplc/mrna-analysis-using-u-hplc-separation-methods.html]
- 49. Photocaged 5′ cap analogues for optical control of mRNA ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7613264/]
- 50. Comparison of capillary electrophoresis-based methods for ... [https://pubmed.ncbi.nlm.nih.gov/39029354/]
- 51. Recent Advances and Innovations in the Preparation and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10536309/]
- 52. Successfully Transitioning mRNA Production To cGMP [https://www.lonza.com/knowledge-center/biologics/a/successfully-transitioning-mrna-production-to-cgmp]
- 53. Insights into mRNA Scale-Up and cGMP Manufacturing ... [https://www.genengnews.com/sponsored/insights-into-mrna-scale-up-and-cgmp-manufacturing-challenges/]
- 54. Five Tips on Scaling Up IVT for mRNA Production [https://www.thermofisher.com/us/en/home/industrial/pharma-biopharma/nucleic-acid-therapeutic-development-solutions/nucleic-acid-therapeutic-case-studies/mrna-scale-up-tips.html]
- 55. Development of mRNA manufacturing for vaccines and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8641981/]
- 56. Cap analogs with a hydrophobic photocleavable tag enable ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10175277/]
- 57. mRNA Capping: Process, Mechanisms, Methods & Applications [https://lifesciences.danaher.com/us/en/library/mrna-capping.html]
- 58. Evaluating mRNA capping systems: experimental ... [https://www.insights.bio/nucleic-acid-insights/webinars/750/evaluating-mrna-capping-systems-experimental-strategies-for-mrna-manufacturing]
- 59. Cell Reprogramming and Differentiation Utilizing ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10971469/]
- 60. Knife's edge: Balancing immunogenicity and reactogenicity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10394010/]
- 61. Cell Reprogramming and Differentiation Utilizing ... [https://www.mdpi.com/2221-3759/12/1/1]
- 62. Current Progress and Future Perspectives of RNA-Based ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12153701/]
- 63. mRNA Post-Transcriptional Enzymatic Capping Market ... [https://www.insightaceanalytic.com/report/mrna-post-transcriptional-enzymatic-capping-market/3104]
- 64. Comparing Capping - Aldevron Thought Leadership [https://www.aldevron.com/blog/comparing-capping]
- 65. Process optimization of mRNA purification for vaccines and ... [https://www.insights.bio/nucleic-acid-insights/webinars/576/process-optimization-of-mrna-purification-for-vaccines-and-therapeutic-applications]
- 66. Potential Unlocked! [https://www.aldevron.com/blog/enable-mrna-therapeutic-development]